Abstract
This practice guideline provides updated evidence-based conclusions and recommendations regarding the effects of antiseizure medications (ASMs) and folic acid supplementation on the prevalence of major congenital malformations (MCMs), adverse perinatal outcomes, and neuro-developmental outcomes in children born to people with epilepsy of childbearing potential (PWECP). A multidisciplinary panel conducted a systematic review and developed practice recommendations following the process outlined in the 2017 edition of the American Academy of Neurology Clinical Practice Guideline Process Manual. The systematic review includes studies through August 2022. Recommendations are supported by structured rationales that integrate evidence from the systematic review, related evidence, principles of care, and inferences from evidence. The following are some of the major recommendations. When treating PWECP, clinicians should recommend ASMs and doses that optimize both seizure control and fetal outcomes should pregnancy occur, at the earliest possible opportunity preconceptionally. Clinicians must minimize the occurrence of convulsive seizures in PWECP during pregnancy to minimize potential risks to the birth parent and to the fetus. Once a PWECP is already pregnant, clinicians should exercise caution in attempting to remove or replace an ASM that is effective in controlling generalized tonic-clonic or focal-to-bilateral tonic-clonic seizures. Clinicians must consider using lamotrigine, levetiracetam, or oxcarbazepine in PWECP when appropriate based on the patient’s epilepsy syndrome, likelihood of achieving seizure control, and comorbidities, to minimize the risk of MCMs. Clinicians must avoid the use of valproic acid in PWECP to minimize the risk of MCMs or neural tube defects (NTDs), if clinically feasible. Clinicians should avoid the use of valproic acid or topiramate in PWECP to minimize the risk of offspring being born small for gestational age, if clinically feasible. To reduce the risk of poor neurodevelopmental outcomes, including autism spectrum disorder and lower IQ, in children born to PWECP, clinicians must avoid the use of valproic acid in PWECP, if clinically feasible. Clinicians should prescribe at least 0.4 mg of folic acid supplementation daily preconceptionally and during pregnancy to any PWECP treated with an ASM to decrease the risk of NTDs and possibly improve neurodevelopmental outcomes in the offspring.
Glossary
American Academy of Neurology
adjusted hazard ratio
autism spectrum disorder
antiseizure medication
conflict of interest
major congenital malformation
neural tube defect
prevalence difference
prevalence ratio
people with epilepsy of childbearing potential
raw mean difference
small for gestational age.
Epilepsy is one of the most common neurologic disorders, affecting more than 50 million people worldwide. One in 5 of those affected are people of childbearing potential, based on extrapolations from the proportion of the 2022 US female population aged 15–45 years. 1 Infants born to people with epilepsy are at increased risk of major congenital malformations (MCMs), adverse perinatal outcomes, and adverse neuro-developmental outcomes. 2 Multiple factors are associated with this risk, including genetic differences, environmental factors, seizure control, and intrauterine exposure to antiseizure medications (ASMs). The role of folic acid supplementation in mitigating these risks is unclear. Optimizing the treatment of epilepsy is necessary to achieve the most favorable outcomes for persons with epilepsy and their offspring.
In 2009, the American Academy of Neurology (AAN) published the guideline “Practice Parameter update: Management issues for women with epilepsy—Focus on pregnancy: Teratogenesis and perinatal outcomes.” 3 The authors concluded that treatment with valproic acid carries a higher risk of MCMs in the offspring of women with epilepsy than treatment with carbamazepine, phenytoin, and phenobarbital, especially if taken in polytherapy. The risk associated with other commonly used ASMs, such as levetiracetam or topiramate, was not evaluated because of limited available evidence. The authors concluded that treatment with valproic acid carried the highest risk of adverse cognitive outcomes in the offspring of women with epilepsy as compared with carbamazepine, although the risk of autism spectrum disorder (ASD) was not addressed because this association was not yet reported in the literature. Infants exposed to any ASM in utero had a higher risk of being born small for gestational age (SGA), but there was no evidence of an increased risk of fetal death.
A separate 2009 practice guideline recommended that preconception folic acid supplementation “may be considered to reduce the risk of MCMs,” but did not provide further guidance on supplementation dosage. 4 Since 2009, new studies have been published related to the risk of MCMs associated with several ASMs, the association between different ASMs and adverse perinatal or neurodevelopmental outcomes, and the effect of folic acid supplementation.
While the 2009 guidelines described the affected population as “women with epilepsy,” this phrasing does not recognize the important difference between biological sex and sociocultural gender. In this update, we refer to the affected population with the gender-neutral language, “people with epilepsy of childbearing potential” (PWECP).
In this practice guideline update, we aim to provide guidance to clinicians when choosing an ASM, in monotherapy or polytherapy, in this patient population. We also aim to clarify the potential role of folic acid supplementation among PWECP. This guideline specifically addresses the following 4 clinical questions:
What is the prevalence of MCMs associated with intrauterine exposure to specific ASMs, and how does this vary between ASMs in monotherapy vs polytherapy, and at high doses vs low-medium doses of ASMs, in children born to PWECP? What is the prevalence of adverse perinatal outcomes associated with intrauterine exposure to specific ASMs, and how does this vary between ASMs in monotherapy vs polytherapy, and at high doses vs low-medium doses of ASMs, in children born to PWECP? What is the prevalence of adverse neurodevelopmental outcomes associated with intrauterine exposure to specific ASMs, and how does this vary between ASMs in monotherapy vs polytherapy, and at high vs low-medium doses of ASMs, in children born to PWECP? What is the effect of intrauterine exposure to folic acid on the prevalence of MCMs, adverse perinatal outcomes, and neurodevelopmental outcomes, and how does this vary by folic acid dose in children born to PWECP treated with ASMs?
Description of the Analytic Process
The development of this practice guideline followed the 2017 edition of the AAN’s guideline development process manual. 5 In March 2018, a multidisciplinary panel was recruited to develop the protocol for this guideline. The authors include content experts, methodologists, Guidelines Subcommittee members, an AAN epilepsy quality measure workgroup representative, physician representatives for the American Epilepsy Society and the Society for Maternal-Fetal Medicine, and patient advocates. In accordance with AAN policy, the current lead developer (A.M.P.), and the majority of the panel, has no conflicts of interest (COIs). Five of the 19 guideline developers (J.F., E.G., K.P., G.S., and T.T.) were determined to have COIs, but each COI was judged to be not significant enough to preclude authorship. These 5 developers were not permitted to review or rate the evidence; they served in an advisory capacity to help with the validation of the key questions, the scope of the literature search, and the identification of seminal articles. They also participated in the recommendation development process. The full author panel was solely responsible for final decisions about the design, analysis, and reporting of this guideline.
This article is a summary of the key findings of the guideline. The complete guideline, including the literature search strategy, details about evidence classification, and the full systematic review of the evidence, is available in eAppendix 1.
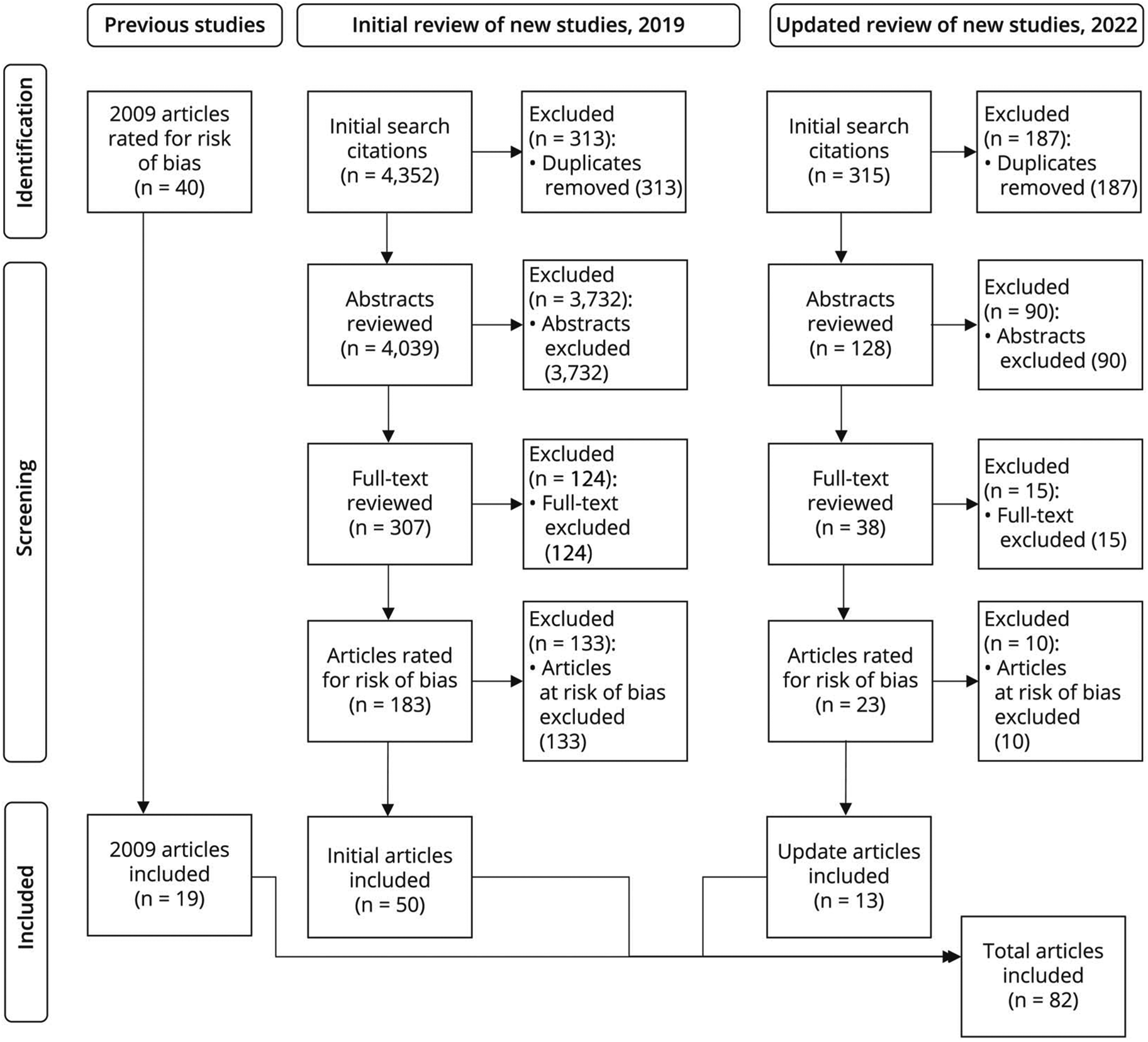
Study Selection.
