Abstract
The risk assessment for research involving rodents housed in colonies must include the potential for transmission of Hantavirus and lymphocytic choriomeningitis virus (LCMV). Various zoonotic strains of Hantavirus are present at varying levels in wild rodent reservoirs around the world; LCMV infects a percentage of the common house mouse population. The infection in rodents for these viruses is generally inapparent, and transmission of both viruses to staff working with the rodents is documented in the literature. Exposure to aerosolized feces, urine, nesting material dust, or bites from an infected rodent can transmit the virus to both the staff and the uninfected rodents in the colony. Infection can also be spread to rodents from implantation of cells passaged in infected rodents, since both viruses retain infectivity during storage of infected cells in liquid nitrogen. This literature survey of occupational infections with Hantavirus and LCMV arising from work with rodent colonies is offered to increase understanding of 4 elements of AAALAC International requirements for rodent colony management: pest control, verification of pathogen status prior to import of rodents, health monitoring of rodent colonies, and pathogen testing of rodent-derived biologicals used in animal protocols. Although published case studies do not provide statistical data, the cases presented here illustrate the importance of adhering to rigorous colony management programs. The pet industry in the United States does not follow these critical standards, as evidenced by the outbreak of Seoul virus, a strain of Hantavirus, in 2018 and a larger outbreak of LCMV virus that occurred in 2012.
Hantaviral Infections
Hantaviruses are a genus within the Bunyavirus family. Unlike other Bunyavirus genera, Hantaviruses are rodent borne, not arthropod borne. These enveloped single-stranded RNA viruses are transmitted by inhalation of aerosolized urine, droppings, saliva, or dust from nesting materials of infected rodents. Transmission may also occur when infectious materials come in contact with broken skin or droplets of infectious material contact mucous membranes. Bites from infected rodents also transmit Hantavirus. Person-to-person transmission has not occurred, except in rare instances with the Andes virus in South America. 1 Arthropod vectors do not transmit Hantavirus. 2
Diseases caused by Hantavirus are now known to exist in various rodent reservoirs in both the New and the Old World, with an estimated 150,000 to 200,000 cases occurring per year in the general population, with more than half occurring in China. 3 It is also now known that there are many strains of Hantavirus, with 21 strains that cause human disease. The diseases caused by Hantavirus are hemorrhagic fever with renal syndrome (HFRS) and Hantavirus pulmonary syndrome (HPS). The Hantavirus strains discussed in this review are listed in Table 1. To date, only the Hantaviruses that cause HFRS have been associated with work in rodent colonies. HFRS begins with a sudden febrile illness, usually 1 to 2 weeks after exposure. The initial phase of the disease includes fever, chills, nausea, and blurred vision and may include a red, flushed face; red eyes; or rash. The second phase may include shock symptoms, vascular leakage, and acute kidney failure. 