Abstract
The ability to develop effective new treatments for epilepsy may depend on improved understanding of seizure pathophysiology, about which many questions remain. Dynamic fluorescence imaging of activity at single-neuron resolution with fluorescent indicators in experimental model systems in vivo has revolutionized basic neuroscience and has the potential to do so for epilepsy research as well. Here, we review salient issues as they pertain to experimental imaging in basic epilepsy research, including commonly used imaging technologies, data processing and analysis, interpretation of results, and selected examples of how imaging-based approaches have revealed new insight into mechanisms of seizures and epilepsy.
Introduction
Epilepsy is a common and potentially devastating neurological illness that affects approximately 4% of all people at some point in their lives and is associated with significant morbidity and mortality. 1 There is a critical need for new and better treatments, achievement of which may require a more thorough understanding of seizure pathophysiology. Yet, many fundamental questions in the field remain unanswered: How does a seizure start? What determines the speed and spatial extent of seizure propagation? What are the specific cellular and circuit elements involved in the generation, propagation, and termination of seizures?
Many features of seizures are likely universal 2,3 across species, meaning that experimental systems such as mouse, zebrafish, and others can serve as useful models of the human disease. Seizures can be produced in model systems using lesional, pharmacologic and genetic approaches. 4 Ongoing work benefits from communities of active researchers and available reagents that enable sophisticated cell-type specific recording (with fluorescent indicators) and manipulation (opto- and chemogenetic agents) to monitor and interrogate epilepsy-related cells and circuits, as well as open-source tools for data processing and analysis. 5 -7
Dynamic fluorescence imaging has been increasingly used to investigate mechanisms of seizures and epilepsy in animal models. These approaches facilitate the interrogation of large neuronal populations (up to ∼one million neurons simultaneously) in living organisms at relatively high spatial and temporal resolution deep in the intact brain. 8 The ability to monitor cellular activity at this scale may prove critical for understanding network-level phenomena. However, application of these techniques to study seizures and epilepsy requires special considerations, which we discuss here.
