Abstract
Introduction:
Severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) is classified as a Risk Group 3 pathogen; propagative work with this live virus should be conducted in biosafety level-3 (BSL-3) laboratories. However, inactivated virus can be safely handled in BSL-2 laboratories. Gamma irradiation is one of the methods used to inactivate a variety of pathogens including viruses.
Objective:
To determine the radiation dose required to inactivate SARS-CoV-2 and its effect, if any, on subsequent polymerase chain reaction (PCR) assay.
Methods:
Aliquots of SARS-CoV-2 virus culture were subjected to increasing doses of gamma radiation to determine the proper dose required to inactivate the virus. Real-time quantitative polymerase chain reaction (RT-qPCR) data from irradiated samples was compared with that of the non-irradiated samples to assess the effect of gamma radiation on PCR assay.
Results:
A radiation dose of 1 Mrad was required to completely inactivate 106.5 TCID50/ml of SARS-CoV-2. The influence of gamma radiation on PCR sensitivity was inversely related and dose-dependent up to 0.5 Mrad with no further reduction thereafter.
Conclusion:
Gamma irradiation can be used as a reliable method to inactivate SARS-CoV-2 with minimal effect on subsequent PCR assay.
Introduction
Researchers worldwide are working tirelessly to develop vaccines and therapeutics to battle the ongoing pandemic caused by SARS-CoV-2. This virus is classified as Risk Group 3; propagative work (eg, virus culture, neutralization assays) should be restricted to BSL-3 containment. 1 However, once inactivated, it can be handled safely in BSL-2 laboratories that are more available in comparison, where much needed viral countermeasure research can be done at an accelerated pace. Researchers use a variety of chemical, heat or irradiation treatments to inactivate viruses, which can then be brought out of high-containment to conduct molecular and immunological analyses in lower containment laboratories.
Gamma radiation is an ionizing radiation and has been commonly used by maximum containment laboratories to render high-risk group viruses inactive. 2 Gamma irradiation is often the preferred method as it is known to preserve viral morphology and viral protein integrity. 3 Here we sought to determine the radiation dose required for complete inactivation of SARS-CoV-2. The primary mechanism of virus inactivation by ionizing radiation is caused by breakage and crosslinking of genetic material. 4 -6 Therefore, we also wanted to see if the gamma irradiation process damaged viral RNA leading to a change in reverse transcription polymerase chain reaction (RT-PCR) sensitivity. A radiation dose of 1 Mrad was required to completely inactivate 106.5 TCID50/ml of SARS-CoV-2 with a calculated D10 value of 0.16 Mrad. The influence of gamma radiation on PCR sensitivity was inversely related and dose-dependent up to 0.5 Mrad with no additional reduction thereafter.
Materials and Methods
Severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2; hCoV-19/Canada/ON-VIDO-01/2020, GISAID accession No. EPI_ISL_425177) was cultured in a high-containment laboratory on Vero cells, CCL-81 (ATCC, USA) grown in minimum essential medium (Hyclone, USA) containing 1% fetal bovine serum and 1% L-glutamine. T150 tissue culture flasks with 80% to 90% confluent cells were infected with SARS-CoV-2 at 1:1000 dilution and incubated at 37°C with 5% CO2 until 90% cytopathic effect (CPE) became evident (approximately 3 days). The flasks were then harvested and clarified by centrifugation at 6000 x
Gammacell 220 Excel (MDS Nordion Inc, ON, Canada), a self-shielded irradiator with a cobalt-60 source, was used for this study. The irradiator’s drawer can accommodate a 2-L beaker where virus tubes were placed along with dry ice. The drawer moves down vertically to carry the samples to the sample chamber for irradiation. The irradiator’s central absorbed dose rate was 0.114 Mrad/h when these inactivation experiments were conducted in early April 2020. We used increasing radiation doses of 0, 0.25, 0.5, 1, 2, 3, 4, and 5 Mrads. After irradiation, samples were taken back to the high-containment laboratory to determine the viable virus titer in median tissue culture infectious dose (TCID50) assay on Vero cells as described previously. 7 Briefly, 100 µL of neat and 10-fold dilutions of each treatment was transferred to 96 well plates and incubated as described and read 3 days later; the plates were read for CPE, and the TCID50 was calculated per Reed and Muench. 8 Negative cultures were confirmed negative by a second passage on Vero cells and monitored for 3 additional days.
