Abstract
Trujillo CA, Gao R, Negraes PD, et al. Cell Stem Cell. 2019;25(4):558-569.e7. doi:10.1016/j.stem.2019.08.002. Structural and transcriptional changes during early brain maturation follow fixed developmental programs defined by genetics. However, whether this is true for functional network activity remains unknown, primarily due to experimental inaccessibility of the initial stages of the living human brain. Here, we developed human cortical organoids that dynamically change cellular populations during maturation and exhibited consistent increases in electrical activity over the span of several months. The spontaneous network formation displayed periodic and regular oscillatory events that were dependent on glutamatergic and GABAergic signaling. The oscillatory activity transitioned to more spatiotemporally irregular patterns, and synchronous network events resembled features similar to those observed in preterm human electroencephalography. These results show that the development of structured network activity in a human neocortex model may follow stable genetic programming. Our approach provides opportunities for investigating and manipulating the role of network activity in the developing human cortex.
Commentary
The brain is such an incredibly complex organ that any attempt at emulation seems utopian. From the diabolical Donovan’s brain to the Turing machine, the imitation game stays unsolved. It always lacks the soul. Our soul has the form of a beat, a river babbling; a cacophony of spikes nested in the electrical waves of the brain rhythms. A silent brain is nothing.
Attempts of growing brain tissue started long ago. 1 In less than a dozen years, research with stem cell–derived brain organoids has faced vertiginous advances in understanding their cellular diversity and development, 2,3 yet the question remains of whether they are able to self-organize functionally. Now, a recent paper in Cell Stem Cell by Trujillo et al 4 found the beat. Oscillatory network activity perhaps similar to preterm neonatal brainwaves can emerge from brain organoids cultured over 10 months.
Using single-cell RNAseq, immunohistochemistry and mass spectrometry, Trujillo et al described the growth and maturation of organoids with cortical specification. They showed the development from an initial condition with the dominance of progenitor cells (expressing Ki-67, SOX2, and Nestin) and intermediate progenitors (expressing TBR2) to a final stage with a proportion of glial cells, glutamatergic neurons, and GABAergic interneurons. Interestingly, neither the presence of GABAergic markers (GAD65, DLX1, and DLX5) nor the GABA neurotransmitter were found in organoids younger than 6 months although there were already glutamatergic neurons expressing subunits of GABAergic receptors. Another recent report with cortical organoids confirmed the appearance of inhibitory interneuron precursors and immature inhibitory interneurons in 6-month-old but not in 3-month-old organoids. 5 In the human brain, it has been described that cortical interneurons expressing DLX2, a transcription factor essential for their differentiation and migration, are present after 10 weeks of conception. 6 Possibly, brain organoids incorporating ganglionic eminence interneuron progenitors in addition to excitatory cell lineages will likely pattern similar to brains.
To address the functional characterization of their cortical organoids, Trujillo et al first performed patch-clamp experiments. Individual neurons showed normal resting potential, fired sodium-dependent action potentials, and presented excitatory postsynaptic potentials that were blocked with N-methyl-
In the real brain, cross-frequency coupling between neuronal oscillations such as θ and γ have been described both in rodents and humans. 9,10 In organoids, Trujillo et al observed that the application of the sodium channel blocker Tetrodotoxin abolished electrical activity and that glutamatergic receptor antagonists and GABAergic agonists reduced the frequency of both spikes and LFP events. Interestingly, the blockade of the GABAergic transmission abolished nested oscillatory activity but not spiking, suggesting that inhibition is critical for pacing the beat in this preparation.
In their report, Trujillo et al compared their results with a public electroencephalogram (EEG) data set from preterm infants. 11 Because the biophysical properties of the scalp EEG are missing in organoids, they rely on basic EEG features such as the frequency of oscillations and duration of events. They found similar developmental trajectories. Using a regularized regression and cross-validated model of brainwaves, they were able to predict the organoid developmental time after 25 weeks of plating. Before that, the model performed suboptimal, yet it captured some trend.
Cerebral organoids are no more than layers of cortex around some choroid plexus. Although they recapitulate basic developmental programs, many critical biological processes are absent. For instance, GABA plays major depolarizing roles early in development, 12 a circumstance that perhaps is missing in cortical organoids. Similarly, appropriate interhemispheric connectivity, which is essential for normal EEG patterns, follows independent developmental programs 13 and requires activity-dependent tuning. 14 In real brains, sensory inputs and feedback from fetal movements contribute to shape brain-wide oscillations and the formation and refinement of cortical connectivity. 15 Every brain needs a body.