Systematic Review of the Evidence
The panel searched Ovid MEDLINE, the Cochrane Central Register of Controlled Trials, the Cochrane Database of Systematic Reviews, Ovid Embase, CINAHL, the Database of Abstracts of Reviews of Effects, ClinicalTrials.gov, and the US Food and Drug Administration literature databases from June 1, 2007, to February 15, 2019, for relevant peer-reviewed articles that met inclusion criteria. The initial search after duplicates were removed yielded 4,039 articles. Using a systematic process detailed in the AAN’s guideline development process manual, 5 2 review panel members (not the same pair for all articles) independently reviewed the article titles and abstracts for relevance and then reviewed the full text of the articles determined to be relevant (Figure). Disagreements about inclusion were resolved through discussion between the 2 panelists, with a third reviewer included to break ties when necessary. One hundred eighty-three articles were selected and rated for risk of bias by 2 panel members using the AAN criteria for the classification of causation studies. Class I studies have the lowest risk of bias, and Class IV studies have the highest risk of bias. As per predefined exclusion criteria that are laid out in the process manual, 5 the panel excluded articles that were assessed as Class IV (n = 133). This left 50 articles for inclusion. Forty articles included in the 2009 guidelines were reviewed by 2 panel members and 19 were selected for inclusion, for a total of 69 articles.
An updated literature search was completed to identify additional relevant articles published between February 15, 2019, and August 1, 2022. The initial search after duplicates were removed yielded 128 articles. The abstracts and full-text articles were reviewed following the same process as the first literature review, which resulted in 13 articles being added to the systematic review (Figure). The primary findings of the systematic review are summarized in Tables 1–7. Additional data are presented in eTables 1 and 2.
Unadjusted Prevalence of Any MCM by ASM in Monotherapy or Polytherapy.
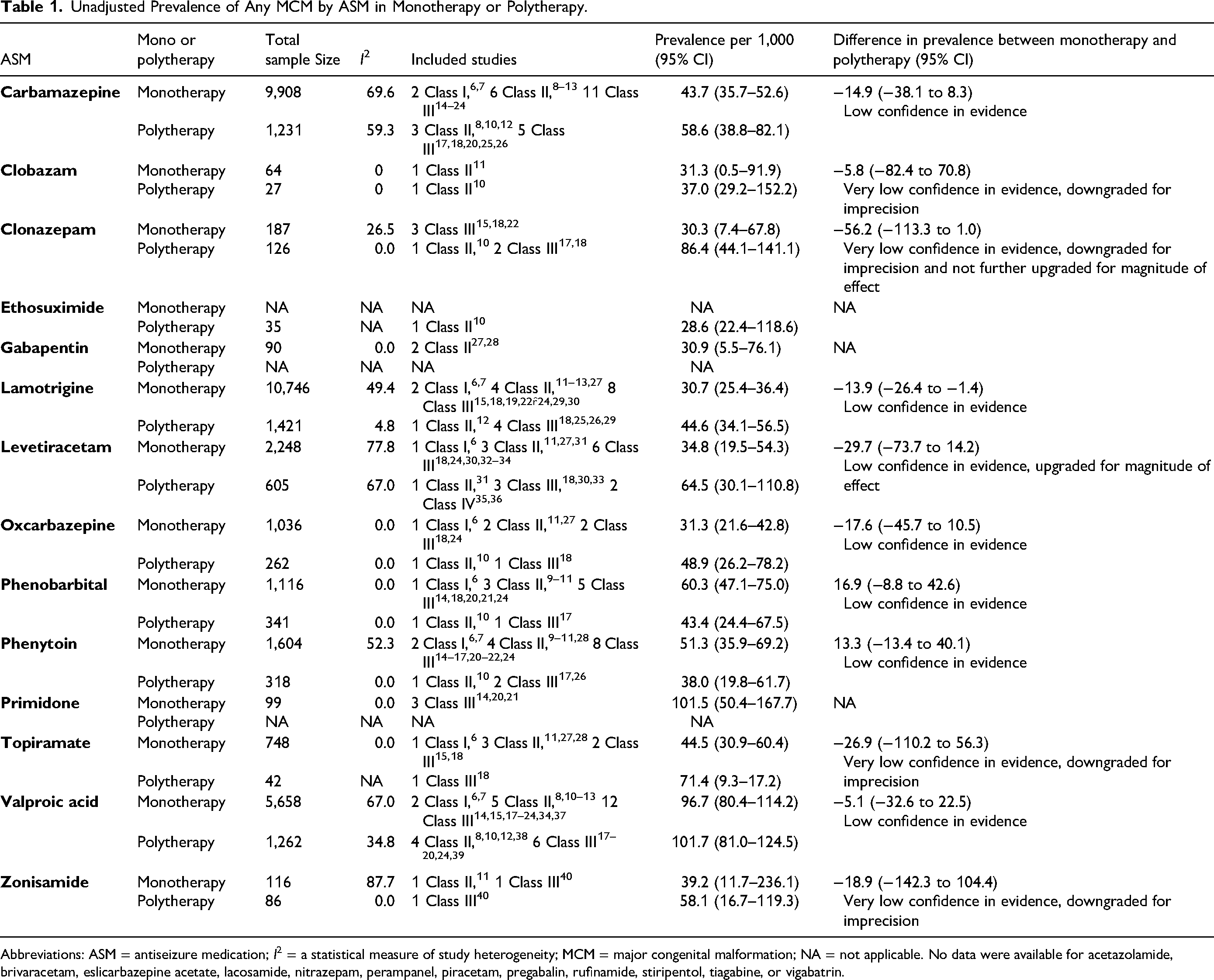
Abbreviations: ASM = antiseizure medication; I2 = a statistical measure of study heterogeneity; MCM = major congenital malformation; NA = not applicable. No data were available for acetazolamide, brivaracetam, eslicarbazepine acetate, lacosamide, nitrazepam, perampanel, piracetam, pregabalin, rufinamide, stiripentol, tiagabine, or vigabatrin.
Unadjusted Prevalence Differences of Any MCM Across ASMs in Monotherapy.
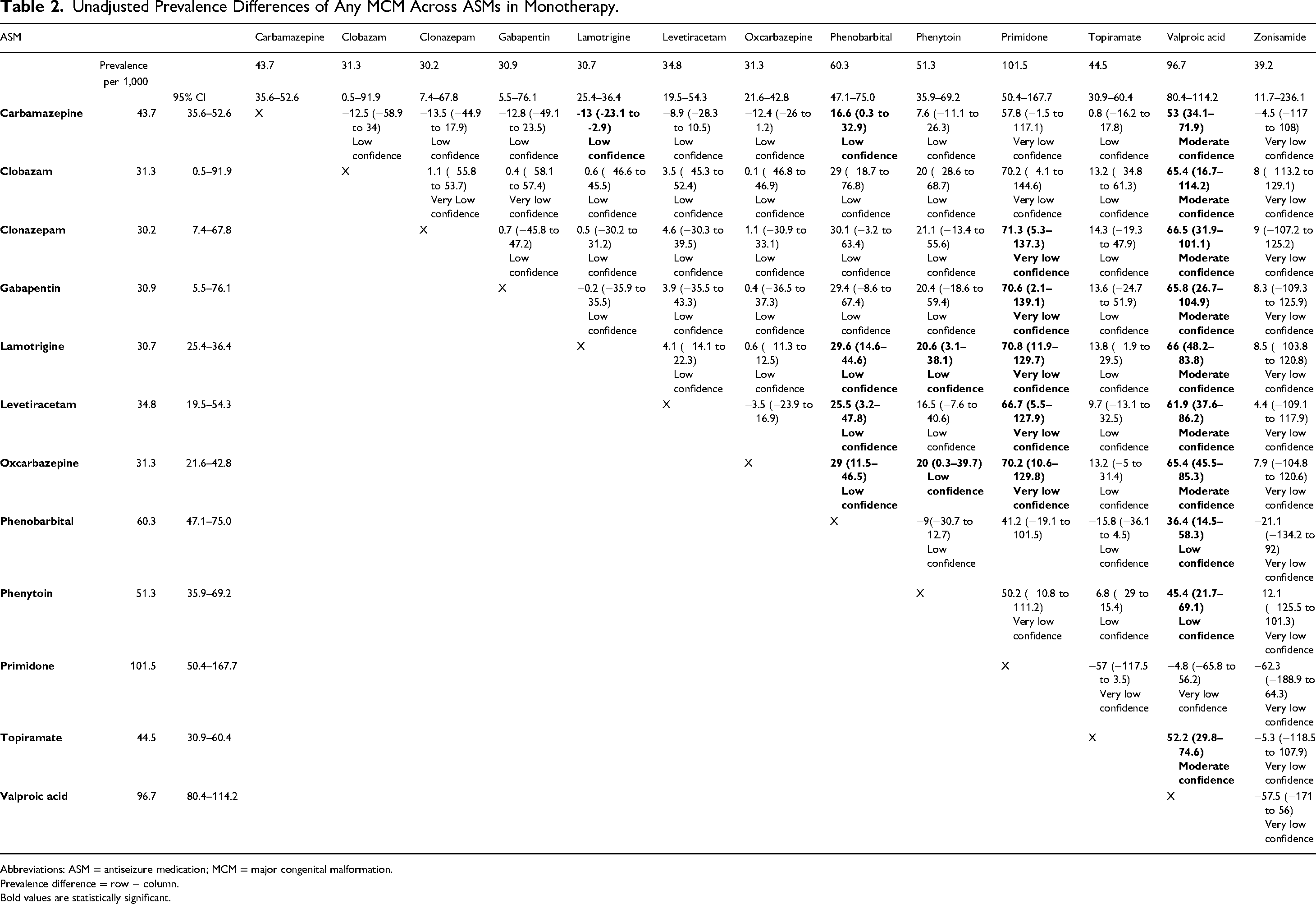
Abbreviations: ASM = antiseizure medication; MCM = major congenital malformation.