1 Long convalescence is often required. In some cases, exposed individuals become seropositive but do not develop clinical symptoms. There is no specific therapy for a Hantaviral infection; however, hospitalization in the early phases of the disease is strongly recommended since intensive supportive care reduces the severity of disease. Rodent exposure must be reported when seeking medical attention, and procedures for physicians to obtain diagnostic testing are available on the Centers for Disease Control and Prevention (CDC) Hantavirus website. 1
Characteristics of Selected Hantaviruses.a
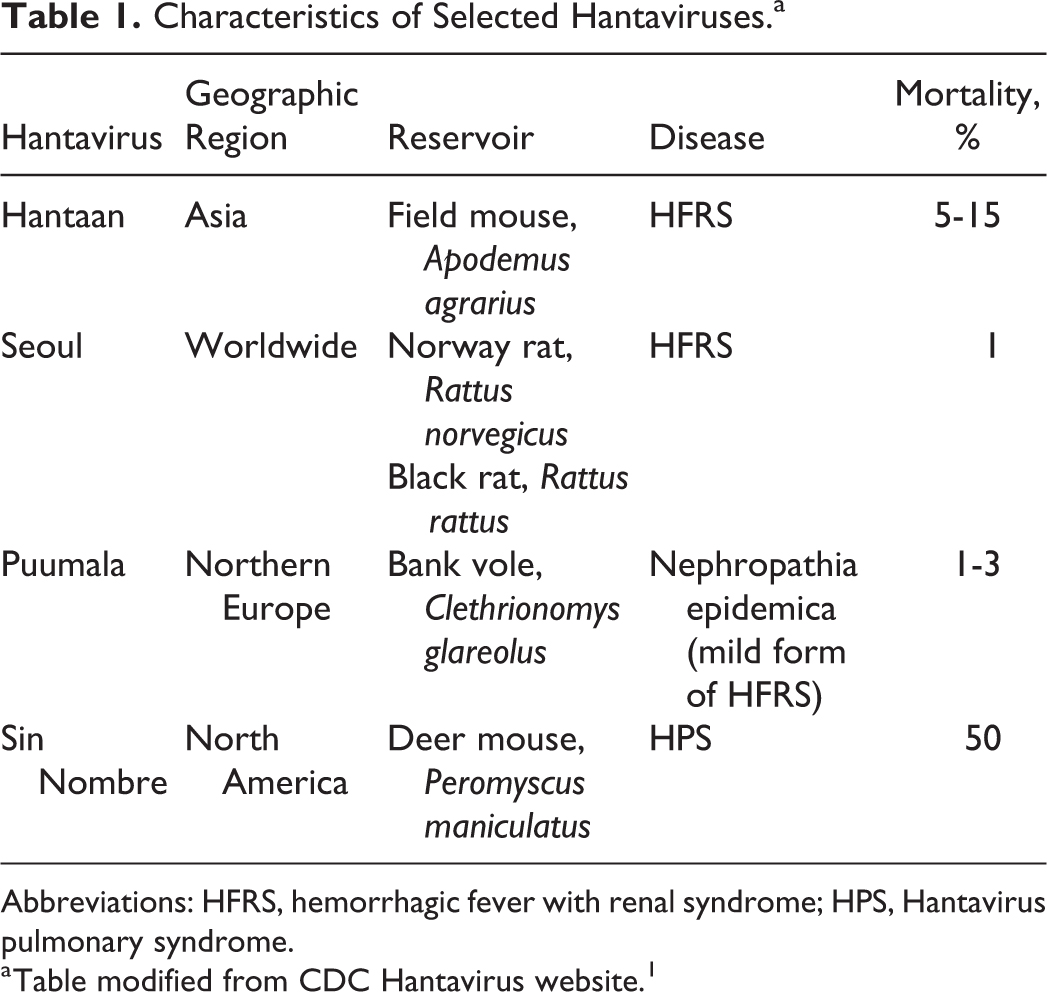
Abbreviations: HFRS, hemorrhagic fever with renal syndrome; HPS, Hantavirus pulmonary syndrome.
Table modified from CDC Hantavirus website. 1
Risk Assessment for Work with Experimentally Infected Rodents
This article focuses on infections acquired when staff were unaware that the rodents under their care or used in their experiments were infected. However, for comparative purposes, the requirements for work with rodents experimentally infected with Hantavirus are described in this section. The ABSA International Risk Group Database indicates that Hantavirus is Risk Group 3 in all countries listed, with 2 exceptions. Puumala virus is classified as Risk Group 2, and approval of the Agri-Food and Veterinary Authority (AVA) of Singapore must be obtained for work with Hantavirus in Singapore. The CDC–National Institutes of Health (NIH) Biosafety in Microbiological and Biomedical Laboratories (BMBL), fifth edition, provides the following containment guidance:
Animal biosafety level 2 (ABSL-2) housing if the rodent species is known not to excrete the virus, with ABSL-2 practices and procedures Biosafety level 2 (BSL-2) with biosafety level 3 (BSL-3) practices for handling serum or tissue samples from potentially infected rodents Animal biosafety level 4 (ABSL-4) for experiments involving inoculation of Hantavirus into species permissive for chronic infection.
2
Hantaviral Infections Acquired from Conducting Research with Colony Rats with Undetected Infection
A total of 185 clinical HFRS cases and 85 subclinical infections detected by serology have been reported in unsuspecting staff who were caring for or using rats in their experiments and were unaware that the rodents were infected. The genus Hantavirus includes pathogenic viruses that may infect rodents for life. Usually, clinical symptoms are not evident in the rodent, but the virus is shed in rodent saliva, feces, and urine. It is easy to visualize how an infected wild rat eating food from the top of an open-wire cage