Abstract
The growth hormone secretagogue receptor 1a (GHSR), also called the ghrelin receptor, is a G protein-coupled receptor known to play an important metabolic role in the regulation of various physiological processes, including energy expenditure, growth hormone secretion, and cell proliferation. This receptor has been implicated in numerous health issues including obesity, gastrointestinal disorders, type II diabetes, and regulation of body weight in patients with Prader-Willi syndrome, and there has been growing interest in studying its mechanism of behavior to unlock further applications of GHSR-targeted therapeutics. In addition, the GHSR is expressed in various types of cancer including prostate, breast, and testicular cancers, while aberrant expression has been reported in cardiac disease. Targeted molecular imaging of the GHSR could provide insights into its role in biological processes related to these disease states. Over the past decade, imaging probes targeting this receptor have been discovered for the imaging modalities PET, SPECT, and optical imaging. High-affinity analogues of ghrelin, the endogenous ligand for the GHSR, as well as small molecule inhibitors have been developed and evaluated both in vitro and in pre-clinical models. This review provides a comprehensive overview of the molecular imaging agents targeting the GHSR reported to the end of 2019.
Keywords
Background
The GHSR and Its Endogenous Ligands
The growth hormone secretagogue receptor (GHSR) is a member of the G protein-coupled receptor (GPCR) superfamily and was first identified in 1996 by Howard and co-workers. 1 It is also commonly referred to as the ghrelin receptor and exists in 2 known isoforms, GHSR-1a and GHSR-1b. The GHSR-1a is made up of 366 amino acids and is the only active form of the receptor. Isoform 1b is 289 amino acids in length with 100% sequence identity to that of the GHSR-1a up to Leu265. The GHSR-1a, hereafter referred to as GHSR, was first discovered in the pituitary and hypothalamus as a receptor that bound growth hormone secretagogues (GHS), a class of synthetic therapeutics developed to stimulate endogenous growth hormone secretion. Many GHS molecules were developed prior to the discovery and identification of the GHSR or its endogenous ligands; some of the many examples include GHRP-6, GHRP-2, hexarelin, MK 0677, G-7039, and ipamorelin. 2 -8 Since that time, the GHSR has been found to be expressed in tissues outside the CNS, including the pancreas, thyroid gland, spleen, adrenal gland, gastrointestinal tract, and cardiovascular system. 9,10 Intracellular signaling of this receptor is mediated by the endogenous ligand for the GHSR, ghrelin.
Kojima and coworkers first identified ghrelin in 1999 as the endogenous ligand for the GHSR (EC50 = 2.5 nM). 11 Later, it was determined that the gene product is preproghrelin, which translates to a 117 amino acid polypeptide that is post-translationally cleaved after a 23 amino acid signal peptide to give the 94 amino acid peptide, proghrelin (Figure 1). 12 Proghrelin may then be processed by prohormone convertase (PC) 1/3 to yield a 28 amino acid ghrelin peptide, and then acylated by ghrelin O-acyl transferase (GOAT) to produce bioactive ghrelin. 13 -15 GOAT is a membrane-bound enzyme known to specifically attach an n-octanoyl fatty acid side chain off the serine residue in position 3 of ghrelin. 13,14 This unique acylation is essential in order for ghrelin to bind and activate the GHSR, whereas the non-acylated form of ghrelin, desacyl ghrelin (DAG), has no affinity for the receptor (IC50 > 10,000 nM). 16 Interestingly, DAG is the predominate form of ghrelin found in circulation, but its particular function in relation to the receptor is still not fully understood. However, DAG does participate in physiological pathways independent of the GHSR including binding to the corticotropin releasing factor receptor type 2a (CRF2a), exerting beneficial actions on vascular function, and cardiac protection. 17 -19 Additionally, a cyclic analogue of DAG, known as Livoletide, is currently in phase III clinical trials for the treatment of Prader-Willi syndrome. 20 The GHSR-1b isoform does not bind ghrelin or growth hormone secretagogues and, therefore, does not share the same signaling profile as the GHSR-1a. 21 The biological function of the GHSR-1b is still not well understood, though it has been shown to modulate the relatively high constitutive activity of the GHSR-1a through proposed hetero-dimerization of the 2 isoforms on the endoplasmic reticulum. 22 -24 Therefore, the majority of the research on ligand development has been devoted exclusively to targeting the GHSR isoform 1a.

Biosynthetic processing from preproghrelin to ghrelin.
