Abstract
Purpose
The kappa opioid receptor (KOR) plays a pivotal role in stress- and anxiety-related behaviors, with growing evidence linking it to stress induced by social isolation and separation. Despite this, tools for studying KOR in clinically relevant social contexts remain limited. The socially monogamous coppery titi monkey offers a translational model for investigating pair bonding. This study evaluated the feasibility of [11C]GR103545 PET imaging to characterize KOR activity
Methods
Adult titi monkeys (N = 6) underwent [11C]GR103545 PET brain scans at baseline and following administration of the KOR antagonist CERC-501. Non-displaceable binding potential (BPND) was calculated across 14 brain volumes of interest (VOIs) implicated in social bonding, using Simplified and Logan reference tissue models (SRTM and LRTM), with the cerebellum as the reference region.
Results
Baseline [11C]GR103545 uptake patterns across VOIs were consistent with reports in humans, other primates and published autoradiography data. CERC-501 pretreatment significantly reduced BPND (SRTM: 55.99%, LRTM: 59.68%) across several, but not all brain VOIs.
Conclusions
This study establishes [11C]GR103545 PET as a viable tool for assessing KOR binding dynamics in titi monkeys, providing new opportunities to explore KOR modulation in social bonding and separation.
Keywords
Introduction
Kappa opioid receptors (KORs), the most abundant subtype of the opioid receptors, play a central role in a wide range of neurophysiological functions pertaining to regulation of mood and motivation.1,2 Given their antidepressant-like effects, KOR antagonists are being actively explored as therapeutics for stress-related disorders,3,4 including in ongoing clinical trials for CERC-501 (NCT06514742). 5 Activation of KOR by its only endogenous ligand, dynorphin, in response to stress has been strongly associated with aversive behavior regulation in humans.6–8 Social stressors, such as social isolation and separation have been shown to trigger KOR activation in preclinical models.9–11 However, the mechanisms by which KOR influences critical social neurotransmitters like oxytocin (OT) remain unclear. The widespread loneliness reported during the COVID-19 pandemic underscored the profound psychological impact of these social stressors on human health.12,13 Therefore, now more than ever, it is crucial to understand the role of KOR in social stress and its potential for pharmacologic targeting.
Positron Emission Tomography (PET) has been widely utilized for imaging the opioid system14–17 with several radiotracers developed to provide a readout for the KOR.18–21 In particular, the radiotracer [11C]GR103545, a highly selective KOR agonist,
22
has provided valuable,
The coppery titi monkey (
To date, no PET studies analyzing the KOR have been conducted in titi monkeys, despite their high relevance to human social bonding. To address this unmet need, the aim of this study was to characterize the distribution and binding of the radiotracer [11C]GR103545, in the titi monkey brain. Additionally, we evaluated radiotracer binding dynamics following pharmacological blockade of KOR with the antagonist CERC-501, providing the first
Materials and Methods
Animals
All experiments with animals were approved by the University of California Davis Institutional Animal Care and Use Committee (IACUC; protocol number: 23483) and used facilities fully accredited by American Association for Accreditation of Laboratory Animal Care (AAALAC) International. Six adult coppery titi monkeys (3 male, 3 female; 7.0 ± 1.5 year old; 1.22 ± 0.15 kg), born and housed at the California National Primate Research Center, were selected for this study. As this was a pilot study designed to assess feasibility, a formal sample size calculation was not performed. All subjects were socially housed in cages (1.2 m × 1.2 m × 2.1 m) under a 12:12 light:dark cycle with temperature maintained at 21 °C.
PET Imaging Protocol and Pretreatment
The PET radiotracer [11C]GR103545 was synthesized as described in 32 with a mean molar activity of 155.02 ± 30.63 PBq/mol at the end of synthesis. Titi monkeys were anesthetized with 1–2% isoflurane and positioned head-first on a custom bed of a dedicated brain PET scanner (PiPET, Brain Biosciences, Rockville, MD). The dynamic 110 min PET acquisition began approximately 15 s prior to injection of [11C]GR103545 (injected activity 48.16 ± 6.27 MBq, injected mass 0.26 ± 0.04 µgm/kg; Figure 1). Data were reconstructed with an isotropic voxel size of 0.8 mm3 using framing of 6 × 10 s, 8 × 30 s, 5 × 60 s, 4 × 300 s, 8 × 600 s. A single-pass “cold” transmission scan was acquired pre-injection for attenuation and scatter correction. This PET imaging protocol was established and validated before the start of the study.
