Abstract
Irrigated fresh vegetables are a potential portal for dissemination of human pathogens that may be associated with irrigation water source. This study investigated the quality of irrigation water and the fresh vegetables produced in regard to pathogenic bacteria found at a selected vegetable farm in southwestern Nigeria. Enumeration and characterization of total heterotrophic bacteria and potential pathogens (mostly enteric bacteria) were done with international procedures. Susceptibility of the potential pathogens to antibiotics was by disk diffusion techniques, while plasmid profile was by polymerase chain reaction–based DNA fingerprinting. The mean values for total heterotrophic bacteria in irrigation water and fresh vegetable samples were, however, statistically comparable (P = .86). The corresponding comparison for coliform also showed no significant difference (P = .07). The isolated pathogenic bacteria genera from the irrigation water and fresh vegetable samples include Citrobacter, Enterobacter, Escherichia, Klebsiella, Pseudomonas, and Salmonella. In sum, 67.2% of the isolates were obtained from irrigation water samples, compared with 32.8% in the vegetable samples; 54.1% of the isolates were resistant to >1 antibiotic with a multiple antibiotic resistance index >0.2, suggesting an antibiotic pressurized environment. Resistance to ampicillin was very high (98.36%), whereas resistance to ciprofloxacin was very low (0.0%); 27.9% of the multiresistant enteric isolates harbored ≥1 plasmids. There is a possibility of increased numbers of pathogens on irrigated vegetables as a direct consequence of poor irrigation water quality in the study site. The use of pathogen-free irrigation water and good agricultural practices has the potential to eliminate microbial hazards in fresh vegetables.
Irrigation is the artificial application of water to land or soil food crop farming; it is used to assist in the growing of agricultural produce since nutrients are made available to plants in liquid form for their maximal growth and development. 1 Irrigation is also by far the largest component of anthropogenic demand for fresh water and, as such, constitutes an essential part of the global hydrologic cycle, as illustrated by global hydrologic model simulations. 2 However, many sources of irrigation water are subject to inputs of pathogenic loads from point and nonpoint sources stemming from multiple land uses in watersheds.3,4
Vegetable plants require irrigation to prevent plant stress during critical growth stages. In this case, irrigation is applied especially during the dry seasons in the tropics in sub-Saharan Africa. The irrigation systems employed, as well as the postharvest washing of vegetables, commonly use untreated surface water or groundwater, which is of objectionable microbial quality. If such water harbors pathogenic microorganisms, there may be health risks to consumers. Consequently, the promise of nutrition and health benefits from consumption of vegetables may be thwarted by infectious disease outbreaks.
Vegetables are among the food groups implicated with greater frequency in recent years as causative agents of enteric diseases. 5 All types of fresh produce have the potential to harbor pathogens, including but not limited to Shigella spp, Salmonella spp, enterotoxigenic and enterohaemorrhagic Escherichia coli, Campylobacter spp, Listeria monocytogenes, Yersinia enterocolitica, Bacillus cereus, Clostridium botulinum, enteric viruses, and parasites such as Giardia lamblia, Cyclospora cayetanensis, and Cryptosporidium parvum. Most of these pathogens have also been associated with food-borne illnesses.6 –9
The potential role of irrigation water in contamination of fresh vegetables is well documented. 10 The overhead spray irrigation systems employed in southwestern Nigeria have the potential for microbial contamination and spread among humans from handling practices.
Gelting and Baloch 4 reported 2 nationwide disease outbreaks in the United States that were directly linked to consumption of vegetables contaminated with pathogens from irrigation water. In the first, fresh bagged spinach from a single farm in California was implicated as the source of a 2006 E coli O157: H7 outbreak that caused >200 illnesses and 5 deaths. Groundwater used as irrigation water and its potential contamination by surface water recharge were identified as the most likely water-related contributing factors involved in this outbreak. In the second, iceberg lettuce served in chain restaurants was identified as the vehicle of transmission for a different E coli O157: H7 outbreak in 2006. Samples from environmental investigations also revealed a genetic match between the outbreak strain and that of the irrigation water systems.
Fresh vegetables are either consumed raw or cooked as part of a main dish in many countries around the world. One would expect that cooking would have effectively eliminated microbial contaminants usually found in agricultural irrigation systems that are capable of causing diseases in consumers. However, passive handling through constant touch during the supply chain before cooking may result in dissemination of potential pathogens.
The passive handling of fresh vegetables is usually performed by (1) farmworkers, who wash produce with irrigation water and pack them in an unhygienic manner for retail and transport to point of sale, or (2) consumers, who may engage in the habit of touching several vegetables before selecting the one they wish to purchase. These methods are especially significant in developing countries where fresh produce sale occurs in open-air markets. The common salutation of shaking hands may lead to the spreading of viruses, bacteria, and other pathogens, especially when improper hand-washing techniques occurred before handling produce and other food items. It may also contribute to cross-contamination of food items that might be consumed unwashed and uncooked. Some food-borne illness outbreaks have been attributed to such poor hand hygiene practices of food handlers. 10
There is likely a large number of unreported cases of gastrointestinal infections that may have been due to contact with irrigation water-related microbial contaminants through passive handling of vegetables. 3 It therefore speaks to the need for more scientific data linking illness to direct consumption of unwholesome food or water.
Research on the potential effects of irrigation water quality on food safety requires a system-based assessment of samples of irrigation water and the corresponding irrigated fresh vegetables. This study investigated the quality of irrigation water used in agricultural settings, the potential pathogenic bacterial load in irrigation water, and the potential contamination of fresh vegetables grown and harvested in a major vegetable farm in southwestern Nigeria.
Materials and Methods
Sampling Site and Sample Collection
The study site is one of several farms selected for the Nigeria-Canada Vegetable Project (NICANVEG Project 106511), an effort to enhance rural food security through sustainable production and consumption of underutilized Nigerian vegetables.
The site is located along Akanran Road (N07.31601º and E003.99900º) in the densely populated city of Ibadan, Nigeria. The site comprised 5 vegetable fields that are laterally arranged around the Omi River. The river water is used for irrigation, postharvest washing of the vegetables, and other domestic activities. There are several other vegetable farms along the river way that are not associated with this study.
