Abstract
The disposal of culture media containing antibiotics is a daily practice in research laboratories at any level (ie, academia, industry, federal/state). Generally, good microbiological practices recommend that the biohazard be treated before disposal of the culture media. This brief summary aims to provide a different angle in the waste management of culture media containing antibiotics from the perspective of handling the biohazard (microbiological agent) and the chemical hazard (antibiotic) before disposal. Available practices and suggested discussion points are presented to initiate conversation among biosafety professionals and within institutions.
As a biosafety professional, have you asked yourself what happens to the antibiotics used for in vitro culture when the culture media is disposed down the drain after inactivation of any biohazard? The goal of this overview is to briefly bring information available to help start the conversation at your institution regarding inactivation of an antibiotic present in culture media before it enters the waste stream.
Why should the biosafety professional worry about disposing of antibiotics present in culture media?
Therapeutic drugs are regularly found in the environment. The most frequent sources of contamination are health care facilities and animal farming. 1 There is evidence that commonly used antibiotics can be found in drinking water, river water, and river sediments. 2 Thus, inactivation or removal of antibiotics in water reservoirs is imperative to avoid harm to human health. The media-antibiotic combination could ultimately become part of the public water streams unless the water treatment plants are able to inactivate the antibiotics as part of the required treatment for organic chemical compounds. Also, from a public health perspective, overuse or abuse of antibiotics is known to influence microbial resistance. 3
Is there any guidance on how to dispose of or inactivate antibiotics in culture media?
Federal Law
In the United States, the Environmental Protection Agency (EPA) charged a working group with the task of compiling information on contaminants of emerging concern. The working group drafted a white paper in response to this request.
4
The draft document starts with the definition of “chemicals of emerging concern” to include 2 characteristics: (1) the chemical is not included in an existing regulation, and (2) the chemical has been “discovered” in natural streams. The draft also has broad categories, 2 of which could include antibiotics used in culture media:
Pharmaceuticals and personal care products, including a wide suite of human prescribed drugs (eg, antidepressants, blood pressure), over-the-counter medications (eg, ibuprofen), bactericides (eg, triclosan), sunscreens, synthetic masks Veterinary medicines such as antimicrobials, antibiotics, antifungals, growth promoters and hormones
4
The EPA requested its Scientific Advisory Board (SAB) to opine on the scientific validity of the white paper. The SAB recommended that the determination of aquatic life criteria be risk based per the criteria included in the EPA’s 1998 guidelines for ecologic risk assessment and the more recent advice to the EPA on the application of the ecologic risk assessment. 5 Furthermore, in a 2009 response to the SAB, the EPA stated that it was planning a “technical support document,” including the aquatic life criteria to be released for comment in 2009. As of July 2017, a notice of proposed rule making has not been published in the Federal Register on this issue. However, the EPA has issued technical guidance for certain chemicals. 6 Notably, the EPA’s technical fact sheets, which are specific for certain chemicals of concern, do not include antibiotic compounds.
Water Treatment Plant Technology
The EPA is charged with ensuring that individuals in the United States have safe drinking water. Congress delegated such authority through the Safe Drinking Water Act (SDWA), which mandates the establishment of a list of contaminants and their corresponding maximum contaminant levels and maximum contaminant level goals. The list of contaminants can be found on the EPA’s website for the national primary drinking water regulations (https://www.epa.gov/sites/production/files/2016-06/documents/npwdr_complete_table.pdf). Within this list, contaminants are classified as disinfectant, disinfectant by-product, inorganic chemical, microorganism, organic chemical, or radionuclide, but antibiotics are not listed as contaminants. Under the SDWA, water treatment plants are required to use appropriate technology to inactivate or remove such contaminants. Generally, antibiotics used in culture media would be classified as organic chemicals. Some of the technologies available to small treatment plants to inactivate organic chemicals, some are already in use, include powdered activated carbon. Studies have already been published showing promising results in the removal of various antibiotics from water to improve its quality; however, the studies also recognize that additional data must be collected.7,8
The EPA has generated 4 contaminant candidate lists since 2003. The purpose of these lists is to periodically review the chemicals that are not subject to the SDWA standards; thus, these chemicals are not listed in any proposed or promulgated SDWA regulation. The third list, promulgated in 2009, included erythromycin based on its “potential adverse health effects and potential for occurrence in public water systems.” 9
States
In the United States, states are encouraged to develop and run their own hazardous waste program; otherwise, the EPA runs the program for the state. Within the scope of the SDWA, water treatment plants must comply with the standards set by EPA. Institutions generating hazardous waste must meet the requirements of the state or federal plan.