We review a selected set of issues in dynamic fluorescence imaging as related to experimental epilepsy. We provide a general overview of available imaging technologies, with a necessary focus on two-photon (2P) calcium imaging in experimental mouse models. We also highlight recent work that has advanced our understanding of the pathophysiology of seizures and epilepsy, including new experimental and analytic approaches. Our goal is to encourage the broader implementation of imaging approaches in the field of basic epilepsy research.
Dynamic Fluorescence Imaging Approaches
In this section, we review and highlight recent innovations in imaging approaches (Table 1), activity-dependent indicators, and use of model systems deemed of greatest relevance to epilepsy research.
Comparison of selected microscopy approaches for imaging of seizures.
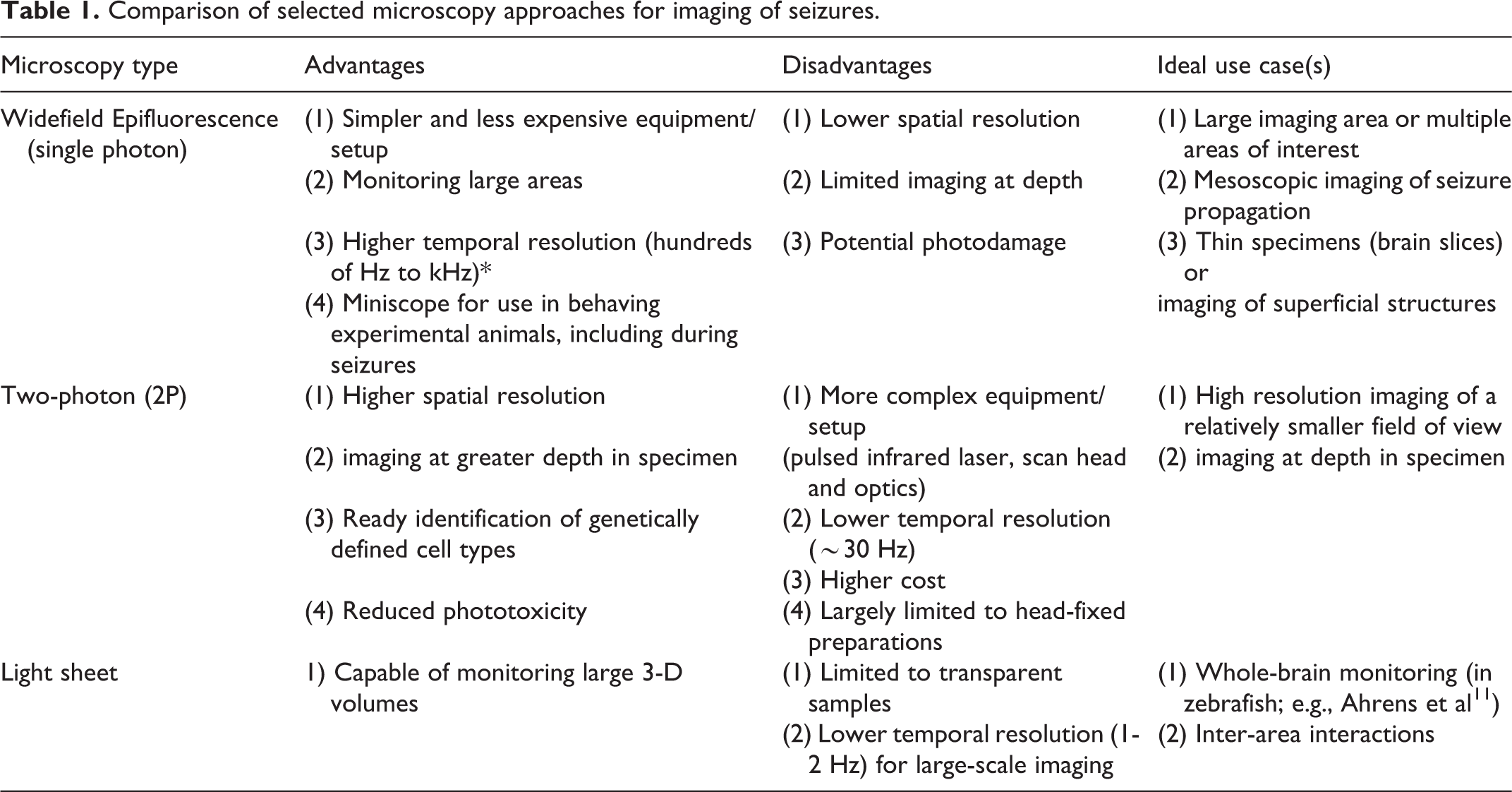
Epifluorescence Microscopy
In single-photon (1P) epifluorescence widefield microscopy, the entire specimen (or brain) is illuminated simultaneously by a stationary light source. This strategy employs excitation of a fluorescent activity-dependent indicator such as a genetically-encoded calcium indicator. Widefield microscopy can be performed in zebrafish and rodents in vivo. In mice, this is performed by exposing and imaging through the intact skull, or by removing and replacing the skull with a transparent, solid material, such as a molded glass coverslip.
Many neurons in the mouse brain can be monitored simultaneously – up to 50,000 in one study that used a “crystal skull” preparation. 9 Temporal resolution is also excellent, with frame rates of up to hundreds of Hz, as determined by sensitivity of the camera and the dimensions of the region of interest selected for imaging. As the main advantage of widefield 1P imaging is the ability to monitor large fields of view, this approach may be an appropriate choice to study seizure spread/generalization across brain areas or functional connectivity in the epileptic brain, or to attempt to image initiation in a model characterized by seizures with neocortical onset. The equipment required for illumination and detection in epifluorescence microscopy can be miniaturized and rendered portable and mounted on a freely-behaving animal as in the “Miniscope,” 10 the general application of which has been reviewed extensively elsewhere.
The major drawbacks of widefield 1P microscopy are background noise/contamination and the inability to image at depth in the brain due to light scattering. Activity cannot be reliably attributed to individual cells as fluorescence emission is also collected from cells outside of the focal plane.