lid can infect the caged rodents. Prior to development of the health monitoring of rat colonies that is practiced today, an index case of HFRS in the staff of a research institution was the first indication that the rats in a research facility were infected. (See compilation of cases in Table 2.) A total of 180 HFRS cases and 85 subclinical human infections acquired from work with rat colonies have been reported from Argentina, 4 Belgium, 5 China,6 -8 France, 9 Japan,10,11 Korea,12,13 Netherlands, 14 United Kingdom, 15 and Singapore. 16
Hemorrhagic Fever with Renal Syndrome (HFRS) Cases Caused by Wild Rat Penetration into Research Animal Colony or Import of Infected Rodents.a
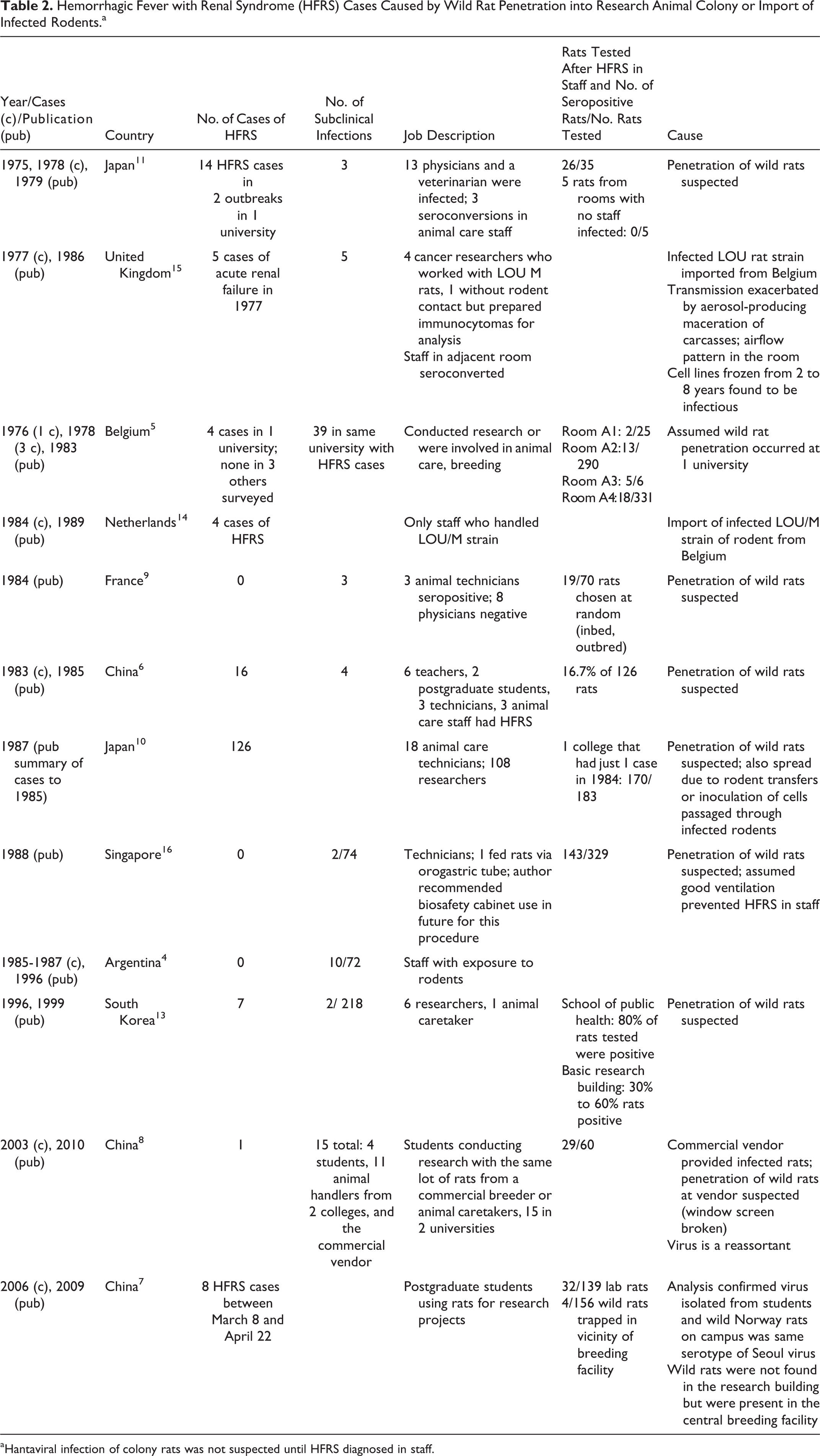
Hantaviral infection of colony rats was not suspected until HFRS diagnosed in staff.
Key Role of the Pest Control Program to Prevent Penetration of Zoonotic Infections
Numerous case studies document the importance of robust pest control programs. For example, 2 separate outbreaks of HFRS occurred at Tokonu University in Japan in 1975 and 1978. In total, there were 14 cases of HFRS (13 physicians and a veterinarian); 3 animal caretakers also seroconverted but did not become ill. 11 The infections were localized in specific rooms; staff who worked in other rooms did not seroconvert. The authors suspected that wild rats had penetrated the specific rooms where infected staff worked, and this was later shown to be a factor in many of the outbreaks. By 1985, the number of laboratory animal–associated HFRS cases in Japan was up to 126. 10 The route of transmission to staff was inhalation of virus-contaminated air.
In 1984, a total of 70 rats chosen at random from various outbred and inbred strains raised in a Paris facility for over 10 years were screened for infection with Hantavirus. Nineteen rats had titers of 8 or more against Hantaan virus strain 76-118. Three animal keepers had titers of 16, and the 8 physicians who were tested were negative. This author recommended routine screening for Hantavirus infection in rat colonies. 9 In 1985, 16 of 60 staff members working in a college laboratory and animal facility in China developed HFRS. In addition, there were 4 seropositive individuals, indicating exposure resulting in an asymptomatic infection. 6
An investigation at the Shenyang Pharmaceutical University in China firmly established the link between the penetration of feral rats and an outbreak. 7 Eight postgraduate students developed HFRS between March 8, 2006, and April 22, 2006. The students were all in the same department and did research studies using the same rat colony. Wild rats were trapped on the grounds of the animal facility, and samples from the infected students, the colony rats, and the wild rats were analyzed. The Hantaan viruses from all 3 sets of samples had sequences that clustered within 1 of the 3 lineages of Seoul virus circulating in wild rats in the outside community in Shenyang, People’s Republic of China.