Institutional
In the United States, the National Institutes of Health posted a waste disposal guide online in 2014 that directly addresses culture media containing antibiotics.
10
The guidance document indicates the following:
Before disposal, cell culture media must be decontaminated (see MPW Section for instructions) either by steam autoclave or adding disinfectant directly to vessel or treating pooled spent media. Decontaminated media must be collected as chemical waste and called for pick up by Chemical Waste Services. Dispose of empty, decontaminated cell culture vessels in the Disposable Labware & Broken Glass Box.
Other resources provide specific recommendations on disposal of cell culture media with antibiotics. For example, the Ecole Polytechnique Federal de Lausanne, Switzerland, has provided such guidance. 11 In the guidelines for the management of biomedical waste, the Ecole Polytechnique Federal de Lausanne has a specific section for treatment and elimination of liquid waste containing antibiotics, hazardous chemicals, or high nucleic acid content. The recommendation provided in this guidance documents specific for antibiotic-containing waste media is that autoclaving is the preferred method for inactivation when certain antibiotics are used and, if autoclaving is not possible (logistically or because of the nature of the antibiotic), then the antibiotic-containing waste must be disposed of as toxic chemical products, after inactivation of the biohazard. Importantly, it reiterated that bleach not be added to cell culture media that requires inactivation by heat. 11
The University of Stockholm published an online document titled “Procedures for the Disposal of Liquid Chemical Residues and Aqueous Solutions.” This guidance document covers laboratory research and teaching laboratories within the university that are connected to a very specific part of the sewage system in the city of Stockholm. The guidance clarifies that, in general, “no hazardous substance may be poured down the drain”. 12 The guidance document includes that antibiotics such as amphotericin (Fungizone), erythromycin, Geneticin (G418), gentamycin, neomycin, puromycin, streptomycin, sulfadoxine, and tetracycline, among other specific chemicals, “may be poured down the drain” after being inactivated by autoclaving. 12
Table 1 has been updated from the original posting at the University of Gothenburg’s website under the sections “Rules for Handling Antibiotics” and “Handling Instructions” to add a few more antibiotics commonly used in culture media. 13 The recommendation provided here is for antibiotics contained in media; it should not be used for disposal of stock solutions.