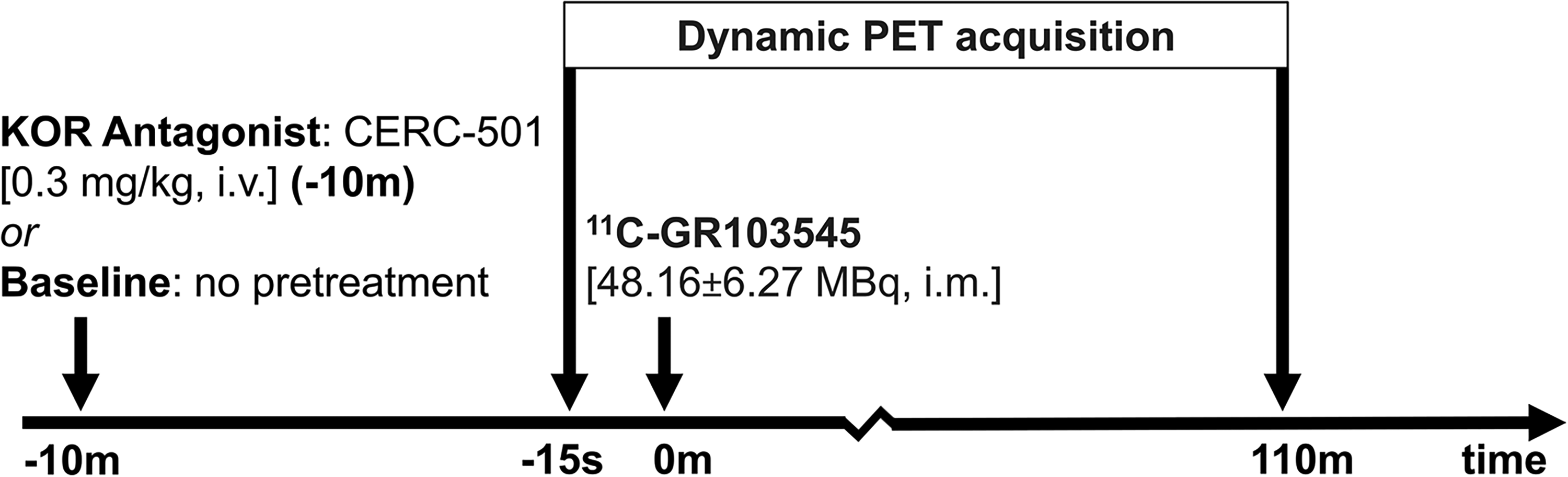
The Scanning Paradigm for N = 6 Titi Monkeys, Imaged for 110-min (PET Emission Scan), at Two Timepoints; Baseline and 10-min After Pretreatment with Antagonist CERC-501. Imaging Sessions for Each Animal Were Conducted with a Minimum Interval of one Month Between Them.
Blocking experiments involved pre-treatment with KOR antagonist CERC-501 (Supplier: MedChemExpress, Monmouth Junction, NJ, Cat. No.: HY-101718), administered (0.3 mg/kg, i.v.) to the same n = 6 titi monkeys, 10-min prior to radiotracer administration. 33 Imaging sessions were spaced at least one month apart per animal.
Magnetic Resonance Imaging (MRI) Acquisition
To aid with regional delineation, anatomical T1-weighted brain MRIs were acquired with a 1.5 T GE Signa LX9.1 scanner (General Electric Corporation, Milwaukee, WI) and a 3” surface coil. The MR acquisition employed a 3D spoiled gradient echo pulse sequence in the coronal plane using the following parameters: echo time (TE) = 7.9 ms, repetition time (TR) = 22.0 ms, flip angle=30.0°, field of view=8 cm, number of excitations=3, matrix=256 × 256, resulting in a pixel size of 0.3125 × 0.3125 mm2 and a slice thickness of 1 mm.
Image Post Processing and Kinetic Modeling
Anatomical MRIs were mapped to a titi monkey reference space using rigid registration and processed with BrainSuite (v23a) 34 for skull-stripping and N4ITK for bias-correction. 35 A titi monkey brain atlas was warped to each MRI using symmetric normalization (Advanced Normalization Tools)36,37 to generate regional labels, refined as needed by a neuroanatomy expert. PET data were motion-corrected and coregistered to MRIs on PMOD (v4.4, PMOD Technologies, Zürich, Switzerland). The image analysis pipeline is shown in Supplementary Figure S1.
