Abstract
A recent assessment of the classification of nonconvulsive status epilepticus (NCSE) has incorporated the specific electroencephalographic (EEG) patterns on a syndromic basis. Such a clinical EEG syndromic approach may enable more accurate and expedited diagnosis of particular subtypes of NCSE so as to improve therapy. Herein, we review the characteristics of subacute encephalopathy with seizures in alcoholics syndrome, a subtype of focal NCSE occurring in chronic alcoholism with specific features, including encephalopathy, lateralized periodic discharges on the EEG, chronic microvascular ischemia on neuroimaging studies, and possible recurrence when chronic antiseizure treatment is stopped.
Keywords
Introduction
Neurologists have long been aware of the diversity of neurological syndromes associated with alcoholism. Traditionally, alcohol withdrawal syndrome (AWS), delirium tremens, hepatic encephalopathy, alcoholic hallucinosis, and Wernicke encephalopathy or Korsakoff psychosis are the best known. Epileptic seizures are frequent clinical features that can occur in different settings. Optimization of treatment and accurate syndromic diagnosis and classification determine the ultimate prognosis.
Alcohol withdrawal syndrome is a well-known and common condition occurring after intentional or unintentional abrupt cessation of alcohol consumption. 1 This picture typically occurs within 24 to 48 hours of stopping alcohol, and signs and symptoms may include tremor, irritability, psychomotor agitation, disorientation, hallucinations, anxiety, and generalized tonic–clonic seizures (GTCSs).
An underrecognized clinical disorder of
Historical Overview of SESA Syndrome
An unusual picture of a subacute encephalopathy in chronic alcoholics characterized by confusion or lethargy, transient neurological deficits, and marked EEG abnormalities was initially characterized by Niedermeyer et al
2
and Freund and Niedermeyer.
3
The EEG findings included focal slowing, spiking, and lateralized periodic discharges (LPDs). Focal motor and GTCSs were common and convulsive status epilepticus was also reported. The patients did not fit criteria for other known neurologic complications in alcoholics, and the authors coined the term
Twenty-five years later, Fernández-Torre et al 7,8 reported cases of SESA syndrome with complex partial status epilepticus (CPSE) that had arisen in temporal and extratemporal regions and that was associated with focal deficits and confusion. They suggested that the syndrome was more common than reported and that the existence of CPSE could readily explain the alteration in mental state. The encephalopathy may have led to underrecognition and possibly masking of an associated nonconvulsive ictal state. As a result, 2 additional cases were used to argue for a revision of the characteristics of SESA syndrome, 11 including supporting evidence from neuroimaging, thus updating EEG and clinical and prognostic syndromic characteristics. Most recently, the full spectrum of neuroimaging features has been described in detail by Drake-Pérez et al 13 expanding the diagnostic criteria for this epileptic entity. In addition, Kaplan et al 15 described a patient who had frontoparietal “ping-pong” nonconvulsive status epilepticus (NCSE) that responded to nonsedating antiseizure drugs (ASDs), while a second patient had focal frontocentral confusional NCSE that was more refractory to treatment.
Clinical Features in SESA
Encephalopathy with lethargy, delirium, confusion, obtundation, agitation, inattention, and disorientation is frequent. In addition, transient episodes of hemiparesis, aphasia, neglect, hemianopsia, and cortical blindness have been described (Table 1). Focal motor and grand mal seizures were reported in the original description. 2,3 However, it was not until 2006 that Fernández-Torre and colleagues described the occurrence of complex partial seizures (CPSs) and episodes of CPSE. 7,8 Nonetheless, the original description in 1981 noted probable subclinical focal seizures in cases 1, 2, and 5. Recurrences occurred in 3 patients. Subsequently, recurrence has been corroborated by other authors. 7,10
Summary of the Clinical, EEG, and Neuroimaging Features of All Published Patients With SESA Syndrome in the English Literature.