Importance of Verifying Health Status of Rodents Prior to Import Into a Colony
An index HFRS case occurred in a college student in Kunming, Yunnan province, China, in 2003. 8 In response, a seroprevalence study in 8 institutions in China was conducted. Fifteen graduate students in 3 institutions were seropositive; the infected students had all worked with rats obtained in March 2003 from the same commercial vendor. Characterization of the virus in that lot of rats indicated that it was a new Hantaan reassortant. When an audit of the commercial source was conducted, it revealed at least 1 broken window screen in the vendor’s animal facility, indicating a likely point of feral rat penetration. In addition, a ventilation fan in the animal room had failed months earlier; this may have facilitated the spread of virus in the colony.
Import of an Infected Rat Strain with Aerosol-Generating Procedures and Ventilation Exacerbating Risk of Personnel Exposure
The Louvain (LOU) rat strain spontaneously developed a tumor that stably secreted immunoglobulins and could be easily transplanted. 15 In a facility in the United Kingdom, between January and July 1977, 4 cancer researchers were working in room A with immunocytomas using the LOU rat model imported from Belgium. 17 Four researchers who worked in room A developed HFRS; 9 of 10 staff who worked in either room A or the adjacent room B seroconverted. In addition, 1 individual who never entered the rat room but prepared the rat immunocytomas for analysis was also infected. Other staff working in the same building, but not with LOU rats or tissue, were not infected. During the investigation to determine the cause of the outbreak, it was noted that viral aerosols and droplets emanated from a macerator without bioseals that was used to dispose of tissue and carcasses in the sink drain; the researchers opened the lid during operation to add tissue as it was generated. Tissue sometimes backed up in the sink. The ventilation in the room flowed from the ceiling supply vents, located above the macerator, to the exhaust over the door. The LOU rat model had been studied by these investigators for 10 years, and tumor samples thawed after being held in liquid nitrogen for 10 years retained infectivity. It is not known why the infectivity peaked in the rat colony during the 6-month period that resulted in the 9 staff seroconversions and 4 HFRS cases. The rats in affected rooms were euthanized, the rooms associated with infection were decontaminated, and clean rats were introduced after 2 months and there were no new staff infections. The LOU/M laboratory rats were also the source of 4 HFRS infections in the Netherlands at the National Institute of Public Health and Environmental Protection; staff who had not handled these rats were seronegative. 14
Infected Rodents Spread Infection, Produce Infected Rodent-Passaged Cells
Three staff who worked in the same room with rats at a Belgian university were hospitalized with acute renal failure in 1978. 5 In 1981, serology retrospectively confirmed that these cases were HFRS; a fourth probable case had occurred in 1976. Due to these results, a seroprevalence study was conducted, and 30 of the 60 staff members working in the rooms housing rats were seropositive. The infected rat strains were not limited to LOU strains; Wistar, R/A, and others were also seropositive. An additional feature of the investigation was the thawing of tumors stored in liquid nitrogen and transplanting of tumors into seronegative rats in an isolated area; this confirmed that transmission of the virus from frozen cells could occur. This study also recommended rat health colony monitoring. 5
Route of transmission
Inhalation was the route of exposure in most cases. Some individuals became infected by visiting an animal room for 5 to 10 minutes.10,12,18 In another case, rats were housed in cages suspended from a rack with feces collected on disposable paper below the cages. An animal caretaker was fatally infected and the exposure was attributed to dust from the aerosolization of rat feces during disposal of the paper. 10 Two staff were infected while cleaning a defrosted morgue freezer. Some of the bags containing infected rat carcasses had leaked, and the cleaning method resulted in aerosolization of the virus. 10
Two references document parenteral exposures that resulted in HFRS. A medical student was drawing blood from a rat when he sustained a needlestick; 2 other students working in the same room who did not have parenteral exposures did not seroconvert. 10 A researcher weighing rats for 3 weeks after intracranial inoculation of the B-1 strain of Hantaan virus was suddenly bitten through his glove and developed HFRS. Testing of the rat’s salivary glands revealed many Hantaan viral antigens. 10 The index case of HFRS and 5 students who seroconverted reported that they had been bitten by rats. 8
Human host factor
Authors noted that clinical infections occurred more frequently in researchers rather than caretakers.6,10,11 Researchers, especially those new to animal research and physically exhausted, were more likely to be infected. Among the 126 cases in Japan between 1970 and 1983, 18 were animal care technicians, and 108 were laboratory researchers performing animal experiments. 10 This was also the case in the outbreak that occurred in the Basic Research Building of the Seoul National University College of Medicine in Korea. There were 2 cases of HFRS; both researchers worked in basic research building BRB. In response, 218 researchers and employees participated in a serological survey; 6 researchers and 1 animal caretaker were seropositive. 13 The authors speculated that clinical infections occurred more frequently in researchers because they had infrequent exposure to contaminated air.
Role of Ventilation in Preventing Hantavirus Transmission: Work with Wild-Caught Rodents
Rodents captured in the wild and maintained in an animal colony present unknown zoonotic risks, and the potential for transmission may be amplified if the facility containment procedures are not commensurate with the potential risk. Case studies involving wild rodents captured for evaluation of their role in disease outbreaks are described below and clearly reinforce the critical importance of laboratory ventilation.