Water samples were drawn from the Omi River, which is used to irrigate and wash the 3 most cultivated vegetables in the NICANVEG: Amaranthus dubius (spinach), Solanum macrocarpon (African eggplant), and Telfairia occidentalis (fluted pumpkin).
Seven surface water samples (approximately 1 L each) were obtained from the river via a dip sampler made of polyvinyl chloride and stored in 1-L sterile plastic containers. Water samples were taken upstream, midstream, and downstream of the vegetable fields. Four composite vegetable samples (approximately 100 g per sample) were obtained: A dubius (1 sample), S macrocarpon (1 sample), and T occidentalis (2 samples). The vegetable samples were stored in sterile resealable bags and labeled appropriately. All the samples were kept in coolers stocked with ice packs before and during transportation to the laboratory where they were maintained at 4ºC before further processing.
Enumeration and Characterization of Bacteria in the Samples
Total heterotrophic bacteria (THB) and coliform bacteria counts in fresh vegetable and irrigation water samples were carried out according to standard methods.11 –13
Vegetable samples were prepared for THB and coliform bacteria count by carefully weighing 25 g of each sample into 225 mL of phosphate-buffered saline, which served as stock for 10-fold dilution in 9 mL of Ringer’s solution. A 100-µL aliquot of 10-3 to 10-6 dilution factors were then spread plated onto nutrient agar (Oxoid, England) for THB and on MacConkey agar (Oxoid) for coliform bacteria in triplicates.
For THB counts in irrigation water samples, 1 mL of each sample was added to 9 mL of Ringer’s solution and then serially diluted to 10-3. A 100-µL aliquot of 10-3 to 10-6 dilution factors was then spread plated onto nutrient agar plates in triplicates. However, a membrane filtration technique was used for coliform bacteria count in irrigation water samples. About 100 mL of the water sample was filtered with 0.45-µm membrane filter (Millipore), and the filter was aseptically transferred onto the MacConkey agar plates in triplicates. All plates were then incubated at 37ºC for 24 to 48 hours. The colony-forming units per milliliter (CFU/mL) was calculated with the following formula:
Discrete colonies were subcultured and purified on nutrient agar plates for the development of axenic isolates, which were stored on nutrient agar slants for subsequent characterization and identification tests at 4ºC ± 2ºC. 11
Identification of all bacterial isolates was by microscopic, morphologic, and biochemical characterization.14 –16 The biochemical tests were interpreted to determine the presumptive nomenclature of the potential pathogenic bacteria isolates (mostly enteric) through Bergey’s Manual of Determinative Bacteriology and ABIS online (Advanced Bacterial Identification Software).
Antibiotic Susceptibility Testing of Bacteria Isolates
The antibiotic susceptibility testing was performed with the Kirby-Bauer agar disc diffusion method.17,18 The antibiotics used in this study (Oxoid) included ampicillin (10 µg), amoxicillin (30 µg), chloramphenicol (30 µg), colistin (10 µg), ciprofloxacin (5 µg), gentamicin (10 µg), nalidixic acid (30 µg), nitrofurantoin (300 µg), tetracycline (30 µg), and trimethoprim/sulfamethoxazole (1.25/23.75 µg).
Five to 8 colonies from 18- to 24-hour-old culture of each isolate were inoculated into 5 mL of sterile Ringer’s solution to give a turbidity equivalent to 0.5 McFarland standards. A sterile cotton-tipped applicator (Evepon, Nigeria) was inserted into each inoculum tube, with excess fluid removed by rotating the cotton swab against the side of the test tube, and swabbed onto the entire surface of the Mueller-Hinton agar plate, which was then incubated at 37ºC for 24 hours.
The antibiotic test disks were then placed aseptically on the surface of the inoculated agar plates via an 8-place disc dispenser (Oxoid) and incubated for 18 to 24 hours at 37ºC. The diameters of the zones of inhibition on the plate were measured to the nearest millimeter and results interpreted through a standard table that relates the inhibition zone diameter to the degree of bacteria resistance. 19 Enterobacter aerogenes (American Type Culture Collection, Rockville, Maryland) was used as control organism. Multidrug resistance among the isolates was defined as resistance to ≥1 agent in ≥3 antibiotic classes.
Plasmid Profiling of Bacteria Isolates
The isolates were subjected to plasmid profile analysis via the TENS/miniprep method.20 –22 An overnight culture of each bacteria isolate was prepared in 5 mL of nutrient broth. The broth culture was properly mixed by vortexing, and 1.5 mL was then transferred into a prelabeled Eppendorf tube.
The tubes were then centrifuged for 2 minutes at 13 000 revolutions per minute (rpm) to harvest the bacterial cells. The supernatant was gently decanted, leaving about 100 μL of the broth culture, which was then vortexed at high speed until the bacterial cell pellet became completely suspended. TENS solution (300 μL; 25mM Tris, 10mM EDTA, 0.1N NaOH, 0.5% SDS) was then added to lyse the bacterial cells. It was mixed by inversion 3 to 5 times until the solution became slimy, after which 150 μL of 3.0M sodium acetate (pH 5.2) was added and again vortexed for about 10 seconds. It was further centrifuged at 13 000 rpm for 5 minutes to pellet out cellular debris and chromosomal DNA.
The supernatant was then transferred into another labeled 1.5-mL Eppendorf tube, and 900 μL of cold absolute ethanol was added. The solution was centrifuged at 13 000 rpm for 10 minutes. The supernatant was discarded, and the white pellet containing the plasmid DNA was rinsed twice with 1000 μL of 70% ethanol.
The pellet was then air-dried, and 40 μL of TE buffer (10mM Tris, 1mM Na2EDTA) was added to resuspend the pellet. Electrophoresis of the extracted plasmid DNA was carried out on 1.0% agarose gel and run for 3 hours at 63 V with HIND III digest of lambda phage DNA (Fermentas) as the molecular weight marker. It was visualized with ethidium bromide via the ultraviolet transilluminator.
