Abstract
Foster E, Conquest A, Ewart C, Nicolo JP, Rayner G, Winton-Brown TT, O'Brien TJ, Kwan P, Malpas C, French J. Neurology. 2025;105(3):e213900. doi: 10.1212/WNL.0000000000213900. Background and Objectives: There is a clear need in epilepsy clinical trials and practice for a measure that captures the tradeoff between seizure and treatment-related adverse effects, which is reliable over time and across different treatment regimens. We aimed to create and validate the Seizure-Related Impact Assessment Scale (SERIAS) to fill this need. Methods: This was a prospective longitudinal study of adults with epilepsy recruited from an Australian comprehensive epilepsy center. Participants completed SERIAS at baseline and 3 and 6 months later. SERIAS has 6 self-report items. Five items record the number of days per month that seizures or treatment-related adverse effects partially or fully affect work/home/school and family/social/nonwork activities. The final item is an epilepsy disability visual analog scale. SERIAS is scored by adding the days per month of disability, with scores ranging from 0 to 150 (higher scores indicate more disability). SERIAS was completed alongside 7 validated instruments measuring seizure-related and treatment-related adverse effects (Work and Social Adjustment Scale [WSAS], Liverpool Adverse Events Profile [LAEP]), mood disorders (Neurological Disorders Depression Inventory for Epilepsy [NDDI-E], Generalized Anxiety Disorder [GAD-7]), somatic symptoms (Somatic Symptom Scale [SSS-8]), and quality of life (Quality of Life in Epilepsy Inventory [QOLIE]-31, EuroQol 5 Dimensions [EQ-5D]). General linear mixed models were used to investigate the relationship between the SERIAS and other relevant clinical and psychometric data. Standardized model coefficients β are presented with 95% confidence intervals. Results: A total of 90 patients (64.4% female, mean age 43.1 years) completed baseline SERIAS. Most patients reported at least 1 day of disability (62%, median SERIAS score = 3, interquartile range = 18.3). Greater disability was negatively correlated with QOLIE-31 total score (β = −0.17, 95% CI −0.27 to −0.07) and positively correlated with scores on 5-level EQ-5D (β = 0.15, 95% CI 0.04-0.25), NDDI-E (β = 0.22, 95% CI 0.13-0.31), GAD-7 (β = 0.21, 95% CI 0.09-0.32), SSS8 (β = 0.29, 95% CI 0.17-0.41), LAEP (β = 0.29, 95% CI 0.20-0.39), WSAS seizure-related adverse events (β = 0.23, 95% CI 0.14-0.33), and WSAS treatment-related adverse events (β = 0.36, 95% CI 0.26-0.46). Higher seizure frequency was associated with higher SERIAS score (β = 0.07, 95% CI 0.03-0.11). Psychometric reliability for the SERIAS was acceptable (all coefficients >0.70) as was test-retest reliability (n = 35 patients, intraclass correlation coefficient = 0.72, 95% CI 0.51-0.85).
Kheder A. Epilepsia. 2025. doi: 10.1111/epi.18471. Epub ahead of print. PMID: 40402032. The Multidimensional Outcome Reporting in Epilepsy (MORE) framework addresses critical limitations in current epilepsy outcome assessment by combining seizure control, quality of life, and patients’ subjective experience. Although seizure frequency is the main emphasis of traditional classifications, they overlook important aspects of patients’ experience such as treatment burden, psychosocial well-being, and functional outcomes. This framework uses a 40-40-20 weighting model in three different domains and includes proven quality of life measures. Implementation issues include consideration of the subjective aspects of assessment, standardizing across settings, and integrating clinical workflow. Eleven subdomains that cover important facets of postoperative epilepsy care are included in MORE. With simple questions, these subdomains can be evaluated effectively at a standard clinic visit. The nature of the framework permits adaption to both adult and pediatric populations while upholding fundamental concepts. MORE is more in line with the principles of patient-centered care and has the potential to influence future consensus-driven outcome definitions in the field, making it an essential consideration given the advancement of epilepsy care.