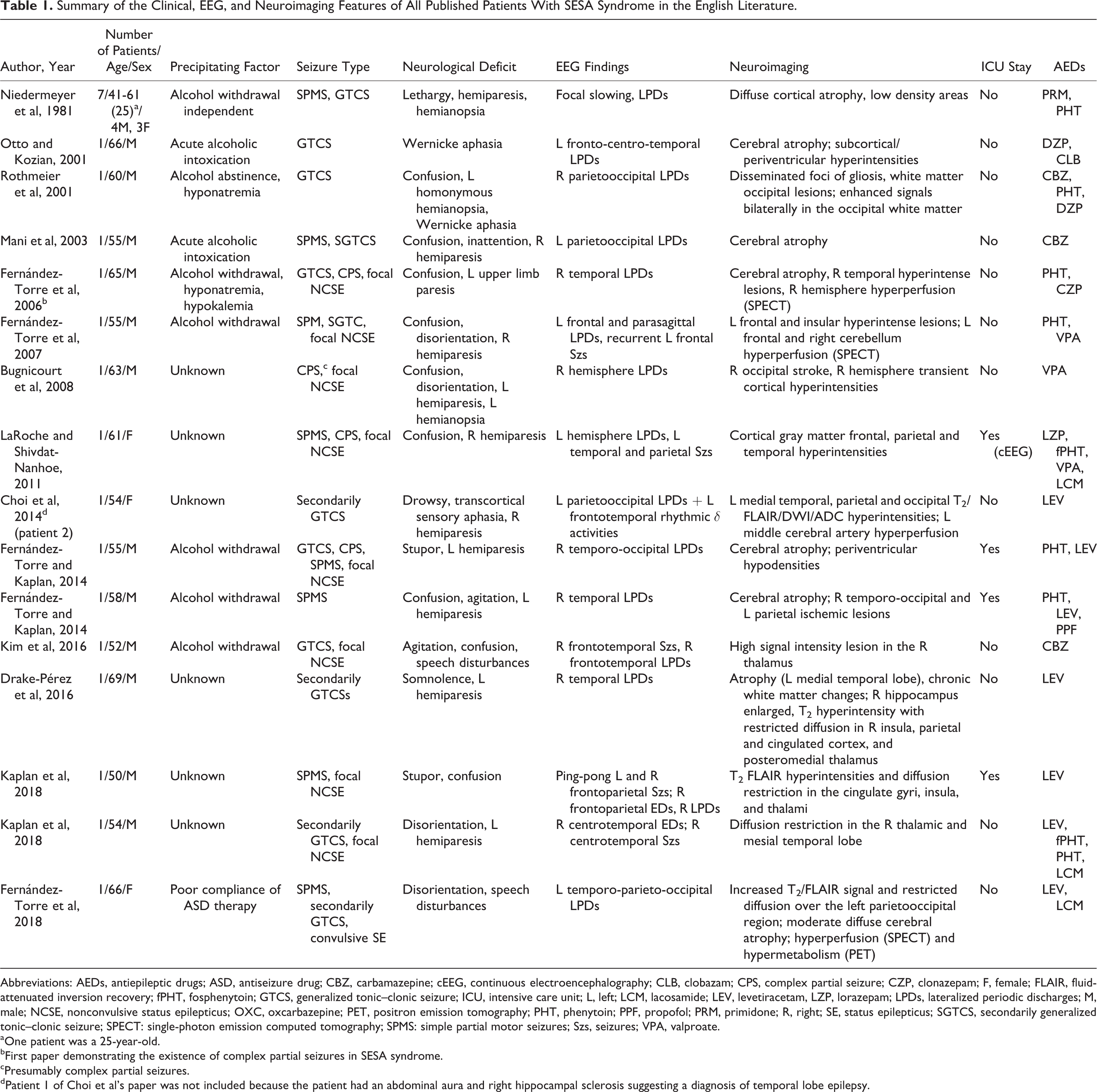
Abbreviations: AEDs, antiepileptic drugs; ASD, antiseizure drug; CBZ, carbamazepine; cEEG, continuous electroencephalography; CLB, clobazam; CPS, complex partial seizure; CZP, clonazepam; F, female; FLAIR, fluid-attenuated inversion recovery; fPHT, fosphenytoin; GTCS, generalized tonic–clonic seizure; ICU, intensive care unit; L, left; LCM, lacosamide; LEV, levetiracetam, LZP, lorazepam; LPDs, lateralized periodic discharges; M, male; NCSE, nonconvulsive status epilepticus; OXC, oxcarbazepine; PET, positron emission tomography; PHT, phenytoin; PPF, propofol; PRM, primidone; R, right; SE, status epilepticus; SGTCS, secondarily generalized tonic–clonic seizure; SPECT: single-photon emission computed tomography; SPMS: simple partial motor seizures; Szs, seizures; VPA, valproate.
aOne patient was a 25-year-old.
bFirst paper demonstrating the existence of complex partial seizures in SESA syndrome.
cPresumably complex partial seizures.
dPatient 1 of Choi et al’s paper was not included because the patient had an abdominal aura and right hippocampal sclerosis suggesting a diagnosis of temporal lobe epilepsy.
Electroencephalography Abnormalities in SESA
As pointed out by Niedermeyer et al, 2 EEG changes in chronic alcoholism are not particularly striking. 1,16 In contrast, in SESA syndrome, EEG abnormalities constitute one of the cornerstones of diagnosis. Focal slowing and spiking and LPDs over the temporal, central, frontal, parietal, and occipital regions were observed by Niedermeyer and associates. They proposed a diverse range of underlying pathogenic mechanisms in their patients, including vascular in 2 patients, traumatic in 1 patient, and “uncertain” in 4 patients.