Multi-Photon Microscopy
In the case of 2P (and 3P) microscopy, longer (infrared) illumination wavelengths are used such that two (or three) photons are required to excite the fluorescent indicator. This highly reduces the probability of excitation outside a thin axial plane of focus, which restricts excitation to a smaller tissue volume within the specimen and limits phototoxicity outside the imaging plane. Furthermore, the longer wavelengths used in 2P excitation are subject to less scattering by brain tissue, allowing for imaging at greater depth in the sample. Because two photons must be absorbed near-simultaneously, illumination requires high-powered pulsed infrared lasers. Illumination is applied by serially scanning small regions of the specimen; therefore, temporal resolution for standard 2P microscopy is typically in the range of 1-30 Hz when imaging hundreds or thousands of cells simultaneously. 12,13 This is lower than what can be achieved for widefield imaging using a CMOS camera; however, in practice, other factors may be more limiting of temporal resolution, such as the decay kinetics of available activity-dependent indicators. Due to the technical requirements of laser scanning, a 2P setup is less portable compared to a head-mounted epifluorescence Miniscope (although miniaturized two- and three-photon microscopes are under development 14 -16 ). In addition, the imaging area is typically limited to a 1 X 1 mm area (but see Table 2) and a depth of approximately 1 mm (without use of a GRIN lens).
Innovations in multi-photon imaging of neuronal activity in vivo.
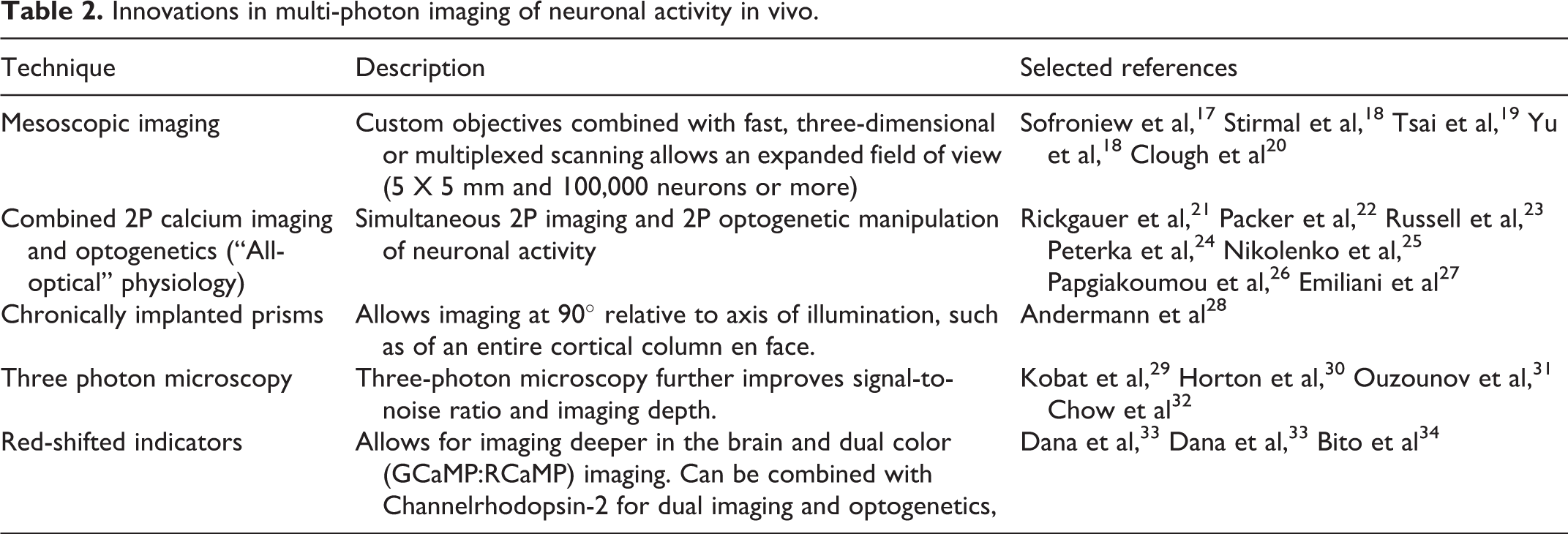
2P microscopy is a strong option for seizure studies requiring dynamic cellular-level imaging of neurons at depth in the intact brain in vivo, and many recent innovations have extended the capabilities of this approach for the functional imaging of neural circuits in experimental model systems (summarized in Table 2).
