Abstract
Introduction:
Chronic limb-threatening ischemia (CLTI) represents the most severe form of peripheral artery disease (PAD). Up to a third of CLTI patients are not eligible to receive first-line treatments such as bypass surgery or endovascular interventions. Epidural spinal cord stimulation (SCS) has been used as a method to improve microcirculatory blood flow and relieve ischemic pain in CTLI patients. The aim of the study was to evaluate limb salvage, ulcer closure, and clinical changes of SCS implanted CTLI patients at 1-year follow-up.
Methods:
Eligible patients had end-stage lower limb PAD unresponsive to medical therapy and not amenable to surgical reconstruction. Patients were candidates for amputation, but limb loss was not inevitable (Fontaine stage III and IV). Pain intensity and skin temperature in the ischemic area (visual analogue scale), quality of life (WHOQoL-BREF), and ankle/brachial blood pressure index (ABI) were recorded at routine follow-up visits. Data were analyzed retrospectively.
Results:
29 patients underwent SCS implantation at one vascular center. The minimum follow-up period was 30 months. Limb survival at 1-year follow-up was 97% (28/29) and 73% (11/15) had complete closure of limb ulcers. Pain intensity, skin temperature, and quality of life progressively improved up to 12 months after implant, with Fontaine stage III patients improving more substantially than Fontaine stage IV patients. SCS therapy did not affect ABI measurement. No complications related to the device or procedure occurred.
Conclusions:
SCS is a valid alternative in patients unsuitable for revascularization. The quality of results depends on both a strict selection of patients by vascular specialists and the frequency of follow-up controls. The therapy may be more beneficial in patients classified as Fontaine stage III.
Introduction
Chronic limb-threatening ischemia (CLTI) represents the most severe form of peripheral artery disease (PAD) and has a large impact on quality of life, morbidity, and mortality. 1 An estimated 5% to 10% of PAD patients aged >50 years develop CLTI within 5 years. 2 Patients with CLTI have severe hemodynamic impairment (ankle pressure are typically <50-70 mmHg) associated with complex anatomical patterns of arterial disease. Severe limb pain, claudication, ulcerations, and limb amputation are common complications. 3,4 The Society for Vascular Surgery Lower Extremity Threatened Limb Classification Scheme describes 4 clinical stages of CLTI based on 3 critical parameters that determine the level of limb threat: the presence and extent of wounds, the severity of ischemia, and associated foot infection (WIfI) staging. 5
The universal treatment approach for CLTI includes risk-factor modification, pharmacologic therapy, and revascularization through endovascular treatment or distal bypass to restore a pulsatile flow as distal as possible. 6 Ideal treatment allows patients to retain their limbs with no or tolerable pain and to regain a satisfactory level of independence. However, up to 34% of CLTI patients are not eligible to receive first-line treatments such as bypass surgery or angioplasty. 7 Patients without further treatment options have extensive and diffuse, often infrapopliteal, atherosclerotic lesions, comorbidity, and/or lack of a suitable bypass graft. 8 In the absence of effective revascularization, almost 25% of patients with CLTI require a major amputation within 12 months. 9 Various revascularization modalities have been used in practice to salvage CTLI patients who are not candidates for bypass or endovascular intervention. However, the efficacy of these interventions is unclear. 10 -13
Spinal cord stimulation (SCS) is a neuromodulator procedure representing an alternative treatment for patients with CLTI who are unsuitable for vascular reconstruction procedures. During SCS therapy, electrical pulses are applied to the dorsal column of the spinal cord via a thin electrical wire (called a lead) placed in the epidural space and connected to an implantable pulse generator (IPG). Although the therapy was first introduced in the 1960s for the treatment of pain, Cook et al. first used SCS in PAD in 1976. 14 They found an improvement in the healing of ulcers as well as in pain relief. In 2013, the Cochrane Collaboration reviewed 6 controlled studies, including a total of 444 patients, comparing SCS with conservative medical treatment in patients with non-reconstructable chronic CLTI (ischemic rest pain and ulceration). 15 The meta-analysis found that patients treated with SCS required less analgesia and had a significantly higher limb salvage rate after 12 months than patients who only had conservative medical treatment. However, SCS has not shown to prevent amputation in trials and cannot yet be recommended as standard treatment for non-reconstructable CLTI. 16 -18 Furthermore, in the previous studies there are no data about how many patients were receiving chronic pain medication at enrollment and how the use of said drugs changed during follow up. The aim of the study was to evaluate limb salvage, ulcer closure, and clinical changes of SCS implanted CTLI patients at 1-year follow-up.