Neuroimaging Findings in SESA
Recently, Drake-Pérez et al 13 reviewed the full spectrum of neuroimaging findings in 10 published cases of SESA. Initial magnetic resonance imaging (MRI) studies revealed cortical–subcortical areas of increased T2/fluid-attenuated inversion recovery (FLAIR) signal and restricted diffusion in 6 patients. In 5 patients, the affected region included the temporal lobe. The areas of abnormal signal correlated with the origin of the LPDs on the EEG for all 6 cases. Hyperperfusion of the region was observed in 3 of the 6 patients (1 patient had increased distal flow on the magnetic resonance angiography [MRA], and the other 2 had a single-photon emission computed tomography [SPECT] revealing hyperperfusion). The other 3 patients did not have confirmatory SPECT or MRAs. Atrophy was present in 62.5% of the patients, with 2 patients showing a temporal predominance, while 3 had diffuse or unspecified atrophy. Chronic microvascular ischemic changes were described in half. Other isolated findings included hydrocephalus, Chiari I malformation, and choroid fissure cyst. Follow-up MRI in 50% of the patients showed resolution of the hyperintense lesions but revealed emerging focal atrophic changes in 75%. Residual T2 hyperintensities were seen in the right temporal lobe and in the splenium of the corpus callosum in other cases.
Kim et al 14 reported a high signal intensity lesion in the right thalamus associated with focal NCSE in a patient with suspected SESA. In 2 new cases, T2 FLAIR hyperintensities and diffusion restriction in the cingulate gyri, insula, and thalami and in the right thalamus and mesial temporal lobe, respectively, were observed. 15 To summarize, neuroimaging findings include transient cortical–subcortical T2-hyperintense areas with restricted diffusion along with atrophy and chronic multifocal vascular lesions.
Recently, Fernández-Torre et al 16 have described both focal hyperperfusion (SPECT) and hypermetabolism (positron emission tomography [PET]), which were strongly suggestive of an epileptic event in the clinical context. The author concluded that cerebral SPECT and PET closely correlated with EEG can play an important role in the optimization of ASD therapy and diagnosis of SESA syndrome.
Distinguishing Between SESA and AWS
The question has been raised whether SESA is a distinct pathological condition or simply whether it represents the spectrum of CPSs, CPSE, and generalized seizures in the context of AWS or in acute intoxication in chronic alcoholic patients. However, there are significant differences between these entities, including the type of seizures, neurological symptoms, EEG abnormalities, neuroimaging features, and clinical evolution (Table 2). Thus, GTCSs, isolated or recurrent, are the most frequent type seen in AWS. 17,18 In these cases, the diagnostic value of the EEG is limited and, generally, epileptiform activity is absent. 19 Conversely, in SESA syndrome, although GTCSs or secondarily GTCSs are frequent, focal motor seizures occur in up to 40% of the cases. Moreover, frank epileptiform abnormalities on EEG constitute one of the major diagnostic criteria. Of note, when a patient has recurrence, clinical presentation, EEG, and evolution are frequently stereotyped.
Differentiating AWS From SESA Syndrome.a
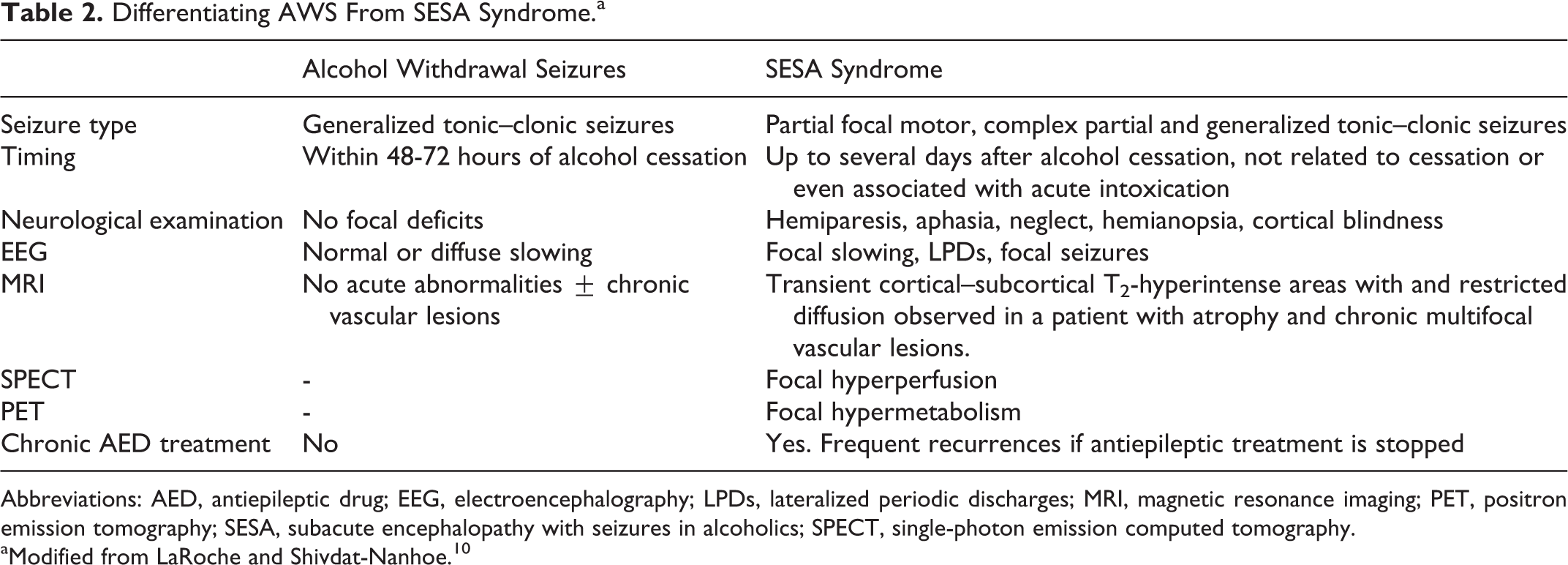