Light Sheet Microscopy
With light sheet microscopy, illumination is provided in a thin plane that is scanned along a single axis. This approach has been used in studies of the nervous system of the transparent larval zebrafish, 11 allowing imaging of up to 80% of all neurons. Temporal resolution at this impressive scale remains relatively slow 11,35 ; the entire volume of the zebrafish has been imaged at a rate of 1-2 Hz. This may preclude study of more fine-grained temporal dynamics yet could remain an appropriate option for investigators interested in imaging initiation and propagation of seizure-like activity across regions of the zebrafish nervous system
Activity-Dependent Indicators
Calcium-Sensitive Indicators
Calcium-sensitive indicators are currently the most popular and flexible indictors for monitoring neuronal activity. The major driver of somatic fluorescence transients is calcium influx attributed to activation of voltage-gated calcium channels during action potentials. However, these indicators exhibit slow kinetics relative to the action potential and hence provide an indirect, low-pass filtered readout of spiking. The specific response characteristics of a given calcium-sensitive indicator (sensitivity; kinetics; baseline fluorescence; saturation; etc.) depend on the chemical properties of the indicator itself as well as the cell type-specific physiology of intracellular calcium.
The GCaMP series of indicators are the most widely used family of calcium-sensitive indicators and are now in their eighth generation of development. 36,37 They vary in kinetics (rise time, decay time) and sensitivity (baseline fluorescence; signal-to-noise ratio, dynamic range), which leads to variation in the ability to report single action potentials. These indicators can be delivered by viral vector or expressed genetically in a constitutive or conditional manner (allowing cell type-specific expression) and can be targeted to subcellular compartments such as the soma or axon. GCaMP-expressing flies, mice, marmosets, 38 and other organisms, have also been generated.
There are important seizure/epilepsy-specific considerations when selecting a calcium-sensitive indicator. Decay is slow compared to the action potential time course, and indicators may not operate within a linear range across relevant firing rates. Therefore, it is not possible to extract an exact number of action potentials from the ΔF/F0 trace, particularly with high firing rates seen during seizures. During depolarization block, 39,40 calcium levels, and thus fluorescence, might remain high even though the neuron is not actively spiking.
Voltage-Sensitive Indicators
Voltage-sensitive indicators – reviewed in depth elsewhere 13 – are highly promising for future epilepsy research yet remain under development. Voltage indicators can report sub-threshold voltage changes, such as synaptic potentials and subthreshold oscillations, which are not detected by calcium imaging. 41 As such, this approach could potentially be used to disambiguate depolarization block from high-frequency bursting.
Given the time course of voltage fluctuations in the cell, voltage imaging requires ultra-fast cameras typically combined with high-power epifluorescence illumination sources. Furthermore, indicators are often subject to bleaching. Hence, most experiments are limited to relatively small populations of neurons over short recording periods. 41 -45 A recent study, 46 however, was able to image voltage across more than one hundred neurons. Rapid advances in chemical and genetically encoded indicators will facilitate improvements in large-scale voltage imaging and 2P voltage imaging in the intact brain in vivo.
Other Indicators
Electrical activity has been a desired output measure in epilepsy research. However, other factors clearly contribute to the pathophysiology of seizures and epilepsy, and can also be monitored via imaging approaches in the experimental setting. Fluorescent indicators have been developed for glutamate, 47,48 GABA, 49 neuromodulators (such as dopamine, 50,51 serotonin, 52 and endocannibinoids 53 ), and for indicators of bioenergetics such as ATP-sensitive indicators. 54,55
Model Organisms
Mouse
The similarities of mouse brain to that of the human combined with genetic and experimental tractability make the mouse model an excellent system for the study of seizures and epilepsy. However, while 1000-fold smaller than the human brain, the mouse brain is sufficiently large that imaging across the entire brain cannot yet be performed.