Plasmid Curing of Bacterial Isolates
Isolates that appeared to possess plasmid (per the plasmid profile data) were selected for curing with acridine orange as described by Silhavvy et al. 23 Overnight cultures of the selected isolates in nutrient broth were subcultured into 5 mL of nutrient broth supplemented with 0.1 mg/mL of acridine orange. It was incubated at 37ºC for 24 hours and plated out on nutrient agar.
Data Analysis
Results are presented with descriptive statistics (mean ± SD). To determine whether there were significant differences between mean bacterial counts obtained from irrigation water and fresh vegetable samples, independent t test and 1-way analysis of variance were used with P ≤ .05 as indicator of statistical significance. SPSS 16.0 (a software package for statistical analysis) for Windows was used to perform the analyses.
Results
Enumeration of THB and Total Coliform in the Samples
The THB count for irrigation water samples ranged from 5.00 × 104 to 8.00 × 105 CFU/mL with a mean of 2.88 × 105 ± 0.35 CFU/mL (n = 21), while the coliform count for irrigation water samples ranged from 1.40 × 103 to 1.40 × 105 CFU/mL with a mean of 1.27 × 104 ± 0.45 CFU/mL (n = 21; Figure 1). For the fresh vegetable samples, the THB count is in the range of 3.30 × 104 to 9.70 × 105 CFU/g with a mean of 3.99 × 105 ± 0.59 CFU/g (n = 12), while the coliform count is in the range of 1.10 × 103 to 3.10 × 105 CFU/g with a mean of 7.73 × 104 ± 0.90 CFU/g (n = 12).
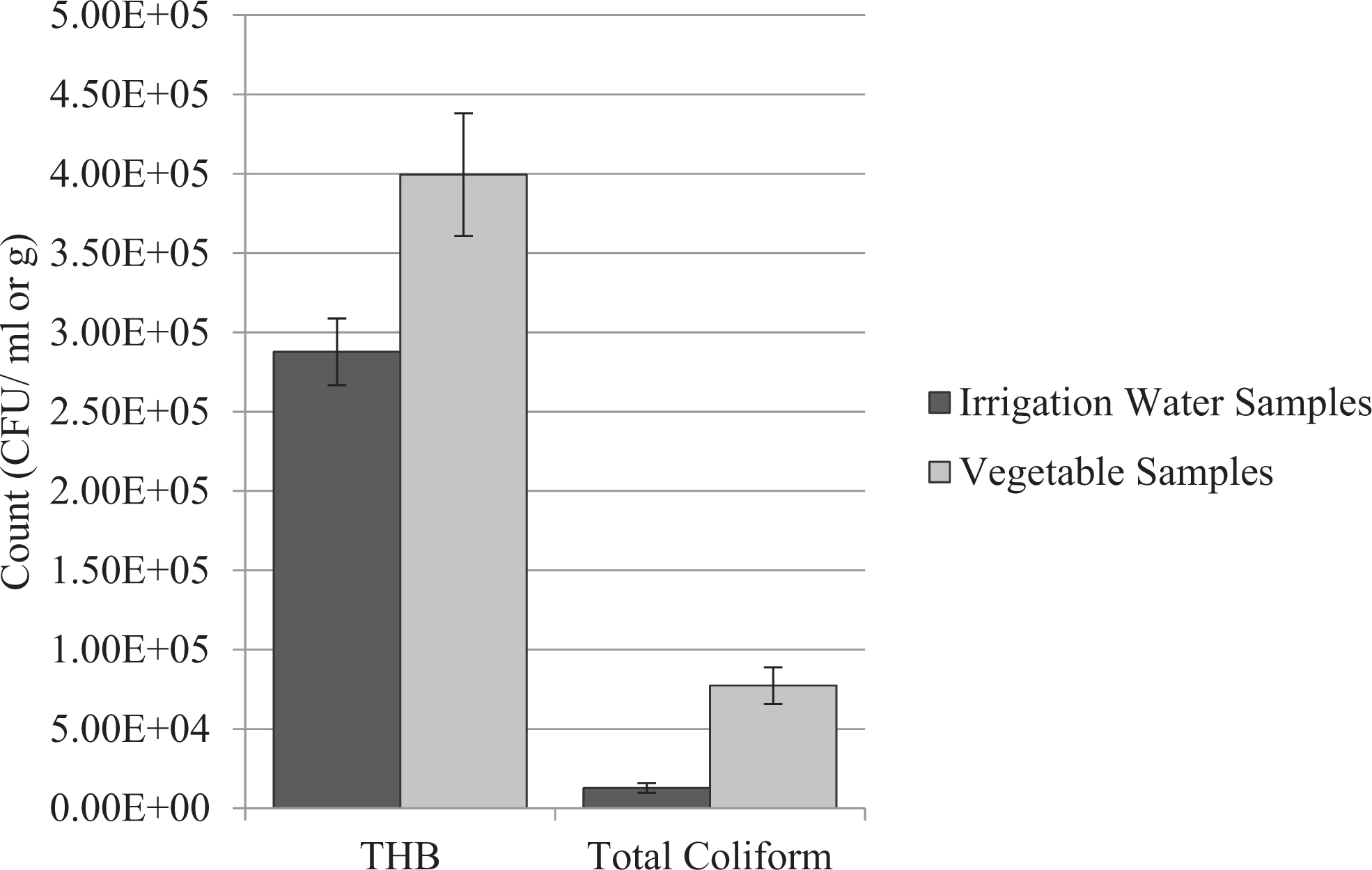
Total heterotrophic bacteria (THB) and total coliform counts in the irrigation water and fresh vegetable samples. CFU, colony-forming units.
Comparison of the mean log10 THB count for irrigation water and fresh vegetable samples shows that the THB count was statistically comparable (P = .86). Also, no significant difference (P = .07) was observed between the coliform count of irrigation water and fresh vegetable samples.
