Abstract
Commentary
Childhood absence epilepsy is a generalized seizure disorder characterized by bilaterally synchronous spike-and-wave discharges (SWD) on EEG and loss of consciousness. These seizures involve widespread neural networks, including the cortex, thalamus, and basal ganglia.
Historically, several theories have competed to explain the mechanisms of absence seizures. 1 Early on a single pacemaker theory was proposed, specifically implicating the midline thalamus, whereby a centralized coordinator drives widespread cortical oscillations. 2 Later studies linked SWDs to thalamocortical mechanisms that produce spindles by demonstrating that spindles can transform to SWDs when cortical excitability is increased by local penicillin application. 3 This concept was subsequently expanded by the finding that an intrathalamic circuit could produce emergent pacemaker activity to drive cortical SWD oscillations. 4 In contrast to pacemaker based theories, the cortical focus theory of rat absence seizures postulates that cortical hyperexcitability, specifically in deep layers, initiates seizures, which then generalize through cortical and thalamic networks.5,6
Recent research has brought into question the core tenets of the cortical focus theory. For example, genetic perturbations specific to either thalamic or cortical regions can cause absence seizures, suggesting cortical hyperexcitability to be only one of several potential mechanisms ([reviewed in 7 ). More strikingly, McAfferty et al (2023), using GAERS rats—a central model for the development of the cortical hyperexcitability hypothesis—found no evidence that seizures begin with increases in cortical firing. 8 Instead, cortical neuron firing decreased during the transition to seizures. This suggests that, although earlier studies in GAERS rats showed higher baseline firing in deep cortical layers than wild-type rats, 6 seizures do not appear to reflect increases of focal firing spreading to other brain regions—rather the opposite seems true.
These findings, derived from multielectrode studies, have limitations. Spike sorting, which is required to putatively identify single neurons, likely overlooks some spikes, especially those occurring during synchronous firing or periods of high activity like bursting. To address this, Khan et al employed juxtacellular recording in C3H/Hej mice, another absence seizure model. 9 This method captures single-neuron activity clearly, avoiding issues with spike sorting, and uses awake, head-fixed mice to eliminate anesthesia-related confounds. This is a commendable and heroic experimental effort. Their findings corroborate recent multielectrode studies: absence seizures are not associated with increases in cortical neuron firing.
If cortical hyperexcitability does not universally underlie absence seizures, what does? The hallmark of absence seizures is the high-amplitude spike-and-wave oscillations in LFP/ECoG signals, used to identify seizures in both patients and animal models. Oscillatory neuronal spiking is consistently observed during these seizures, most notably in animal models, in which high precision single unit recordings can be reliably obtained. The alignment of neuronal spiking with the spike component of the spike-and-wave discharge has to our knowledge never been controversial.
How is this oscillatory spiking measured? A common measure of spiking regularity is to examine interspike intervals and calculate the coefficient of variation (CV-ISI). For typical cortical firing in vivo, this value is close to 1, reflecting a Poisson-like process where intervals are independent. Values >1 indicate irregular, clustered spiking, while values <1 suggest higher regularity, perhaps rhythmic firing. In the extreme of perfect rhythmicity, as with a metronome, the CV-ISI is 0. In McAfferty et al (2023) and Khan et al, the reciprocal is used as a rhythmicity index. However, both the CV-ISI and its reciprocal have caveats; this “rhythmicity index” can be high in the absence of rhythmicity and low even with a strong rhythm. For example, a spike train with slightly noisy intervals near 100 ms has a low CV-ISI and high rhythmicity index as predicted (Fig. 1, orange). Surprisingly, introducing spike bursts with the same rhythm abolishes all rhythmicity by this measure (Fig. 1, red). Importantly, both patterns exhibit similarly high periodicity in their spike time autocorrelations. Additionally, low ISI variability does not itself quantify rhythmicity, as shown by spike trains with a high rhythmicity index (Fig. 1, blue), showing no rhythm in their spike time autocorrelation. We therefore suggest alternative measures for measuring rhythmicity such as autocorrelation, Fourier-based methods, or Poincare plots among others.