Acute focal seizures can be readily elicited and imaged in the mouse with local application of ictogenic compounds such as 4-aminopyridine 56,57 (4-AP) or GABAA antagonists, 58 facilitating study of propagation speed and spatial extent and architecture of seizure spread. This approach has the advantage of enabling acute provocation of seizures, which are otherwise unpredictable events. In addition, the site of seizure onset is known (presumably the site of pro-convulsant compound administration), and can be generated in a brain area that is easily accessible to imaging (such as superficial neocortex). Alternatively, spontaneous seizures can be imaged in mouse models of chronic epilepsy, such as chronic acquired temporal lobe epilepsy (TLE) after brain injury. However, if seizure frequency is low, spontaneous seizures may be difficult to capture during an imaging session.
Zebrafish
Zebrafish has also become an important model organism in dynamic fluorescence imaging of experimental epilepsy. Zebrafish have approximately 100,000 neurons, with a telencephalon (analogous to the cerebral cortex) comprised of excitatory, inhibitory, and neuromodulatory cell types. 59 The major benefit of larval zebrafish is experimental tractability. Due to its translucency and small size, the entire nervous system can be imaged simultaneously 60 (see above for light-sheet microscopy). This benefit may be critical for understanding seizure spread and the functional connectivity between brain areas that are difficult to study in the mouse. Seizure-like episodes can be induced that resemble human seizures behaviorally and electrographically, and in their response to anti-seizure medications. 59,61,62
For example, the GABAA antagonist pentylenetetrazole (PTZ) is a commonly employed model of acute seizure induction in zebrafish. 63 -65 Genetic epilepsy syndromes can also be modeled in zebrafish using a variety of approaches and such models have been successfully used to screen for anti-seizure medications that reduce the frequency of spontaneous or PTZ-induced seizure-like events. 62,66,67,68,69
Insights Into Seizure Mechanisms Revealed by Imaging
In this section, we review specific imaging studies that have contributed to our understanding of seizure mechanisms.
Seizure Onset Zone Versus Distant Areas of Spread
Basic mechanisms of seizure propagation remain unknown, but dynamic large-scale imaging studies have contributed to our understanding of this process by facilitating the high-speed monitoring of large areas of brain tissue at cellular resolution in experimental model systems in vivo.
Early studies used dynamic fluorescence imaging of neurons in acute slices of mouse brain to investigate spread of seizure-like events. Such studies highlighted the role of the “inhibitory surround” provided by interneurons (INs). 41,70 More recent studies have extended the use of fluorescence imaging in vivo. For example, Liou et al 71 induced seizures pharmacologically with focal injection of 4-AP in anesthetized mice. Using both widefield 1P and 2P imaging, they showed that the zone of seizure spread was limited to within 2-3 mm of the 4-AP injection site. Application of bicuculline led to spread of seizure activity beyond this 2-3 mm zone, which could be tracked with imaging. Application of 4-AP to one site and bicuculline to a distant site led to seizure spread from the onset zone (at the site of 4-AP injection) as well as to the site of bicuculline injection, but not to the region between the two zones. This study, enabled by monitoring of distant sites, suggests that seizure spread need not occur along contiguous areas of cortex.
Wenzel et al 57 used 2P calcium imaging (in conjunction with electrical recording) to monitor 4-AP-induced focal seizure-like events either at the initiation site or at the site of spread. They found that neurons were recruited continuously at the site of distant seizure spread, but that neurons at the site of initiation were recruited stepwise in a “saltatory” fashion. In another study by the same group, 58 neurons were recruited into the seizure “elastically” – the sequence of neuronal recruitment was reliable, but the absolute timing varied from one seizure to another. Furthermore, on average, the activity of labeled parvalbumin (PV)-INs increased leading up to seizures. While there was considerable heterogeneity in the responses of individual neurons: some were recruited early in the seizure, some were recruited late in the seizure, and some did not participate at all. Overall, these studies demonstrate fascinating details of seizure initiation and spread that would have been difficult to reveal with electrical recording alone, capturing active and silent neurons and definitively identifying subsets of seizure-related cells such as PV-INs.