Materials and Methods
Study Population
This was a single-center retrospective study of CLTI patients who were implanted with an SCS system at the Vascular and Endovascular Surgery Division of the University Hospital Essen, Germany, between January 2010 and December 2017. All of the study procedures adhered to the Declaration of Helsinki and were approved by the Institutional Ethics Committee of the Medical Faculty of the University of Duisburg-Essen (20-9609-BO). All patients provided written informed consent for SCS implantation. Inclusion criteria for the study were age >18 years, CLTI (Fontaine grade III-IV/Rutherford stage IV-V), ankle-brachial index (ABI) <0.6 or unreliable ABI, and ineligibility for surgical or endovascular intervention. All patients were candidates for amputation and had a life expectancy of 1 year or more. Exclusion criteria were a history of neoplasm or malignancy in the past 10 years, concomitant disease with life expectancy of less than 1 year and an inability to complete follow-up. Patients with rapidly progressing ischemia, gangrene of more than one toe, extensive infection and/or extensive non-healing ischemic ulcerations, or the presence of associated diseases prohibiting the use of SCS and patients with inadequate compliance due to psychological or social incompetence were also excluded from the study. The total length of follow-up was chosen at 1 year for a cutoff in the primary analyses.
All patients underwent digital subtraction arteriography. A multidisciplinary council, consisting of multiple vascular surgeons and interventional radiologists, determined if the patient was inoperable and without revascularization options. Reasons for the no-option designation included distal lesions not amenable to revascularization, no available autologous venous material, and lack of endovascular options.
Patient characteristics including age, gender, nicotine use, hypertension, diabetes mellitus and coronary artery disease were obtained from clinical charts and operation records. For all patients medication adjustments were allowed at any time during the study, if needed. All changes in medication in terms of frequency of administration and type of pain medication (narcotic analgesics were considered major analgesics, whereas nonsteroidal anti-inflammatory drugs were minor analgesics) were registered. However, reduction and changes in medication were not part of the study protocol.
Patients with isolated resting limb pain were diagnosed as Fontaine grade III (Rutherford stage IV), and those with resting limb pain together with ulcers that are less than 3 cm in diameter were diagnosed as Fontaine grade IV (Rutherford stage V). Operative data were collected at the time of surgery on standardized data forms.
Wounds were graded using the Wagner scale and they were included if they measured ≥ 1 cm in any dimension at the initial evaluation. This protocol is a standard protocol for care of limb ulcers used by many wound healing centers. Chronic wounds were defined as full-thickness ulcers extending through the entire dermis into the subcutaneous tissue present for a minimum of 6 weeks before initiation of treatment.
Follow-Up and End Points
Patient assessments were planned before intervention, at discharge, after 3, 6, 12 and 24 months, and annually thereafter or until a major endpoint (major amputation or death) was reached. No patients were lost to follow-up until 12 months after inclusion. These controls include a physical examination and technical controls of the stimulation system. Patients who did not attend follow-up visits were contacted by phone. Patient data were collected until the last follow-up visit or date of death.
ABI was measured whenever possible and were performed in accredited vascular laboratories. In general, a single pressure measurement of each limb vessel was performed. The ABI was considered immeasurable when inflation of the blood pressure cuff to a pressure of >250 mm Hg was insufficient to compress the arteries at ankle level.
Pain intensity was measured across a continuum of values on a visual analogue scale (VAS). VAS scores are recorded by making a handwritten mark on a 100-mm line that represents a continuum between 0 (no pain at all) and 100 (unbearable pain). The patients also assessed skin temperature in the ischemic area using the VAS from 0 (maximally cold) to 100(maximal warm). This parameter has been validated with a direct temperature. All patients were also asked to fill out the World Health Organization Quality of Life-BREF (WHOQOL-BREF). This is a self-report questionnaire that assesses 4 domains of quality of life: physical health, psychological health, social relationships, and environment. Items scored 1-5. Raw domain score is the sum of respective item scores. All domain scores are then normalized to a range of 0-100.