Region-specific, non-displaceable binding potential (BPND) was calculated on PMOD utilizing the Simplified Reference Tissue Model (SRTM) 38 and Logan Reference Tissue Model (LRTM), 39 each with the cerebellum as the reference region. Reference tissue modeling has been validated for this radiotracer in previous studies across species.22,33,40,41 SRTM and LRTM were compared to identify the best fit for the dynamic data. For LRTM, the average tissue-to-plasma clearance k2’ was estimated from SRTM and t* was set at 40 min. The primary analysis included 14 brain volumes of interest (VOIs) relevant to social bonding, namely, nucleus accumbens, caudate, hypothalamus, cingulate, hippocampus, amygdala, claustrum, orbitofrontal cortex, thalamus, septum, superior frontal gyrus, superior temporal gyrus, insula, and putamen. Additionally, the pituitary gland, showing high PET signal, was delineated with a spherical VOI.
Statistical Analysis
BPND differences between baseline and antagonist conditions were analyzed using one-way nonparametric ANOVA for matched pairs (Friedman test; GraphPad Prism v10.0.2). 42 Multiple comparisons were accounted for by controlling the false discovery rate (FDR). 43 Pearson's correlation assessed agreement between BPND calculated from the SRTM and LRTM. Goodness of fit for kinetic models was evaluated using the Akaike Information Criteria (AIC) 44 reported in PMOD. The significance threshold was set at 0.05.
Results
Baseline BPND Concurred with Reported Distribution of KOR in the Titi Monkey Brain
Representative [11C]GR103545 uptake patterns at baseline and after pre-treatment by CERC-501 are shown in Figure 2. Regional time-activity curves are shown in Figure 3. Baseline uptake patterns were consistent with autoradiography studies of KOR density in the titi monkey brain. 45 Correspondingly, high BPND (> 1.3) was observed in the pituitary gland followed by the insula, claustrum and orbitofrontal cortex. Moderate BPND (0.9-1.3) was detected in VOIs including (but are not limited to) the nucleus accumbens, amygdala, and hippocampus, while the thalamus exhibited low BPND.