Identification and Characterization of Bacteria Isolated From the Samples
The occurrence and distribution of the bacteria isolated from vegetable and irrigation water samples are shown in Figure 2. A total of 61 gram-negative bacteria, mostly enteric rods, were isolated. These included 20 isolates from the vegetable samples and 41 isolates from the irrigation water samples. The bacterial isolates belonged to 8 predominant genera, which included Klebsiella pneumoniae (27.9%), Pseudomonas aeruginosa (26.2%), and E coli (8.2%); the rest are shown in Table 1.
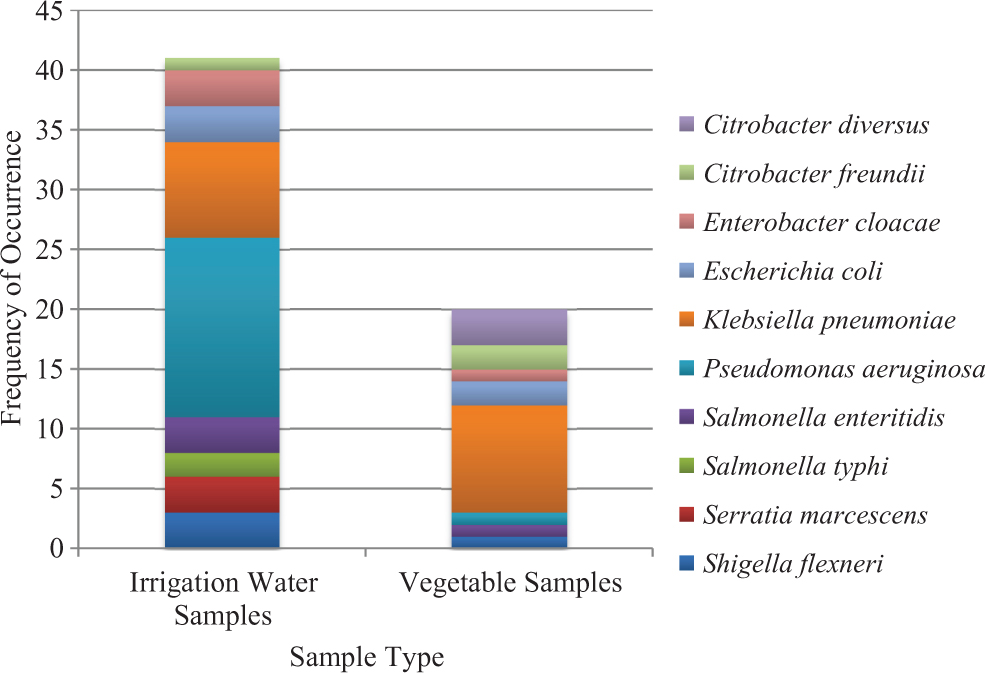
Distribution of gram-negative bacteria isolated from irrigation water and vegetable samples used for the study.
Antibiotic Resistance Patterns of Recovered Isolates From Vegetable and Irrigation Water Samples.
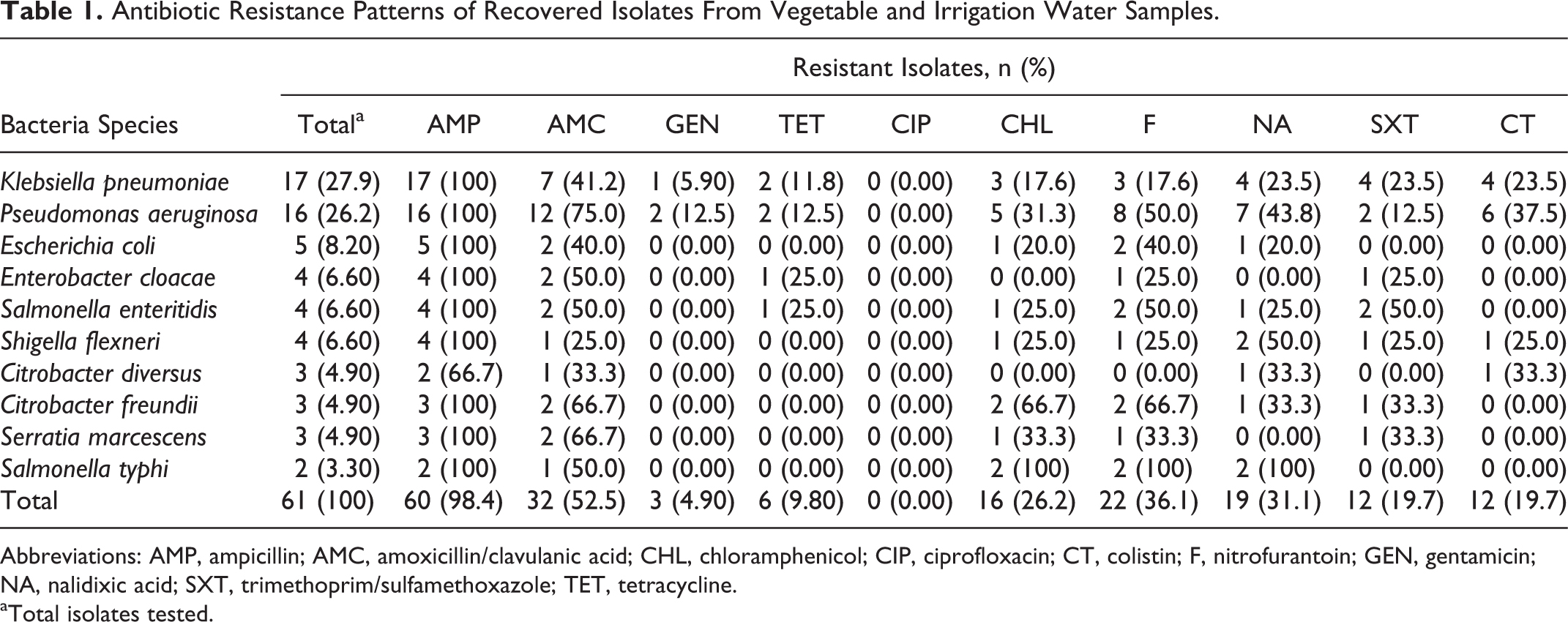
Abbreviations: AMP, ampicillin; AMC, amoxicillin/clavulanic acid; CHL, chloramphenicol; CIP, ciprofloxacin; CT, colistin; F, nitrofurantoin; GEN, gentamicin; NA, nalidixic acid; SXT, trimethoprim/sulfamethoxazole; TET, tetracycline.