Recently in 2018, Ge et al. reported their discovery of liver-expressed antimicrobial peptide 2 (LEAP2) as a second endogenous ligand for the GHSR. 25 Mature LEAP2 is a 40 amino acid bicyclic peptide with 2 disulfide bridges, originally isolated in 2003 from human blood ultrafiltrate (Figure 2). 26 It was found to function as an antagonist for the receptor, causing inhibition of ghrelin-induced GHSR activation. 25,27 Later reports demonstrated that LEAP2 also behaved as an inverse agonist toward GHSR constitutive activity. M’Kadmi et al. found that LEAP2 displaces ghrelin from the orthosteric binding site of the GHSR (Ki = 1.26 nM) and substantially decreased ghrelin-independent receptor signaling (EC50 = 22.8 nM) in an inositol phosphate (IP1) production assay. 28 Similar to ghrelin, the full peptide sequence of LEAP2 is not necessary for the ligand to affect the receptor. Rather, the N-terminal region, LEAP2(1-14), which contains no disulfide bridges, is sufficient to maintain strong receptor binding (Ki = 3.66 nM) and activity as an inverse agonist (EC50 = 76.4 nM). 28 Furthermore, blocking endogenous LEAP2 was found to enhance ghrelin-induced GHSR activation in vivo. 25 The ability of LEAP2 to tune the ghrelin-GHSR system makes it an interesting therapeutic target for the treatment of metabolic diseases, such as obesity.

Amino acid sequence of mature LEAP2.
Clinical Relevance of the GHSR Expression and Ghrelin Secretion
Activation of the GHSR by ghrelin results in a variety of physiological functions including regulation of appetite, energy homeostasis, growth hormone secretion, cell proliferation and survival, glucose and lipid metabolism, blood pressure regulation, and the protection of cells in the nervous and cardiovascular systems. 29 -33 Research into perturbing the expression of ghrelin for the treatment of metabolic disorders, including anorexia, cachexia, obesity, and diabetes has resulted in the rapid expansion of reports of ghrelin receptor agonists, antagonists, and inverse agonists. 34 -37 Several review articles exist summarize many of these ligands and their journey toward the clinic. 8,38 -40 Additionally, altered GHSR and ghrelin expression has been observed in many cancers including pancreatic cancer, breast cancer, prostate cancer, ovarian cancer, gastric cancer, colon cancer, thyroid cancer, pituitary adenoma, and lung cancer. 33,41 -46 Particularly, differential expression of the GHSR in prostate cancer tissues compared to benign hyperplasia was demonstrated by Lu et al, indicating a potential for use of the GHSR as a diagnostic biomarker for such cancers. 47 Therefore, targeted imaging of this receptor could provide a minimally invasive method for monitoring cancer treatment and progression. Moreover, the GHSR is abnormally expressed in cardiac pathology; increased expression has been demonstrated in the myocardium of patients with chronic heart failure and decreased expression in patients with end-stage cardiomyopathy. 41,48 Related to these findings, ghrelin administration has been shown to have several therapeutic effects in cardiac disease including improving cardiac function in models of chronic heart failure. 33 As such, there is growing interest in developing ghrelin analogues for the purpose of imaging the biological processes related to cardiac GHSR. Current research to image the GHSR is underway through the development of probes derived from ghrelin, LEAP2, growth hormone secretagogues, and small molecules. While other reviews have provided overviews of ligands targeting a variety of GPCRs including the GHSR, this review seeks to provide a focused, stand-alone synopsis of molecular imaging agents targeting the GHSR reported to the end of 2019. 49,50
Review
Molecular Imaging Agents Based on Ghrelin
Soon after the discovery of ghrelin, researchers were interested in elucidating the structure activity relationship (SAR) between this peptide hormone and its receptor. Early work focused on investigating the role of the aliphatic side-chain of ghrelin, the biological importance of the ester linkage to this side-chain, and the minimum sequence length required to maintain activation of the receptor. 16,51 These studies, reported in the early 2000s, determined that the octanoyl side-chain, while crucial for binding, could tolerate various structural modifications provided that sufficient hydrophobicity is retained. The ester group linking the aliphatic side-chain to the ghrelin peptide can be replaced by substituting serine-3 with diaminopropionic acid (Dpr), which affords a more chemically robust amide linkage with no detriment to the binding affinity. Furthermore, it was found that the N-terminal tetrapeptide, Gly-Ser-Ser(n-octanoyl)-Phe-NH2, was the smallest truncated analogue of ghrelin that could still activate the GHSR, albeit with low potency. Binding affinity and activation was stronger with longer analogues, such as ghrelin(1-10) and ghrelin(1-14). An alanine scan of ghrelin(1-14) systematically confirmed the importance of a positive charge at the N-terminus, identified Phe 4 as a critical residue for binding, and suggested that most other amino acids in the sequence could be replaced to further optimize affinity and potency. 52
The last decade has seen a substantial increase in the number of imaging probes targeting the GHSR with 2009 marking the first publication investigating the potential to image this receptor as a cancer target. In this preliminary investigation, Rosita et al. reported on the development of ghrelin analogues bearing PET and SPECT imaging moieties.