Abbreviations: AED, antiepileptic drug; EEG, electroencephalography; LPDs, lateralized periodic discharges; MRI, magnetic resonance imaging; PET, positron emission tomography; SESA, subacute encephalopathy with seizures in alcoholics; SPECT, single-photon emission computed tomography.
aModified from LaRoche and Shivdat-Nanhoe. 10
Timing of the EEG is important. In some cases, 7,8,11 recurrent CPSs were recorded during the first hours or days of symptom onset. Often, in the subsequent hours after GTCSs in alcoholic patients, a delirium is attributed to a postictal state or withdrawal syndrome. Underlying conditions along the ictal–interictal continuum, ranging from LPDs, to focal CPSE, should be considered. Both LPDs and NCSE in other settings have long been recognized as being associated with impaired cognition, focal neurologic signs, and a decreased level of consciousness, but the diagnosis of these conditions is subject to EEG sampling error, and routine 20-minute recordings might not capture them. Generally, LPDs disappear during focal CPSs and reappear after seizure resolution during which time the patient may remain confused. This dynamic represents an excellent example of the hypothesis of Pohlmann-Eden et al, 20 in which LPDs form part of a continuum between ictal and interictal states. Continuous EEG appears ideal for revealing this pathophysiological electroclinical evolution. A meticulous clinical evaluation and a high level of suspicion during the first 24 to 48 hours after admission are then essential for an expedited diagnosis and optimal management.
Patients who develop SESA syndrome frequently have preexisting cerebral lesions which, in the setting of alcohol withdrawal, acute intoxication, metabolic disturbances, or a combination, produce LPDs and recurrent focal seizures. While focal motor and GCTCs are the cause of the hospital admission, CPSs and CPSE remain underdiagnosed and hence undertreated.
Treatment and Response to ASDs in SESA
Patients with SESA syndrome respond well to ASDs. In some cases, confusion is prolonged and requires intensive care unit (ICU) management. 10,11,15 Several ASDs have been used, including phenytoin, valproate, benzodiazepines, and, more recently, levetiracetam and lacosamide. There are no studies that indicate which agent is most effective. Despite a good prognosis, SESA syndrome may warrant chronic treatment with ASDs and cessation of alcohol use to prevent recurrence.
Establishing a diagnosis of SESA syndrome contributes to the treatment and management. First, because we are defining what will probably be the patient’s natural history and clinical course. Second, because diagnosis points to specific ancillary tests (eg, clinical EEG [cEEG] monitoring, MRI, SPECT, PET) that will help define the pathophysiology (ictal–interictal) and which will help optimize ASD therapy. Third, because knowing that recurrences are frequent may help convince the patient of the need for strict compliance and alcohol cessation.
Conclusions
Subacute encephalopathy with seizures in alcoholics (SESA) syndrome should now include the spectrum of conditions that lie along an ictal–interictal continuum and that may require cEEG monitoring and ICU management. The syndrome encompasses focal NCSE in alcoholic individuals who manifest transient neurologic deficits, interictal LPDs on the EEG, and transient cortical–subcortical T2-hyperintense areas with restricted diffusion and multifocal chronic cerebrovascular abnormalities. Chronic treatment with ASDs is necessary as recurrence is common.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