The primary endpoint of the study was major amputation and all-cause mortality. The secondary endpoint was the clinical outcome (Fontaine classification, pain sensation, quality of life, surgical complications) and wound closure 1 year after initiation of treatment. Death from progressive gangrene, sepsis, complications and 30-day perioperative mortality after amputation or implantation was classified as disease-specific mortality. Mortality was defined as post intervention ≥1-year all-cause mortality. Wound closure was defined as complete epithelial coverage.
Indications for amputation were progressive gangrene, intractable pain, or extensive infection and/or non-healing ulcerations. Limb salvage was defined as no amputation. The extent of amputation was classified in order of increasing handicap as none (no amputation, or minor amputations on the forefoot only), moderate (unilateral below knee amputation), or major (at or above knee level).
Surgical Procedures
One of 3 vascular surgeons carried out the implantation of the SCS system in each patient. A preoperative stimulation trial was not performed. The dorsal epidural space was punctured under local anesthesia, and a thin multielectrode lead was introduced under radiological control, between the thoracic 10-12 levels. Intra-operative paresthesia mapping confirmed coverage of the ischemic areas. The lead was anchored to the supraspinal fascia. A subcutaneous pouch was established in the left iliac fossa for the pulse generator (Genesis™ Implantable Pulse Generator, St. Jude Medical/Abbott, Plano, Texas, U.S.A.). A subcutaneous extension wire connected the lead and the pulse generator. All patients had an antiplatelet therapy with aspirin at enrollment and all patients have been treated preoperative successfully with prostaglandins. Prophylactic antibiotic treatment was administered. Patients were usually discharged a few days later after external telemetric programming of the pulse generator. The stimulation design consisted of pulses repeated at a 50 Hz frequency, pulse width 210/∼s and a stimulating current (mA) giving comfortable paresthesia in the ischemic area. One amplitude was programed for the upright position and a lower amplitude for horizontal bed rest positions.
The patients could start or stop the stimulation as well as switch between the 2 stimulation intensities and were encouraged to use the stimulator as often as they wished. During follow-up visits, the patients were interviewed regarding function of the stimulator, with emphasis on the adequacy of paresthesia in the ischemic area. The patients were instructed to contact the responsible physician between follow-up visits if the paresthesia coverage did not overlap the ischemic pain area any more. The safety and tolerability of SCS therapy over time was evaluated using information on treatment-emergent adverse events and device complications.
Statistical Analysis
Numerical variables are presented as mean ± SD. Two-sided Student’s t-test was used to determine the differences in continuous variables. Discrete and categorical variables are presented as percentages. Survival estimates were performed using Kaplan-Meier analyses with the use of log-rank test. A p-value <0.05 was considered statistically significant. Data were analyzed using IBM SPSS Statistics version 24 (SPSS, Chicago, IL, USA). All data were analyzed retrospectively.
Results
Between January 2010 and January 2017, 29 patients underwent SCS implantation. The average age was 64 years (range, 48-76) and 72% of the patients were male. The minimum follow-up period for all patients was 30 months. Medical and vascular history at baseline for both groups are presented in Table 1.
Medical and Vascular History at Baseline.
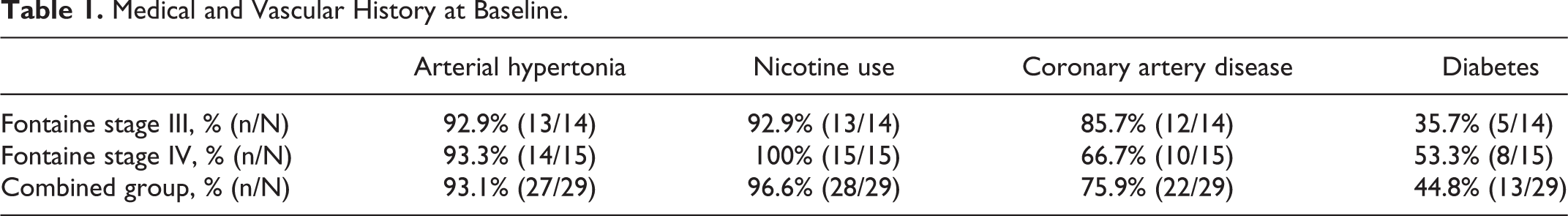
All patients were considered unsuitable for vascular reconstruction. Two patients (7%) had no autologous vein, 22 patients (76%) did not have a suitable artery, and 5 patients (17%) had neither. Angiographic assessment showed that 20 patients (69%) had stenoses in more than 65% of the vessels or occlusion in the upper or lower leg and 26 patients (90%) had a closure of at least one vessel as well as no suitable connection vessel or effluent vessel.