53
The authors utilized structure-activity knowledge reported in the early years of ghrelin research to investigate 2 classes of ghrelin analogues. First, peptides containing fluorine within the aliphatic side chain of ghrelin were investigated as potential probes for 18F-PET imaging. Peptides were synthesized and their binding affinities toward the GHSR were assessed via an in vitro radioligand-displacement binding assay. The resulting best candidate was a ghrelin(1-14) analogue bearing fluorine at the end of a 12C side chain, ghrelin(1-14)-12C-F

Fluorine and rhenium functionalized ghrelin analogues as potential GHSR-targeting PET and SPECT imaging probes.
Later in 2015, Koźmiński and Gniazdowska continued work on developing SPECT imaging probes for the GHSR labeled with technetium-99 m (Figure 4).
59
The authors reported 3 analogues of ghrelin(1-7) conjugated to polydentate 99mTc-complexes via a bifunctional coupling agent (BFCA), isocyanobutyric acid succinimidyl ester, on a lysine residue at the C-terminus of the peptide. All analogues were produced in 85%-95% radiochemical yields and molar activities in the range of 20-25 GBq/µmol. First, a positively charged analogue, [Lys
7
(99mTc(CO)3LN, OCN-BFCA)+]ghrelin(1-7)
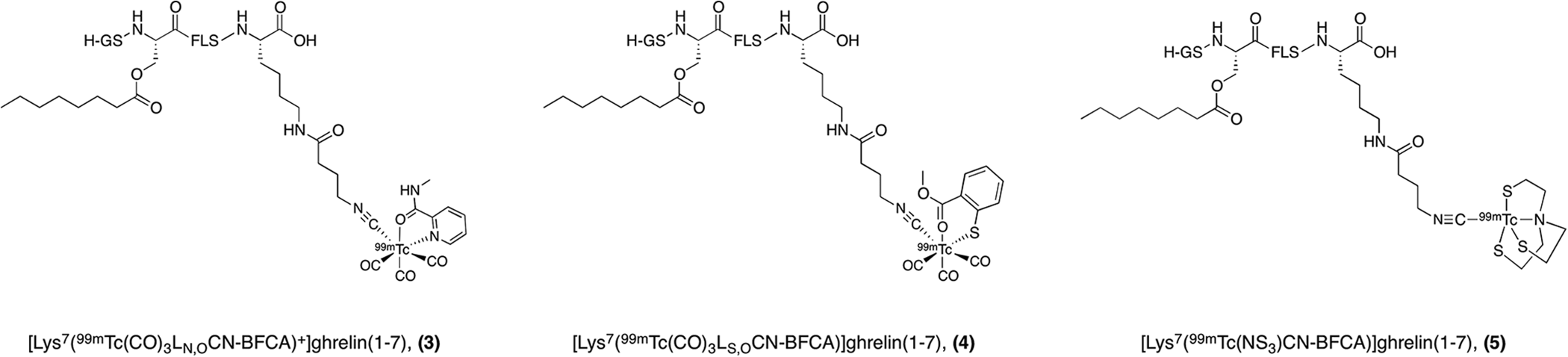
99mTc-SPECT imaging probes for the GHSR.
During the early 2000s, the development of GHSR targeted probes was also underway outside the realm of nuclear imaging modalities. The first fluorescently labeled ghrelin analogue was reported by Enderle et al. with the goal of developing a non-radioactively labeled compound that could identify the GHSR and measure the binding affinity of other test compounds in cell-based assays. 60 The resulting analogues featured ghrelin truncated to the first 18 amino acids in the peptide sequence. A variety of maleimide-conjugated fluorophores, including Texas Red, tetramethyl rhodamine, BODIPY FL, and MR121, were conjugated to an additional cysteine residue on the C-terminus of the peptide to give analogues of the form: [Dpr(octanoyl) 3 , Cys(dye) 19 ]ghrelin(1-19) amide. 60 In 2011, Leyris et al. developed a high-affinity red fluorescent ghrelin analogue, known as red-ghrelin, for use in a homogenous time-resolved fluorescence (HTRF) assay to screen ligands for the GHSR. 61 The biological properties of red-ghrelin were characterized and validated through a competitive radioligand displacement assay and inositol phosphate (IP1) accumulation. While the precise structure of this probe was not disclosed in the article, it was reported to bind to the GHSR with a Ki value of 19 nM and induce IP1 accumulation as efficiently as ghrelin with an EC50 value of 88 nM, approximately half as potent as their evaluation of native ghrelin. In addition to its use in the HTRF assay, red-ghrelin has also been used as a probe to localize GHSR expression and identify GPCR heteromerization in mouse brain. 62,63
Later that year, McGirr et al. reported another fluorescently labeled ghrelin analogue for optical imaging of the GHSR (Figure 5).