Total isolates tested.
Antibiotic Susceptibility Testing of Bacteria Isolates
Results for the resistance to antibiotics of bacteria isolates recovered from vegetable and irrigation water samples are presented in Tables 1 and 2. The results show that a very high proportion of recovered isolates were resistant to ampicillin (98.4%) with Citrobacter diversus being the exception; this was followed by resistance to amoxicillin (52.5%) and nitrofurantoin (36.1%). Ciprofloxacin proved to be highly effective against all isolates, as none of the 61 isolates screened for antibiotic susceptibility was resistant to it (0.0%); this was closely followed by gentamicin (4.9%) and tetracycline (9.8%). The resistance patterns of the remaining isolates are shown in Table 1. The results featured on Table 2 show that, all together, 33 (54.1%) of 61 bacteria isolates from both vegetable and irrigation water samples were resistant to ≥3 classes of antibiotics.
Multiple Resistance Profile of Recovered Bacteria Isolates.
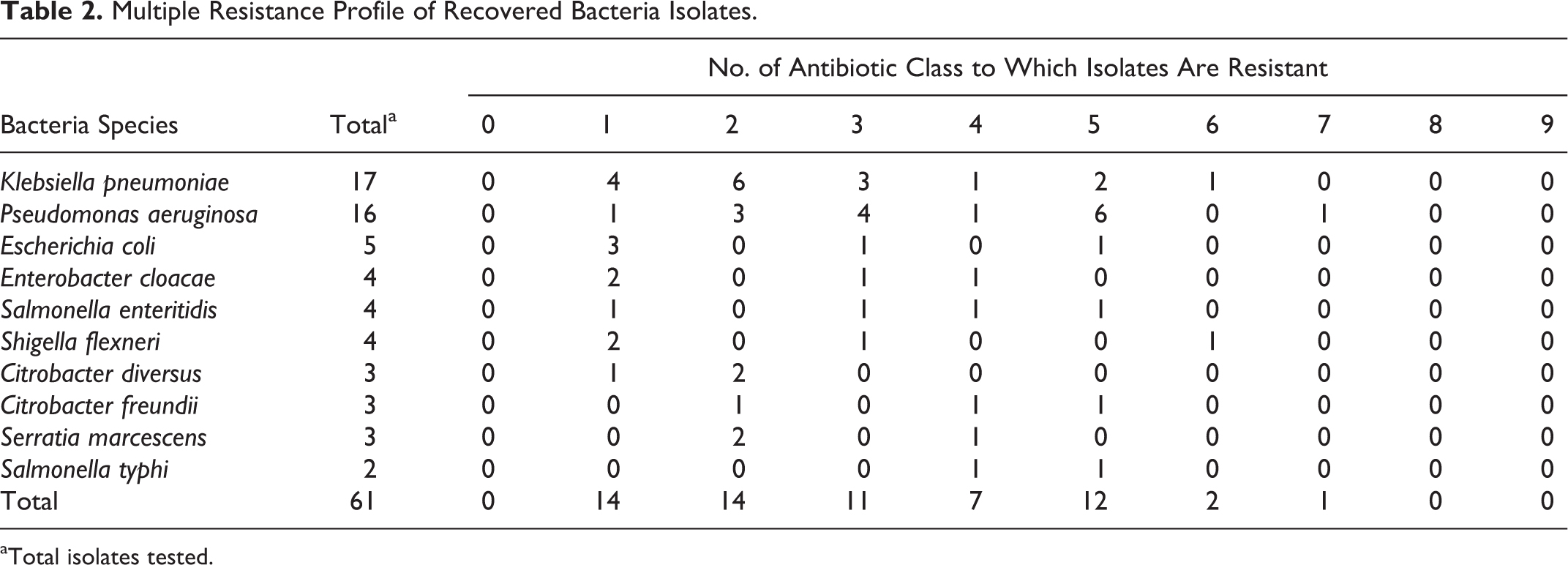
Total isolates tested.
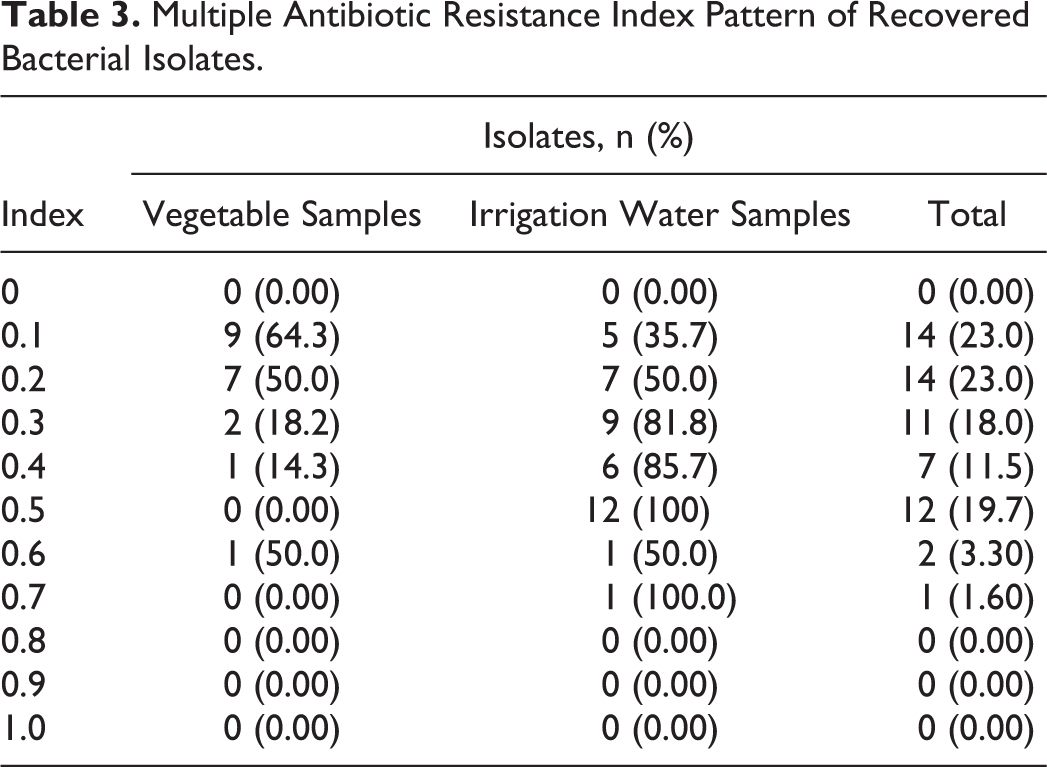
The multiple antibiotic resistance (MAR) index pattern of the bacterial isolates is shown in Table 3. Based on MAR index ≥0.3, the antibiotic resistance of isolates shows that 29 of 41 (70.7%) irrigation water samples were resistant to ≥3 classes of antibiotics, as compared with 4 of 20 (20%) isolates of the vegetable samples.
