Abstract
Research programs utilizing animal models present a wide variety of risks to personnel safety. These risks stem from a range of hazards including well-recognized physical, chemical, or infectious hazards to novel or less-well defined hazards associated with new and emerging technologies. Institutions must provide appropriate oversight of occupational health and safety programs to help prevent and recognize personnel injury or illness. In this article, we review institutional responsibilities pertaining to animal research safety programs including their regulatory basis and practices necessary for their effective oversight.
Keywords
A variety of risks are encountered in animal research programs. Ergonomic injuries can result from handling equipment, supplies, and animals. Strain secondary to repetitive motions (eg, cage changing activities) can result in chronic injury. Physical injuries can be sustained directly from animal bites or scratches. As with any work environment, slips, trips, and falls as well as electrical, fire, explosion, noise, and heat hazards need to be considered. Ionizing and ultraviolet radiation used in experimental modalities present potential physical hazards to staff. 1 The use of chemicals for cleaning and disinfection and in experimental procedures are prevalent in animal research programs. Likewise, infectious agents are of concern, including those used experimentally as well as those infecting research animals or contaminating biological materials. 2 Personnel exposed to animals and their environment are at risk of developing laboratory animal allergies, which, if not prevented or appropriately managed, can become severe or career-ending. While most research activities utilize common species such as mice within controlled environments, risks inherent to less commonly utilized animals (eg, birds, aquatics, amphibians, and reptiles) may be poorly described and should not be overlooked when developing and managing safety programs. In addition, newer technologies, such as the use of nanomaterials, can present novel or undefined risks that must be investigated and assessed.
This article focuses on institutional responsibilities for occupational health and safety oversight in animal research programs. The presence of a robust institutional occupational health and safety program (OHSP) is central to preserving personnel safety from the complex and varied hazards that can be encountered in the conduct of animal research. Management of OHSPs generally requires the interaction of numerous offices, units, and individuals, each responsible for a component of the larger, comprehensive program. Essential program elements include appropriately charged and empowered oversight entities, recognition and assessment of risk in the work environment, well-designed and achievable operating practices, thoughtful disaster planning, effective staff training, efficient and clear documentation systems, and comprehensive occupational health programs. Ultimately, an institutional culture driven by leadership that expects and supports safety practices and responsibility from all levels of personnel is integral to achieving an efficient and successful OHSP. 3
Regulations and Guidelines
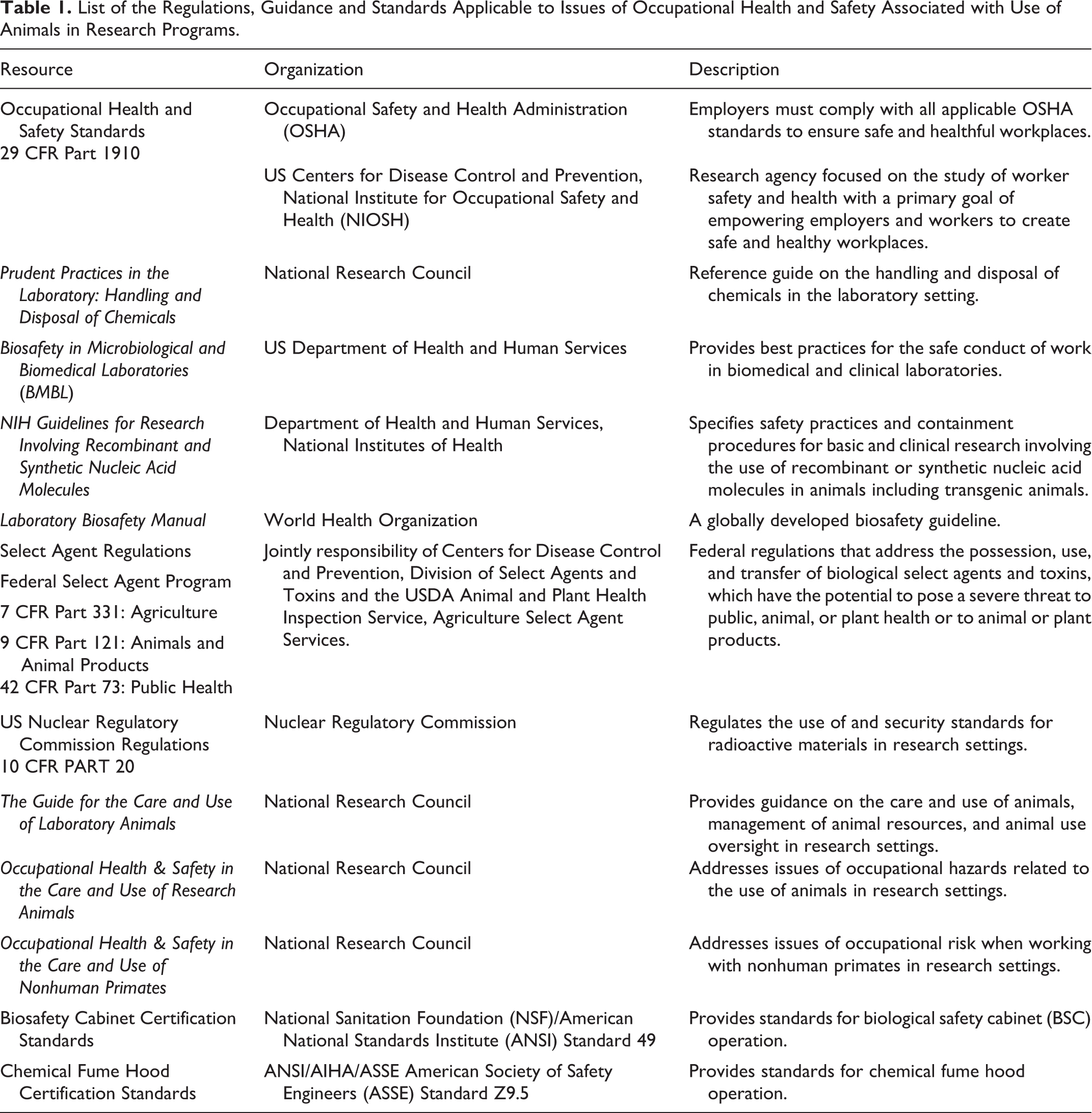