64
The imaging label, fluorescein isothiocyanate (FITC), was incorporated onto the peptide via amide conjugation to the C-terminal lysine side chain, giving [Dpr(octanoyl)
3
, Lys(fluorescein)
19
]ghrelin(1–19)

Ghrelin-derived fluorescent probes for optical imaging of the GHSR.

Differential ghrelin probe binding in prostate cancer ex vivo. Ghrelin probe binding was evaluated in benign tissue subdivided into normal and hyperplasia, prostatic intraepithelial neoplasia (PIN), and prostate cancer using the hapten amplification approach. Negative control was processed in the absence of ghrelin probe. Grayscale = ghrelin probe, Blue = DAPI. Scale bar = 10 µm. 47 Reprinted with permission from John Wiley and Sons.
The success of the fluorescein ghrelin probe in GHSR detection prompted its use in other biological studies. Indeed, since its development, this probe has been used on numerous occasions in the identification of ghrelin binding sites in genetically engineered cells and in mouse brain tissue.
65
-68
The brain areas accessible to ghrelin have been elucidated by systematically mapping the distribution of centrally or peripherally administered tracer
However, due to interference by tissue autofluorescence and the dye’s susceptibility to photobleaching, the fluorescein dye was replaced with a more stable, far-red sulfonated cyanine 5 dye (SulfoCy5) producing the ghrelin-based probe, [Dpr(octanoyl)
3
, Lys(SulfoCy5)
19
]ghrelin(1–19)
The increasing promise of fluorescently labeled ghrelin analogues contributed to a desire to develop ghrelin imaging probes with improved metabolic stability. Recently, ghrelin(1-20) was cyclized through a lactam bridge between Glu
12
and Lys
16
for improved stability of secondary structure, and labeled with the SulfoCy5 dye off the C-terminal lysine residue (Figure 5).
80
This stapled peptide, cyclo-12, 16[Dpr(octanoyl)
3
, Glu
12
, Lys(SulfoCy5)
20
]ghrelin(1–20)
The ability to incorporate a bulky dye off the C-terminus of truncated ghrelin opened up the possibility of implementing other imaging moieties such as radionuclide-containing prosthetic groups or radiometal chelators in that position, while maintaining desirable binding affinities in the low nanomolar range. Chollet et al. developed full length ghrelin(1-28) and truncated ghrelin(1-16) agonists labeled with gallium-68 via a 7-triazacyclononane,1-glutaric acid-4,7-acetic acid (NODAGA) chelator for PET imaging (Figure 8).
57
Highly potent [Ga]-NODAGA peptide conjugates were synthesized and their ligand-receptor interactions were characterized through an inositol triphosphate (IP3) turnover assay resulting in EC50 values in the 0.7-2.0 nM range. Initially, the chelator was conjugated to the peptide at the N-terminus, and, unsurprisingly, the modification was not well tolerated and N
α-NODAGA(Ga)-ghrelin(1−28) lost all activity toward the receptor. It is well established in the literature that an available N-terminus is necessary for ligand-receptor binding between ghrelin and the GHSR.