For the RT-qPCR assay, viral RNA from nonirradiated and irradiated samples was extracted with the Viral RNA Mini kit (Qiagen, Germany), serially diluted from 10–1 to 10–7 in 10 mM Tris EDTA and run on a LightCycler 96 (Roche, Germany). The EXPRESS One-Step Superscript RT-qPCR Universal Kit (Invitrogen, USA) was used with primers and probes targeting the envelope (E) and the nucleocapsid (NP) genes. 9,10 Thermal cycling conditions were 50°C for 15 minutes for reverse transcription, followed by 95°C for 20 seconds and then 40 cycles of 95°C for 3 seconds, 60°C for 30 seconds.
Results
Irradiation Dose Required for SARS-CoV-2 Inactivation
Complete inactivation of SARS-CoV-2 was achieved with 1 Mrad of radiation (Figure 1), which is consistent with previously published data for SARS-CoV-1. 2 The dose required to reduce the viral titre by one log (D10 value) was determined from the slope of the regression line best fitting the dose curve of the virus from the TCID50 units versus radiation dose (in Mrads). GraphPad Prism (GraphPad Software Inc.) was used for plotting, calculations, and statistical analysis. Since a dose of 1 Mrad completely inactivated the virus, data from 2 Mrad and higher were not used for the D10 value calculations. The calculated D10 value for SARS-CoV-2 was 0.16 Mrad.
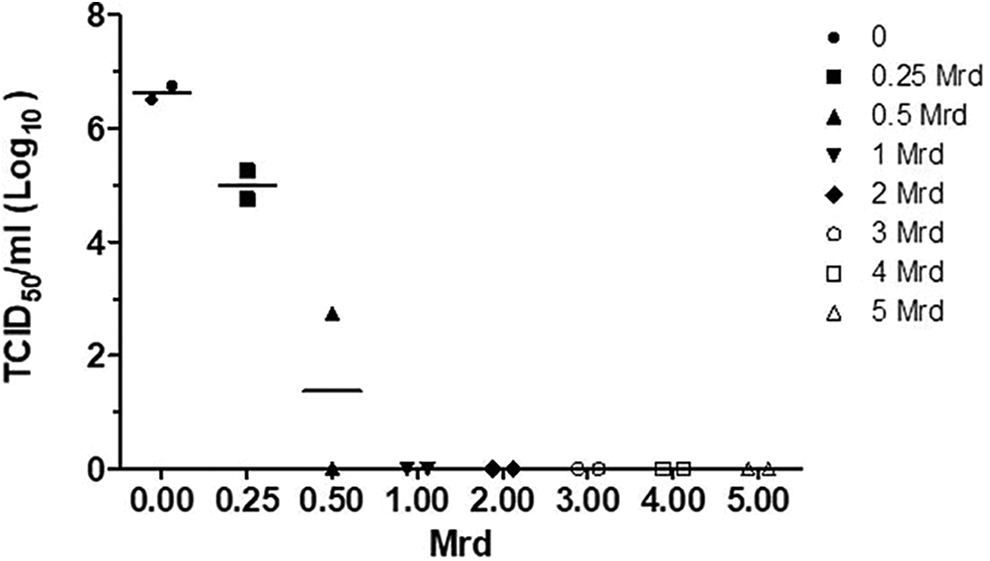
Inactivation of SARS-CoV-2 One-ml frozen samples containing 106.5 TCID50/ml of SARS-CoV-2 virus were exposed to increasing doses of gamma radiation on dry ice.
Effect of High-Irradiation Dose on PCR Results
Researchers often expose their high-risk viral samples to many-fold higher radiation dose than required for a “complete kill” for added safety. Such high doses of radiation can induce severe damage to the viral RNA, leading to unexpected PCR results. Even though our virus sample was completely inactivated after exposure to 1 Mrad, we subjected our samples to increasing doses of radiation up to 5 Mrad. Viral RNA preparations made from these samples were 10-fold serially diluted and tested by RT-qPCR for NP and E genes and plotted against cycle threshold (Cq) (Figure 2).