Cell Type-Specific Dysfunction
Seizures may be associated with elevated and/or hypersynchronous neuronal firing, which can be due to increased excitability of principal cells, impaired activity of inhibitory cells, or both. Imaging experiments have made important contributions towards answering questions related to cell type-specific recruitment in the preictal and ictal period. The identification of genetically or functionally defined cell types that can be integrated with dynamic fluorescence imaging approaches highlights cell type-specific roles in seizure pathophysiology.
Tran et al 12 used 2P calcium imaging to record naturalistic seizures in a mouse model of Dravet syndrome (Scn1a+/- mice), taking advantage of the fact that such mice exhibit seizures in response to elevation of core body temperature (as in human patients) which can be experimentally controlled. This study recorded from putative excitatory neurons, while specifically labeling PV-INs. Prior to temperature-sensitive seizures, there was decreased PV-IN:PV-IN synchronization, despite higher mean neocortical PV-IN activity in Scn1a+/- relative to wildtype mice during quiet wakefulness. In the same model system, Somarowthu et al 72 examined three major types of GABAergic interneurons, and found that PV, somatostatin (SST), and vasointestinal peptide (VIP)-INs were recruited at different phases of the pre/peri-ictal period.
In a recent study, Mattis et al 73 leveraged the same mouse model of Dravet syndrome to investigate the role of the dentate gyrus (DG) in filtering excitatory input to the hippocampus. In acute brain slices, the authors stimulated the perforant path and used 2P imaging to record DG granule cells in Scn1a+/- and wildtype animals during the active phase (juvenile mice) and chronic phase of the disorder (young adult mice). Recruitment of granule cells was markedly increased in Scn1a+/- mice relative to wildtype in young adult mice but was essentially normal in juvenile mice. This approach facilitated the large-scale recording of hundreds of DG granule cells in the cortico-hippocampal circuit simultaneously and revealed enhanced excitatory input to the DG in Scn1a+/- mice relative to wildtype during the chronic phase of the disorder, while excitability of PV-INs and excitatory input onto PV-INs was near-normal.
Interictal Discharges
Interictal discharges (IIDs) are brief, large-scale electrical discharges that occur between seizures. The significance of these events and pathophysiological link to seizures remains incompletely understood, but such events in experimental animal models are amenable to study using dynamic fluorescence imaging.
Muldoon et al 74 performed 2P calcium imaging in dentate gyrus in acute hippocampal brain slices prepared from mice with pilocarpine-induced chronic temporal lobe epilepsy (TLE). The authors found that, across repetitive interictal discharges, the active cluster of cells was highly variable. A follow-up study 75 using 2P imaging in hippocampal area CA1 in vivo found that interictal discharges were correlated with synchronous activity of INs.
Aeed et al 56 combined 2P imaging with the use of implanted microprisms (which allow for imaging at 90° relative to the illumination source and hence across layers of the neocortex). To investigate how interictal discharges and seizures propagate across cortical layers, they applied 4-AP to the pial surface. Both interictal discharges and seizures quickly recruited excitatory cells and PV-INs in layer 2/3. However, seizures differed from IIDs due to imbalanced activity of excitatory cells and PV-INs. Seizures, but not IIDs, then gradually spread to layer 5, facilitating subsequent propagation to other brain areas.
The studies highlighted above leverage the capabilities of dynamic fluorescence imaging to determine cell-type specific activity patterns and presumptive roles in IIDs, and how IIDs may relate to seizures.