Prevalence difference = row − column.
Bold values are statistically significant.
Unadjusted Prevalence of Specific MCM, by Individual ASMs in Monotherapy.
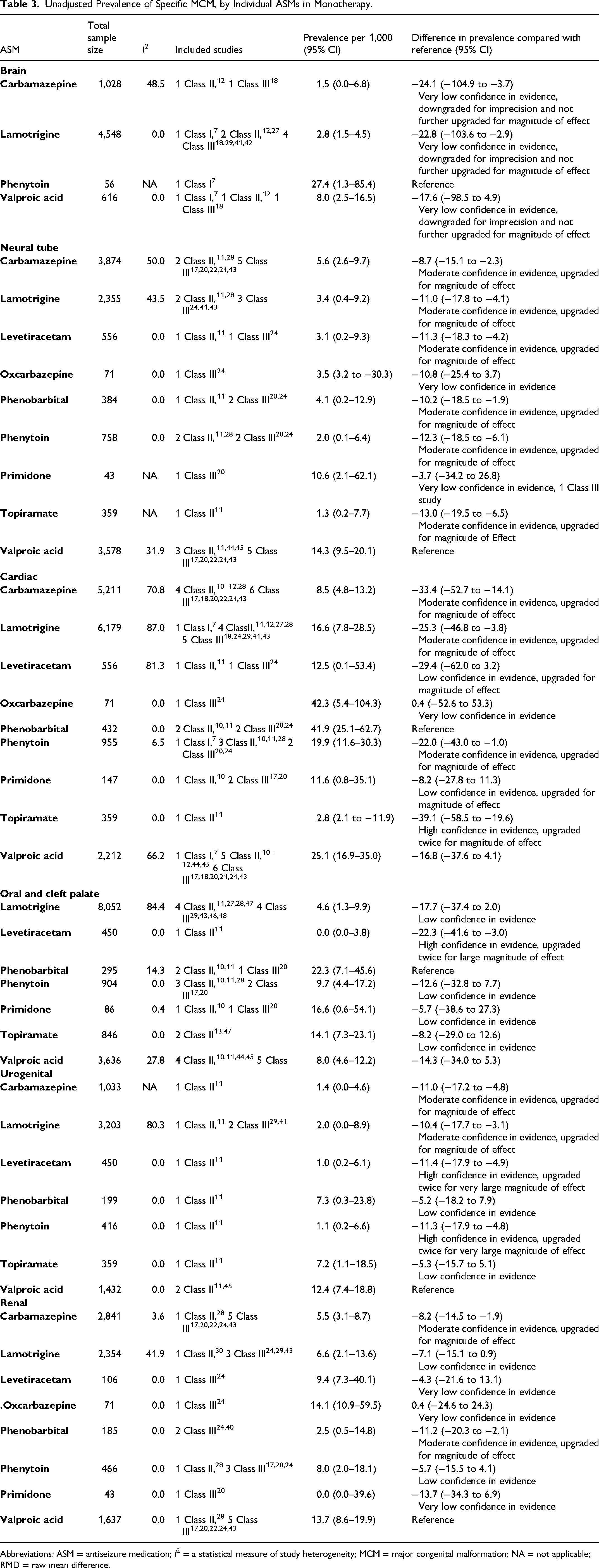
Abbreviations: ASM = antiseizure medication; I2 = a statistical measure of study heterogeneity; MCM = major congenital malformation; NA = not applicable; RMD = raw mean difference.
Global IQ With Exposure to ASM Monotherapy.
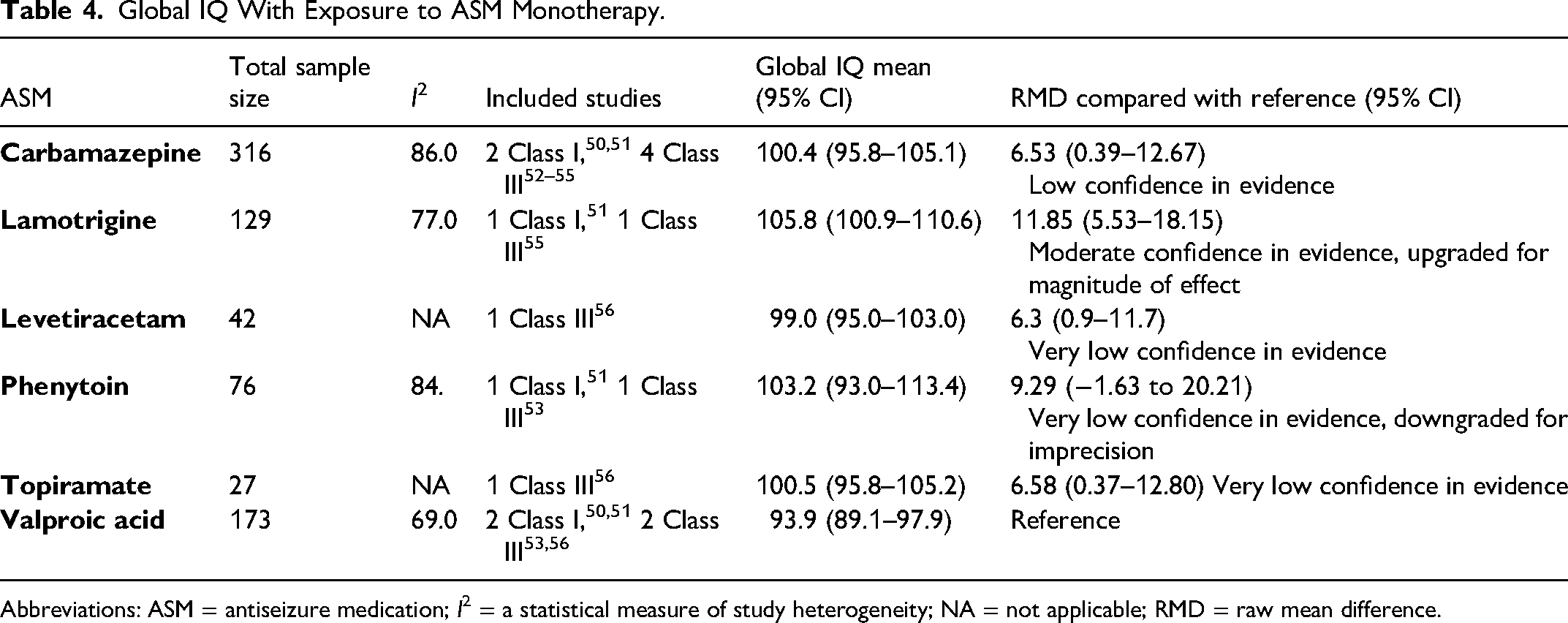
Abbreviations: ASM = antiseizure medication; I2 = a statistical measure of study heterogeneity; NA = not applicable; RMD = raw mean difference.
Verbal and Non-Verbal IQ With Exposure to ASM Monotherapy.