The Lee laboratory, in 1978, published the groundbreaking research that identified the striped field mouse (Apodemus agrarius) as the rodent reservoir and developed assays that enabled detection of Hantaviral infection. 12 Although 3000 United Nations troops were infected with Korean hemorrhagic fever during the Korean War, the virus, now called Hantaan virus, was not identified as the source of the disease prior to the publication of this research. However, all 7 of the Lee laboratory staff and 2 casual visitors to that lab were infected and developed HFRS. The study was conducted with wild-caught rodents or experimentally infected Wistar rats at the Korea University Virus Institute in Seoul between 1971 and 1979. All of the staff infections occurred in the winter months, when windows in the facility were sealed against the cold and a coal burner provided heat in a single room. In the summer, the rats were held in 2 rooms, but the rats were consolidated into 1 room in the winter. The lack of ventilation and low humidity contributed to the transmissibility of the aerosols produced by the chronically infected rodents. Although the laboratory staff were immune as a result of their previous Hantavirus infection, the husbandry practices were upgraded to require filter bonnets on all cages and high-efficiency particulate-filtered cage rack enclosures. No additional cases were reported after these improvements were made.
In October 1958, a total of 235 wild-caught rodents from the endemic area of Tula, in the former Soviet Union, were brought into the Institute of Poliomyelitis and Viral Encephalitis laboratory. All worked in the animal colony without respirators. By December 28, 1958, 12 staff members had HFRS, including 4 staff who did not handle the rodents but worked in an adjacent area. Fortunately, all recovered. 18
On September 24, 1961, field-caught rodents (Clethrionomys glareolus, Clethrionomys rutilus, Apodemus sylvaticus, Apodemus flavicollis, and Microtus oeconomus) were brought to a different laboratory in Moscow. Between October 18 and November 29, 113 out of the 186 employees or visitors to the facility developed HFRS. There were 81 infrequent visitors, and of the 24 who had been in the facility for 3 to 4 hours on a single day, 18 were infected, including 1 who was in the facility for only 5 minutes. 19
In 1967, 3 pairs of bank voles trapped in Kirov were brought to the D. I. Ivanovsky Institute of Virology. 18 The animal room had an anteroom with double doors that were always closed, but the seal was not airtight and directional airflow into the room was deficient. Ten days after the voles arrived, illness struck. The first to succumb, with severe infections, were the 2 staff who took care of the voles. The additional 17 HFRS infections occurred in staff whose only exposure was walking down the corridor past the animal room.
Current Practice for Colonization of Wild-Caught Rodents
In 1995, the CDC recommendations for housing wild-caught Peromyscus and Sigmodon were published and included requirements for quarantine in physically separate housing with no air exchange with areas where humans work or other animals are housed. Additional recommendations include the following: barrier clothing and respirators are required for the staff, and rodents are tested on initial introduction to the facility and again in 30 days. If any rodents test positive, the entire colony in quarantine is euthanized and disposed of properly. If all the rodents are seronegative, they may be removed from quarantine but maintained in a separate room to prevent transmission of other zoonotic pathogens to other animals in the facility. 20
Outbreak of Seoul Virus in the Pet Industry
Hantaviral infections were in the news recently due to the February 2018 report of 17 infections with Seoul virus in the United States and 1 in Canada; the infections were associated with the home businesses that breed rats for the pet industry. 21 The source was linked to 31 home ratteries in 11 states; Canadian public health authorities were contacted when it was determined that 6 US ratteries had shared stock with Canadian ratteries. In the United States, the reservoir for Seoul virus is the wild Norway rat. Serological monitoring was offered and accepted by 183 individuals associated with the home rat breeding industry; 24 had antibodies to Seoul virus, and 3 were hospitalized. The mother of 1 rat breeder, who did not work directly with the rodents, was hospitalized and reported that her exposure to rodents was cleaning some rat feces out of a bathtub, one time, about 3 weeks before becoming ill. 22
The public health response included an alert to US Health Departments on January 24, 2017, and notification of the World Health Organization, as required by international public health laws, on February 20, 2017. In the United States, a quarantine was imposed on ratteries with confirmed or suspected infections; rats could not be added or shipped out from the quarantined facility. The number of individuals caring for the rats was limited to reduce the number of potential exposures. Testing of the rats was offered to the rattery owners; this allowed the culling of infected rats only, and the quarantine was lifted if all rodents were negative 4 weeks after the last rodent was added to the colony. If the owners did not consent to testing of the rats, the options were (1) quarantine for the life of all rats or (2) euthanasia of all rodents. In Canada, the public health response was education of rattery owners with voluntary testing and culling of the colonies.
The CDC website (www.cdc.gov) is updated regularly with information about Hantavirus infections in the United States. The website includes information for the public on exposure prevention and for human health care professionals on treatment. Information on testing rats for Seoul virus is also included for both rat pet owners and veterinarians.