At 3 months follow-up, all patients with Fontaine grade III showed Fontaine grade improvement from stage III to stage II or I. At 1-year, limb salvage was achieved in all but 1 patient (97%; 28/29). 15 patients had limb ulcers (Fontaine stage IV); complete closure occurred in 73% (11/15). No patients died during this period.
During long-term follow-up, a total of 4 patients (14%) were amputated at or above the knee (“major amputation”); all were Fontaine grade IV and diabetic. Amputation occurred at 6, 18, 42, and 48 months and all were performed as the disease progressed. Amputations classified as minor or moderate were not performed. A total of 7 patients died; 2 were previously amputated. Cause of death was cardiac (n = 4) and cancer (n = 3; 2 bronchial and 1 prostate), and time of death ranged between 17 to 68 months after SCS implant. Kaplan-Meier overall and limb survival curves for all patients and according to Fontaine stage III and stage IV are shown in Figure 1.
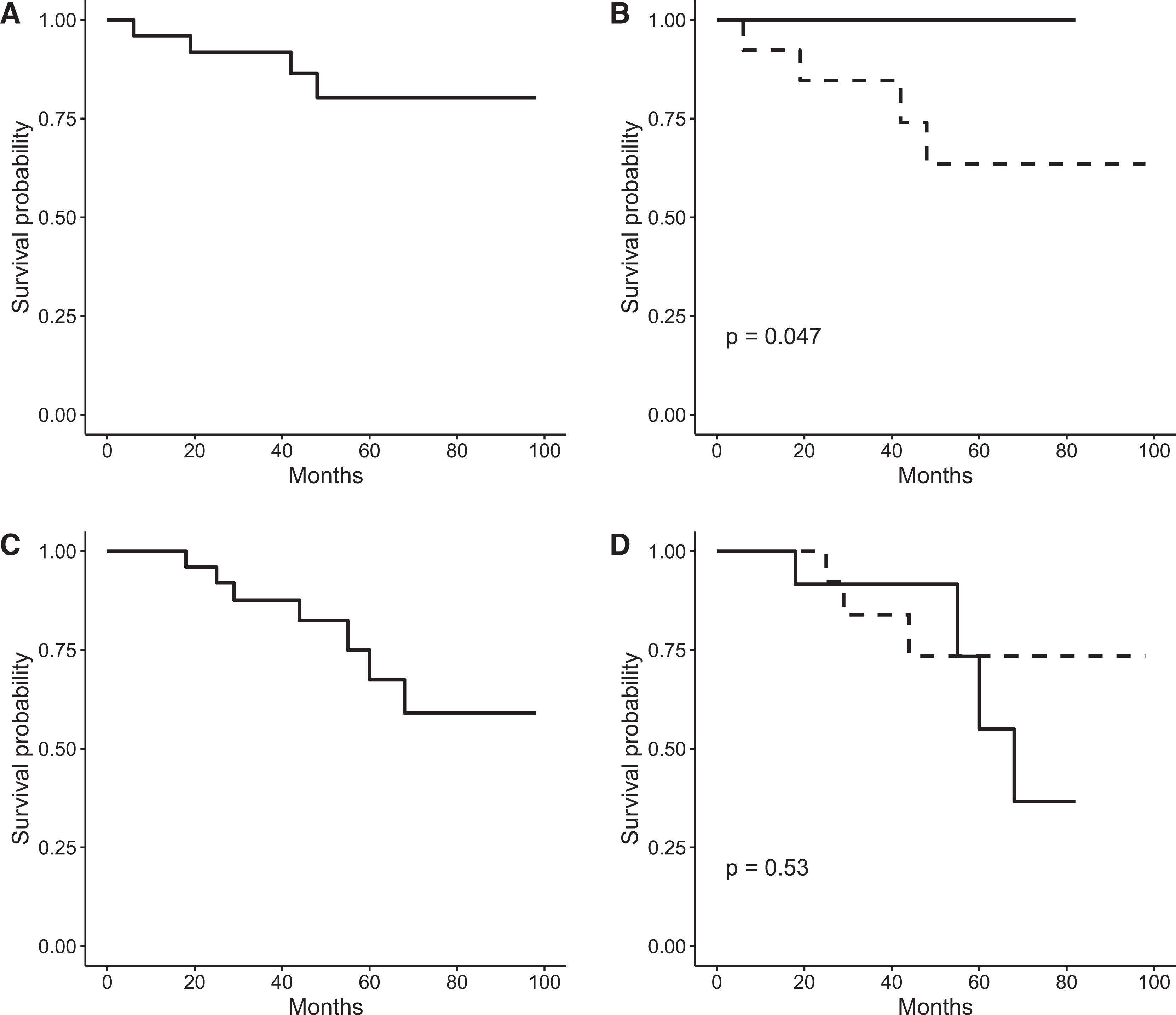
Kaplan-Meier estimator. The visit at which the spinal cord stimulator was implanted was counted as time point 0; individuals were subsequently followed until a major end point (major amputation or death) was reached. Panel A and C show the Kaplan-Meier analysis for overall and limb survival, respectively. Panel B and D show the Kaplan-Meier analysis for overall and limb survival according to Fontaine stage, respectively. Full line represents the outcome for Fontaine stage III patients, while the dotted line represents the outcome for Fontaine stage IV patients.