Commentary
When defining epilepsy treatment outcomes, seizure frequency is often the first thing we focus on as it drives much of the disease burden. However, when patients have been asked what concerns them most about living with seizures, their top concern was driving then employment then independence; treatment-related adverse effects and seizure unpredictability emerged further down the list. 1 Emphasis on seizure freedom often fails to adequately represent the broader effects that epilepsy and seizure treatment may have on patients’ lives. While seizure frequency has been the primary focus of traditional clinical outcomes assessments, such assessments may overlook other equally important aspects of patient experience such as treatment burden, psychosocial well-being, and functional considerations. In a narrative review, where four electronic databases were queried, identifying 20 articles for review, key issues identified both seizure-specific priorities (such as frequency, duration, severity, warning) but also non-seizure medical priorities (such as cognition, mood, physical side effects), supporting the importance of incorporating individual priorities to promote patient-centered care and improve patient outcomes and overall well-being. 2 Epilepsy outcome measures that capture the full spectrum of disability, attributable to both seizure and treatment-related adverse effects, would be beneficial for both epilepsy research and clinical practice. Ideally these measures would directly assess the “trade-off” between seizure control and reducing treatment adverse effects. Yet currently validated instruments for epilepsy clinical outcomes are not optimized to comprehensively capture the patient's perspective (which may differ from provider) on tradeoffs between seizures and treatment-related adverse effects, incorporating aspects of treatment burden, psychosocial well-being, and functional outcomes to accurately measure outcomes.
The two publications reviewed in this commentary underscore that the change in seizure frequency cannot be the only marker of treatment success. First of all, seizure frequency does not necessarily translate directly to disease severity and impact on quality of life (QoL). While existing established patient-reported outcome measures (PROMs) specifically assess objective features (eg, seizure duration, loss of consciousness), they may not capture the full spectrum of seizure severity or disabling seizure features. Additionally, they do not collectively assess independence, mental health (depression and anxiety), QoL, and cognitive function in one comprehensive measure. The two articles reviewed below both strive to address these gaps in assessment, aiming to more accurately and properly capture the degree of disability experienced by the patient and how seizures, treatment burden, QoL, and the experience of living with epilepsy are applicable across all treatment modalities, medical, surgical, and neuromodulatory.
The first article by Dr Foster et al, aimed to create and validate the Seizure-Related Impact Assessment Scale (SERIAS) to address critical gaps in epilepsy PROMs and to capture burden related to seizure versus treatment adverse effects to provide an overall sense of epilepsy disability. 3 SERIAS was designed to overcome the inadequacies of “seizure counting,” as SERIAS converts seizure frequency and ictal features into a common denominator of “time lost.” Thus seizure-related disability is quantified as the time seizures take away from people's daily activities and explores the degree to which a person is limited in carrying out activities of daily living due to disease-related impairment. Additional goals were to delineate a severe disability that is less frequent from a milder disability that is common, and to determine how much that disability resulted from seizures versus from the treatment of the seizures. This is important because modifications to treatment regimens may improve seizure control but come at the expense of substantial adverse effects; a treatment that reduces seizures by half, but greatly increases sleepiness, may not improve QoL overall.
The SERIAS study was a prospective longitudinal study of adults with epilepsy recruited from an Australian comprehensive epilepsy center. Participants completed SERIAS at baseline and 3 and 6 months later. SERIAS has 6 self-report items (see Figure 1). 3 Five items record the number of days per month that seizures or treatment-related adverse effects partially or fully affect work/home/school and family/social/nonwork activities. The final item is an epilepsy disability visual analog scale. SERIAS scores range from 0 to 150 with higher scores indicating greater disability. The mean seizure score, based on an established seizure frequency scale, was 7, which correlated to a seizure frequency of 1 to 3 seizures per month. Higher SERIAS scores (indicating worse disability) were associated with higher seizure burden and higher medication adverse effects as measured via existing validated instruments.

Text version SERIAS. 3 .
The major limitation of the SERIAS study was a mean score of 2 in the study population (possible range 0-150), which indicated that most participants reported no or little treatment-related or seizure-related disability. One reason proposed by the authors for this finding was that the sample comprised of patients who generally had well-controlled epilepsy. Specifically, 58% of the study sample were seizure-free at the time of enrollment. It will be important to investigate the performance of the SERIAS in patient populations with higher seizure frequency and poorly controlled disease. Another limitation was participant recruitment from a comprehensive epilepsy center which may restrain generalizable to other settings. Additionally, the SERIAS relies on a 30-day recall period which may be affected if people do not recall events accurately. Lastly, a potential issue with the SERIAS is that it relies on patients to be able to distinguish effects from a seizure versus medication related side effects as an etiology for their symptoms. This can be very difficult to differentiate for certain symptoms, such as cognitive dysfunction, fatigue, or depressed mood.