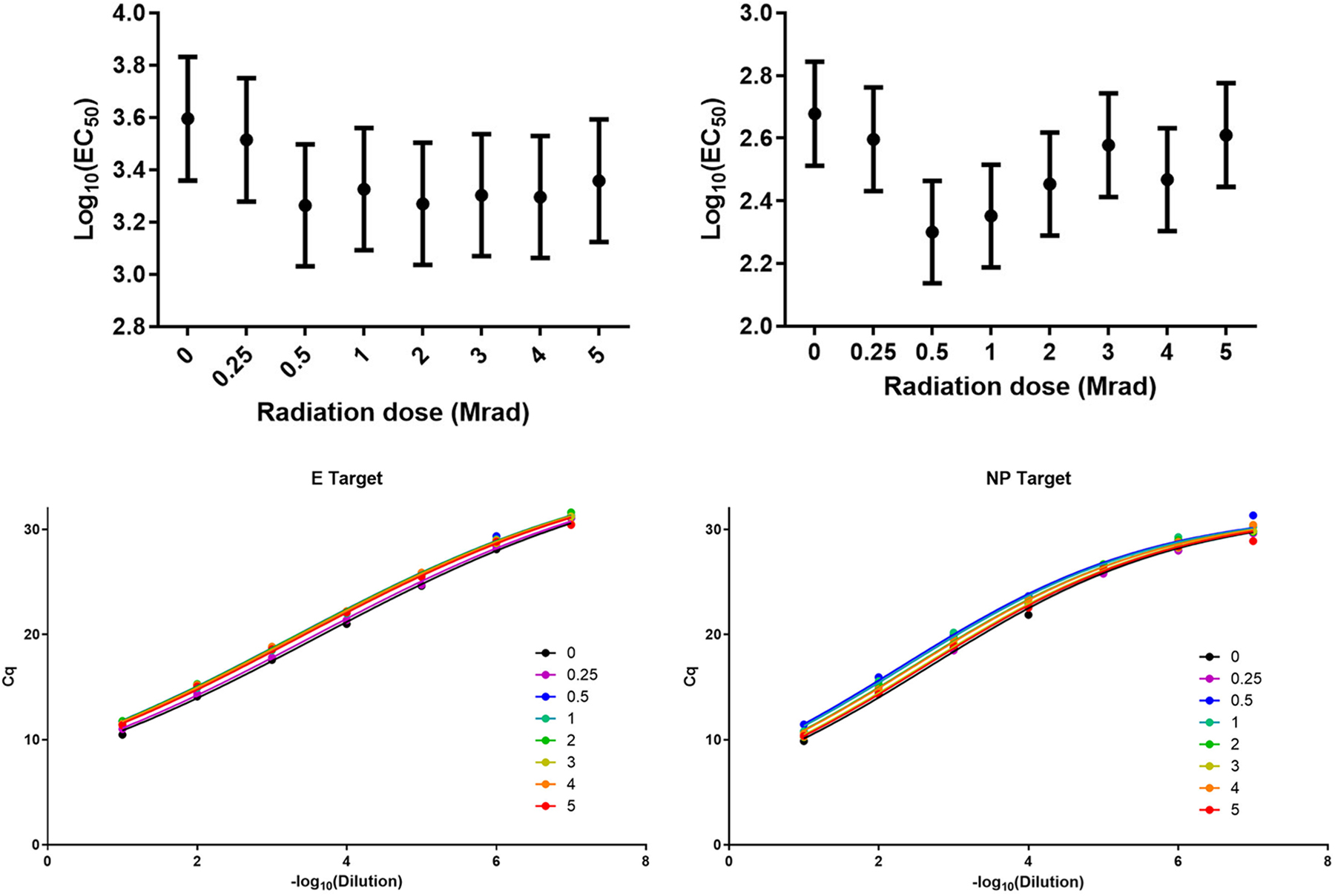
Influence of increasing doses of gamma radiation on PCR sensitivity. SARS-CoV-2 viral samples were exposed to increasing doses of gamma radiation; RNA extracted from the irradiated samples were serially diluted (10-fold) and subjected to PCR testing. Dilution versus Cq for E and NP genes on the left panel; radiation dose versus midpoint on the right panel.