Patients reported a significant decrease in pain intensity on VAS from 81 mm ± 7 at baseline to 45 mm ± 17 at 3 months, to 23 mm ± 16 at 6 months, and to 16 mm ± 15 at 12 months (p < 0.001 for all time points). A similar progressive improvement was reported for skin temperature in the ischemic area and quality of life. At 1-year follow-up, skin temperature on VAS more than doubled and quality of life score was 66% higher. No significant increase in ABI value with time was observed (Table 2).
Secondary Outcome Measures for All Subjects, Reported as Mean ± SD (n = 29).
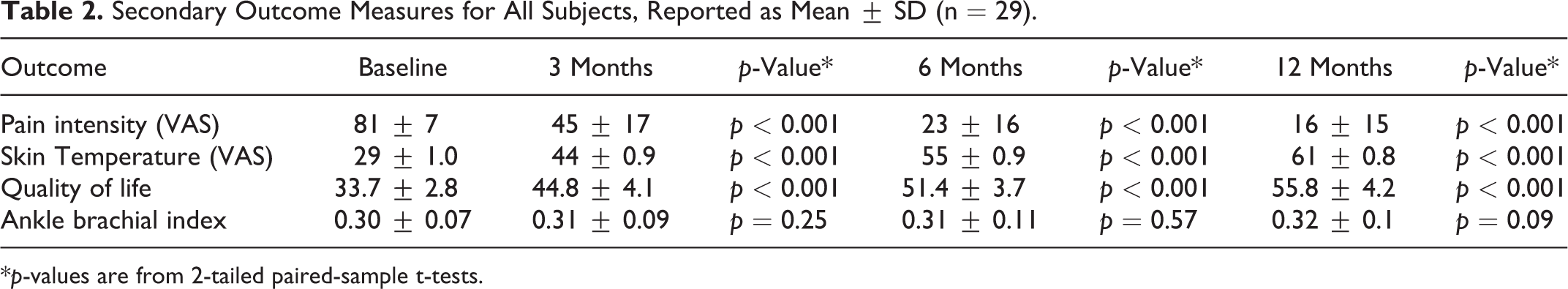
*p-values are from 2-tailed paired-sample t-tests.
A total of 14 patients were diagnosed as Fontaine stage III and 15 patients were diagnosed as Fontaine stage IV. Baseline data was similar between the 2 groups for pain intensity and quality of life but differed significantly for ABI and skin temperature in the ischemic area. At follow-up, patients with Fontaine stage III reported further improved pain scores at 6 and 12 months; only 3 patients reported any pain at 1-year follow-up, and further improved quality of life at all timepoints. The difference in ABI between Fontaine stage III and stage IV remained at follow-up but was no longer present at 6 and 12 months for skin temperature in the ischemic area. Within each Fontaine stage, results were in line with the combined group; significant improvement in pain intensity, quality of life, and skin temperature. Similarly to the combined group, ABI measurement in Fontaine stage III and IV patients did not reveal any differences from baseline; only patients with Fontaine stage III at 12 months after SCS implant showed a significantly improved ABI measurement to 0.4 (p = 0.003), the lower limit for moderate PAD (Table 3).
Secondary Outcome Measures According to Fontaine Stage, Reported as Mean ± SD.