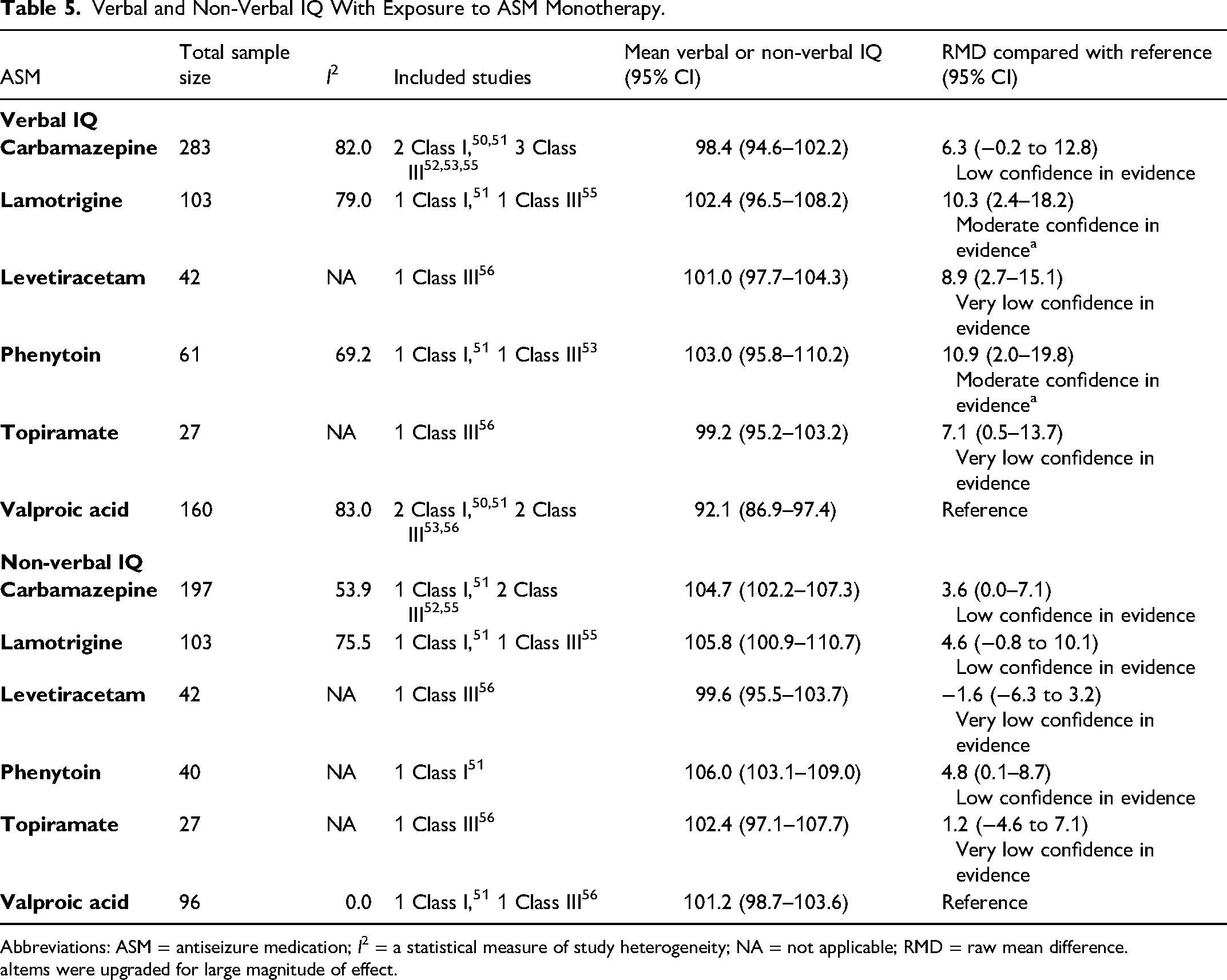
Abbreviations: ASM = antiseizure medication; I2 = a statistical measure of study heterogeneity; NA = not applicable; RMD = raw mean difference.
Items were upgraded for large magnitude of effect.
Unadjusted Prevalence of ASD, PDD, or ASD Traits by ASM Monotherapy.
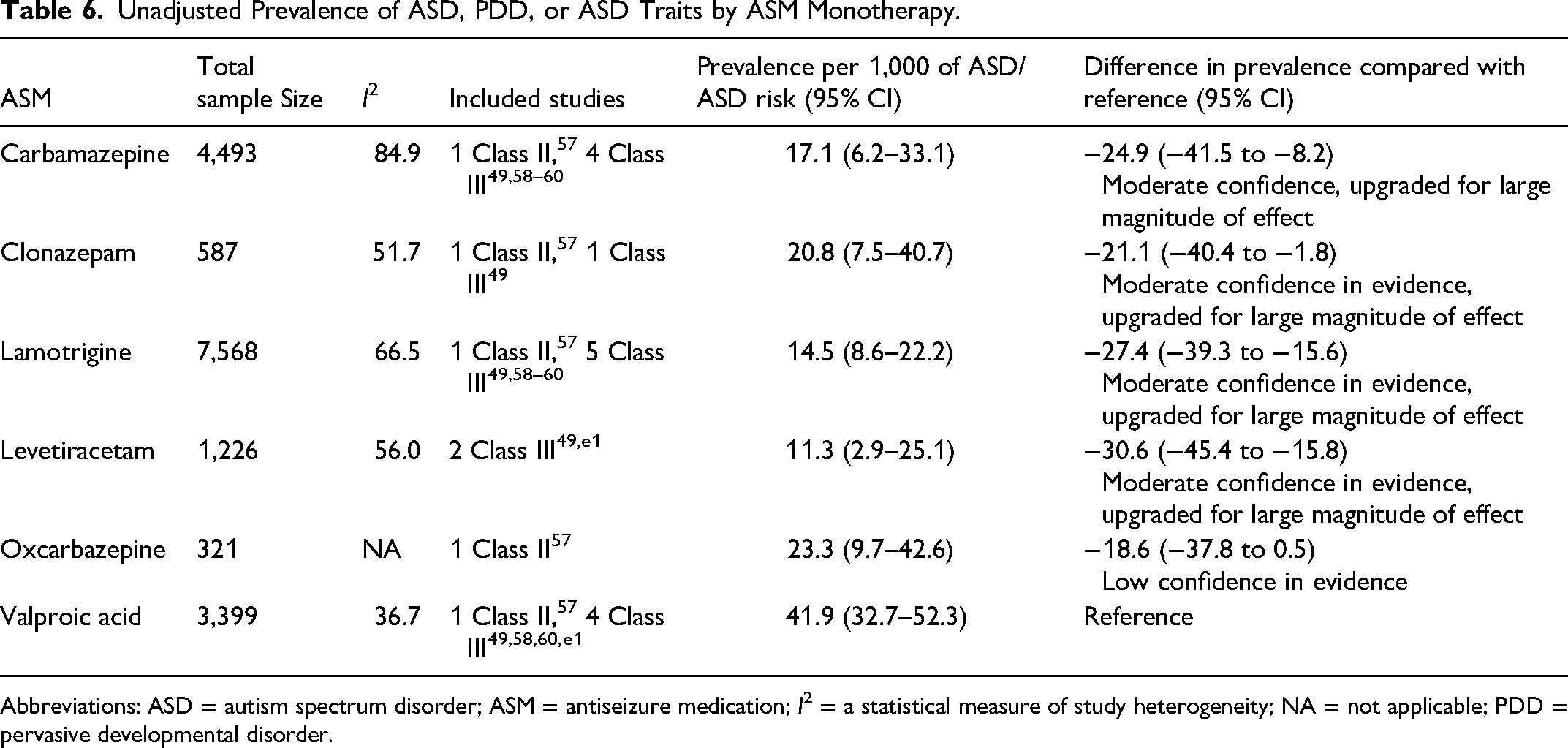
Abbreviations: ASD = autism spectrum disorder; ASM = antiseizure medication; I2 = a statistical measure of study heterogeneity; NA = not applicable; PDD = pervasive developmental disorder.
Unadjusted Prevalence of SGA by ASM Monotherapy.
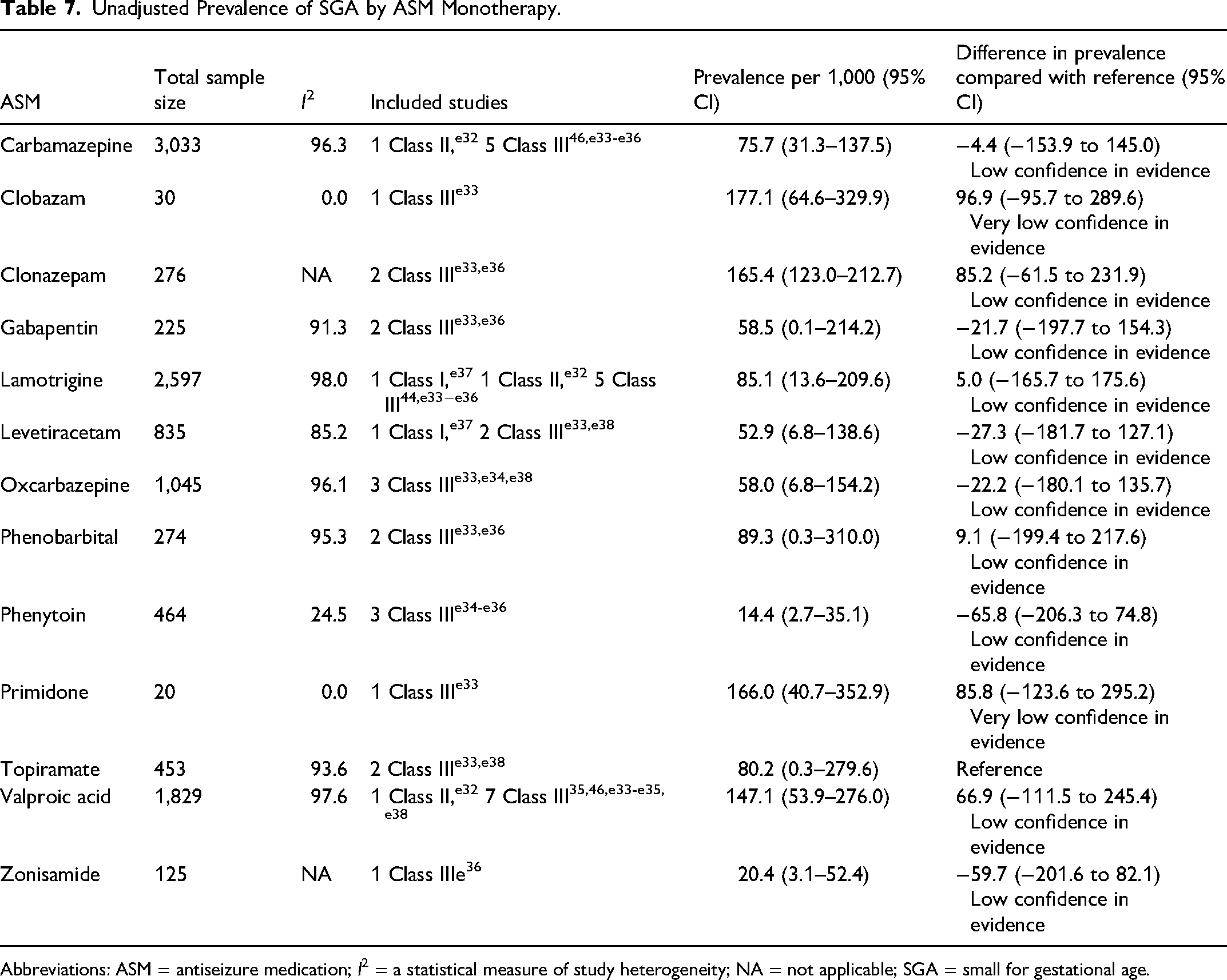
Abbreviations: ASM = antiseizure medication; I2 = a statistical measure of study heterogeneity; NA = not applicable; SGA = small for gestational age.