Multiple Antibiotic Resistance Index Pattern of Recovered Bacterial Isolates.
Plasmid Isolation and Characterization
Attempts were made to isolate plasmids from all 61 bacteria isolates recovered from vegetable and irrigation water samples. The plasmid profile of the gram-negative bacteria isolated from vegetable and irrigation water samples is as shown in Figures 3 and 4. All the bands at 23,130 base pairs (bp) are chromosomal DNA. However, 17 of the 61 isolates revealed the presence of at least 1 plasmid DNA band at ≤2027 bp, except isolates on lanes 19 and 25 (Figure 3). The isolate on lane 19 had 2 bands with 1 of the bands at 2322 bp, while the isolate on lane 25 had only 1 plasmid DNA band at 2322 bp.
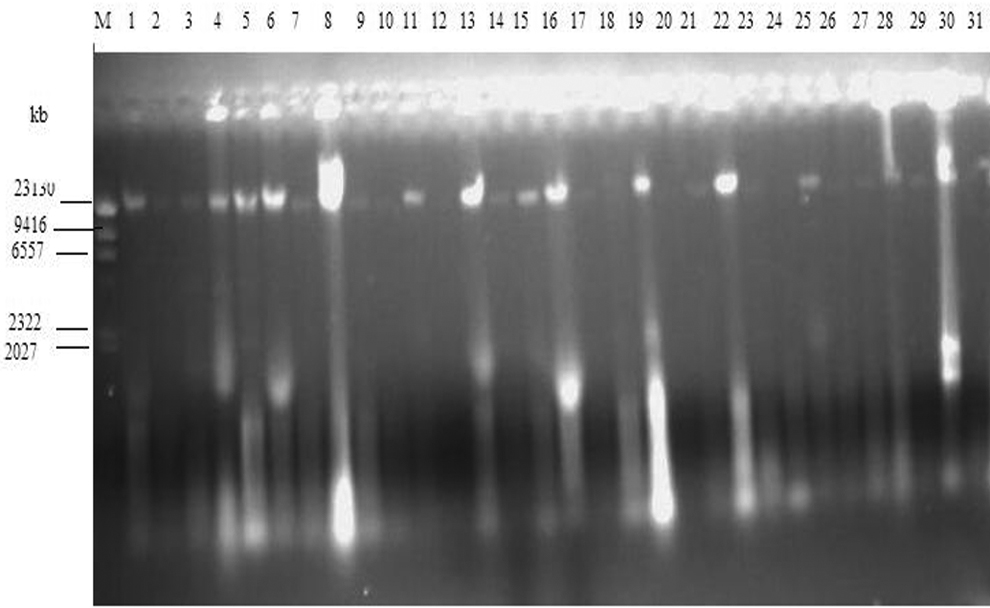
Plasmid profile analysis of gram-negative isolates from water and vegetables: samples 1-31. Lane M: molecular weight marker. Lanes 1-31: gram-negative isolates.
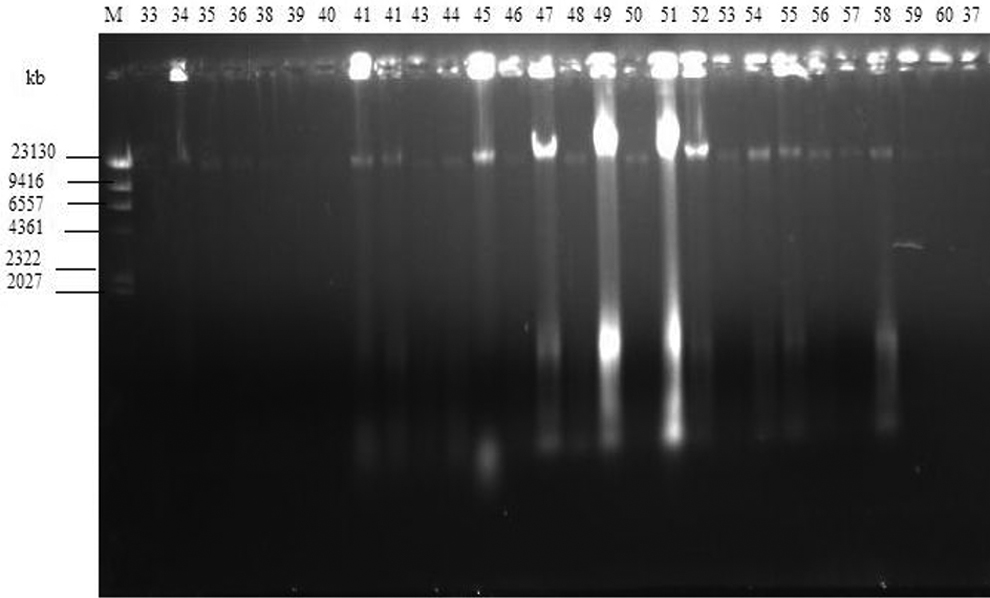
Plasmid profile analysis of gram-negative isolates from water and vegetables: samples 33-60. Lane M: molecular weight marker. Lanes 33-60: gram-negative isolates.