According to the Occupational Safety and Health Administration (OSHA), employers are required to provide safe and healthy working conditions for their employees. 4 In addition, institutions are responsible for identifying and minimizing workplace risks and ensuring compliance with multiple local, regional, state, and federal regulations designed to protect employee health. 1 Federal regulation is provided for occupational health and safety issues in general industry, hazardous substances, the use of select agents, and the use of radioactive materials.4 -9 In addition, a variety of guidelines and informational resources address risks to personnel safety in animal research programs (Table 1).1,10 -17
List of the Regulations, Guidance and Standards Applicable to Issues of Occupational Health and Safety Associated with Use of Animals in Research Programs.
Assessment and Management of Risk
Institutions have numerous responsibilities for assessing and managing risks associated with animal research activities. Risk assessment and management activities should be done in collaboration with researchers (principal investigators and research technicians), animal resources, and veterinary personnel; environmental safety and occupational health professionals; and institutional committees including the Institutional Animal Care and Use Committee (IACUC), IBC, and safety committee(s). The safe conduct of animal research requires that potential sources of risk be identified, quantified, and effectively controlled to safeguard human health and to protect the environment. Risks should be assessed for individuals with direct animal contact as well as for those exposed to animal environments, equipment, or waste. Nevertheless, while the protection of human health is paramount, concurrent support of animal health and welfare and facilitation of research are vital and should not be overlooked.
Hazard Identification and Risk Assessment
The provision of a safe work environment first requires identification of potential hazards followed by an assessment of their associated risks to personnel. As described above, a wide range of hazards may be encountered in the conduct of animal research, including hazards specific to experimental procedures and the research setting (eg, field studies, animal facility), as well as those inherent to animals and animal handling. All potential hazards should be evaluated by appropriate institutional review entities prior to their use in an institution’s animal research program. Multiple factors should be considered when estimating the level of risk posed by an identified hazard. These include the severity of the hazard; the intensity, duration, and frequency of personnel exposure to the hazard; the health status, work practices, laboratory and animal-handling experience, knowledge, and attitudes of personnel; and the history of occupational illness and injury in the same or similar workplaces.1,14
Risk assessment requires a balanced approach based on available data and avoids an overestimation of risk leading to unnecessary burden and expense. However, while assessing the risks inherent to common or well-characterized hazards may be clear and straightforward, assessing risks from novel or poorly characterized substances (eg, reemerging or novel microbes) or procedures can be especially challenging. In these situations, information should be sought from many resources including the literature, subject-matter experts, professional organizations, and other institutions. When insufficient information exists to fully define the hazard, professional judgement and a conservative approach should be applied to protect human health and the environment.
