Abstract
D’Gama AM, Mulhern S, Sheidley BR, Boodhoo F, Buts S, Chandler NJ, Cobb J, Curtis M, Higginbotham EJ, Holland J, Khan T, Koh J, Liang NSY, McRae L, Nesbitt SE, Oby BT, Paternoster B, Patton A, Rose G, Scotchman E, Valentine R, Wiltrout KN; Gene-STEPS Study Group; IPCHiP Executive Committee; Hayeems RZ, Jain P, Lunke S, Marshall CR, Rockowitz S, Sebire NJ, Stark Z, White SM, Chitty LS, Cross JH, Scheffer IE, Chau V, Costain G, Poduri A, Howell KB, McTague A. Lancet Neurol. 2023;22(9):812-825. doi:10.1016/S1474-4422(23)00246-6
Most neonatal and infantile-onset epilepsies have presumed genetic aetiologies, and early genetic diagnoses have the potential to inform clinical management and improve outcomes. We therefore aimed to determine the feasibility, diagnostic yield, and clinical utility of rapid genome sequencing in this population. We conducted an international, multicentre, cohort study (Gene-STEPS), which is a pilot study of the International Precision Child Health Partnership (IPCHiP). IPCHiP is a consortium of four paediatric centres with tertiary-level subspecialty services in Australia, Canada, the UK, and the USA. We recruited infants with new-onset epilepsy or complex febrile seizures from IPCHiP centres, who were younger than 12 months at seizure onset. We excluded infants with simple febrile seizures, acute provoked seizures, known acquired cause, or known genetic cause. Blood samples were collected from probands and available biological parents. Clinical data were collected from medical records, treating clinicians, and parents. Trio genome sequencing was done when both parents were available, and duo or singleton genome sequencing was done when one or neither parent was available. Site-specific protocols were used for DNA extraction and library preparation. Rapid genome sequencing and analysis was done at clinically accredited laboratories, and results were returned to families. We analysed summary statistics for cohort demographic and clinical characteristics and the timing, diagnostic yield, and clinical impact of rapid genome sequencing. Between Sept 1, 2021, and Aug 31, 2022, we enrolled 100 infants with new-onset epilepsy, of whom 41 (41%) were girls and 59 (59%) were boys. Median age of seizure onset was 128 days (IQR 46-192). For 43 (43% [binomial distribution 95% CI 33-53]) of 100 infants, we identified genetic diagnoses, with a median time from seizure onset to rapid genome sequencing result of 37 days (IQR 25-59). Genetic diagnosis was associated with neonatal seizure onset versus infantile seizure onset (14 [74%] of 19 vs 29 [36%] of 81; p = 0.0027), referral setting (12 [71%] of 17 for intensive care, 19 [44%] of 43 non-intensive care inpatient, and 12 [28%] of 40 outpatient; p = 0.0178), and epilepsy syndrome (13 [87%] of 15 for self-limited epilepsies, 18 [35%] of 51 for developmental and epileptic encephalopathies, 12 [35%] of 34 for other syndromes; p = 0.001). Rapid genome sequencing revealed genetic heterogeneity, with 34 unique genes or genomic regions implicated. Genetic diagnoses had immediate clinical utility, informing treatment (24 [56%] of 43), additional evaluation (28 [65%]), prognosis (37 [86%]), and recurrence risk counselling (all cases). Our findings support the feasibility of implementation of rapid genome sequencing in the clinical care of infants with new-onset epilepsy. Longitudinal follow-up is needed to further assess the role of rapid genetic diagnosis in improving clinical, quality-of-life, and economic outcomes.Background:
Methods:
Findings:
Interpretation:
Commentary
The recent article about feasibility, diagnostic yield, and clinical utility of rapid whole genome sequencing (rWGS) in pediatric epilepsy is sure to garner varied degrees of interest across different tiers of readership in the world of epilepsy. D’Gama and coauthors from 4 premier institutions have published their findings on the feasibility of doing
How do findings from this study affect the clinical practice of pediatric epilepsy? They don’t—not today—but they open doors for the future!