In the second article by Dr Kheder, he proposed the Multidimensional Outcome Reporting in Epilepsy (MORE), a multifaceted framework that combines QoL metrics with seizure outcomes to offer a more comprehensive evaluation of the effectiveness of treatment. 4 The three main domains balanced within the MORE are: Seizure Control Domain (40%): measures seizure-free intervals, rescue therapy uses, and seizure frequency reduction; QoL Domain (40%): Assesses daily functioning, emotional health, cognitive function, and medication burden; and Patient-Reported Experience Domain (20%): Documents social engagement, side effect burden, treatment satisfaction, and overall perceived impact. Eleven subdomains were integrated into the MORE framework (see Figures 2 and 3). 4 This method provides a more patient-centered and clinically meaningful evaluation of outcomes by capturing not only seizure freedom but also functional, cognitive, and psychosocial changes. It documents independence, cognitive function, and QoL and considers functional recovery, ASM burden, and ongoing seizure control. It also considers the use of rescue therapy which enhances QoL and lessens harm associated with seizures. The MORE assessment explores intervention risk–benefit profiles by quantifying potential harms in addition to benefits. It also aims to be a more comprehensive method of assessment across all treatment modalities including neuromodulation.
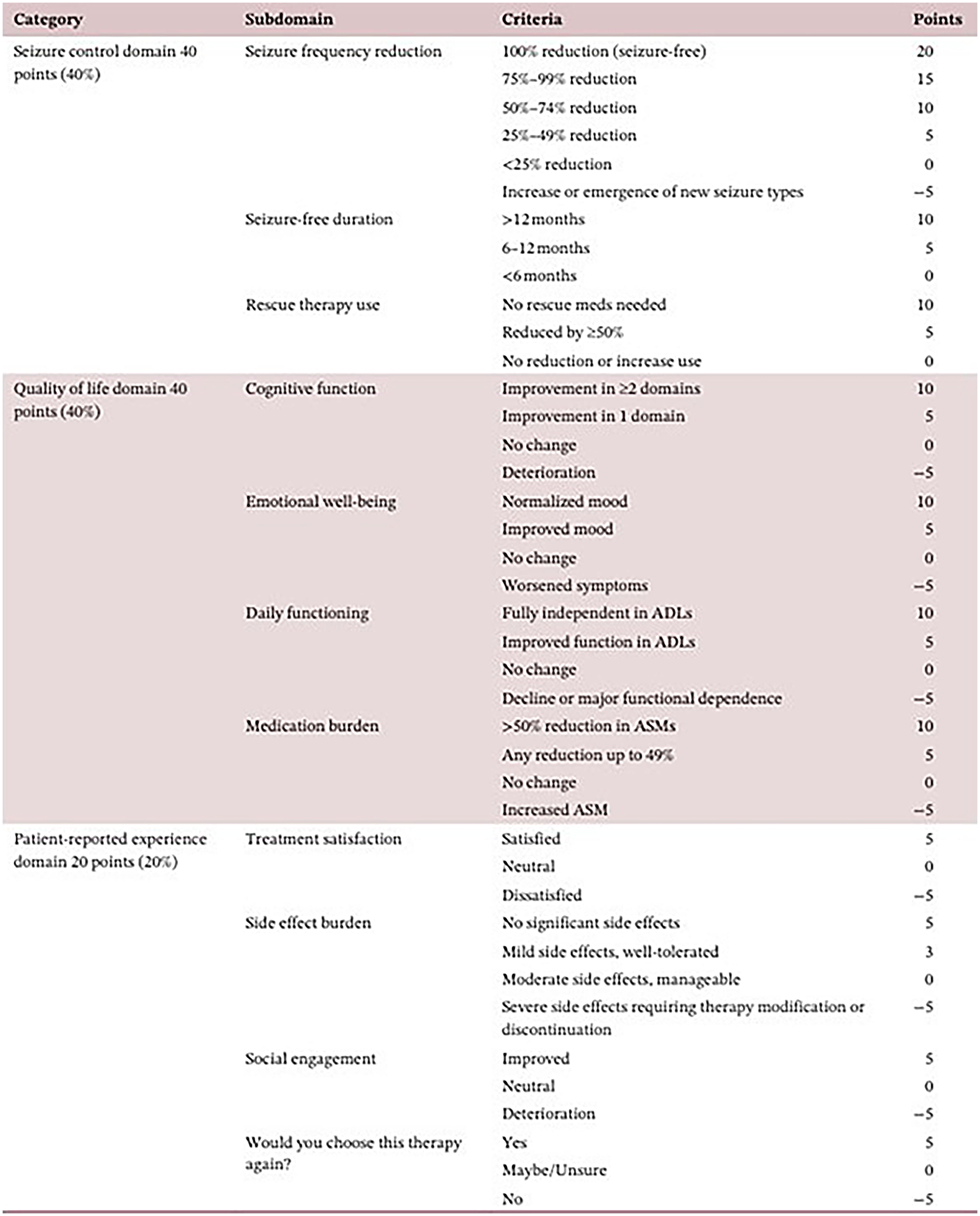
The MORE (Multidimensional Outcome Reporting in Epilepsy) Scale. 4 .