Lymphocytic Choriomeningitis Virus
Lymphocytic choriomeningitis virus (LCMV) is a single-stranded RNA virus member of the family Arenaviridae. It is usually spread by the common house mouse, Mus musculus, and the CDC estimates that about 5% of the mice in the United States are infected. The virus has also been found in the wild mouse populations of Europe, the Americas, Australia, and Japan. Guinea pigs, hamsters, humans, and nonhuman primates are not natural reservoirs, but they can be infected by exposure to infected wild mice. Humans are infected by inhaling infectious aerosolized particles of rodent urine, feces, or saliva or nesting materials; by ingesting food contaminated with virus; by exposing cuts or open wounds to infectious materials; and by rodent bites. In immunocompetent individuals, the virus may cause asymptomatic development of antibodies, flu-like illness, aseptic meningitis, encephalitis, or meningoencephalitis; most immunocompetent individuals usually recover completely after several weeks. Mortality from lymphocytic choriomeningitis (LCM) is less than 1%; however, the CDC notes that, as with any central nervous system infection, temporary or permanent neurological damage is possible. According to the CDC fact sheet on LCM, deafness and arthritis may also be associated with previous LCMV infection. The immunosuppressed, however, are at greater risk. 23 Receiving organs from an LCMV-infected donor has proven fatal to 14 out of 17 organ recipients since 2005. 24 Person-to-person transmission does not occur, with the exception of transmission from an infected mother to a fetus. When a pregnant woman is infected during the first or second trimester, LCMV infects the fetal central nervous system. Fetal death, congenital hydrocephalus, chorioretinitis, and retardation may result. 23
Risk Assessment for Rodents Experimentally Infected with LCMV
According to the ABSA International Risk Group Database, LCMV (nonneurotropic) is classified as Risk Group 2 in all countries listed except Canada, which lists this virus as Risk Group 3; Singapore requires AVA approval for work with this virus. LCMV (neurotropic) is listed as Risk Group 3.
Containment guidelines for animal work in the fifth edition of the BMBL:
ABSL-3 for work with rodents infected with strains lethal for non-human primates ABSL-2 for adult mice with mouse-brain passaged strains requiring BSL-2-containment ABSL-3 for work with infected hamsters Containment and practices for work with known or potentially LCMV-infected source materials (cell lines, tissues, fluids) are recommended at BSL-2
3
.
Case Studies of LCMV Infections Due to Infected Hamsters, Infected Cell Lines
Before 1975, there were 3 outbreaks of LCMV in US academic institutions. 25 In 1965, there were 10 confirmed cases at the NIH; this was the first report of LCMV in hamsters infected with a tumor cell line. The first 5 staff who had a flu-like illness were animal caretakers; 5 additional staff infections occurred in 3 locations that received the infected hamsters or their tissues for study. 25 In 1969, a surgical resident developed aseptic meningitis attributed to work with hamsters implanted with an LCMV-infected tumor cell line. 27 Forty-eight cases of LCMV occurred between 1971 and 1973 at the University of Rochester medical school; those infections were also linked to a contaminated tumor cell line. 26 The contaminated cell line that caused the University of Rochester outbreak was traced to 1 biological cell line supplier. Further investigation revealed that 13 of the 22 cell lines that were propagated for distribution by this supplier were contaminated. All recipients of the cell line were notified, and the supplier voluntarily stopped providing any tumors that tested positive. That same cell line injected in Syrian hamsters caused 7 cases of LCM in the staff at a New York state hospital. 27 That outbreak is evidence of the airborne route of transmission for LCMV; most of the infected staff reportedly were not involved in husbandry or research but had used the copier located in a room that housed 200 Syrian hamsters. It is assumed that the same biological supplier that produced the LCMV-contaminated cell line was inadvertently also the source of the 1974 pet hamster outbreak that resulted in 181 cases of LCM in 12 states. The infections were traced to a single supplier who bred hamsters as a home business but was also the full-time employee of the biological supply service that produced the tumor cell line linked to the outbreak at the University of Rochester. The breeder stopped supplying pet shops, the shops were notified, and hamsters ordered from that breeder were euthanized. 26
Colony Health Monitoring for LCMV
A cancer research program changed the rodent animal model from hamsters to the smaller nude mice to reduce dose of radioisotopes required for tumor labeling. In 1986, the nude mice census was 760; in 1989, it was 4146. There was a lapse in the health monitoring program from August 1988 to March 1989. When LCMV testing was resumed, the oldest sentinel hamsters were positive, and this information was conveyed to the physician caring for a staff member with aseptic meningitis. Once the index case was diagnosed, the CDC offered serological testing, and 82 of the 90 staff participated. There were 7 definite seroconversions and 1 probable, for a 10% seroconversion rate. The infected individuals had all worked with nude mice that were injected with the same LCMV-contaminated proprietary cell line. Cleaning cages and changing the water bottles for nude mice were the activities most associated with infection. In the follow-up investigation, deficiencies were noted in the facility, including inadequate exhaust ventilation and inappropriate liquid disposal. However, restricted access was effective in restricting the outbreak; no researchers or animal care staff who did not work with nudes were infected, and none of the 29 staff without animal contact were infected. 28
Two Outbreaks of LCMV in Rodents in a Facility with an Active Serological Monitoring Program
In January 1980, in a facility with a rigorous serological monitoring program, 3 of 26 sentinel mice became seropositive for LCMV in a first-floor room. This prompted testing of 110 mice from the same room and 218 mice along the first-floor corridor; 17 mice tested positive. The source was not known at that time. On the fourth floor of the same building, in March 1980, an experiment using chemotherapy agents in rodents yielded improbable results; this prompted the researchers to question whether the rats were infected, since it is known that LCMV infection affects the development of tumors. When the test results were analyzed in terms of the spatial distribution in the room, 5 sentinel mice in an open cage on the bottom rack were positive, and the highest concentration of seropositive rats was on the bottom shelves of an adjacent rack. The authors assume that the lower shelves were accessible to wild mice; there was ongoing construction in the medical school between 1979 and 1980. Also, a wild mouse caught in a tunnel connecting medical school buildings was positive for LCMV. 29
The GLO tumor line had been propagated in mice on the fourth floor and, in January, had also been injected into mice on the first floor. The GLO tumor line was not infected when it was initially received by the researchers; this was confirmed by thawing cells frozen prior to the outbreak. Passage in the fourth-floor room must have resulted in infection of the cell line. Fortunately, no staff were infected, but the authors note that a “large and valuable animal facility was depopulated for safety reasons.” 29 Depopulation is also required to eliminate the effect of LCMV on research results.