Institutions should ensure that the hazard identification and risk assessment processes are conducted by personnel who independently or collectively possess the appropriate knowledge and experience relevant to the research activities, the animal subjects, and the animal and research environments. Furthermore, institutions should ensure that the hazard identification and risk assessment processes are repeated at sufficient intervals so that continuing advances in both scientific knowledge and recognized safe work practices can be applied to both new and ongoing research activities.
Risk Management
Following the identification and assessment of risks, a combination of methods can be implemented to minimize and manage risks to below an acceptable level. As detailed in the Centers for Disease Control and Prevention BMBL 5th edition, three main strategies are utilized to manage risks associated with biological hazards: engineering controls, administrative controls, and use of personal protective equipment (PPE). 11 These strategies can often also be applied to manage risks from other sources including chemical and physical hazards such as radiation, compressed gases, lasers, electrical equipment, and thermal sources.
Engineering controls “protect workers by removing hazardous conditions or by placing a barrier between the worker and the hazard” and include primary containment devices such as biological safety cabinets and fume hoods; waste anesthetic gas scavenging systems; sharps management devices such as rigid, sealed sharps disposal containers and self-sheathing needles; and building-level airflow controls such as airlocks and directional airflow. 1 Institutions are responsible for ensuring that engineering controls function properly and are tested and certified as required. For example, in the United States, biological safety cabinets and chemical fume hoods should be certified in accordance with the National Sanitation Foundation (NSF)/American National Standards Institute (ANSI) Standard 49 and ANSI/AIHA/American Society of Safety Engineers (ASSE) Standard Z9.5, respectively.16,17
Administrative controls include work practices designed to minimize exposure risks and consequences to personnel. Personnel must have sufficient knowledge and training to work safely with animals, to recognize risks associated with the hazards they may encounter, to understand the rationale behind prescribed safety practices, and to effectively implement practices and standard operating procedures. Institutions should ensure that personnel possess these skills and knowledge and receive sufficient training to maintain and expand them, as needed. When possible, institutional procedures and policies should be documented and easily accessible to all personnel in written form. 14
PPE should be used when both engineering and administrative controls are insufficient to adequately minimize risks. Institutions should ensure that sufficient supplies of appropriate PPE are available to personnel and that personnel are adequately trained in their use, including appropriate donning and doffing techniques and procedures. When directed by regulations or standards, institutions must provide employees access to health screening, monitoring and protection programs. For instance, institutions may be required to provide enrollment in respiratory protection programs to employees who wear respiratory protection, or implement a hearing conservation program for personnel who wear hearing protection in noisy environments. 4 As with administrative controls, training should be provided to personnel to ensure their understanding and proper utilization of protections provided through use of PPE.
Physical injuries such as ergonomic and repetitive motion injuries, as well as injuries associated with equipment use (eg, bulk cage wash and sterilization equipment, robotic devices, mechanical lift devises, and ladders) and from slips, trips, and falls are of significant concern in animal research. Specially trained occupational health professionals, such as ergonomists and industrial hygienists, can help animal care and research personnel avoid and minimize these injuries. Injuries may be further reduced or avoided through changes to the workplace environment, use of specially designed equipment, or modified operational practices. Institutions should recognize the risk of physical injuries in the animal research environment and ensure that sufficient resources are available to minimize them.
While personnel are themselves responsible for following institutional safety practices and utilizing required safety equipment, institutions are responsible for ensuring that personnel’s use of administrative controls, PPE, and safe work practices are consistent with institutional requirements. To support personnel in these efforts, institutions should provide facilities and supplies to facilitate personal hygiene, such as changing rooms and personnel showers. 14 Institutions should also ensure that risk management practices are periodically reassessed and refined as needed in light of current scientific knowledge, technological advances, personnel experience, and modifications to laboratory practices and methods.