Information Encoding
Developmental delay and intellectual disability are common comorbidities of epilepsy, suggesting overlapping pathophysiological mechanisms. Shuman et al 76 investigated information encoding by studying hippocampal place cells using miniscope imaging combined with silicon probe recording in mice with pilocarpine-induced TLE. This arrangement facilitated the recording of hippocampal place cells in awake behaving experimental animals. Place cell representations were disrupted six weeks after pilocarpine injection (and weeks after seizure onset), which the authors propose is due to altered timing/synchronization secondary to epilepsy-related hippocampal circuit dysfunction.
Imaging of Non-Neuronal Cells
Glia clearly play important roles in the pathogenesis of seizures and epilepsy and are also amenable to study using 2P calcium imaging. 77 Diaz Verdugo et al 78 studied glial contributions to PTZ- and pilocarpine-induced seizures in zebrafish. The authors observed increases in positive correlations between neurons in the pre-ictal period that was difficult to explain by synaptic transmission alone. Large-scale 2P imaging revealed rapid-onset, large-scale synchronization between glial cells which was attributed to electrical coupling via gap junctions. Furthermore, during the pre-ictal period, glial networks had the general effect of dampening neuronal activity, and seizures occurred following erosion of this protective seizure-suppressing effect.
Data Processing
A critical step in imaging studies is data processing. Vast amounts of data can be generated from hundreds to tens of thousands of neurons across many hours of recording. Optical signals in general cannot be directly translated into the primary output measure interest. Software pipelines must be constructed that account for the data collection process, correct for motion artifacts, identify individual neurons, and subtract noise where possible; ideally in a semi-automated or automated fashion. The most sophisticated tools (for example, Suite2p 5 and CaImAn 6 ) have been developed for calcium imaging, as this is by far the most common type of fluorescent imaging performed in neuroscience. These tools, however, were not developed for the study of seizures. Seizure-specific factors such as high firing rates, high levels of synchrony, depolarization block, and high neuropil signal contamination make data processing further challenging. These factors could be addressed by future work.
Modeling Perspectives
Here we briefly highlight current modeling approaches to dynamic fluorescence imaging of large neuronal populations in studies of the mechanisms of seizures and epilepsy.
One approach attempts to identify functional connectivity motifs that might be involved in seizure pathophysiology. Using PTZ-induced seizures in zebrafish (and validated in mouse), Hadjiabadi et al 79 identified “superhub” neurons in epileptic animals that may predispose to seizure. These neurons were not just highly connected but were embedded within feedforward connectivity patterns that could amplify neuronal activity. Such theories could be further tested using optogenetic manipulation or other targeted cell type-specific perturbations.
Insight into neuronal network function and dysfunction requires consideration of mechanisms at multiple scales and levels of abstraction. Rosch et al 64 built such a hierarchical model of the zebrafish nervous system, and fit the model to data recorded during light-sheet imaging of PTZ-induced seizures to suggest key changes in synaptic strength that may underlie features of the observed events.
Discussion and Future Directions
Dynamic fluorescence imaging of neuronal populations in experimental systems has enabled a new scale of neuroscientific research – the mesoscale – which will be critical for future study of epilepsy mechanisms. Key phenomena such as seizures and interictal discharges operate on the level of neuronal circuits constructed by diverse cell types and the connections between them, with contributions of glia, neuromodulation, and other elements. Here, we reviewed important aspects of commonly used imaging technologies including methods of illumination, indicators of neuronal activity, and model systems, as well as insights gained from previous studies of epilepsy mechanisms using dynamic imaging of neural activity. Finally, we provided an overview of modeling strategies to leverage this imaging data towards an improved understanding of the pathophysiology of seizures and epilepsy. Future technical developments including in imaging hardware, indicators, and data analysis, will enable larger-scale imaging at greater depth in the brain and at higher speeds, with enhanced capabilities for fine-grained monitoring and manipulation of neurons and circuits in awake, behaving experimental models. It is absolutely critical that such advances are leveraged towards a greater understanding of seizures and epilepsy.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Institute of Neurological Disorders and Stroke (R01 NS110869, R25 NS065745), Dana Foundation (David Mahoney Neuroimaging Award).