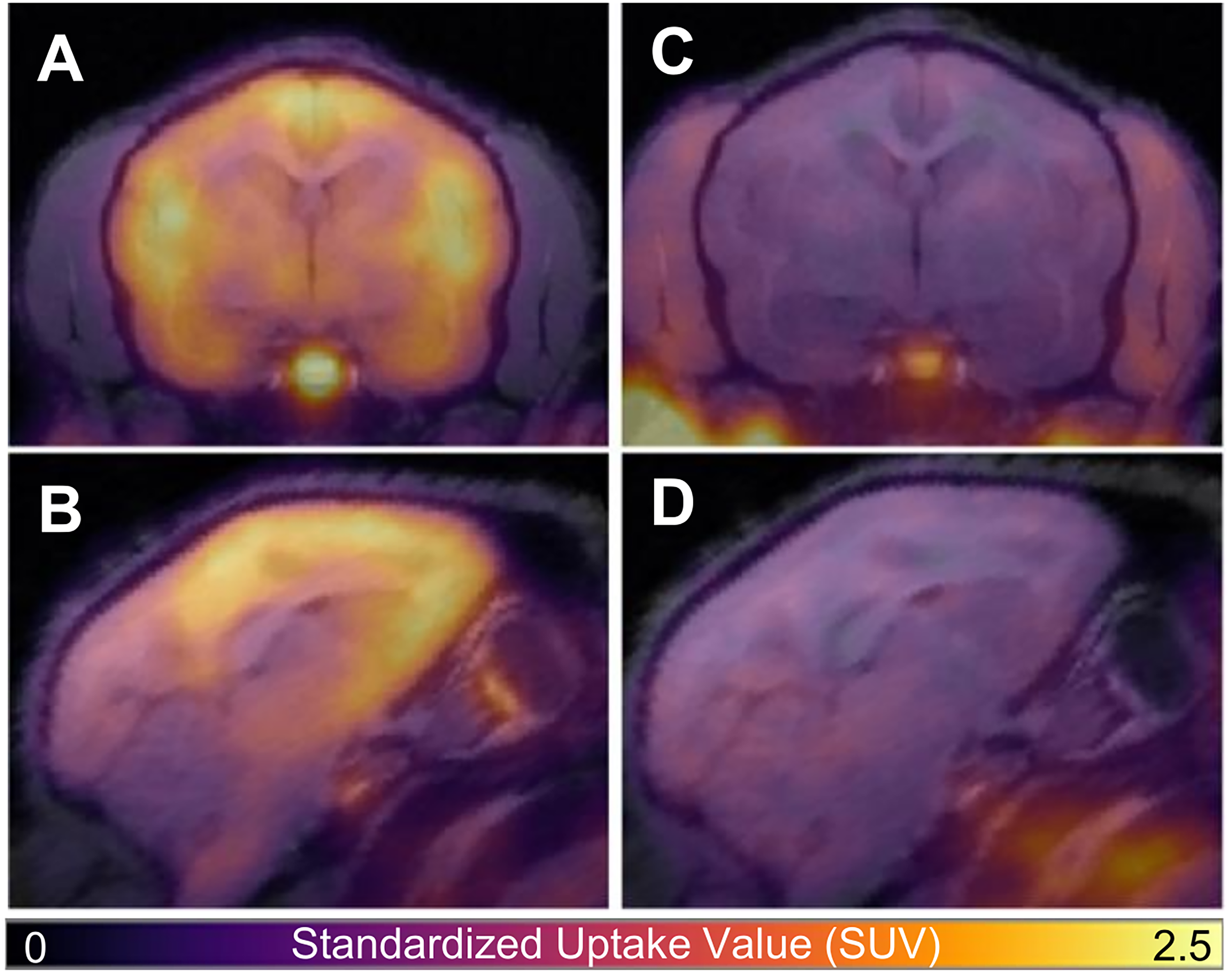
[11C]GR103545 PET (Color) Overlaid onto T1-weighted MRI of a Titi Monkey in the Axial (A, C) and Sagittal (B, D) Plane for Baseline (A, B) and CERC-501 Pretreatment (C, D) Scans. Reduced Radiotracer Uptake in the Brain is Apparent after Pretreatment with CERC-501 (C, D). PET Data are Presented as Standardized Uptake Value (SUV) Images Calculated from Data 80-110 min Post-radiotracer Injection.

[11C]GR103545 PET Time Activity Curves Across Selected Volumes of Interest (VOIs) for a Representative Titi Monkey at Baseline. Points Indicate Measured Standard Uptake Value (SUV) While the Solid Line Indicates the fit with Simplified Reference Tissue Model (SRTM).
Model fits based on SRTM showed a median AIC of −10.73 (range: −32.41 to 36.47), while LRTM exhibited a median AIC of 68.55 (range: 59.02 to 78.15) (Supplementary Table S1). These results suggest that SRTM provided a better fit for the dynamic PET data compared to LRTM.
Pretreatment with CERC-501 Significantly Reduced Binding Across Several, but not all, VOIs When Compared to Baseline BPND
CERC-501 administration significantly reduced BPND across most VOIs, with an average reduction of 55.99% (SRTM) and 59.68% (LRTM). For SRTM, significant reductions were observed in all VOIs except the claustrum, hypothalamus, and septum (Figure 4). The superior frontal gyrus showed the largest decrease (76.25%, p = 0.004) while the pituitary gland the smallest (27.69%, p = 0.021) (Table 1). For LRTM, binding reductions were significant across all VOIs except the nucleus accumbens, claustrum, septum and pituitary (Figure 5, Supplementary Figure S2). Similar to SRTM, the superior frontal gyrus showed the largest decrease (78.79%, p = 0.021) and the pituitary the smallest (23.21%, p = 0.149) (Table 2).