Disaster Planning and Emergency Response
Animal research facilities must possess a written emergency response plan to appropriately manage research animal populations and minimize animal pain, distress, and deaths in the event of a disaster or emergency. 14 These plans should be consistent with and developed in accordance with institutional emergency response plans designed to preserve human health and safety, institutional assets, and security. Plans should emphasize those events most likely to occur in the respective geographic region and include considerations specific to the types of animals, facilities, and research at the institution (eg, wildlife species, biocontainment facilities). Events include extreme weather-related events (eg, floods, tornados), facility system failures (eg, loss of facility ventilation function), excessive employee absenteeism, terrorism, illegal activities, and accidents (eg, chemical, biologic, or radioactive spills). Facility personnel should be trained to effectively implement emergency response plans and be periodically assessed for their ability to do so. Plans should clearly define the roles and responsibilities of individuals and groups that are internal as well as those that are external to the institution, such as emergency responders and law enforcement. When participation by external entities is anticipated, it is of paramount importance that institutions preemptively establish contracts or agreements with the entities and that emergency plans specify designated points of contact and expected means of communications during an emergency event. Institutions should evaluate animal facility emergency response plans and ensure that they are effectively integrated into and supported by the institution-wide emergency plan. This includes ensuring that environmental health and safety personnel, medical professionals, emergency responders, and law enforcement are adequately trained to provide an appropriate emergency response and are sufficiently familiar with the physical structure of the facility and surrounding areas to do so.
Institutional Occupational Health and Safety Programs
Institutions that care for, use, or produce animals for research, testing, or teaching must establish and maintain an institutional OHSP designed to protect employees from work-related injury and illness.14,18 The structure and components of an OHSP should be customized to an institution’s needs and resources and reflect the size and scope of the animal care and use program as well as the risks posed by research activities. 1 While differences exist across institutions, common emphases of OHSPs for animal research programs include the prevention of laboratory animal allergen sensitivity and allergies, zoonotic disease transmission, ergonomic and physical injuries, and chemical or radioactive substance exposure. Strong emphasis is also placed on personnel education. In addition, oversight of programs for waste management, regulatory compliance, and emergency management are also commonly encompassed by OHSPs.
Enrollment of individuals in an OHSP program should be determined based on a risk assessment of their potential exposure to hazards. Individuals to be considered include animal care, veterinary, and research personnel, as well as students, volunteers, building maintenance personnel, janitorial staff, security personnel, and vendors. All individuals with significant exposure to animals, their tissues, their environments, or their waste should be included. Furthermore, service provided to individuals should be consistent across those with similar levels of risk. 19
Another key component of an OHSP is an institution’s occupational health care services that function to minimize risks to personnel health and to promptly detect and effectively treat injuries or illnesses when they occur. To this end, implementation of a robust illness and injury reporting system and active health surveillance strategies can be instrumental in detecting trends of injuries (eg, needlesticks) or illnesses (eg, allergy development) that may otherwise be overlooked within a population. Common elements of occupational health care services include personnel medical and health history evaluations, medical case management, adverse health outcome analyses, nonoccupational health care services, vaccination programs, and medical recordkeeping.1,14
In the context of occupational health care services, institutions are responsible for ensuring that health care providers are sufficiently qualified for their roles through prior training and experience in occupational health and either possess or have means to access appropriate expertise relative to the expected hazard types present at the institution. 1 Moreover, the level of health care services provided to personnel should be consistent with risk assessment findings. 2 Institutions are responsible for communicating to external employers the risks, potential hazards, and necessary safety measures pertinent to contract personnel delivering services to the institution. Furthermore, institutions should confirm that external employers provide a comparable level of occupational health care services to these contract personnel as the institution provides to their own employees with similar duties. 20 It is also an institution’s responsibility to protect personnel’s medical records and health information consistent with the Health Insurance Portability and Accountability Act. 18
To support an effective OHSP, institutions should actively support and promote incorporating safety into the organizational culture and provide clear procedures for personnel to report injuries, illnesses, near misses, and changes in their health status (eg, pregnancy, immunosuppressive disease) without fear of disciplinary actions or loss of employment. Reported events should be promptly evaluated by stakeholders to identify the root cause of the incident and, when indicated, a job safety analysis performed by safety personnel.