16,51
Conversely, by placing the chelator on the Lys
16
residue, away from the N-terminal region of the peptide

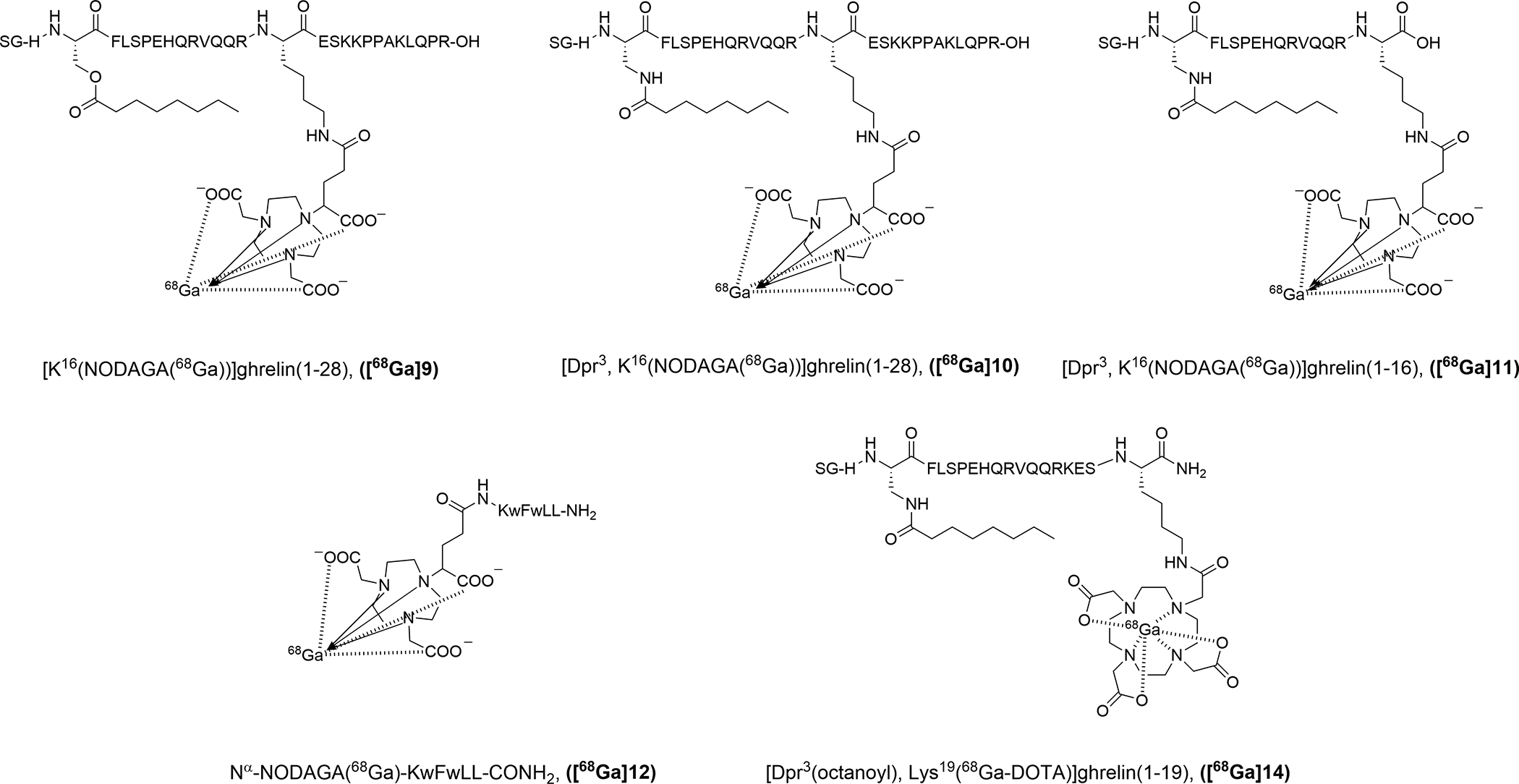
Gallium-68 labeled imaging probes for PET imaging of the GHSR.
Another example of incorporating imaging moieties off the C-terminus of ghrelin was reported by Murrell et al.
82
The authors developed a novel 18F-prosthetic group based on an azadibenzocyclooctyne (ADIBO) scaffold and demonstrated its ability to be incorporated into biomolecules through successful conjugation to ghrelin(1-19) with minimal effects on the binding affinity (Figure 9). The resulting peptide analogue, [Dpr
3
(octanoyl), Lys
19
(triazole-ADIBO-F)]ghrelin(1–19) (
Charron et al. continued with this theme and reported a probe containing a 1,4,7,10-tetraazacyclododecane-1,4,7,10-tetraacetic acid (DOTA) chelator off the C-terminal lysine residue (Figure 8), [Dpr
3
(octanoyl), Lys
19
(Ga-DOTA)]ghrelin(1-19) (
Indeed, the same group conducted an extensive structure-activity relationship campaign to develop a truncated ghrelin(1-8) analogue that maintained high affinity for the GHSR.
83
This SAR investigation on the N-terminal ghrelin(1-8) sequence identified favorable modifications to residues 1, 3, 4, and 8 resulting in the peptide analogue, [Inp
1
, Dpr
3
(6-fluoro-2-naphthoate), 1-Nal
4
, Thr
8
]ghrelin(1−8) amide

Peptidic and peptidomimetic probes for fluorine-18 PET imaging of the GHSR.
Molecular Imaging Agents Based on Other Peptides and Peptidomimetics
Until 2018, most reported peptidic GHSR targeted imaging probes were derived from endogenous ghrelin (Table 1). Fowkes et al. sought to develop a growth hormone secretagogue-based probe for PET imaging of the GHSR.