As detailed in the AAN’s guideline development process manual, a modified version of the Grading of Recommendations Assessment, Development and Evaluation process was used to develop conclusions after the analysis of evidence. 5 The evidence was analyzed based on parameters pertaining to risk of bias, consistency, directness, precision, and publication bias, providing transparency of the classification of evidence. As all comparisons included indirect data (comparisons between results reported in different studies) and, at best, classified as Class III evidence to address causation, the initial confidence rating for most conclusions was anchored as low if at least 2 Class III or at least 1 Class I or II studies informed each estimate used in the comparisons. The initial confidence rating was set to very low if one of the contributing estimates was informed by a single Class III study.
In the second step, the classification of evidence was upgraded or downgraded according to criteria specified in the process manual (e.g., upgraded for large magnitude of effect, downgraded for lack of statistical precision). 5 For estimates obtained through indirect comparisons, confidence in the evidence was downgraded for precision when the width of the 95% CI for any prevalence difference (PD) for MCMs or ASD was greater than 100 per 1,000 live births or greater than 300 per 1,000 live births for perinatal outcomes. Confidence was also downgraded for precision when the width of the 95% CI raw mean difference (RMD) for IQ was greater than 20 points. For indirect comparisons, although we present the PD in the synthesis of evidence and conclusions, our assessment of magnitude of effect was based on the corresponding prevalence ratio (PR). Confidence in the evidence was upgraded by 1 level for large magnitude of effect if the calculated PR was greater than 2 or lower than 0.5. Confidence in the evidence was upgraded by 2 levels for very large magnitude of effect if the calculated PR was greater than 10 or lower than 0.1. Confidence in the evidence was upgraded by 1 level for large magnitude of effect for IQ if the RMD was greater than 10 points and by 2 levels if greater than 20 points. For estimates drawn from adjusted PR (relevant to the perinatal and neurodevelopmental outcomes), confidence in evidence was downgraded for precision if the width of the CI was greater than 2. If the confidence in the evidence was very low, it was not upgraded for other factors. Estimates not reaching statistical significance were not upgraded for magnitude of effect.
The authors formulated a rationale for each recommendation based on the evidence systematically reviewed and stipulated axiomatic principles of care, related evidence, and inferences. The recommendation development process is described in further detail in the complete guideline (eAppendix 1) and the AAN’s guideline development process manual. 5
Clinical Context
The goal of this guideline is to assist clinicians (e.g., physicians, nurses, and advanced practice providers) in the pharmacologic management of PWECP to limit risk of adverse congenital, perinatal, and neurodevelopmental outcomes. Given the many variables that may confound the outcomes we examined (e.g., genetic conditions, pregnancy conditions, and socioeconomic contexts), we weighted evidence more strongly where analyses could be adjusted for these and other potential confounders (i.e., Class I studies). Demonstration of a dose effect can further support a causal relationship between an exposure and an outcome. Although our preplanned analyses using external comparisons could not reach a level of evidence suffcient to drive recommendations, a statistically and clinically important difference in prevalence of MCMs was found for valproic acid and phenobarbital between high and low-dose exposures (eTable 1). The only Class I study addressing this question from EURAP demonstrated a dose effect for carbamazepine, lamotrigine, phenobarbital, and valproic acid. 6 To reduce the risk of MCMs, it is reasonable practice to use the lowest appropriate dose of ASMs in PWECP, if clinically feasible.
The available evidence on the association between in utero ASM exposure and neurodevelopmental outcomes is rapidly expanding. Although valproic acid exposure shows a strong effect, data from our preplanned analyses on adverse neuro-developmental outcomes were insuffcient to demonstrate an effect; thus, caution in counseling is warranted. While we could not extract sufficient data on topiramate exposure, the SCAN-AED study 49 found even higher prevalences of ASD and intellectual disability with exposure to topiramate than valproic acid. Their adjusted hazard ratios (aHRs), however, used prevalence in the general population of children as a comparator group (aHRs for ASD and intellectual disability after topiramate exposure were 2.8 [95% CI 1.4–5.7] and 3.5 [95% CI 1.4–8.6], respectively). Further studies are needed to replicate these findings and examine these outcomes across other ASMs.
Folic acid prescribing practices for PWECP are variable.e2,e3 One much anticipated outcome from the current systematic review was clarification of the optimal folic acid dosage to reduce potential negative effects of ASMs in pregnancy. As discussed, the data do not find that folic acid supplementation reduces the risk of MCMs among PWECP. However, improved neurocognitive outcomes have been observed in offspring of PWECP who received folic acid supplementation before and throughout pregnancy. The analysis does not support a more specific dosage recommendation beyond at least 0.4 mg/d. There is limited evidence from a published analysis of 27,784 children born to people with epilepsy that exposure to periconceptional folic acid ≥1 mg/d was associated with a 0.9% absolute increase in the risk of childhood cancer before age 20 years, resulting in an HR of 2.7 (95% CI 1.2–6.3). Subanalysis restricted to exposure to maternal epilepsy and supplemental folic acid doses <3 mg/d was not significant when compared with maternal epilepsy without a prescription for high-dose folic acid (aHR 2.6, 95% CI 1.0–6.9).e4 A study of 1,257 mother-child pairs from the general population found that very high maternal serum folic acid concentrations (≥60.3 nmol/L) at birth had a 2.5 times increased risk of ASD (95% CI 1.3–4.6) compared with those with lower folic acid concentrations.e5 These results are concerning, but the studies have limitations, including their high risk of confounding by indication. The dose chosen should balance demonstrated benefits of supplementation and potential negative consequences of high doses. Future well-designed (preferably randomized) studies are needed to better define optimal folic acid dosing for PWECP.
Practice Recommendations
General
Recommendation 1 Rationale
The overarching goals of care for PWECP are to optimize health outcomes both for individuals and their future offspring. In many cases, in utero ASM exposure may be associated with increased risks to the fetus. There are also risks associated with discontinuing or changing ASMs in PWECP.53,e6-e8 A shared decision-making process leads to more informed choices, a better understanding of available options, a more accurate risk perception, and improved decision quality grounded in individual values.e9 This decision-making process may take into account an individual’s plans for pregnancy. However, according to the Epilepsy Birth Control Registry of 1,114 PWECP in the United States, more than 65% of pregnancies among PWECP are unintended.e10,e11 The ASM regimen used for a PWECP when pregnancy is not planned is thus very often the regimen used at the time of conception.
Recommendation 1 Statements
1(A) Clinicians should engage in joint decision-making with PWECP, taking individual preferences into account when selecting ASMs and monitoring their dosing (Level B).
1(B) When treating PWECP, clinicians should recommend ASMs and doses that optimize both seizure control and fetal outcomes should pregnancy occur, at the earliest possible opportunity preconceptionally (e.g., at the time of starting an ASM in a person post-menarche) (Level B).
