Abstract
Whyte-Fagundes PA, Vance A, Carroll A, Figueroa F, Manukyan C, Baraban SC. Brain Commun. 2024;6(3):fcae135. Dravet syndrome is a severe genetic epilepsy primarily caused by de novo mutations in a voltage-activated sodium channel gene (SCN1A). Patients face life-threatening seizures that are largely resistant to available anti-seizure medications. Preclinical Dravet syndrome animal models are a valuable tool to identify candidate anti-seizure medications for these patients. Among these, scn1lab mutant zebrafish, exhibiting spontaneous seizure-like activity, are particularly amenable to large-scale drug screening. Thus far, we have screened more than 3000 drug candidates in scn1lab zebrafish mutants, identifying valproate, stiripentol, and fenfluramine e.g. Food and Drug Administration-approved drugs, with clinical application in the Dravet syndrome population. Successful phenotypic screening in scn1lab mutant zebrafish is rigorous and consists of two stages: (i) a locomotion-based assay measuring high-velocity convulsive swim behaviour and (ii) an electrophysiology-based assay, using in vivo local field potential recordings, to quantify electrographic seizure-like events. Historically, nearly 90% of drug candidates fail during translation from preclinical models to the clinic. With such a high failure rate, it becomes necessary to address issues of replication and false positive identification. Leveraging our scn1lab zebrafish assays is one approach to address these problems. Here, we curated a list of nine anti-seizure drug candidates recently identified by other groups using preclinical Dravet syndrome models: 1-Ethyl-2-benzimidazolinone, AA43279, chlorzoxazone, donepezil, lisuride, mifepristone, pargyline, soticlestat and vorinostat. First-stage locomotion-based assays in scn1lab mutant zebrafish identified only 1-Ethyl-2-benzimidazolinone, chlorzoxazone and lisuride. However, second-stage local field potential recording assays did not show significant suppression of spontaneous electrographic seizure activity for any of the nine anti-seizure drug candidates. Surprisingly, soticlestat induced frank electrographic seizure-like discharges in wild-type control zebrafish. Taken together, our results failed to replicate clear anti-seizure efficacy for these drug candidates highlighting a necessity for strict scientific standards in preclinical identification of anti-seizure medications.
Commentary
Ninety percent of drug candidates identified in preclinical models do not make it to the market, and CNS drugs are particularly prone to fail.1,2 In a comparison of compounds that entered clinical trials between 1990 and 2012, Phase 3 CNS drugs were 45% less likely to progress to regulatory filing than non-CNS drugs, and 45% of discontinuations were due to failure to demonstrate efficacy. The high failure risk has been attributed to both inappropriate judgment by sponsors regarding success in Phase 2 and low predictive value of animal models. 1
In epilepsy research, rodents are the most common animal models, but other species, including flies, worms, dogs, and primates have been utilized as well. More recently, new genetic model organisms have emerged, expending the tool kit for modeling epilepsy in animals. 3 Despite these advances, several investigational antiseizure medications (ASMs) have recently failed Phase 2 or 3 clinical trials, 4 highlighting the importance of better lead candidate selection and rigorous validation of preclinical models.
The featured article by Whyte-Fagundes et al presents the outcomes of a study aimed to replicate findings obtained in other preclinical models of Dravet syndrome in mutated zebrafish. 5 The authors used the previously-described mutant scn1lab zebrafish line that exhibits spontaneous seizure-like activity, sleep disturbance, early fatality, metabolic deficits and pharmacoresistance to ASMs. In previous studies, this line correctly identified fenfluramine and cannabidiol as potential therapies for Dravet syndrome. The screening protocol of the lab consists of two stages. The first stage is a high-throughput assay in 96-well plate wells that involves tracking of single-point locomotion in freely swimming zebrafish larvae. Scn1lab mutants have elevated levels of swim activity and compounds with antiseizure effects reduce swim velocity. The second is an electrophysiology-based in vivo assay that identifies antiseizure activity based on suppression of epileptiform events. The molecules were also screed for potential toxicity.