86
The authors studied an extensive number of growth hormone secretagogues including GHRP-1, GHRP-2, GHRP-6, ipamorelin, and G-7039 as potential 18F-PET imaging agents. The fluorine-modified secretagogue derivative with the strongest binding affinity for the GHSR was [1-Nal
4
, Lys
5
(4-fluorobenzoate)]G-7039
In vitro Properties and Radiosynthetic Results of Peptidic GHSR Imaging Probes.
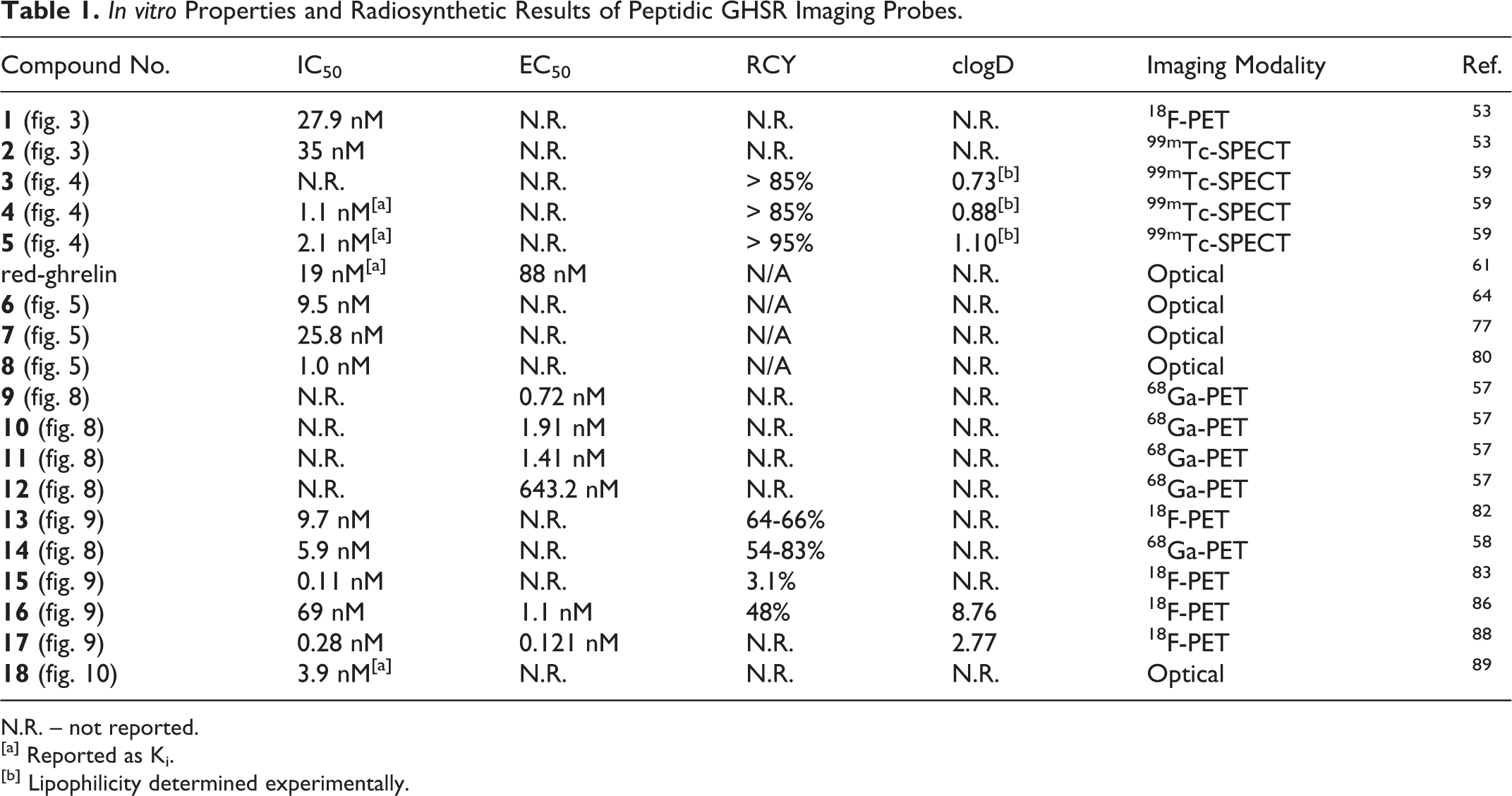
N.R. – not reported.
[a] Reported as Ki.
[b] Lipophilicity determined experimentally.
The recent identification of LEAP2 as a second endogenous ligand for the GHSR prompted a desire to develop a fluorescent analogue of LEAP2 suitable for studying the pharmacological behavior of GHSR: LEAP2 complexes compared to those of GHSR: ghrelin.