The paper by Trujillo et al is a potential game changer. The use of organoids as models for neuropsychiatric and developmental diseases opens new opportunities to evaluate the underlying physiopathology and to assay new therapeutic strategies. They can be combined with organotypic cultures of cortical surgical samples given recent advances in protocols allowing for optimized transduction of transgenes and viral vector strategies. 16 –18 Together, these 2 in vitro tools may transform the study of the human brain ex vivo in a more personalized way (Figure 1). Oscillations not only represent the basic functional mode of the brain but they are essential for a proper construction. By focusing in oscillatory patterns as organizational principles of developmental physiological programs, we can take a big leap in our understanding of brain function and dysfunction. 19
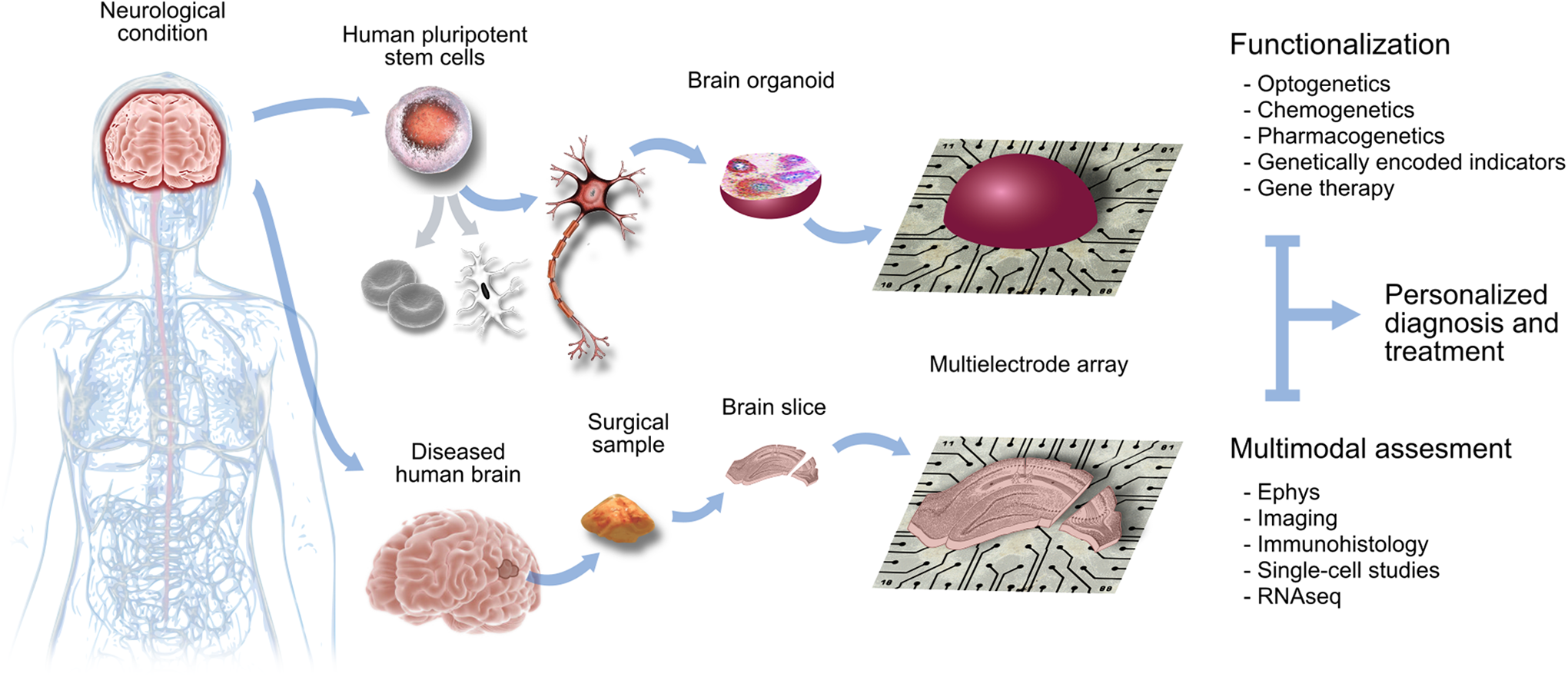
Organoid and organotypic preparations of the human brain are revolutionizing biomedical research. Stem cell–derived organoids plated over multielectrode arrays allow studying physiopathological aspects of many neurological conditions. Whenever possible, they can be combined with organotypic cultures of human brain surgical samples, such as in cases of epilepsy or brain tumors. Both preparations can be functionalized with novel cell-type-specific opto- and chemogenetic approaches to test the physiological impact of novel genetic and pharmacological therapeutic approaches in a more personalized way. Illustrated by Andrea Navas-Olive.