Roles and Responsibilities
Due to the complexities and often-broad range of animal care and research activities within animal research programs, utilization of a team approach is recommended for the design and implementation of OHSPs. As stated in the publication Occupational Health and Safety in the Care and Use of Research Animals, “An effective occupational health and safety program depends on the involvement and commitment of program managers at all levels. Key managers [are] those who have specific expertise in health and safety issues or who [are] charged with and have the authority to implement and enforce components of the program these may include; health professionals, safety professionals, veterinarians, animal-facility managers, research directors and scientists, laboratory supervisors, human resource and financial personnel, legal advisors, environmental experts, and facility engineers.” 1 The collaborative efforts of these individuals are critical to implementing and maintaining a functional OHSP that satisfies the program objectives. Below we review a number of key individuals and committees integral in the operation of OHSP function and management.
Principal investigators (PIs) are responsible for identifying potential hazards associated with their research. PIs should communicate hazard information to relevant groups (eg, EHS, IBC, veterinary, and animal resources personnel) and collaborate with them to effectively mitigate risks while maintaining scientific validity and animal welfare. As the leader of the research team, the PI is also responsible for ensuring that research personnel are appropriately trained before conducting research activities.
Environmental health and safety (EHS) professionals provide technical services that assist the institution in meeting its regulatory and legal responsibilities associated with health and safety. As a group, EHS professionals possess expertise in multiple disciplines such as chemical safety, biological safety, physical safety, industrial hygiene, health physics and radiation safety, engineering, environmental health, fire safety, and toxicology. As a result, they are instrumental in the design and implementation of an institution’s OHSP through their central roles in hazard identification, risk assessment, and development of risk mitigation practices.
Health care professionals, such as physicians and nurses, participate in many levels of the OHSP. They have valuable expertise that is important in assessing the risk of hazards and their impact in human health. They are essential contributors to the development of medical surveillance programs and perform medical evaluations of personnel. Health care professionals can also significantly contribute to development of training materials regarding occupational health risks.
Animal resource personnel, including the veterinary team and husbandry staff, provide expertise on issues related to risks posed to personnel through animal handling and housing environments. For instance, veterinary expertise is integral in assessing an animal’s potential to cause physical injury, or be infected with or shed a zoonotic organism. Veterinarians oversee diagnostic and preventative health programs to monitor for zoonotic disease. Animal resource personnel also have expertise in specialized animal housing and containment practices that are integral to mitigating risk to employees. Last, animal resource personnel often provide training to animal users regarding safe animal handling practices.
The NIH Guidelines for Research Involving Recombinant or Synthetic Nucleic Acid Molecules (NIH Guidelines) mandates that institutions establish an institutional biosafety committee (IBC) to oversee research involving recombinant or synthetic nucleic acid molecules. 12 The NIH Guidelines specifically identifies individuals with unique expertise to constitute the IBC, including individuals with animal expertise. Although not federally mandated, some institutions designate the IBC to evaluate and risk assess research involving other potential hazards including biological agents, toxins, chemicals, or nanoparticles in biological systems
The primary role of the IACUC is to oversee all components of an institution’s animal care and use program. Since the OHSP is an essential part of the animal care and use program, the IACUC must ensure it functions according to the pertinent standards, and facilitates a safe and healthy work environment for animal handlers. 14 Accordingly, it is the IACUC’s responsibility to evaluate an institution’s OHSP as part of its regular schedule of program review and facility inspection. 14
In accordance with federal law, institutions are required to establish a radiation safety committee (RSC) to oversee research involving radioactive materials. 8 The RSC for research establishes and oversees compliance with policies or standards of practice; reviews and approves proposed use of radioactive materials, sources, location, and training; and reviews safety incidents and dose records for personnel.
Many institutions find establishment of a safety committee, while not a required entity, to be a valuable component of OHSP oversight. Safety committees may provide a holistic and collaborative insight into all of the risks present at the institution that are otherwise overseen by separate entities.