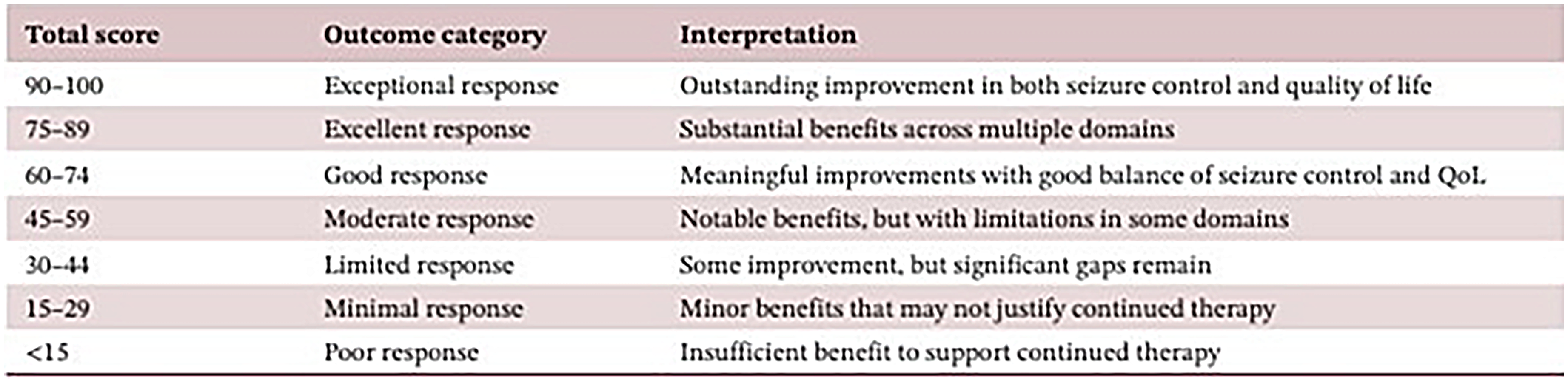
The MORE Final Score Interpretation Guide. 4 .
Unlike the SERIAS article, the MORE assessment is a proposed approach that has not been tested in patients. Proposed limitations of the MORE assessment include the feasibility of integration into current workflow. In time-constrained settings, comprehensive outcome documentation may create additional burden for practitioners. While many providers do acquire this information during their interviews and assessments, standardization across different clinical environments also poses another challenge. Lastly, the weighed model of assessment provides guidance but still contains subjective interpretation which may also be hard to standardize.
These two publications do an excellent job highlighting the current gap in patient-reported outcomes and both support the charge of reevaluating and improving our current epilepsy outcomes assessments. Currently employed measures have limited focus on seizure frequency, lack systematic integration of QoL measures, and fall short in assessments of noncurative surgical procedures and neuromodulation treatments. The SERIAS score captures the “trade-off” between seizure control and treatment-related adverse effects, not readily measurable with our existing instruments and strives to provide an overall sense of epilepsy disability to drive outcome assessments and conversations. The MORE provides a comprehensive measure that focuses on patient-centered and clinically meaningful evaluations, and incorporates functional, cognitive, and psychosocial aspects across care. These two measures appropriately refocus epilepsy care to consider outcomes most important to patients and their families, supporting the growing move to patient-centered care and the need to increase integration of these care principles into clinical practice.
There is an ongoing need for simple, but more comprehensive PROMs in epilepsy clinical trials and practice. The PROMS presented in these two articles spark a crucial conversation about what defines treatment success. These publications are a call to action and encourage providers to evaluate social involvement, medication and rescue therapy changes, cognitive and emotional well-being, functional recovery, and an overall sense of disability while integrating QoL and demonstrating treatment efficacy and patient-related impact across all treatment modalities.
Footnotes
Declaration of Conflicting Interests
The author declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author received no financial support for the research, authorship, and/or publication of this article.