Recommendation 2 Rationale
The odds of mortality during pregnancy is 5–12 times greater among PWECP as compared with pregnant people without epilepsy, according to an analysis of a Danish cohort of more than 2 million pregnancies and a US cohort of more than 20 million participants.e12,e13 Among 202 pregnancy-related deaths in the United Kingdom from 2013 to 2015, most of the 13 epilepsy-related deaths were from sudden unexpected death in epilepsy. All participants with prepregnancy data had uncontrolled seizures. Five of the participants who died had stopped taking their ASMs during pregnancy.e14
In an analysis of the EURAP study including 1,956 pregnancies among 1,882 participants, there was no statistical association between seizures during pregnancy and spontaneous abortion or stillbirth. However, the 1 stillbirth that occurred soon after a seizure was an episode of convulsive status epilepticus.e15 The frequency of generalized tonic-clonic seizures or focal-to-bilateral tonic-clonic seizures may also be a risk factor of lower IQ in children born to PWECP. 53
Valproic acid is one of the most effective ASMs at obtaining adequate seizure control among people with idiopathic generalized epilepsy.e7,e8 An analysis of the EURAP cohort of PWECP treated with valproic acid at the onset of pregnancy showed that generalized tonic-clonic seizures or focal-to-bilateral tonic-clonic seizures during pregnancy were twice as likely to occur when valproic acid was removed or replaced with another ASM, compared with when it was maintained throughout the pregnancy.e6
The serum concentration of most ASMs has a defined therapeutic window for effective seizure control. The serum concentration of some ASMs (in particular, lamotrigine and levetiracetam) decreases during pregnancy. These decreases may occur at any point during the pregnancy.e16-e18
There are limited data available on epilepsy-related outcomes during pregnancy among PWECP for numerous ASMs, including but not limited to acetazolamide, eslicarbazepine, ethosuximide, lacosamide, nitrazepam, perampanel, piracetam, pregabalin, rufinamide, stiripentol, tiagabine, and vigabatrin.
Recommendation 2 Statements
2A. Clinicians must minimize the occurrence of convulsive seizures (generalized tonic-clonic seizures and focal-to-bilateral tonic-clonic seizures) in PWECP during pregnancy to minimize potential risks to the birth parent (e.g., seizure-related mortality) and to the fetus (Level A).
2B. Once a PWECP is already pregnant, clinicians should exercise caution in attempting to remove or replace an ASM that is effective in controlling generalized tonic-clonic or focal-to-bilateral tonic-clonic seizures, even if it is not an optimal choice with regards to the risk to the fetus (e.g., valproic acid) (Level B).
2C. Clinicians should monitor ASM levels in PWECP throughout pregnancy as guided by individual ASM pharmacokinetics and patient clinical presentation (Level B).
2D. Clinicians should adjust the dose of ASMs at their clinical discretion during the pregnancy in response to (1) decreasing serum ASM levels or (2) worsening seizure control (observed or anticipated based on the clinician’s judgment and known pharmacokinetics of ASMs in the pregnant state) (Level B).
2E. Clinicians treating PWECP using acetazolamide, eslicarba-zepine, ethosuximide, lacosamide, nitrazepam, perampanel, piracetam, pregabalin, rufinamide, stiripentol, tiagabine, or vigabatrin should counsel their patients that there are limited data on pregnancy-related outcomes for these drugs (Level B).
Antiseizure Medications: Major Congenital Malformations
Recommendation 3 Rationale
The unadjusted birth prevalence of any MCM among children born to people without epilepsy is approximately 2.4%–2.9%.e19 Of the ASMs with sufficient numbers of exposures to draw reliable conclusions (greater than 1,000 exposures), lamotrigine, levetiracetam, and oxcarbazepine are associated with the lowest unadjusted birth prevalence of any MCM in monotherapy (3.1%, 3.5%, and 3.1%, respectively) among children born to PWECP. Valproic acid exposure is associated with the highest unadjusted birth prevalence (9.7%) of any MCM among children born to PWECP as compared with other ASMs.
Valproic acid is associated with the highest unadjusted birth prevalence of neural tube defects (NTDs) (1.4%) as compared with other ASMs. Phenobarbital is associated with the highest unadjusted birth prevalence of cardiac malformations (4.4%) as compared with other ASMs. Phenobarbital and topiramate are associated with the highest unadjusted birth prevalence of oral and cleft palate (2.2% and 1.4% respectively) compared with other ASMs. Valproic acid is associated with the highest un-adjusted birth prevalence of urogenital (1.2%) and renal (1.4%) malformations compared with other ASMs.
A detailed anatomical ultrasound of the fetus can enable earlier diagnosis of MCMs.e20-e24 Early detection of severe congenital heart defects, especially those requiring surgery in the early postnatal period, has been shown to improve morbidity and mortality in affected newborns.e25-e28 Detection of MCMs can also inform an early pregnancy termination decision or guide perinatal management, including giving birth in specialized pediatric centers, while a normal ultrasound may offer reassurance to expecting parents. This needs to be balanced with differences in individual preferences.
Recommendation 3 Statements
3A. Clinicians must counsel their patients with epilepsy that the birth prevalence of any MCM in the general population is approximately 2.4%–2.9%, providing a comparison frame-work for their individual risk (Level A).
3B. Clinicians must consider using lamotrigine, levetiracetam, or oxcarbazepine in PWECP when appropriate based on the patient’s epilepsy syndrome, likelihood of achieving seizure control, and comorbidities, to minimize the risk of MCMs (Level A).
3C. Clinicians must avoid the use of valproic acid in PWECP to minimize the risk of MCMs (composite outcome) or NTDs, if clinically feasible (Level A).
3D. Clinicians must counsel PWECP who are treated with, or are considering starting, valproic acid that the risk of any MCM is the highest with valproic acid as compared with other studied ASMs (Level A).
3E. To reduce the risk of cardiac malformations, clinicians must avoid the use of phenobarbital in PWECP, if clinically feasible (Level A).
3F. To reduce the risk of oral clefts, clinicians should avoid the use of phenobarbital and topiramate in PWECP, if clinically feasible (Level B).
3G. To reduce the risk of urogenital and renal malformations, clinicians should avoid the use of valproic acid in PWECP, if clinically feasible (Level B).
3H. To enable early detection and timely intervention of MCMs, obstetricians should recommend fetal screening for MCMs (e.g., a detailed anatomical ultrasound, where available) for PWECP who are treated with any ASM during pregnancy (Level B).
3I. To enable early detection and timely intervention of congenital heart defects, obstetricians should recommend screening cardiac investigations of the fetus among PWECP who are treated with phenobarbital during pregnancy (Level B).
Antiseizure Medications: Perinatal Outcomes
Recommendation 4 Rationale
Among children exposed to ASMs in utero and born to PWECP, the prevalence of intrauterine death is highly likely not to differ across ASMs when used in monotherapy and the prevalence of prematurity is possibly no different across ASMs when used in monotherapy (eTable 2). The risk of intrauterine death is likely higher with polytherapy exposure compared with monotherapy exposure. Fetal growth restriction increases the risk of perinatal morbidity and mortality.e29,e30 The prevalence of children born SGA is possibly greater after exposure to valproic acid or topiramate compared with lamotrigine. Prenatal identification of fetuses at risk of being born SGA leads to improved perinatal outcomes by informing timely delivery.e31
Recommendation 4 Statements
4A. Clinicians should counsel PWECP that the prevalence of intrauterine death does not differ among different ASM exposures in monotherapy (Level B).
4B. Clinicians should avoid the use of valproic acid or topiramate in PWECP to minimize the risk of offspring being born SGA, if clinically feasible (Level B).
4C. To enable early identification of fetal growth restriction, obstetricians should recommend screening of fetal growth throughout pregnancy among PWECP who are treated with valproic acid or topiramate (Level B).
Antiseizure Medications: Neurodevelopmental Outcomes
Recommendation 5 Rationale
Among children born to PWECP, in utero exposure to valproic acid is likely associated with a decrease in full scale IQ at age 6 years compared with gabapentin and lamotrigine in monotherapy; valproic acid is possibly associated with a decrease as compared with carbamazepine, levetiracetam, and topiramate in monotherapy; and there is possibly no difference in full scale IQ with valproic acid as compared with phenytoin in monotherapy.
Among children born to PWECP, in utero exposure to valproic acid is likely associated with a decrease in verbal IQ at age 6 years compared with gabapentin, lamotrigine, levetiracetam, and phenytoin in monotherapy, and possibly associated with a decrease as compared with carbamazepine and topiramate in monotherapy.
Among children born to PWECP, in utero exposure to valproic acid is possibly associated with a decrease in non-verbal IQ at age 6 years compared with carbamazepine and phenytoin in monotherapy, but there is possibly no difference as compared with gabapentin, lamotrigine, levetiracetam, and topiramate in monotherapy.
Among children born to PWECP, in utero exposure to valproic acid throughout the pregnancy is possibly associated with an increased risk of ASD and autistic traits compared with other studied ASMs (i.e., carbamazepine, clonazepam, lamotrigine, and levetiracetam) used in monotherapy.
Numerous ASMs have limited available data on neuro-developmental outcomes. These neurodevelopmental out-comes are determined during both early and later stages of pregnancy.e39 Early screening for neurodevelopmental disorders in children enables early diagnosis, facilitating access to early interventions where available. Early interventions in children with neurodevelopmental disorders optimize developmental trajectories.
Recommendation 5 Statements
5A. To reduce the risk of poor neurodevelopmental outcomes, including ASD and lower IQ, in children born to PWECP, clinicians must avoid the use of valproic acid in PWECP, if clinically feasible (Level A).
5B. Clinicians must counsel PWECP who are treated with, or are considering starting, valproic acid that in utero exposure to valproic acid is likely or possibly associated with a decrease in full scale, verbal, and non-verbal IQ, as compared with other studied ASMs (i.e., carbamazepine, gabapentin, lamotrigine, levetiracetam, phenytoin, and topiramate) (Level A).
5C. Clinicians must counsel PWECP who are treated with, or are considering starting, valproic acid that in utero exposure to valproic acid is possibly associated with an increased risk of ASD as compared with other studied ASMs (i.e., carbamazepine, clonazepam, levetiracetam, and lamotrigine) (Level A).
5D. Clinicians should implement age-appropriate developmental screening in children exposed to any ASM in utero born to PWECP (Level B).