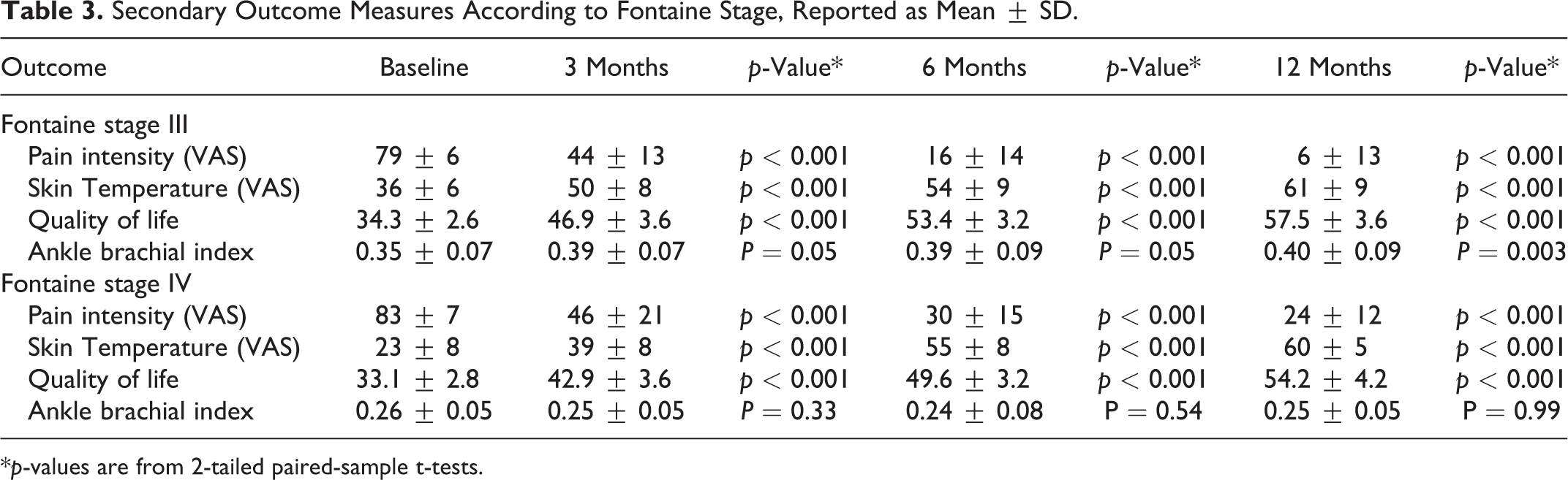
*p-values are from 2-tailed paired-sample t-tests.
A second sub-analysis was performed in diabetic and non-diabetic patients. No differences in outcome were observed between the 2 groups at baseline, but non-diabetic patients had a significantly lower VAS score at 6 and 12 months (VAS of 30 and 23 mm in the diabetic group vs 18 and 9 mm in the non-diabetic group, respectively) and a significantly higher skin temperature in the ischemic area at 6 months (VAS of 50 mm in the diabetic group vs 58 mm in the non-diabetic group). No differences between the 2 groups were observed for quality of life and ABI measurement at follow-up.
There were no infections or other complications related to procedure or device during follow-up. Among all cases, 8 (29%) SCS devices were replaced due to battery life.
Discussion
Patients with CLTI present multiple comorbidities and experience significant pain, impaired wound healing, decreased mobility, and lower quality of life. Revascularization is the first-line therapy; although some patients are poor candidates due to physiologic or anatomic reasons. As a result, alternative noninvasive forms of treatment that can ameliorate the debilitating effects of CLTI, such as SCS, are of interest because medical therapy is considered largely palliative.