Effect of LCMV Infection on Research
Viral stocks and Toxoplasma gondii strains grown in LCMV-infected rodents may be cross-contaminated with LCMV. 30 Cancer research is affected by the blocking of tumor induction by polyoma and mammary tumor viruses, interference with the development of transplantable leukemia, and delay in the rejection of transplantable tumors and cutaneous grafts. The effect of endotoxins and susceptibility to ectromelia is also enhanced in the LCMV-infected mouse. 30
The Importance of Health Status Verification of Mice Imported Into a Facility
The most compelling argument for this practice is the 2012 outbreak of LCMV in the feeder-rodent industry. The CDC investigation involved 2 commercial facilities in Indiana and 1 in Kentucky. Ninety-seven employees became seropositive, 9 became ill, 5 sought medical care, and 4 had aseptic meningitis, confirmed by lumbar puncture. There was 1 pregnant staff member; fortunately, she was not infected. 24 LCMV was suspected and confirmed by the CDC in 2 cases of aseptic meningitis; both individuals worked in a rodent breeding facility in the state of Indiana.24 Testing confirmed LCMV infection, and serological monitoring was offered to the facility staff. The results of testing are summarized in the test results column of Table 3, depicting the transfer of infected animals between facilities. The investigation of the Indiana outbreak led to offering testing of staff and rodents in 2 additional facilities. The Indiana facility, facility A, shipped mice to a distributor (facility B) where live rats and mice were handled and packaged as live and frozen animal food and shipped to 21 states. Facility B had also received 90,000 mice from a breeding facility in Kentucky (facility C) and shipped them to facility A to replenish breeding stocks.
Summary of US Rodent-Feeder Industry Outbreak in 2012.
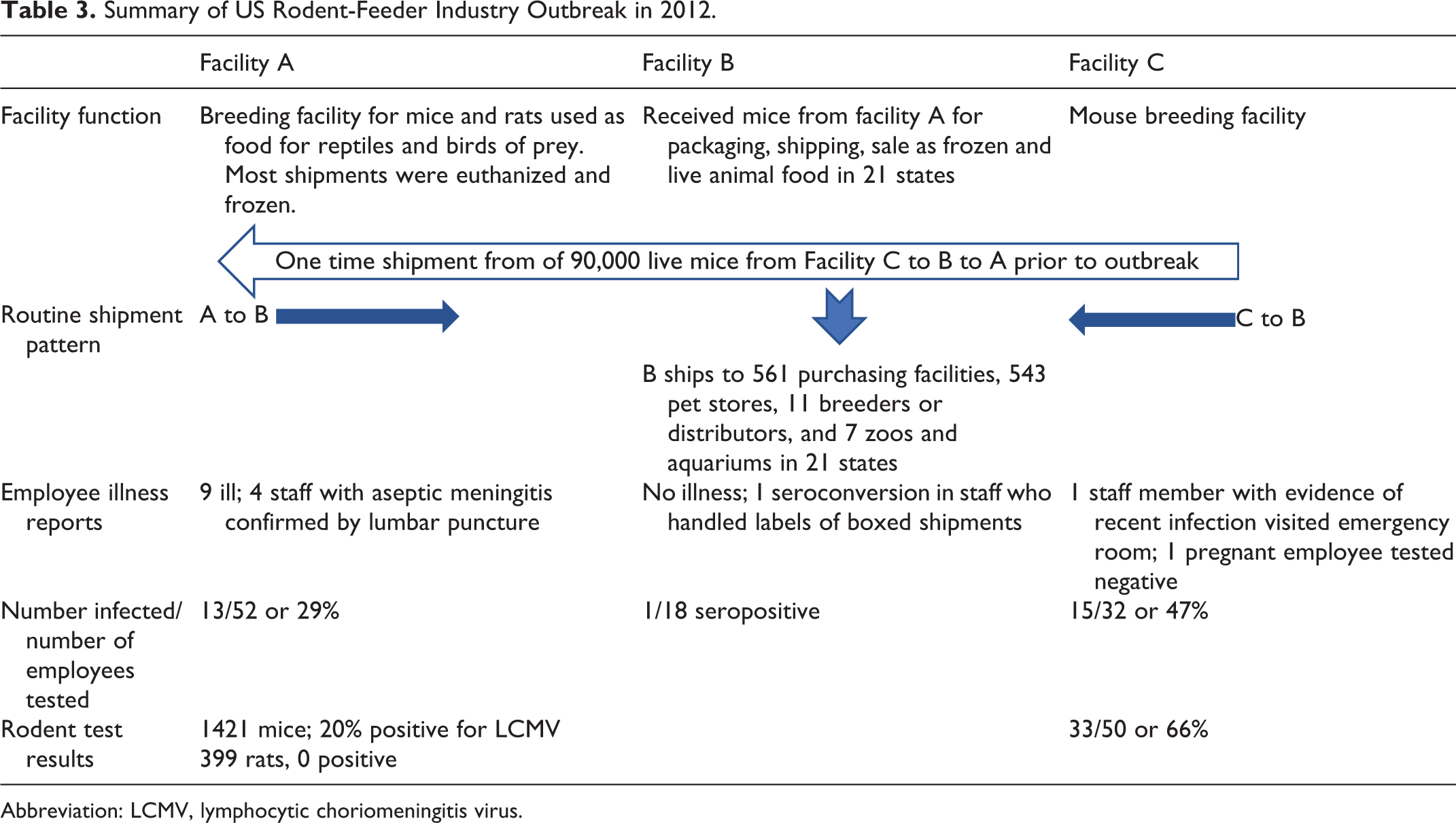
Abbreviation: LCMV, lymphocytic choriomeningitis virus.
Root cause of the 2012 outbreak: import of infected mice
The rodent food storage area of facility C had been infested with wild mice 6 months prior to the outbreak, and some litters of mice were born with black eyes, indicating the interbreeding of wild mice and the albino colony mice. Facility C had 50 mice tested by a commercial testing company when the outbreak at facility A was reported. Thirty-three or 66% of the 50 mice tested were infected. Facility C then proceeded to depopulate the mice in the facility.
Personal protective equipment and practices in facilities A, B, and C
All 97 seropositive employees reported handwashing after handling rodents and use of personal protective equipment (PPE), although some admitted that their use of PPE began after the LCMV outbreak was suspected. One of the infected individuals had worked at facility A for 2 days. Smoking was a risk factor identified in the analysis of the facility A outbreak; it is not known whether this is due to changes in the lung or to the physical act of bringing hands to the mouth.
End of the LCMV outbreak?
