Abstract
Acker DWM, Wong I, Kang M, Paradis S. Epilepsia. 2018;59(6):1257-1268 We previously discovered a role for the extracellular domain of the transmembrane protein semaphorin 4D (Sema4D) as a fast-acting, selective, and positive regulator of functional γ-aminobutyric acid (GABA)ergic synapse formation in hippocampal neuronal culture. We also demonstrated that Sema4D treatment increases inhibitory tone and suppresses hyperexcitability in an organotypic hippocampal slice culture model of epilepsy. Here, we investigate the ability of Sema4D to promote GABAergic synapse formation and suppress seizure activity in vivo in adult mice. We performed a 3-hour, intrahippocampal infusion of Sema4D or control protein into the CA1 region of adult mice. To quantify GABAergic presynaptic bouton density, we performed immunohistochemistry on hippocampal tissue sections isolated from these animals using an antibody that specifically recognizes the glutamic acid decarboxylase isoform 65 protein (GAD65), which is localized to presynaptic GABAergic boutons. To assess seizure activity, we employed 2 in vivo mouse models of epilepsy, intravenous pentylenetetrazol (PTZ) and hippocampal electrical kindling, in the presence or absence of Sema4D treatment. We monitored seizure activity by behavioral observation or electroencephalography. To assay the persistence of the Sema4D effect, we monitored seizure activity and measured the density of GAD65-positive presynaptic boutons 3 or 48 hours after Sema4D infusion. Sema4D-treated mice displayed an elevated density of GABAergic presynaptic boutons juxtaposed to hippocampal pyramidal neuron cell bodies, consistent with the hypothesis that Sema4D promotes the formation of new inhibitory synapses in vivo. In addition, Sema4D acutely suppressed seizures in both the PTZ and electrical kindling models. When we introduced a 48-hour gap between Sema4D treatment and the seizure stimulus, seizure activity was indistinguishable from controls. Moreover, immunohistochemistry on brain sections or hippocampal slices isolated 3 hours, but not 48 hours, after Sema4D treatment displayed an increase in GABAergic bouton density, demonstrating temporal correlation between the effects of Sema4D on seizures and GABAergic synaptic components. Our findings suggest a novel approach to treating acute seizures: harnessing synaptogenic molecules to enhance connectivity in the inhibitory network.Objective:
Methods:
Results:
Significance:
Commentary
Inhibition has been a focus of antiepileptic drug (AED) pharmacology since the discovery of the anticonvulsant actions of phenobarbital, a barbiturate that acts on GABAA receptors (GABAARs), in the early 20th century. Recently, efforts have diverted from classic pharmacological targeting of GABAARs toward manipulating interneurons and their function. For instance, cell-based therapies involving transplantation of interneuron progenitors have shown promise in animal models of epilepsy, 1,2 and this approach is under further development for translational implementation. In addition, optogenetic manipulation of interneurons has demonstrated utility for seizure control in animal models and the therapeutic potential is currently being explored. 3 Here, Acker et al demonstrate a novel antiseizure approach involving rapid inhibitory synaptogenesis, which may be a novel mechanism for enhancing inhibitory signaling to exploit for therapeutics.
Insight into the mechanisms regulating inhibitory synaptogenesis has lagged behind our knowledge of excitatory synapse formation 4 and has largely focused on the role of neuroligin-2. Interestingly, a class 4 semaphorin, Sema4D, was identified through an small interfering Ribonucleic Acid-based screen to identify candidate genes involved in excitatory and inhibitory synapse formation, with the knockdown Sema4D selectively impairing inhibitory synapse formation. 5 The research team responsible for the highlighted article went on to demonstrate that the extracellular domain of Sema4D can induce rapid and selective GABAergic synapse formation, 6 suggesting that Sema4D is one of a limited number of molecules that selectively regulate inhibitory synaptogenesis. In culture, Sema4D rapidly recruits inhibitory synaptic elements, significantly increasing the density of GABAergic synapses within 30 minutes and forming functional synapses within 2 hours, 7 an effect which was shown to involve signaling through Plexin-B1 (Figure 1A). 7 These exciting findings could have major implications for the numerous disorders associated with inhibitory GABAergic dysfunction, including but not limited to schizophrenia, depression, neurodevelopmental disorders, autism, Alzheimer disease, and epilepsy.
In the highlighted article, the research team involved in discovering and characterizing the role of Sema4D in inhibitory synapse formation furthers these important studies to show the therapeutic utility of this approach, demonstrating that in vivo treatment with Sema4D exerts antiseizure effects in 2 independent epilepsy models. Intrahippocampal administration of Sema4D nearly doubled the number of inhibitory synapses and increased pentylenetetrazol- and hippocampal kindling-induced seizure threshold in vivo (Figure 1B). These data demonstrate robust anticonvulsant effects of Sema4D even when the treatment is localized unilaterally to a small region of the CA1 subregion of the hippocampus, an effect which is quite remarkable. The authors note that the anticonvulsant effect of Sema4D is comparable to other anticonvulsants, such as valproate and levetiracetam; however, it should be noted that, unlike these known anticonvulsants, Sema4D is likely to be an excellent add-on therapy with the potential to amplify the effects of first-line anticonvulsants. For example, let us consider the potential utility of Sema4D in combination with diazepam. Benzodiazepines, in particular diazepam, remains the first line of treatment for status epilepticus (SE). 8 However, up to 40% of cases are refractory to current treatments. 9 The prevailing theory regarding the mechanisms contributing to pharmacoresistant SE is a downregulation of benzodiazepine-sensitive GABAARs. 8 However, more recently, it has also been proposed that disruptions in chloride homeostasis may compromise GABAergic signaling contributing to pharmacoresistance. 10 The data presented in the highlighted article demonstrate the ability of Sema4D to increase inhibitory synapses, which could potentially restore the expression of GABAARs and diazepam sensitivity (Figure 1C). Although this is currently just speculation, this should be a high priority for the field given the significant translational implications of this work.