Diagnostic Yield
There are various ways in which a person might investigate the cause of infantile epilepsy. The traditional, stepwise, sequential, investigative method includes a thorough history and clinical examination followed by neuroimaging, cerebrospinal fluid analysis, metabolic, and then genetic testing. The traditional diagnostic method for sequential genetic tests might include an initial karyotype (to look at all the chromosomes) followed by chromosomal microarray (CMA-looking for deletions and duplications) followed by individual gene testing if there is a high degree of clinical suspicion or a gene panel with variable number of genes according to the laboratory used (if unsure about a particular gene based on patient’s phenotype), or, whole exome sequencing (WES) to check all the coding exons—using singleton (patient); duo- (patient and one parent) [or sibling]; trio- (patient and both parents) [or patient, one parent, and sibling with similar phenotype] or as a final measure—doing a WGS where available (both coding and non-coding regions are investigated). A systematic analysis suggests increasing returns on diagnosis as we climb up the tree of sequential genetic tests from 9% for CMA, 24% for WES, and 48% for WGS. 2 Papers contributing to this meta-analysis however had variable criteria for inclusion/exclusion.
Currently, most children in the United States with epilepsy under the age of 8 years can avail of an epilepsy gene panel through a sponsored program called “Behind the Seizure” (Sponsored genetic testing | Behind the Seizure | Invitae). Trio WES is ordered when gene panels don’t provide answers. While most practitioners including myself face insurance, organizational, or institutional barriers in providing clinical WES for their patients, the American Epilepsy Society endorses genetic testing with either multigene panel (>25 genes), WES or WGS as first line of investigation in undiagnosed epilepsies irrespective of age of the patient. 3
Feasibility
Authors belong to an international consortium created to advance precision medicine called the International Precision Child Health Partnership (iPCHP) (authors have reported 15 funding sources) across 4 hospital systems in the United Kingdom, Canada, United States, and Australia. This study called the Gene Shortening Time of Evaluation in Pediatric Epilepsy Services (Gene-STEPS) is their pilot study. Between September 2021 and August 2022, authors screened 147 patients and of those were able to enroll 100 patients for this study. The results are certainly aspirational for any other health care system capable of delivering along these timelines. Authors show us that such rWGS is feasible across several health care institutions and across tiers of referral bases whether inpatient ICU, inpatient non-ICU, or outpatient clinic settings.
Clinical Utility