Folic Acid
Recommendation 6 Rationale
The optimal dosing and timing of folic acid supplementation are unknown in PWECP. There is likely no demonstrated benefit of folic acid supplementation (at least 0.4 mg/d) specifically for the prevention of MCMs in children born to PWECP. Randomized controlled trials conducted before widespread folic acid fortification of foods in the United States demonstrated a reduction in NTDs among the offspring of the general childbearing population receiving periconceptional multivitamin supplementation.e40 A systematic review of 14 studies of folic acid supplementation (up to 1 mg/d) among pregnant people in the general population (generally without epilepsy), including 1,053 participants (some being control participants without folic acid supplementation) estimated that folic acid supplementation of 0.2 mg/d (the United States’ level of folic acid fortification) would reduce the risk of NTDs by 23%.e41 This protective effect was greater in pregnant people with an initial low serum folate concentration than in those with higher serum folate concentrations.e41 Although valproic acid exposure in utero is associated with the highest prevalence of NTDs, the teratogenic causal pathway is not exclusively through the disruption of folic acid metabolism.e42
Preconception folic acid supplementation is possibly associated with better neurodevelopmental outcomes among children born to PWECP. Folic acid supplementation of at least 0.4 mg/d is possibly associated with reduced autistic traits at 3 years (OR 7.9, 95% CI 2.5–24.9) and likely associated with a higher global IQ (on average 6 points) at 6 years in children born to PWECP exposed to ASMs in utero. Lower plasma concentrations of folic acid at gestational weeks 17–19 among pregnant people with epilepsy exposed to ASMs is correlated with a higher risk of autistic traits at 3 years. Higher exposure levels of folic acid from diet and supplements is associated with statistically significant increases in IQ at age 6 years; this association is not seen among PWECP who only received dietary folic acid and not periconceptional folic acid supplements. Higher doses of folic acid supplementation result in higher serum concentrations of folic acid.e43,e44 There is in-conclusive evidence for an increased risk of adverse events with folic acid supplementation for the PWECP and the child (e.g., increased occurrence of twins, asthma, masking vitamin B12 deficiency, new or worsening of preexisting neoplasia).e40,e45,e46 In a recent analysis of 27,784 children born to people with epilepsy, exposure to periconceptional folic acid greater than 1 mg/d was associated with a 0.9% absolute increase in the risk of childhood cancer before age 20 years, resulting in an HR of 2.7 (95% CI 1.2–6.3).e46 There are potential pharmacokinetic interactions where folic acid can decrease phenytoin serum concentrations.e47 Adherence to folic acid supplementation is generally poor among PWECP, even during pregnancy.e48 ASM polytherapy is associated with decreased folic acid adherence among PWECP.e49 In the United States, where there is no high-dose folic acid formulation, higher doses of folic acid require a large number of tablets, potentially reducing adherence to folic acid supplementation.
Recommendation 6 Statements
6A. Clinicians should prescribe at least 0.4 mg of folic acid supplementation daily preconceptionally and during pregnancy to any PWECP treated with an ASM to decrease the risk of NTDs in the offspring (Level B).
6B. Clinicians must prescribe at least 0.4 mg of folic acid supplementation daily preconceptionally and during pregnancy to any PWECP treated with an ASM to possibly improve neurodevelopmental outcomes such as ASD and global IQ in the offspring (Level A).
6C. Clinicians should counsel PWECP treated with an ASM that adherence to recommended folic acid supplementation preconceptionally and during pregnancy is important to minimize the risk of MCMs and poor neurodevelopmental outcomes (Level B).
Suggestions for Future Research
The findings of this systematic review highlight several knowledge gaps that should be addressed in future research to optimize reproductive outcomes for PWECP. The risks of MCMs and adverse perinatal outcomes for newer and understudied ASMs (e.g., lacosamide, zonisamide, clobazam, and perampanel) require further research. Future guidelines should consider even newer ASMs, such as cenobamate and fenfluramine, which were not included in our search strategy. Longitudinal studies evaluating long-term neuro-developmental outcomes in children with in utero exposure to ASMs other than valproic acid are necessary to inform ASM choice among PWECP, developmental screening requirements, and resource planning. The risk of MCMs, adverse perinatal outcomes, and adverse neurodevelopmental out-comes in polytherapy is a complex picture that merits further clarification. Importantly, an improved understanding of the pathophysiologic mechanisms underlying teratogenic effects of some ASMs will guide rational development of therapeutic strategies. Clarification of factors affecting the pharmacokinetics and pharmacodynamics of ASM metabolism in PWECP during pregnancy and postpartum will inform dosing regimens. Future studies should work to use more uniform definitions for exposures (e.g., high vs low doses of ASMs) and outcomes, as well as which adjustment variables are included in any multivariable analyses, to facilitate the discovery of important findings and their interpretation.
There is considerable practice variation in the dosing of folic acid supplementation. High-quality studies, including randomized controlled trials where possible, will be required to definitively clarify the optimal dose and timing with respect to conception.
The impact of screening for fetal anomalies and growth restriction on perinatal outcomes needs to be established. Clarification of the impact of socioeconomic status on pregnancy outcomes in PWECP will inform social service priorities. To better clarify the potentially diverse needs of underrepresented groups, future studies should work to include diverse ethnic and racial groups, people from low and middle-income countries, as well as transgender, nonbinary, and intersex PWECP. Altogether, these lines of research will help identify pregnancies at greatest risk of adverse outcomes and inform new, targeted interventions to improve parental, fetal, perinatal, and neurodevelopmental outcomes.
Disclaimer
Clinical practice guidelines, practice advisories, systematic reviews, and other guidance published by the American Academy of Neurology (AAN) and its affiliates are assessments of current scientific and clinical information provided as an educational service. The information (1) should not be considered inclusive of all proper treatments or methods of care or as a statement of the standard of care; (2) is not continually updated and may not reflect the most recent evidence (new evidence may emerge between the time information is developed and when it is published or read); (3) addresses only the question(s) specifically identified; (4) does not mandate any particular course of medical care; and (5) is not intended to substitute for the independent professional judgment of the treating provider because the information does not account for individual variation among patients. In all cases, the selected course of action should be considered by the treating provider in the context of treating the individual patient. Use of the information is voluntary. The AAN provides this information on an “as is” basis and makes no warranty, expressed or implied, regarding the information. The AAN specifically disclaims any warranties of merchantability or fitness for a particular use or purpose. AAN assumes no responsibility for any injury or damage to persons or property arising out of or related to any use of this information or for any errors or omissions.
Conflict of Interest
The American Academy of Neurology (AAN) is committed to producing independent, critical, and trustworthy clinical practice guidelines (CPGs) and evidence-based documents. Significant efforts are made to minimize the potential for conflicts of interest to influence the recommendations of this evidence-based document. Management and disclosure of document developer relationships is conducted in compliance with the 2017 AAN process manual section titled, “Implementing the AAN Conflict of Interest Policy for Guidelines and Case Definitions,” which can be viewed at aan.com.
Footnotes
Acknowledgment
Coauthor Sanjeev V. Thomas, MD, died February 4, 2024. The authors are grateful for his contributions to this guideline and to the field of neurology. The authors thank former lead developer Cynthia L. Harden, MD, for drafting the protocol and clinical questions and former AAN staff member Shannon Merillat, MLIS, for her assistance during the guideline development process.
Study Funding
This practice guideline was developed with financial support from the American Academy of Neurology (AAN). Authors who have served as AAN subcommittee members (A.M.P., M.O., S.W.R., D.K.D., J.F., K.S., M.K.), or as methodologists (M.O., D.B.S.), or who are or were AAN staff members (M.D.O., K.B.D., H.M.S.) were reimbursed by the AAN for expenses related to travel to subcommittee meetings where drafts of manuscripts were reviewed.