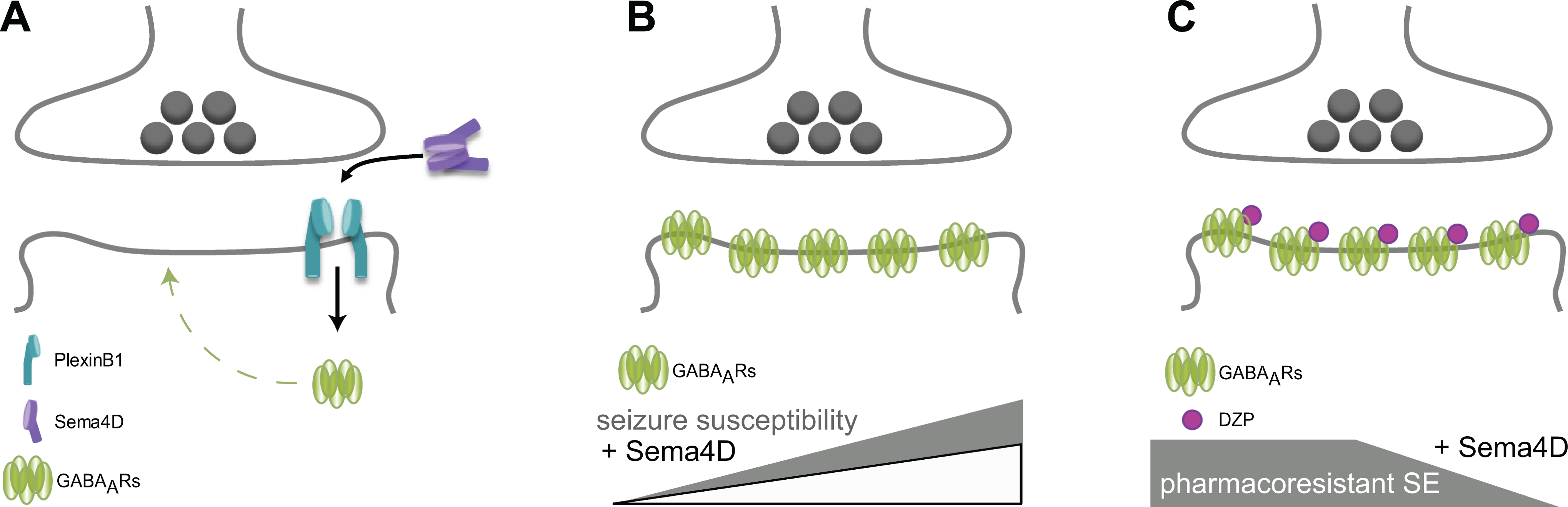
Sema4D promotes inhibitory synapse formation, decreases seizure susceptibility, and has the potential to reverse pharmacoresistant SE. (A) Sema4D signals through PlexinB1 to increase the density of inhibitory synapses. (B) Sema4D exerts anticonvulsant actions, decreasing seizure susceptibility to PTZ- and kindling-induced seizures. (C) Sema4D-mediated increase in inhibitory synapses may reverse the downregulation of benzodiazepine-sensitive GABAARs associated with pharmacoresistant SE, thereby restoring diazepam (DZP) sensitivity.
A potential caveat to the translational relevance of Sema4D is the transient effect. The effects of Sema4D on inhibitory synapse formation and the anticonvulsant effects were observed 3 hours but not 48 hours following treatment. These data suggest that the effects of Sema4D on inhibitory synaptogenesis are transient, which may be viewed as a limitation of this approach. However, there may be benefits of the reversible effects of Sema4D treatment on inhibitory synaptogenesis in regard to treatment approaches, which is unique from other strategies involving cell-based therapies or delivery of opsins. Being able to rapidly and transiently enhance GABAergic signaling may be beneficial in restoring healthy brain activity without long-term consequences and potential adverse side effects, tolerance issues, and/or abuse potential.
Although few laboratory test results have focused on the mechanisms regulating inhibitory synaptogenesis, this pioneering work may contribute toward novel antiseizure drug development, focused on selectively enhancing the inhibitory network. This work supports a novel mechanism for AED targets, facilitating an increase in the expression of inhibitory synapses rather than trying to target existing receptors which may be compromised. There is much work yet to be done, but this preclinical study provides hope that enhancing inhibitory function through noninvasive means remains possible and effective.