Amputation rate at 1-year follow-up in our study was 3% with no mortality. In a larger study of 169 patients with CLTI and uncomplicated ulcers treated with wound care, antibiotics and optimization of medical management, the amputation rate was 23% at 12 months. Among patients with ABI < 0.5, however, the amputation rate increased to 35% at 12 months. 19 This is more in line with the ABI measurements reported in our patient population; only 5 patients had an ABI > 0.4 at 12 months. Non-healing ischemic ulcers, typical of Fontaine stage IV, were completely closed in 73% of patients at 1-year follow-up. Previous studies of limb loss in patients with limb ulcers have defined diabetes mellitus and renal insufficiency as risk factors associates with amputation. 3,4,20 We did not identify these risk factors, but this may be related to an inadequate patient number, although SCS might be less effective in diabetic subjects, most notably in terms of reduction in pain intensity. SCS therapy did not affect ABI measurement. This is not unexpected because patients treated with SCS had crural vessels that were unsuitable for reconstruction. We did not perform any micro-circulatory assessments in this study and no significant macro-circulatory effects were observed. A recent report did show significant improvement in ABI in SCS-treated CLTI patients, although the proportion of patients with Fontaine stage III was higher and normalized measurements 1-year after SCS were not obtained. 21
Most patients with Fontaine stage III reported no pain 1 year after implant and their quality of life was considerably higher than in Fontaine stage IV patients. These outcomes suggest that treatment of CLTI with SCS is advantageous earlier rather than later in the stage III to IV disease progression, although both groups showed progressive and meaningful improvements up to 1-year follow-up. These results are in line with contemporary literature reports that demonstrate the efficacy of SCS in CLTI patients. 16,17,21 -25 The beneficial effects of spinal cord stimulation in patients with CLTI were also an increase in skin temperature in the ischemic area, consistent with results obtained by other groups. 22,26 Finally, the aim of SCS in PAD patients is not only to achieve effective analgesia, but also to promote the trophic-functional recovery of the body segment affected by an advanced ischemic process. In our study, we found that all Fontaine stage III SCS implanted patients showed stage improvements after 3-months follow-up.
The natural history of the disease and the possibility of placebo effect must be considered when interpreting the response to a new form of treatment. It is well known that intermittent claudication tends to improve with time, but this usually occurs over a period of months rather than days and tends to occur in the early stages of the illness. 27,28 The normal clinical course of patients with severe persistent rest pain is to remain stable or deteriorate. How SCS is able to improve clinical outcomes in vascular disease is not yet fully understood and is thought to be complex and multifaceted. Recent research suggests that SCS induces vasodilation in the peripheral microcirculatory system by a combination of antidromic activation of sensory fibers, inhibition of autonomically mediated vasoconstriction, and activation of vasoactive substances. 26,29
The estimated overall complication rate of SCS is 17% and includes hardware failure (lead migration, generator failure) and infection at the epidural lead site or generator site. 15 Ubbink and Vermeulen reported that the risk of infection of the lead or generator pocket is less than 3%. 15 We did not observe complications related to the SCS procedure or device. Infections at the SCS electrode implant site or of the subcutaneous wall pocket are perhaps the most relevant side-effects, usually requiring explant of the SCS device. Infections seem to be more frequent when SCS device implantation is performed in 2 separate sessions (i.e., during a trial followed by the permanent IPG implant procedure), especially in PAD patients which may have compromised immune systems.
Limitations
Interpretation of the outcomes of this study is limited because the study design was not controlled, and data were not collected prospectively. Our results are derived from a real-world patient population of truly refractory patients and add to the growing body of evidence that is broadly supportive of treating selected CLTI patients with SCS therapy. However, further randomized controlled studies are required to confirm these findings, as well as to establish the additional benefits of TcPO2 measurements. These measurements were not performed in our study as they are time-consuming and technically challenging to implement in everyday practice. The outcome may also be influenced by cardiac and pulmonary function, skin thickness, edema, and obesity. Finally, other important outcomes in CLTI patients not collected in the study, including claudication distance and medication intake, could provide a broader view of the effectiveness of this therapy.
Conclusions
SCS is safe and could be considered a valid alternative for patients affected by PAD with refractory ischemic pain and tissue loss when all other forms of treatment have failed. The selection of the right candidate for SCS among patients with PAD is important to have clear criteria for patient selection. 26 All our patients had severe symptoms and had either failed to respond to conventional surgical and medical treatment or were unsuitable for such treatment. This method improves limb survival in patients with critical limb ischemia, gives significant pain control and improves patient’s quality of life. Patients with CLTI show a positive clinical dynamics 1 year after SCS. This is reflected in the improvement of the clinical stages of lower limb from III to II or I according to the Fontaine classification.
However, SCS will have no influence upon the underlying pathology and cannot be expected to reverse gangrene but it improves wound healing, most likely due to the increase of micro-perfusion. The therapy may be more beneficial in patients classified as Fontaine stage III. The present results indicate that SCS is a promising method for the treatment of CLTI, a difficult patient group when first-line treatments are not possible or feasible, and suggest the utility of evaluating the long-term effects of SCS as well as limb salvage in randomized controlled studies.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