To end the outbreak at the rodent-feeder breeding facilities, mice were euthanized, the used food and litter were disposed of, and the facility and all equipment was cleaned and disinfected.26 Respirator protection and training were provided to the staff, and no new cases resulted from the depopulation process.26 Facility A euthanized 400,000 mice; facility C euthanized 380,000 mice. Facility B disposed of 810,000 frozen mice and what remained of the facility A frozen stock. There is no other option when LCMV has infected a facility. However, a CDC trace-forward investigation revealed that approximately 304,000 live mice from facility B were shipped to 561 purchasing facilities, 543 pet stores, 11 breeders or distributors, and 7 zoos and aquariums in 21 states before the problem was identified. 31 The authors concluded that thousands of employees and pet shop customers were potentially exposed to LCMV.
Conclusion
The advancement of science requires studies to be conducted with rodents housed in colonies; however, these rodents are vulnerable to infection by wild rodents. The published outbreak reports promote understanding of the zoonotic risks involved in rodent research and support efforts to minimize that risk. An investigator who is eager to begin work and does not appreciate the importance of testing cell lines for rodent pathogens prior to use can inadvertently introduce LCMV into the rodent colony. An incident of this type was presented at the ABSA International Conference in 2004. An animal caretaker was ill, and the sentinel animals tested positive for LCMV. The facility veterinarian communicated this result to animal caretaker’s physician, and testing confirmed LCMV infection. Incident reports are useful for training on zoonotic risks and biohazard communication. Recently, ABSA International made available the lab-acquired infections database (https://my.absa.org/LAI), which includes documented cases of LCMV and Hantavirus. Another key point is that research and animal care staff who work with rodents must be trained to report rodent exposure if they become ill. Diagnostic procedures for the diseases caused by Hantaviruses and LCMV are not initiated unless exposure to rodents is reported to the health care provider, who can then obtain testing from the CDC by contacting their state public health department. 1
When colony animals become infected with LCMV or Hantavirus, the solution employed is depopulation of the colony and decontamination of the facility. This is required to eliminate the risk of zoonotic infection, but it is an overwhelming loss of animal life. In addition to economic losses, valuable research models may be at risk, and depending on the nature of the research, some studies conducted on infected rodents may have to be repeated. Ultimately, a constant, concerted effort is required to minimize zoonotic risk to staff working with rodents housed in colonies and to support critical rodent research activities that generate uncompromised data.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