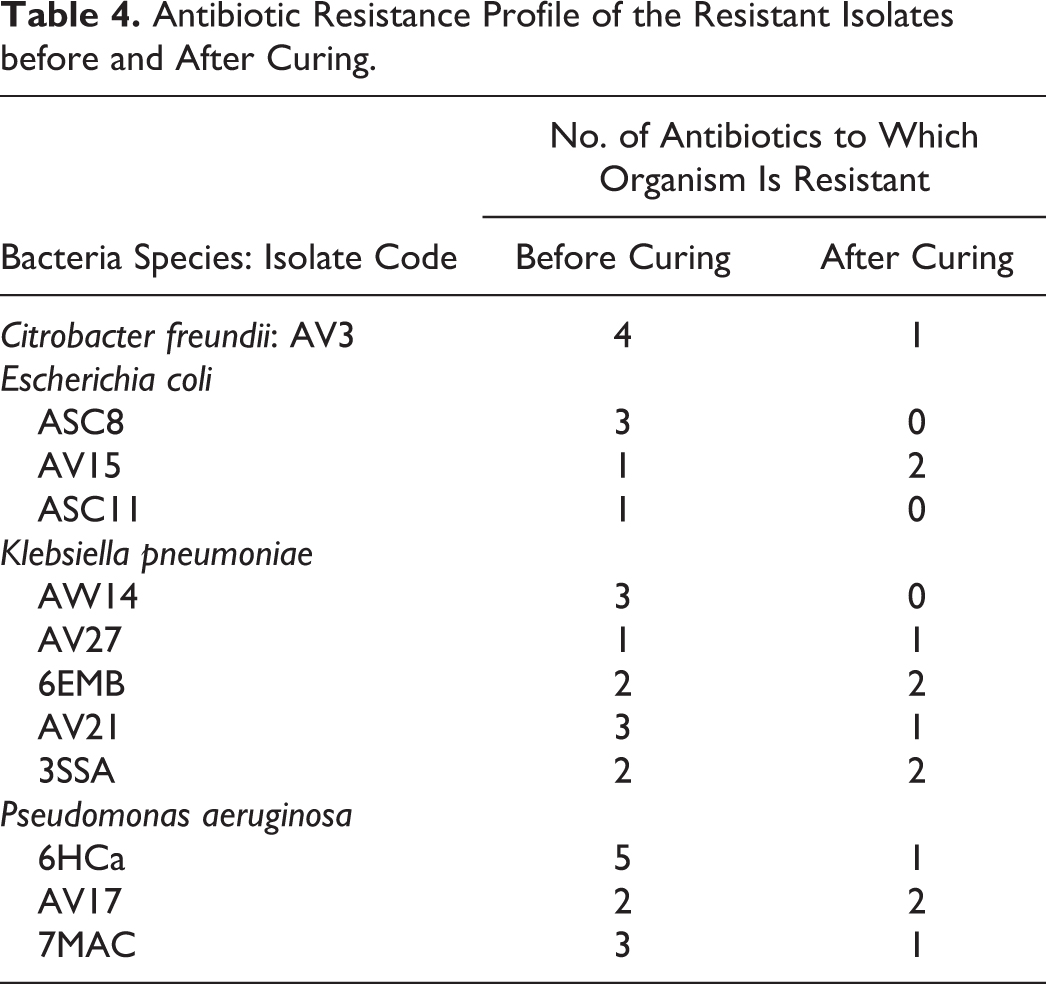
After the isolates were cured of their plasmids, the antibiotic susceptibility testing of the 12 isolates that showed plasmid presence revealed a reduction of resistance in 58.3% of the isolates, which became resistant to a lesser number of antibiotics than that before the curing experiment (Table 4).
Antibiotic Resistance Profile of the Resistant Isolates before and After Curing.
Discussion
The main objective of this study was to investigate the quality of irrigation water used in agricultural settings, the potential bacterial load in irrigation water and/or soil, and the potential contamination of fresh vegetables grown and harvested in a major vegetable farm in southwestern Nigeria. The data in this study revealed culturable bacterial populations (THB) of 8.00 × 105 CFU/mL in irrigation water and 9.70 × 105 CFU/g in vegetable samples obtained from the study site.
THB and total coliform counts, reported by numerous authors, varied widely for irrigation water sources and vegetable types.24 –26 The observed mean counts in both irrigation water and the fresh vegetable samples were higher than the recommended standards of 1.0 × 102 CFU/mL (or CFU/g) for THB and 1.0 × 101 CFU/mL (or CFU/g) for total coliform.27 –29 Similar THB values had been previously reported.25,30,31
There was no significant difference in the THB and total coliform values reported for the irrigation water and vegetable samples; however, higher mean values were recorded for the vegetable samples. Microbial contamination on fresh vegetables occurs throughout the life cycle, from cultivation to distribution, and the degree of contamination depends on several environmental factors, including the irrigation water source. 24 High total coliform count in the irrigation water samples from the Omi River was indicative of contamination with human feces and other anthropogenic activities.32,33
The isolates recovered from the irrigation water and vegetable samples used in this study are, in order of prevalence, K pneumoniae (27.9%), P aeruginosa (26.2%), and E coli (8.2%). Others include Enterobacter cloacae, Salmonella enteritidis, Shigella flexneri, C diversus, Citrobacter freundii, Serratia marcescens, and Salmonella typhi. The preponderance of K pneumoniae and P aeruginosa in all the samples analyzed is of great importance epidemiologically because the organisms have been known to cause nosocomial and opportunistic infections in debilitated individuals.
Pathogenic bacteria K pneumoniae, Pseudomonas sp, E coli, Salmonella spp, Shigella sp, and E cloacae isolated from the water used for irrigation and harvested vegetable samples have been implicated in various human health conditions, including salmonellosis, shigellosis, nosocomial, and opportunistic infections.34,35
The probability is high that the bacteria from the contaminated water used for irrigation and postharvest washing of vegetables in the study site may survive on the vegetables in the field, as well as when the produce is delivered to retail outlets or other local markets. Improper storage and handling in the markets could increase the bacterial load and increase the chance of dissemination of the pathogens.