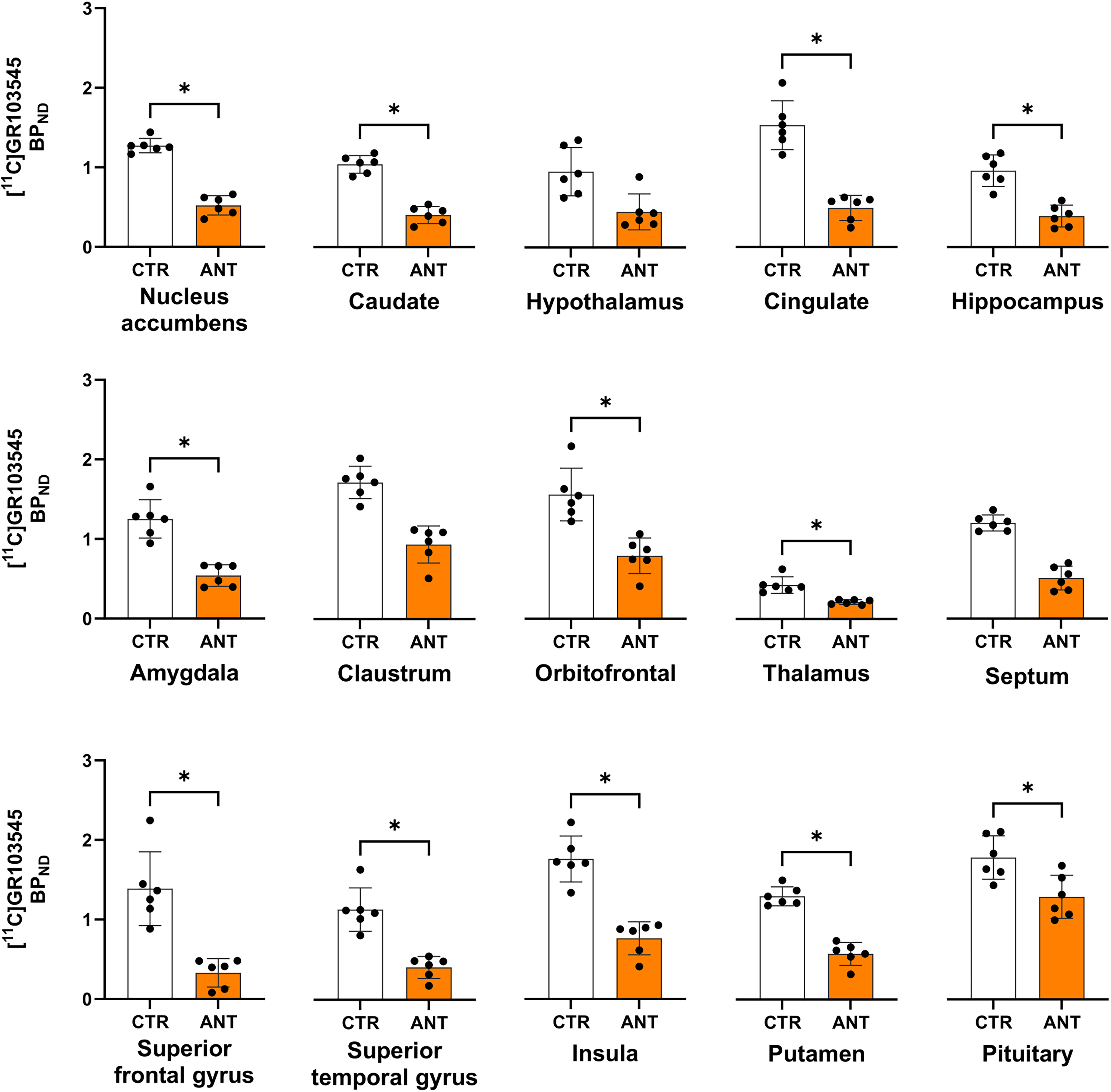
BPND Across the 15 VOIs Calculated with SRTM for Baseline (CTR) and after CERC-501 Pretreatment (ANT). * Indicates Significant Difference after FDR Correction. The Points Indicate Individual Datapoints, While the Bars Indicate the 95% Confidence Interval.

BPND Across the 15 VOIs Calculated with Logan Reference Tissue Model (LRTM) for Baseline (CTR) and After CERC-501 Pretreatment (ANT). * Indicates Significant Difference After FDR Correction. the Points Indicate Individual Datapoints, While the Bars Indicate the 95% Confidence Interval.
BPND Calculated with SRTM Averaged Across N = 6 Titi Monkeys in the 15 VOIs Scanned at Baseline and After CERC-501 Pretreatment, Their Corresponding p Values upon Comparing Baseline BPND to BPND After CERC-501 Pretreatment. the Reported p-Values are Before FDR Correction, While the Asterisk Denotes Comparisons That Survived FDR Correction.