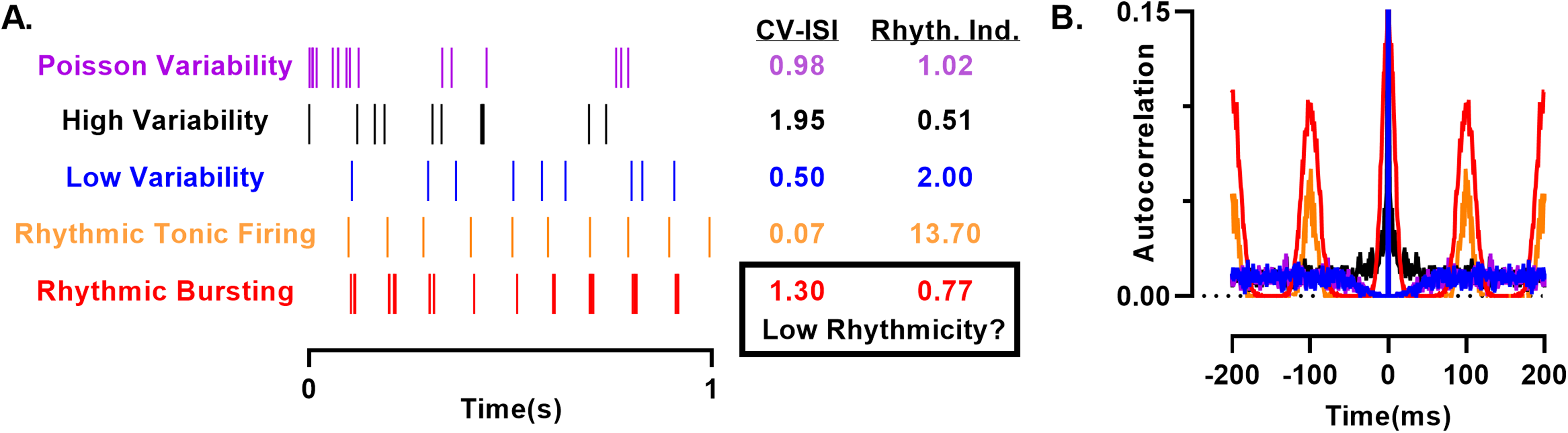
Comparison of spiking patterns and quantification of rhythmicity. A. Various spiking patterns were generated with ISIs sampled from a gamma distribution consistent with a Poisson point process (purple) and either higher ISI variability (black) or lower (blue) than a Poisson process. For comparison, rhythmic spike trains with a 100 ms interval were generated with a jitter sampled from a Gaussian distribution with a standard deviation of 5 ms. Bursting (red) was generated by sampling as above with a 15 ms window around each spike with a mean and standard deviation ISI of 5 ms. B. Autocorrelation computed from the spike trains to the left.
Khan et al found an increased rhythmicity index during absence seizures, although the effect is likely dramatically underestimated due to the presence of multiple clustered spikes per oscillation cycle as evident in their example traces. The lack of increased firing along with high oscillatory spike patterning suggests we should shift our focus to understanding the causes of oscillatory spiking, harkening back to the centrencephalic and thalamic pacemaker theories. The absence of cell-specific labeling or recording depth in McAfferty (2023) and Khan et al, means that at this point we cannot completely rule out a potential role for increased deep-layer cortical firing in causing absence epilepsy, but it appears that the concept of hyperexcitability spreading from a cortical focus relies on an increasingly shaky foundation.
The cortical focus theory is built on the idea of rapidly spreading spike and waves, with extremely fast generalization (<10 ms) between the cortex, thalamus, and various cortical areas. 5 While these delays might be relevant for identifying the initiating neurons in an avalanche of spreading hyperexcitability, they are much less meaningful if the organizing principle is cyclic oscillations, where one cycle's end is another's beginning. More importantly, in coupled oscillator populations, the leading oscillator is often merely the fastest, not one with a special causal significance. 10
Where does this leave the cortical focus theory? If we try to understand the concept of a cortical focus in terms of oscillatory firing, as suggested by Khan et al, we lose the temporal precision (∼10 ms delay) that forms the foundation of the cortical focus theory. Instead, we must account for oscillatory intervals that last for hundreds of ms, which are approximately 10 times longer than the interregional delays proposed to be central to the cortical focus theory. In the context of cycles of rhythmic firing across multiple neuronal populations, the precision of these delays may be below a comfortable threshold for defining the temporal boundaries of the oscillation, leading to cracks in the foundation of the cortical focus theory. If hyperexcitability indeed spreads so rapidly and robustly through widespread networks as the theory seems to require, why are we repeatedly failing in recent studies to observe even moderate firing rate changes at seizure onset? Finally, if cortical hyperexcitability is present in only some models, might finding mechanisms for the emergence of oscillations be a stronger foundation for understanding absence seizures?