The nine investigational compounds that were screened have previously demonstrated effectiveness in murine or zebrafish models of Dravet syndrome. One, soticlestat, has been on two Phase 3 trials in patients with Dravet syndrome (SKYLINE, NCT04940624) and Lennox-Gastaut syndrome (SKYWAY, NCT04938427) and on the open-label ENDYMION extension study (NCT05163314). 6 Vorinostat was tested in a Phase 2 safety and efficacy trial in pediatric patients with drug-resistant epilepsy (NCT03894826).
In contrast to findings from the previous preclinical studies, the scn1lab mutant zebrafish did not identify any of the nine candidates as a promising ASM: only three compounds were effective in locomotion suppression and none significantly suppressed spontaneous electrographic seizure activity. Soticlestat not only failed to abolish electrographic seizure activity in the mutant larvae, but also adversely affected the wild type controls: swim velocities were higher, survival rates lower, and the larvae demonstrated seizure-like activity.
One major limitation, also common to many zebrafish studies, is the lack of information to address the question of whether the non-promising findings were related to pharmacokinetic or pharmacodynamic factors. The study was conducted on day post-fertilization (DPF) 5, and the screening assays in which the tested molecules demonstrated effectiveness were performed on DFP 5-7, during blood-brain barrier (BBB) maturation in Zebrafish. In wild type zebrafish, some but not all cerebral vessels express the key BBB proteins claudin-5 and ZO-1 from DPF 3, coinciding with the retention of certain dyes and tracers within those vessels. Sodium fluorescein (molecular weight 376 Da) can cross the zebrafish BBB until DPF 10. In contrast, Evans blues (961 Da) is already excluded from the brain on DPF 5. 7 Interestingly, heterogeneity of the vasculature has also been reported for the human epileptogenic brain, 8 suggesting that ASM concentrations in “islets” of brain tissue might not suffice for seizure suppression. The heterogeneity in vasculature might also be one explanation for the inconsistency among the screening assays. Several key questions yet to be answered are whether the BBB of the mutant larvae follows the wild-type pattern of development, to what extent it models the human BBB, and what properties other than molecular weight can determine whether a molecule can cross the BBB. For example, the molecular weight of soticlestat is 373 Da, similar to fluorescein, but the molecule is three-fold longer as compared to fluorescein.
The paper by Whyte-Fagundes et al was first published online on April 16, 2024; two months later, on June 17, 2024, Ovid Therapeutics reported on the outcomes of the two Phase 3 soticlestat trials. 6 In SKYLINE (144 patients with Dravet syndrome), soticlestat met the secondary endpoints but missed the primary endpoint of reduction from baseline in convulsive seizure frequency as compared with placebo (P = 0.06). In the SKYWAY trial (270 patients with Lennox–Gastaut syndrome), soticlestat missed the primary endpoint of reduction in major motor drop seizure frequency in comparison with placebo. Unlike in fish, soticlestat demonstrated in humans a safety profile consistent with the findings of earlier studies. The vorinostat study was expected to be completed in 2020 but as of 2024 its status is unknown.
Recently we published an open-access utility for calculating the risk of candidate ASMs failing during drug development or drug repurposing, based on their physicochemical properties. 4 The algorithm consists of six properties such as molecular weight and length. Four of the nine compounds that were screened in the featured study, including soticlestat, were assigned in the in silico analysis a score lower than 4, predictive of a high risk of failure due to efficacy reasons. One (vorinostat) achieved a borderline score (Figure 1). In contrast, no issues were identified for cannabidiol and fenfluramine. The in silico tool is not meant to predict toxicity and cannot explain the emergence of soticlestat-induced seizure-like activity in wild type fish, nor can it clarify whether non-efficacy is related to pharmacokinetics, pharmacodynamics, or both.