In a study by Afolabi and Oloyede, 36 bacterial pathogens isolated from the vegetables irrigated with water from polluted stream included E aerogenes, E coli, Salmonella paratyphi, Shigella dysenteriae, K pneumoniae, among others. All these organisms are of public health significance, and their presence in any produce is an indication that contamination has occurred, thereby making it unsafe for human consumption in the raw, unprocessed form. Although the frequency of Salmonella sp was low in this study, its presence is important because it has been implicated in food poisoning and food-borne infections. Awe et al 37 reported high frequencies of Salmonella sp on vegetables irrigated with contaminated river water and stream water.
The antibiotic susceptibility testing to all 61 isolates revealed that 54.1% of the isolates were multidrug-resistant isolates. Despite the relatively high frequency of multidrug resistance observed in this study, it is interesting to note that a high number of the isolates were still highly sensitive to ciprofloxacin as well as gentamicin. None of the isolates were resistant to ciprofloxacin, while only 4.9% were resistant to gentamicin. It may be attributed to the high cost of ciprofloxacin. Ciprofloxacin is a relatively expensive drug when compared with most antibiotics frequently used in Nigeria, including the β-lactams and tetracycline.
Gentamicin, though cheaper, is available only for intravenous administration. The observed susceptibility of isolates to ciprofloxacin may be related to the high cost of ciprofloxacin, which may have restricted its procurement, misuse, and abuse by the populace, thereby reducing emergence of resistant bacterial strains. The low level of resistance to gentamicin has been recorded in other studies.38 –42 All these cases suggested that the efficacy of gentamicin may be related to the mode of its administration, which has limited its misuse.
Ampicillin proved highly ineffective, as 98.4% of the isolates were resistant and the remaining 1.6% exhibited intermediate interpretation and were not totally susceptible. This high level of resistance to a β-lactam antibiotic suggests that the isolates were likely to be β-lactamase producers. 43 The reason for this high-resistance profile in isolates recovered from samples is not fully understood, but it could be due to the misuse or abuse of antibiotics among healthy individuals and subsequent discharge of fecal and other bodily metabolic wastes into water bodies. The water sources are often used for agricultural irrigation.
In our study, the recovery of bacteria with multidrug-resistant characteristics from vegetables that are sometimes eaten uncooked portends great danger to the populace who eventually consumes the produce. Bacteria that are multidrug resistant have been reported to have a high clinical and economic impact because of limitations in therapeutic options in the treatment of infections that they cause. This may be compounded by the need for an extended stay in the hospital plus a higher cost of treatment. Furthermore, these multidrug-resistant isolates can have a higher virulence, which may increase the severity of infections.
Gram-negative bacteria have been reported to possess several mechanisms of resistance to antibiotics, such as overexpression of the intrinsic AmpC-type cephalosporinase and inactivation of the OprD porin. The expression of the efflux pump has also been implicated in resistance of gram-negative bacteria to beta lactam antibiotics. Resistance to carbapenems, quinolones, and, sometimes, aminoglycosides has been attributed to upregulation of the efflux pumps of the resistance-nodulation-cell division family. 44 This multidrug system is known to provide resistance to a very wide range of compounds in gram-negative bacilli.45,46 These traits—coupled with a low permeability of the outer membrane, 47 as well as an ability to acquire further resistance mechanisms to multiple groups of antimicrobial agents, including β-lactams, aminoglycosides, and fluoroquinolones 48 —often present simultaneously, thereby conferring multiresistant properties to the organism.
Multiple antibiotic resistance (MAR) indices obtained in this study show that 54.1% of the bacterial isolates screened for antibiotic sensitivity in this study had an index ≥0.2. MAR indexing is an effective tool for evaluating the spread of bacterial resistance in a given population. 42 A MAR index >0.2 implies that the strains of such bacteria originate from an antibiotic pressurized environment, where several antibiotics are used. The high MAR indices appear to suggest a need for continuous monitoring of bacteria antibiotic susceptibility from various samples and environments before antibiotic prescription to ensure the reduction in the spread of resistant bacteria strains.
Globally, resistance to antibiotics is an increasing problem. The emergence of antibiotic-resistant phenotypes is due to mutations at different chromosomal loci or acquisition of resistance brought about by horizontal transfer. 49 Multidrug resistance in gram-negative bacteria can often be attributed to gene cassettes that are located within a site-specific recombination system called the integron. 50 These genes are transferred horizontally through mobile genetic elements such as plasmids, transposons, and integrons.51,52
As observed in this study, the possession of plasmids is vital in conferment of characteristics such as antibiotic resistance to organisms. 53 Curing of selected isolates brought about a reduction in the degree of resistance to antibiotics in the cured isolates, as 58.3% of the isolates became resistant to a lesser number of antibiotics when compared with the wild type (Table 4). Our data therefore suggest that the vegetable produce from the study site may harbor pathogens of public health concern.
Conclusion and Recommendations
Surface waters, such as rivers, lakes, and ponds, as well as groundwater, are common sources of agricultural irrigation water in southwestern Nigeria. However, most of these water resources are affected by water-based anthropogenic activities that result in various degrees of contamination or pollution with pathogenic microorganisms, chemical contaminants, and other hazardous substances and, as such, have the potential to increase dissemination of microorganisms onto growing vegetables and crops that are of public health importance. Farmers require quantifiable information on the cultivation of produce using agricultural irrigation sources.
This study shows that several potentially pathogenic and antibiotic-resistant bacteria are present on vegetables as well as in irrigation water. As such, there is need for treatment of the surface water used for irrigation and postharvest washing in this field. Enactment and implementation of appropriate government policies and legislation prohibiting the disposal of waste into the river way will likely achieve lower levels of contaminants in water systems.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by NICANVEG Project 106511, jointly funded by the International Development Research Centre and the Canadian International Development Agency under the Canadian International Food Research Fund.