Spent Culture Media Containing Antibiotics Can Be Inactivated by Heat or Disposed of as Chemical Waste.a
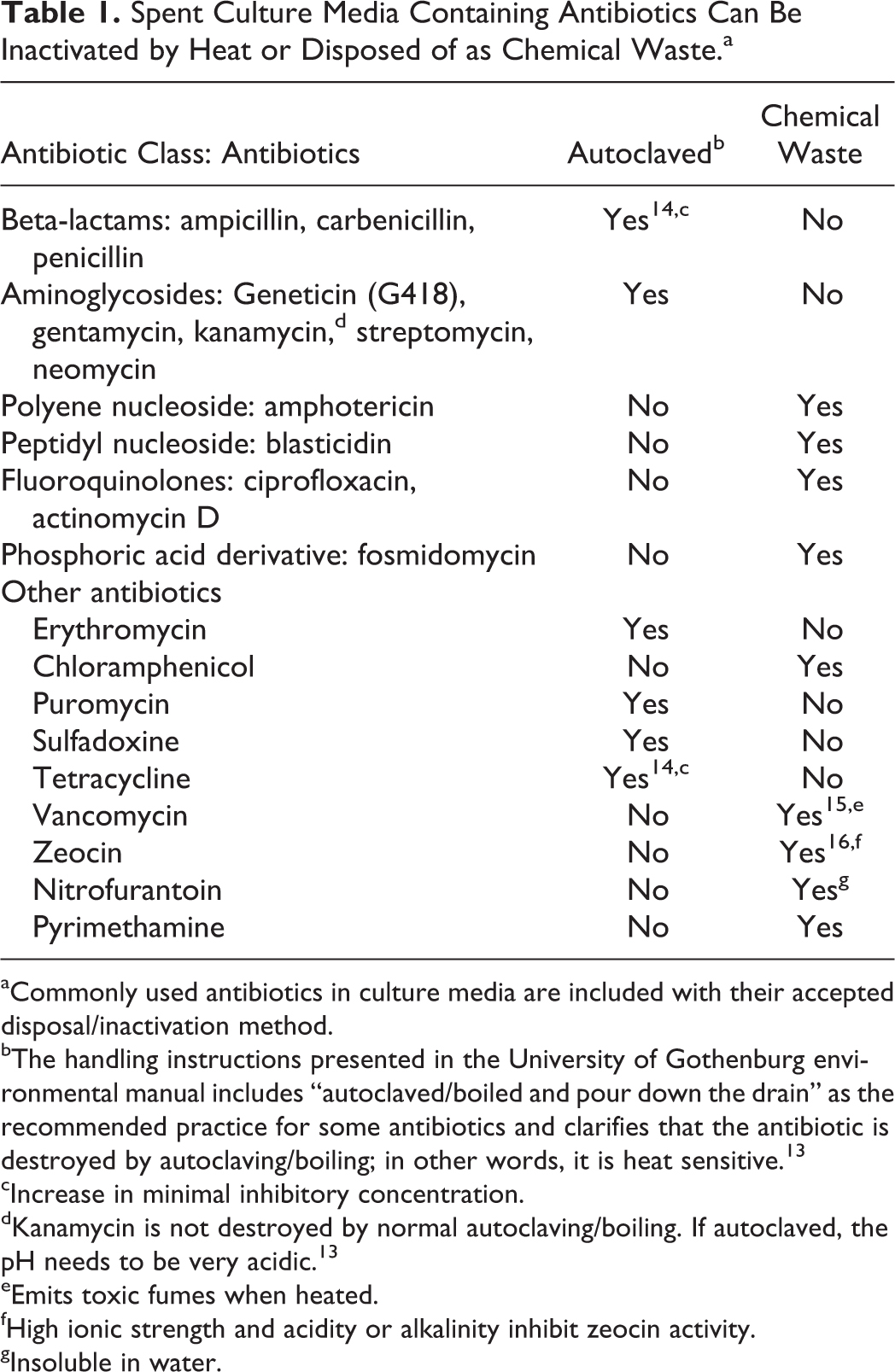
Commonly used antibiotics in culture media are included with their accepted disposal/inactivation method.
The handling instructions presented in the University of Gothenburg environmental manual includes “autoclaved/boiled and pour down the drain” as the recommended practice for some antibiotics and clarifies that the antibiotic is destroyed by autoclaving/boiling; in other words, it is heat sensitive. 13
Increase in minimal inhibitory concentration.
Kanamycin is not destroyed by normal autoclaving/boiling. If autoclaved, the pH needs to be very acidic. 13
Emits toxic fumes when heated.
High ionic strength and acidity or alkalinity inhibit zeocin activity.
Insoluble in water.
What Can We Do as Biosafety Professionals?
There are options, and some are easier to adopt than others. The following suggestions may guide the biosafety professional on how to initiate the discussion:
Risk assessment: A conversation between the research team and the biosafety officer to evaluate the risk of disposing culture media down the drain containing various types of antibiotics (what type of antibiotic?), the biological agents (what risk group? what is the nature of the pathogen?), the volumes of culture produced (is the pathogen being propagated? is it a diagnostic test?), and the infrastructure (new facility vs old facility, availability of autoclave). Evaluate risks outside biosafety: Consultation with chemical and environmental safety colleagues to ensure that local, state, and federal requirements are being followed. Initiate a guidelines draft involving the research community: The ultimate goal is to establish standard procedures for laboratories to follow when disposing antibiotic-containing culture media
While there are no specific requirements in the United States for the management of antibiotics and other pharmaceuticals in cell culture media generated in the laboratory, it could be considered a best practice to start the conversation in your institution. Disposal of culture media is going to be complex, because it contains biological and chemical hazards, it will add cost to waste management, and there will be unintended consequences. The summary provided here is just the beginning of the conversation. The community of biosafety professionals will benefit from continued sharing of lessons learned and challenges encountered while dealing with this issue and will ultimately own this subject matter.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