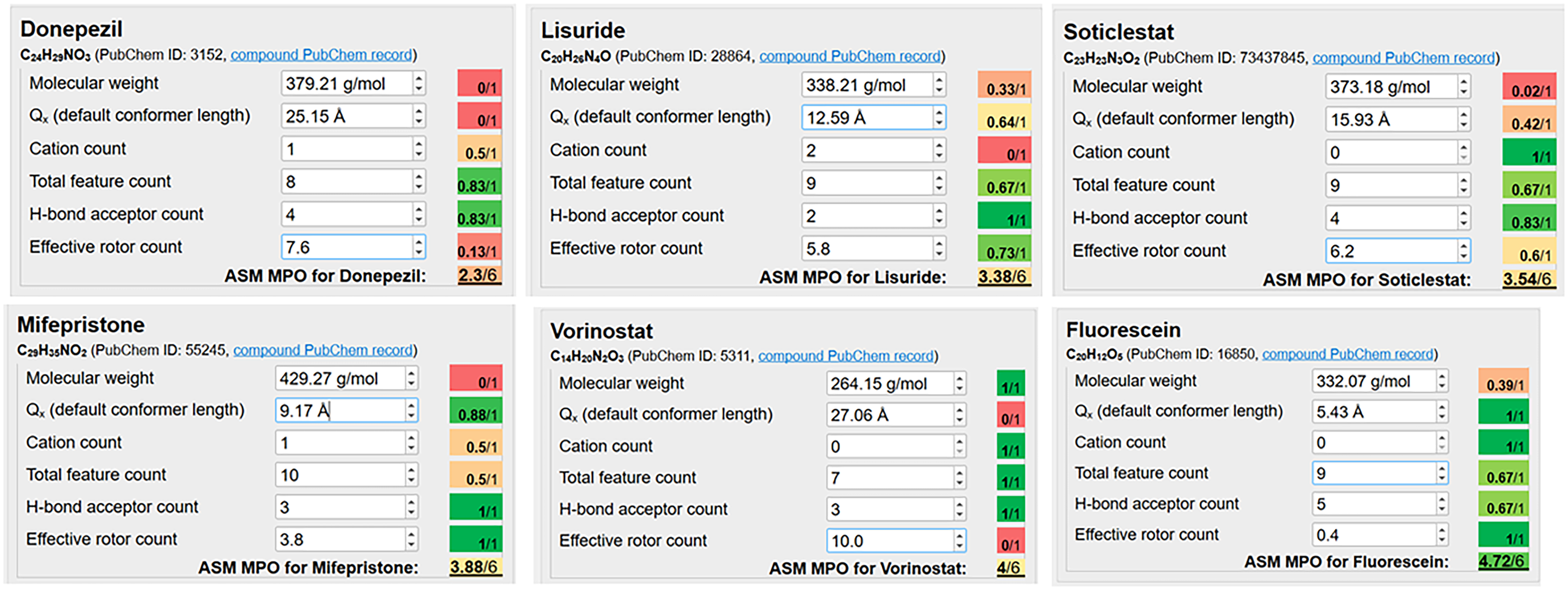
Physicochemical properties of ASM candidates that have not shown efficacy in the study by Whyte-Fagundes et al. and scored low in silico. 4 A score lower than 4 is associated with an increased risk of failure due to insufficient efficacy. Also shown is fluorescein, for comparison.
In analogy with the zebrafish model, too little is known about exposure to ASMs and investigational agents at the human epileptogenic brain. This is demonstrated by the case of padsevonil. The developers conducted positron emission tomography (PET) imaging studies with radiolabeled padsevonil to identify optimal target occupancy. 9 However, the studies were conducted in healthy volunteers, not necessarily reflecting the drug levels achievable in patients with epilepsy. The drug did not achieve the efficacy endpoint Phase 3 trials in adult patients with drug-resistant epilepsy, but the missing patient imaging data complicates the interpretation of the causes for failure.
Back to zebrafish, high-resolution microscopy studies with fluorescently-labeled small molecules can add information on the brain vasculature in the scn1lab model and on heterogeneity in drug distribution. The outcomes of the soticlestat and possibly vorinostat trials support the importance of strict scientific standards in preclinical ASM identification. Additionally, they suggest that higher reliance on in silico tools during lead candidate selection may minimize failure rate in clinical trials.
Footnotes
Declaration of Conflicting Interests
The author declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: The author has served as a consultant for Dexel, TrueMed, and BioPass, Israel.
Funding
The author received no financial support for the research, authorship, and/or publication of this article.