Because the plots followed a sigmoidal shape (s-shaped curve) that can be defined with four parameters (top and bottom asymptotes, slope, and midpoint, EC50), we used GraphPad Prism to fit a four-parameter curve to the Cq versus −log10 dilution. The three parameters, top, bottom, and slope are dependent on the RT-qPCR assay itself, not on the radiation dose; therefore, those parameters were shared across radiation doses within each assay. The EC50 was then the parameter that allowed us to evaluate the effect of radiation on the sensitivity of the RT-qPCR assay. If the EC50 decreased, more RNA would be required to reach the same Cq due to damaged RNA targets being undetected in the assay. If the EC50 increased, then less RNA would be required to reach the same Cq. A slight dose-dependent inhibition of PCR up to 0.5 Mrad of radiation dose was obvious (Figure 2, right panel). It is also evident that the PCR sensitivity for the NP target (Figure 2, right bottom panel) increased after increasing doses of radiation, a sign that the radiation may have eliminated some secondary structures of the viral nucleic acid molecules.
Conclusions
Ionizing radiations interact with matter to generate free electrons and unstable ions, which in turn break the structures of the nucleic acids. In addition, ionizing radiation of biological material results in the formation of reactive hydroxyl radicals, which cause further damage to RNA, DNA, and other cellular macromolecules resulting in cell death. 11 Microbial agents with larger genome are more sensitive to ionizing radiation compared to those with smaller ones. Because viruses have smaller genome in comparison to other infectious agents such as bacteria, they generally require higher doses of irradiation. 12 A measure called D10 value or D value (decimal reduction value) is used to describe the radiation dose required to inactivate 90% (ie, one log or one decimal) of the microbial agents in a given sample. The D10 value that was calculated for SARS-CoV-2 was 0.16 Mrad with complete inactivation achieved with 1 Mrad of absorbed radiation dose. Coronaviruses in general have a D10 value of <0.2 Mrads. 13
Sterilization is often described as the inactivation of all microorganisms; however, it is in fact a probability. The effectiveness of the sterilization process is measured in terms of the reduction of microbial population in a given sample over a set period or dose in case of radiation. One Mrad of radiation was sufficient to inactivate 106.5 TCID50/ml SARS-CoV-2 virus with a calculated D10 value of 0.16 Mrad. This calculated D10 value can be used to determine the dose required to achieve a predetermined sterility assurance level (SAL) in a sample with known microbial concentration. SAL is frequently used to describe the likelihood of a single microbial agent present in an inactivated sample. 14 For example, if a sample that contained 106 viruses is exposed to 6 times the D10 radiation dose (ie, a dose required to inactivate 106 viruses), there is a chance that 1 viable virus will remain in that sample. A sample is considered sterile if it is treated to achieve a sterility assurance level of 10–6, where the chances of finding a viable microbial agent would be one in a million. In the case of a sample containing 106 TCID50/ml SARS-CoV-2 a gamma radiation dose of 1.92 (0.16 × 12) Mrad would achieve a SAL of 10–6. There are several factors that could influence D10 values; frozen samples on dry ice require more radiation dose than samples on wet ice or at room temperature. Because our radiation treatments were done using frozen samples on dry ice, the D10 value determined here would represent the worst-case scenario for sample temperature during irradiation.
Researchers often expose high-risk virus samples from maximum-containment laboratories to excessive amounts of radiation to achieve much higher SALs to be safe; an excessive radiation dose has the potential to damage the integrity of viral RNA or DNA. In this case, we wanted to test what effect, if any, such excessive radiation doses would have on PCR sensitivity. We found excessive radiation, up to 5 Mrads or >31 D10 radiation doses, did not affect PCR results. However, there was discernable incremental reduction in PCR sensitivity up to 0.5 Mrad of radiation. Interestingly, however, a dose of 0.5 Mrad or less would not have been sufficient to inactivate all the viruses in the sample; 1 Mrad was required for complete inactivation. After all the viruses were inactivated at a required minimum dose of 1 Mrad, there was no further reduction in PCR sensitivity observed up to a dose of 5 Mrad. Gamma radiation is often the preferred choice for viral inactivation because it is known to preserve the integrity of viral morphology 15 and protein structures; whether this holds true for SARS-CoV-2 requires further research.
Footnotes
Ethical Approval Statement
Not applicable to this study.
Statement of Human and Animal Rights
Not applicable to this study.
Statement of Informed Consent
Not applicable to this study.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Government of Canada, through the Public Health Agency of Canada.