25,27,28
Barrile et al. recently reported the resulting fluorescent ligand, LEAP2(1-17)-DY-647P1 (

Fluorescently labeled LEAP-2 derivative for optical imaging of the GHSR.
Molecular Imaging Agents Based on Small Molecules
In 2007, Bayer pharmaceuticals reported a number of quinazolinone derivatives as a new class of small molecule GHSR antagonists for the treatment of obesity and diabetes. 90 The piperidine-substituted quinazolinone scaffold (Figure 11) showed inherently nanomolar affinity for the receptor, while alkylating the piperidine ring nitrogen (R1) gave rise to the functionally antagonistic activity.

General scaffold for quinazolinone-derived small molecule GHSR imaging probes.
In 2011, Potter et al. sought to utilize this class of high-affinity small molecules to develop GHSR targeting PET radioligands.
91
One of the molecules reported by Bayer, racemic 6-(4-fluorophenoxy)-3-(piperidin-3-yl)methyl)-2-o-tolylquniazolin-4(3 H)-one (

Quinazolinone derivative reported by Bayer pharmaceuticals (
Later in 2017, Kawamura et al. continued the endeavor of labeling small molecule GHSR antagonists with PET radionuclides and reported 3 new radioligands:

Structures of small molecule GHSR antagonists for 18F-PET and 11C-PET imaging reported by Kawamura et al. 92
In vitro Properties and Radiosynthetic Results of Small Molecule GHSR Imaging Probes.

N.R. – not reported.
[a] Reported as Ki.

Representative PET images of mice using
Another group seeking to generate compounds suitable for GHSR imaging in the brain reported a SAR study on fluorinated quinazolinone derivatives with low nanomolar affinities.
94
Substitution at the piperidine nitrogen is known to have an effect on binding to the receptor; therefore, the authors chose a known, high affinity, quinazolinone derivative as a starting point for SAR studies, compound
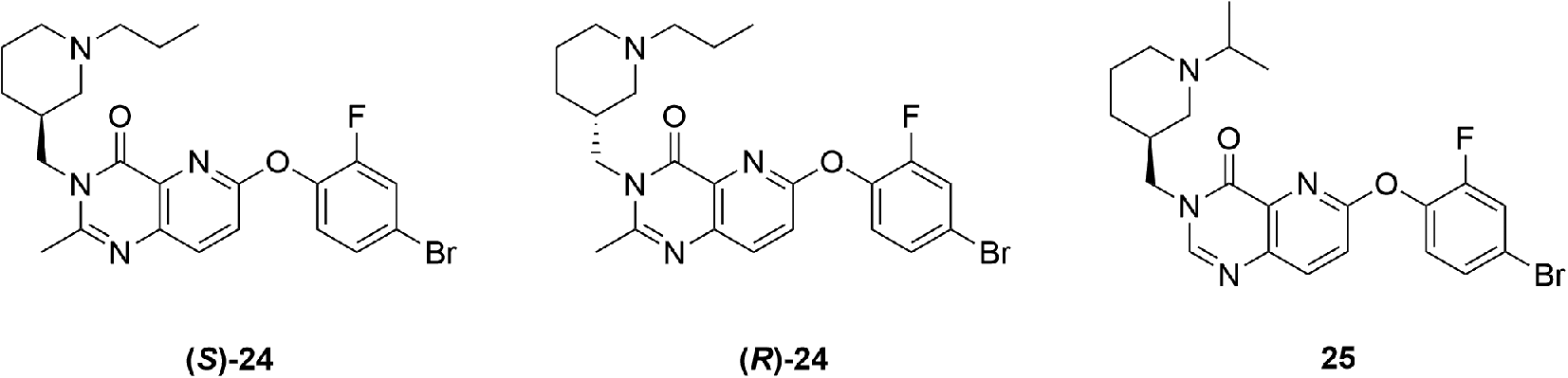
High affinity quinazolinone-derived partial agonists for the GHSR reported by Moldovan et al. 94
The most recent attempt to develop quinazolinone derivatives for 18F-fluorine PET imaging of the GHSR was reported in 2018 by Hou et al.