Disclosure
A.M. Pack serves on the editorial board for the journal Epilepsy Currents, receives royalties from UpToDate, receives funding from the NIH for serving as coinvestigator and site PI for the Maternal Outcomes and Neurodevelopmental Effects of Antiepileptic Drugs (MONEAD) study, and receives funding from Bayer for serving as a co-investigator on a study on women with epilepsy initiating a progestin IUD. An immediate family member of A.M. Pack has received personal ompensation for serving as an employee of REGENEXBIO. M. Oskoui has received personal compensation in the range of $500–$4,999 for serving as an officer or member of the Board of Directors for the Association des Neurologues du Quebec. The institution of M. Oskoui has received research support from Biogen, Roche Genetech, Muscular Dystrophy Canada, and the Canadian Institutes of Health Research. M. Oskoui has received personal compensation in the range of $50,000–$99,999 for serving as an evidence-based medicine methodologist with the AAN. M. Oskoui has a non-compensated relationship as a Member of the Medical and Scientific Advisory Committee with Muscular Dystrophy Canada. S. Williams Roberson receives research funding from the National Institute on Aging for a study related to ICU delirium and associated cognitive decline, serves on the editorial board for Neurology Today, and has a non-compensated relationship as a Physician Advisory Board Member with Epilepsy Foundation of Middle and Western Tennessee. D.K. Donley’s immediate family member has received compensation in the range of $10,000–$49,999 for serving as the vice president of Novello Physicians Organization. J. French’s institution has received research support from the Epilepsy Study Consortium, the Epilepsy Foundation (funded by UCB), GW Pharmaceuticals, the One8 Foundation, FACES (Finding a Cure for Epilepsy and Seizures), the National Institute of Neurological Disorders and Stroke (NINDS), Xenon, and Cerevel. J. French has received personal compensation in the range of $100,000–$499,999 for serving as a Chief Medical and Innovation Officer with the Epilepsy Foundation. J. French has had a non-compensated relationship serving as a consultant or scientific advisory board member for Alterity Therapeutics Limited, Angelini, Arvelle Therapeutics, Autifony Therapeutics Limited, Baergic Bio, Beacon Biosignals, Biogen, Biohaven Pharmaceuticals, Bloom Science Inc., BridgeBio Pharma Inc., Bright minds Biosciences, Camp4 Therapeutics Corporation, Cerebral Therapeutics, Cerecin Inc., Cerevel, Coda Biotherapeutics, Cognizance Biomarkers, Crossject, Eisai, Eliem Therapeutics, Encoded Therapeutics, Engrail, Epalex, Epihunter, Epitel Inc., Equilibre BioPharmaceuticals, Genentech Inc., Grin Therapeutics, GW Pharmaceuticals, iQure Pharma, Janssen Pharmaceuticals, Jazz Pharmaceuticals, Knopp Biosciences, Korro Bio Inc., Leal Therapeutics, Lipocine, LivaNova, Longboard Pharmaceuticals, Lundbeck, Marinus, Mend Neuroscience, Modulight Bio, Neumirna Therapeutics, Neurelis, Neurocrine, Neuroelectrics USA Corporation, NeuroPro Therapeutics, Ono Pharmaceutical Co, Otsuka Pharmaceutical Development, Ovid Therapeutics Inc., Paladin Labs Inc., Pfizer, Praxis, PureTech LTY Inc., Rafa Laboratories Ltd., Rapport Therapeutics, Receptor, Sage Therapeutics, SK Life Science, Stoke, Supernus, Takeda, Third Rock Ventures, UCB Inc., Ventus Therapeutics, Vida Ventures Management, Xenon, and Zogenix. J. French is on the editorial board of Lancet Neurology and Neurology Today. J. French has received travel and/or meal reimbursement related to research, advisory meetings, or presentation of results at scientific meetings from the Epilepsy Study Consortium, the Epilepsy Foundation, Angelini Pharma, Biohaven Pharmaceuticals, Cerebral Therapeutics, Neurelis, Neurocrine, Praxis, Rapport, SK Life Science, Stoke, Takeda, and Xenon. E.E. Gerard has received personal compensation in the range of $500–$4,999 for serving as a consultant for Greenwich Pharmaceuticals. The institution of E.E. Gerard has received research support from NIH/NINDS, Xenon Pharmaceuticals, and Eisai, Inc. (via Stanford University). E.E. Gerard has received travel reimbursements from the American Clinical Neurophysiology Society (ACNS), various institutions for CME lectures, and from the One 8 Foundation. E.E. Gerard’s institution has received compensation from the One 8 Foundation for research coordinator time. The institution of an immediate family member of E.E. Gerard has received research support from NIH and Novo Nordisk. D. Gloss has received personal compensation in the range of $500–$4,999 for serving as a Drug Utilization Review Board Member with the West Virginia Department of Health and Human Resources. W.R. Miller has received research support from Indiana University School of Nursing as part of the Ethel Clarke Fellowship for serving as a PI for Patient Preferences for Delivery of a Web-Based Epilepsy Self-Management Intervention, has received research support from Indiana University Networks Institute for serving as a co-PI (no effort) for the study Sudden Un-expected Death in Epilepsy: Identifying Risk Factors with Social Media Mining, and receives research support from the NIH/NINDS for serving as a core investigator (P30 investigator) for the development of a brain safety lab. H.M. Munger Clary’s institution has received research support from the NIH, the U.S. Department of Defense, Duke Endowment, the Susanne Marcus Collins Foundation, and Eysz, Inc. H.M. Munger Clary has received personal compensation in the range of $500–$4,999 for serving as a speaker with the American Epilepsy Society (AES). H.M. Munger Clary has received personal compensation in the range of $500–$4,999 for serving as a speaker with J. Kiffin Penry Epilepsy Education Programs. H.M. Munger Clary has received personal compensation in the range of $500–$4,999 for serving as a topic editor for DynaMed. H.M. Munger Clary has had non-compensated relationships as Chair of the Psychosocial Comorbidities Committee with the American Epilepsy Society, as Co-Chair of the Integrated Mental Health Care Pathways Task Force with the International League Against Epilepsy, and as Co-Chair Elect of the Resident and Fellow Education Committee with the American Clinical Neurophysiology Society. S.S. Osmund-son’s institution has received research support from NIH. B. McFadden serves as Executive Director for My Epilepsy Story and is uncompensated for this role, serves as an uncompensated member of the ELC Group, serves as an un-compensated patient advisor, serves as an uncompensated Patient-Centered Outcomes Research Institute (PCORI) Ambassador, and receives travel reimbursement from PCORI for attending meetings. K. Parratt has received personal compensation in the range of $500–$4,999 for serving on a speakers bureau for Eisai and for UCB. K.
Parratt receives funding from Zynerba for serving as a sub-investigator for the study Cannabidiol ZYNN2-CL-04 and ZYNN2-CL-04 for artial onset seizures, receives funding from SK Life Science for serving as a subinvestigator for the study Cenobamate YKP3089C021 for partial onset seizures, has received funding from Eisai Inc. for the study Perampanel E2007-G00-335 for partial onset seizures, has received funding from Marinus Pharmaceuticals for the study Ganaxolone 10420603 for partial onset seizures, and has received honoraria from Esai for a dinner meeting lecture. P.B. Pennell’s institution has received research support from the NINDS and the Eunice Kennedy Shriver National Institute of Child Health and Human Development for observational studies of people with epilepsy of childbearing potential and their children. The institution of an immediate family member of P.B. Pennell has received research support from the U.S. Department of Defense, the Environmental Protection Agency, the NIH, and Advanced Energy Consortium. P.B. Pennell has received publishing royalties from UpToDate, a publication relating to health care. P.B. Pennell has received honoraria and/or travel reimbursements from the AES, the AAN, and various academic medical institutions for CME lectures. She has received honoraria for grant reviews from the NIH and Harvard Catalyst. She has received honoraria for serving on the scientific advisory board for BRAINS, an NIH-funded study. G. Saade has received personal compensation in the range of $500–$4,999 for serving as a Consultant for GestVision. G. Saade has received personal compensation in the range of $500–$4,999 for serving on a scientific advisory or data safety monitoring board for CooperSurgical. G. Saade has served on scientific advisory boards for Sage Therapeutics and GestVision. G. Saade has received personal compensation in the range of $5,000–$9,999 for serving as an editor, associate editor, or editorial advisory board member for Thieme Publishing. The institution of G. Saade has received research support from Sera Prognostics, and from NICHD for clinical obstetrics issues. G. Saade has received research support from the NIH for studies related to chronic hypertension and pregnancy, human placenta evaluation, and pregnancy and cardiovascular health. G. Saade has received honoraria for speaking engagements at multiple universities and has given expert testimony, prepared an affidavit, and acted as a witness for legal proceedings regarding preeclampsia. D. Smith has received personal compensation in the range of $10,000–$49,999 for serving as an evidence-based medicine methodologist for the AAN. K. Sullivan has received intellectual property interests from a discovery or technology relating to health care. S.V. Thomas is deceased; to the best of our knowledge, the relevant disclosures are as follows: S.V. Thomas served as a PI of a pregnancy registry in India that has generated clinical data pertaining to the use of anti-epileptic drugs during pregnancy, received honoraria for BMJ Masterclasses on epilepsy, and received research grants from the Indian government. S.V. Thomas received personal compensation in the range of $0–$499 for serving as an editor, associate editor, or editorial advisory board member for Wiley India, and served on the editorial board of the journal Epilepsy Research. T. Tomson’s institution has received personal compensation in the range of $500–$4,999 for serving on a scientific advisory or data safety monitoring board for Angelini and GW Pharmaceuticals. The institution of T. Tomson has received research support from Eisai, GSK, UCB, Bial, Sanofi, Angelini, GW Pharmaceuticals, Teva Pharma, Zentiva, Accord, Ecu Pharm, SF Group, and Glenmark (for serving as a PI in the EURAP study and the International Antiepileptic Drugs and Pregnancy Registry). T. Tomson has received personal compensation in the range of $500–$4,999 for serving as a speaker with Angelini, Sanofi, Eisai, Sun Pharma, and UCB. T. Tomson has received funding from GSK for serving as a PI for a study on sudden unexpected death in epilepsy; has received research funding from Stockholm County Council; and has received research funding from the European Union and Nordforsk. M. Dolan O’Brien was an employee of the AAN. K. Botchway-Doe is an employee of the AAN. H. Silsbee is an employee of the AAN. M.R. Keezer’s institution has received research support from UCB and Eisai. M.R. Keezer serves on the editorial board for the journals Epilepsia and Neurology: Clinical Practice. M.R. Keezer has received a salary award from the Fonds de Recherche du Québec Santé and research grants from the Centre, the Savoy Foundation, the Canadian Frailty Network, the Fonds de Recherche du Québec Santé, TD Bank, and the Canadian Institutes of Health Research. Go to ![]() for full disclosures.
for full disclosures.
Publication History
Received by Neurology October 17, 2023. Accepted in final form February 21, 2024. Submitted and externally peer reviewed. The handling editor was Editor-in-Chief José Merino, MD, MPhil, FAAN.