Harmonization of Efforts
Clearly, a robust OHSP requires the coordinated efforts of many individuals, programs, committees and oversight bodies. It is critical to harmonize the efforts of specific roles within the program. For example, a PI may propose a project that involves the use of radionuclides in transgenic animals. This research requires input and interaction from multiple groups who contribute to OHSP management, including (1) the IBC to consider transgenic animal use; (2) the RSC to review the use of radioactive materials; (3) health professionals and veterinarians to consider the potential risks of zoonosis; (4) EHS, veterinary, and animal resources personnel to identify the safety practices and equipment for use when handling the animals; and (5) the IACUC to review animal research proposals. It is essential that effective communication and clear roles and responsibilities are established to support a smoothly functioning system and successful incorporation of safety and compliance into the institutional culture. 2
Training Programs and Competency Assessment for Personnel
A successful training program should include assessment of who needs training, training content, when and how training should be delivered, standards for successful completion of training, how training is documented, and how training effectiveness is assessed. “Occupational Health & Safety objectives of an institution can be achieved only if employees know the hazards associated with their work activities; understand how the hazards are controlled through institutional policies, engineering controls, work practices, and personal protective equipment; and have sufficient skills to execute safe work practices proficiently.” 1 Personnel performing procedures and those with direct exposure to potentially hazardous activities are clearly the primary target of safety training programs. Ideally, all levels of personnel involved in supporting animal research programs should have training to understand institutional expectations and safety oversight entities, and to understand their role and potential risks. “A critical component of a successful safety program is designing and delivering effective training for employees at all levels.” 3 An institutional safety program is most likely to succeed when risks, responsibilities, communication networks, and emergency response plans are well understood and supported by all personnel within the organization.
Training content should communicate safety risks present in the program; appropriate work practices to prevent injury or illness; regulatory compliance expectations and entities; rights and resources for occupational health care; methods for reporting of unsafe situations; practices, illness, or injury; and emergency response plans. The Guide for the Care and Use of Laboratory Animals states that “personnel at risk should be provided with clearly defined procedures and, in specific situations, personal protective equipment to safely conduct their duties, understand the hazards involved, and be proficient in implementing the required safeguards. They should be trained regarding zoonoses, chemical, biological, and physical hazards (eg, radiation and allergies), unusual conditions or agents that might be part of experimental procedures (eg, the use of human tissue in immunocompromised animals), handling of waste materials, personal hygiene, the appropriate use of PPE, and other considerations (eg, precautions to be taken during pregnancy, illness, or immunosuppression) as appropriate to the risk imposed by their workplace.” 14
Training programs should include both initial onboarding training and continuing education programs. Ideally, training should occur prior to provision of access to animals, facilities, and hazardous situations or materials. Training should be oriented to adult learners and be tailored to the expected audience, taking into account such things as their level of understanding, previous experiences, and job duties. 4 It should be recognized that the time dedicated to training personnel is a valuable investment in long-term work productivity and compliance.
Evaluation methodologies should be planned along with the assessment of training needs and development of content. As such, criteria for competency should be defined as part of the design of the training program. Comprehension and competency may be assessed in the form of written or verbal tests, or by successful performance of a required task. Evaluating comprehension and competency during or immediately following training allows for immediate reinforcement of materials provided to learners. It also allows for assessment of effectiveness of the training experience. Further evaluation occurs subsequent to initial training events. On-the-job practices can vary a great deal in the active workplace compared to a training environment. Worksite assessments gathered subsequent to initial training can assess the long-term retention of knowledge and skills. Training programs should also evaluate instructors, training content, and training structure on a regular basis to ensure that training fulfills the need of the organization and individuals. 4
Documentation of training is an essential component of any training program. 21 Documentation should include individual personnel identification, training topic, date, and assessment results. Ideally, documentation should be available to the learner and to compliance bodies, and shared among responsible divisions within the learner’s organization. 21
Institutional recognition of the value of training and support for enriching training activities helps to encourage engagement of personnel and educational programs. “In an environment where a ‘safety culture’ exists, training is considered a critical part of the job.” 3
Institutional Support
Oversight of safety in animal research programs is an institutional responsibility that entails not only accountability for appropriate practices but also robust support for effective program implementation and management. To accomplish the mandate to maintain an OHSP, the institution must commit both fiscal and human resources to the process. The institutional leadership must establish and empower oversight entities and personnel (eg, IACUC, IBC, PI, EHS) with responsibility and authority for direction and management of the safety program. Engagement of professionals with expertise in risks present at the institution as well as administrative and support staff should be provided to support program activities. Program evaluation should occur on a routine basis to ensure appropriate compliance and successful programmatic outcomes. Ultimately, OHSPs thrive in an institutional culture that values and supports safety initiatives.