96
The authors also used the compound initially reported by Bayer (

Small molecule quinazolinone analogues reported by Hou et al for 18F-PET imaging of the GHSR. 96
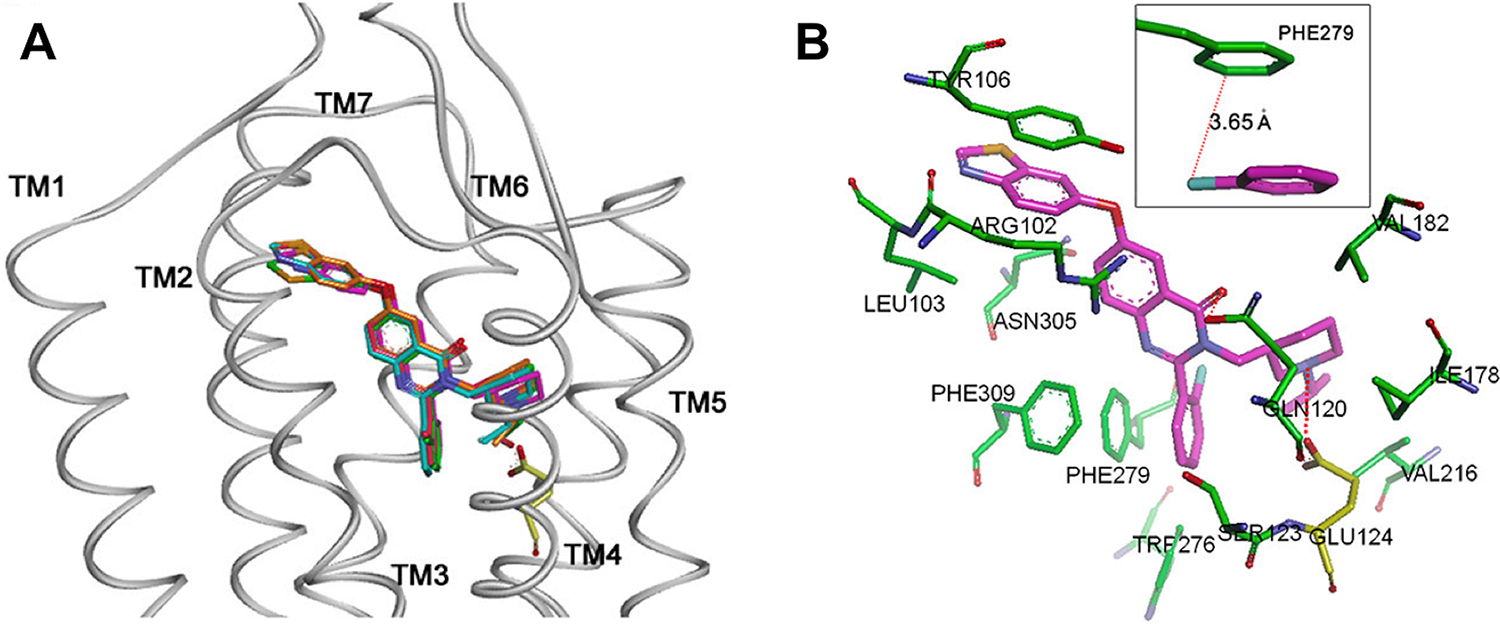
The complex structures from molecular docking studies using a homology model of GHS-R1a. (A) Side view of quinazolinone derivatives superimposed in the GHS-R1a; (B) A view from extracellular side; 3D depiction of the binding mode and surrounding residues for compound
Conclusions
Since the discovery of the GHSR, there has been a growing interest in advancing our understanding of this receptor’s dynamic processes and exploring the therapeutic potential of pharmaceuticals for this target. Molecular imaging provides a minimally invasive window into investigating GHSR expression in normal and disease states. In the last decade, the number of molecular imaging agents targeting this receptor has steadily grown resulting in probes for nuclear imaging modalities (SPECT/PET) using 11C, 18F, 68Ga and 99mTc radioisotopes, and optical imaging through fluorescent dyes. This development has resulted in novel ligands with high affinity for the GHSR based on ghrelin, growth hormone secretagogues, and small molecules previously discovered for therapeutic applications. These probes continue to evolve as researchers seek to optimize their physical and biological properties and proceed to in vivo evaluation. The optical imaging probes have been used for cellular and tissue imaging to investigate the GHSR as a potential biomarker for cardiac disease, and PET imaging probes are being developed for potential cancer and brain imaging. In addition to the incorporation of imaging moieties onto known GHSR compounds, novel ligands with imaging in mind have resulted from structure-activity relationship studies. The acquisition of knowledge regarding the dynamic function of this receptor and ghrelin is continuing to unfold, revealing numerous therapeutic possibilities. The integration of GHSR targeted imaging probes with ligands currently under investigation as potential therapeutics could be a step toward a more harmonized approach in drug research providing valuable information on biodistribution, kinetics, and metabolic behavior of such drug molecules and the treatment monitoring of receptor-influenced disorders.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: We gratefully acknowledge the support of the Natural Sciences and Engineering Research Council of Canada (NSERC) and the Molecular Imaging Collaborative Program at the University of Western Ontario.