Is identifying cause of epilepsy enough? Diagnostic utility can be captured as clinical utility (clinician's view point) as well as personal utility (a patient's/parents’ view point). 4 D’Gama et al in the featured study did not stop at genetic diagnosis as the measure of diagnostic utility but also measured influence on treatment, potential for precision therapy, additional investigations indicated or avoided, additional prognostic information, influence on goals of care or influence on genetic counseling (Table 1). Redirection of care was noted in 2 patients in this cohort but is likely to fluctuate across different geographic and cultural landscapes with differing/ variable access to care.
Overall Utility of This Study: Diagnostic Utility Plus Treatment Implications.
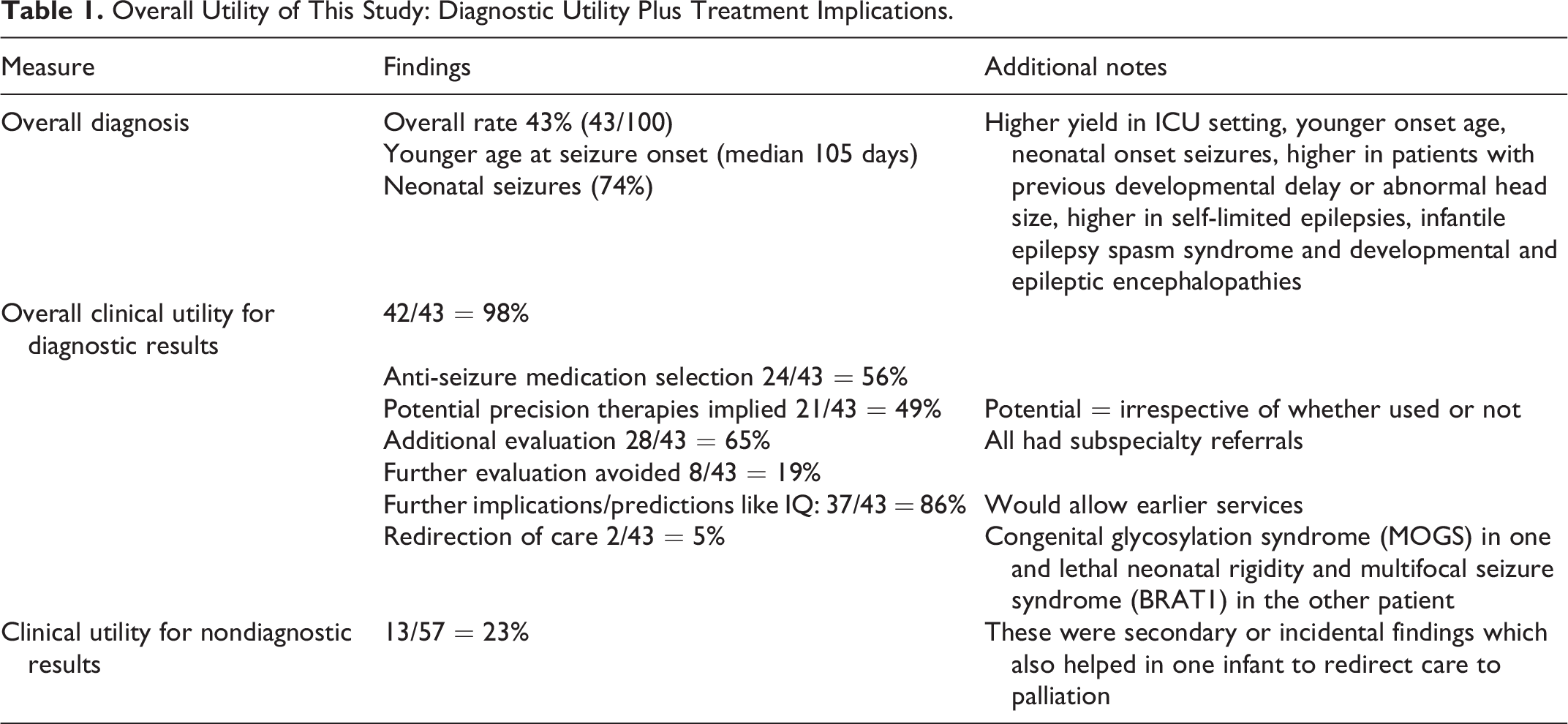
Authors have created a set of parameters that can be used by others in the future when assessing utility. This type of standardization of clinical outcomes empowers future studies and the collective information in turn helps advocacy and arguments toward insurance/pay or reimbursements for testing that is not currently available.
Economic Impacts
Economic impacts of a non-commercially available test must be considered and currently the literature on rWGS is limited. In a recent study from Nicklaus Childrens Hospital that enrolled 65 critically ill pediatric patients with unknown cause; using Incremental Cost Effectiveness Ratio of rWGS, authors showed mean savings of ∼ US$100 000 per patient and a collective ∼US$6 M savings across the 65 patients. 5 While D’Gama et al do not address any economic impacts due to the short duration of follow-up in their study (September-December 2022); future prospective follow-up of this cohort of patients will be more informative.
Access, Standardization, and Implementation in Pediatric Epilepsy
Main barrier to WGS implementation is the cost of the test. Even if a patient were willing to pay out of pocket; without an infrastructure that includes presence of collaborative practitioners (who see the patient at presentation), neurologists (who recognize need for testing and will follow the patient), geneticists (who can rule out other overt genetic causes of seizures), genetic counsellors (who can perform adequate pretest and posttest counselling), and processes that ensure seamless transition from blood draw to laboratory transfer, resulting, posttest counseling, and follow-up; rWGS is bound to fail. Authors have acknowledged challenges to the implementation of this study despite having access to the best possible clinical and research conditions. However, all is not doom and gloom. Using implementation science principles, California and Michigan have successfully launched rWGS in their ICUs. 6,7
Conclusions
Authors have set the stage for future research with standardized protocols and uniform outcome measurement. They remain cautious in their conclusions about needing long-term follow-up in determining clinical, quality of life, and economic outcomes.
Therefore, outside of a research setting, rWGS remains a pipe dream for my patients in the outpatient clinic, AND…A LOT of work is still required for further implementation that is equitable and accessible across geographic and cultural divides.