While structure and conduct of the OHSP are often executed by EHS and occupational health services personnel, the IACUC is ultimately responsible for its oversight in support of animal research programs. To accomplish this expectation, the IACUC reviews the components of the OHSP at least once per year or as often as required. 14 In addition, EHS and other responsible entities should also conduct regular assessments to ensure relevant personnel adhere to OHSP requirements.
The OHSP must include individuals who are qualified to identify, assess, and implement appropriate measures to mitigate potential risks associated with animal research programs. For example, engineering controls used to minimize exposure to potential hazards require certified professionals that ensure relevant systems and equipment are appropriately maintained and effective. Health professionals should be available to perform medical evaluations and provide preventative health care modalities and develop educational material. 14 Individuals qualified to train animal handlers on program-related risks should be part of the overall OHSP team. Human resources and legal personnel should also be identified and engaged to provide consultation when required. 1
A diversity of documents such as institutional policies and practices, research proposals and committee approvals, personnel training and medical records, disaster plans, and injury and illness reports are often maintained by separate responsible entities that are part of an OHSP. It is essential that the program be supported by robust and agile administrative support staff and information management processes. Clearly defined roles and responsibilities, and effective and timely information sharing can best support a well-integrated and successful program. The lack of an effective workflow between entities responsible for aspects of the OHSP may cause unnecessarily interruption of research progress and in effect dissuade individuals from fully engaging in the program. For example, PIs who experience excessive delays when requesting assistance from institutional research oversight entities (eg, IBC, IACUC, EHS, RSC) or who receive conflicting information from multiple entities may be less likely to engage with the entities when assistance is needed for new projects. Personnel who experience poor support or inconsistencies in information regarding risks and appropriate practices may be less likely to engage in appropriate safety practices or report concerns with safety programs. As reported by Kinman et al, “persons involved in laboratory accidents tend to have low opinions of safety programs, to take excessive risks, to work too fast, and to be less aware of the infectious risks of the agents they are handling.” 22 The complexity of oversight mechanisms, management, and administrative support necessary for an OHSP can create a burdensome administrative process if roles and responsibilities are not clearly defined and efficiently executed. At a minimum, the system should have the capability to track past and planned activities (eg, trainings and surveillance events), facilitate exchange of information between necessary individuals (eg, medical and health and safety professionals), and permit data analysis and retention. 2
Incorporating Safety and Compliance into Institutional Culture
Institutions should establish practices that encourage and ultimately reflect a culture of partnership and mutual respect among individuals actively engaged in animal research activities. An institutional culture driven by leadership that expects and supports safety practices and responsibility from all levels within an organization is necessary to achieve an efficient and successful safety program. 3 For an institution to perpetuate an enduring incorporation of safety into the culture rather than simply an expectation for ongoing compliance, individuals associated with the safety program must recognize and understand the identified potential hazards, risks and related mitigation practices. The various stakeholders of a safety program must support institutional decisions on policies and mitigation practices employed to protect personnel from potential hazards. When safety policies and practices are developed by teams consisting of members of the scientific community along with safety professionals, those standards are more likely to be employed by personnel in laboratory settings. Ultimately, personnel performing activities must be well informed in expected practices and feel empowered to report health or safety concerns without fear of discipline or reprisal.
Conclusion
Research programs utilizing animal models represent a variety of potential risks to personnel safety. These risks range from standard physical, chemical, or infectious hazards to more modern and less defined concerns associated with new technologies. Institutions must provide appropriate oversight, personnel empowerment, and fiscal and administrative support to ensure effective OHSPs. Multiple individuals and groups serve key roles in supporting safety within animal research programs. The establishment of clear roles and responsibilities as well as effective communications between these individuals and groups are essential factors for the success of an OHSP. In addition, safety programs should not be regarded as static, but rather as dynamic and continually evolving to best serve the needs of both the institution and its personnel. 2 Above all, incorporating safety and compliance into the institutional culture is of paramount importance and requires the commitment and support of individuals at all levels within an institution including senior leadership and health, safety, research, veterinary, and animal care personnel.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