Indicates significance after FDR correction.
BPND Calculated Using with LRTM Averaged Across N = 6 Titi Monkeys in the 15 VOIs Scanned at Baseline and After CERC-501 Pretreatment, Their Corresponding p Values upon Comparing Baseline BPND to BPND After CERC-501 Pretreatment. the Reported p-Values are Before FDR Correction, While the Asterisk Denotes Comparisons That Survived FDR Correction.

Indicates significance after FDR correction.
BPND Measured from SRTM Correlated with That from the LRTM Analysis
BPND values from STRM and LRTM showed a strong positive correlation (R = 0.94, p < 0.001; Supplementary Figure S3). Reductions in the claustrum and septum were not significant for both models. Unlike SRTM, LRTM reported no reduction in the nucleus accumbens and pituitary gland while SRTM reported no reduction in the hypothalamus. Overall, LRTM estimated a slightly greater reduction in BPND following CERC-501 pretreatment (59.68%) compared to SRTM (55.99%).
Discussion
This study is the first to use PET imaging to analyze KOR binding in titi monkeys, a species uniquely suited for translational studies due to their physiological similarities to humans and complex social behaviors.14,16 The observed distribution of [11C]GR103545 in the titi monkey brain aligned with previously reported autoradiographic findings.
45
Furthermore, significant reductions in [11C]GR103545 BPND following CERC-501 administration confirmed the utility of this radiotracer for assessing KOR modulation
Most brain VOIs showed reduced BPND after CERC-501 pretreatment, a compound that is highly selective to the KOR.5,46,47 This is consistent with findings from previous [11C]GR103545 antagonist blocking experiments performed with naltrexone (an opioid antagonist) and PF04455242 (a KOR antagonist) in baboons
41
and humans,
48
respectively. Self-blocking of [11C]GR103545 in rhesus monkeys,
22
and blocking using a range of antagonists and agonists in rats,
33
have also reported significant reductions in BPND. These results combined with the high selectivity of CERC-501 for mammalian KOR,
5
support the utility of [11C]GR103545 PET for
The highest BPND in the brain was reported in the insula, a finding consistent with its involvement in experiences of pain, 49 anxiety 50 and addiction 51 in humans. Preclinical studies have implicated insular KOR activation 52 in addiction circuits, particularly in alcohol use disorder. 53 Correspondingly, reduced uptake of the KOR antagonist radiotracer [11C]LY2795050 in the insula has been reported in alcohol-dependent individuals compared to healthy controls. 54 Another region of interest, the nucleus accumbens also demonstrated BPND reduction in our study after CERC-501 administration. Dysregulation of KOR in this region has been linked to stress-related decreases in social motivation.9,55–57 Conversely, the septum did not exhibit significant reductions following CERC-501 treatment with either the SRTM or LRTM fits. Being a moderate KOR binding region, it may have been less responsive to the blockade. Additionally, functional compensation by other opioid systems could have also contributed to the diminished KOR activity in this region.58,59 Similarly, the claustrum, a region with dense KOR expression 45 also did not show significant reductions with either modeling method. This outcome could be influenced by the study's small sample size and limitations in segmentation accuracy due to the scan resolution for this anatomically narrow region. Given the roles of these regions in pair-bonding,57,60–62 further investigation into KOR activity under various social experimental conditions in titi monkeys could provide valuable insights into the neurobiological underpinnings of social bonding in these brain regions.
Several radiotracers have been developed to evaluate KOR fluctuations
The use of reference tissue models for our study effectively addressed the challenges associated with arterial blood sampling in a longitudinal experimental paradigm for a monkey that weighs about 1 kg on average and therefore has limited blood volume compared to rhesus monkeys or baboons. Among the models tested, SRTM demonstrated a better fit to [11C]GR103545 kinetics based on AIC metrics. While easier to implement, LRTM may be more sensitive to noise than SRTM and can introduce bias into the dataset.40–42 Notably, it requires a careful selection of the linear phase (t*).67–69 SRTM's ability to model the entire kinetic curve offers improved accuracy for smaller brain regions, 70 consistent with our results. However, due to the small brain size of the titi monkeys and resolution limit on the PET scanner, partial volume effects likely contributed to a likely underestimation of BPND in both methods. This phenomenon has also been reported in rhesus macaques using similar methods.22,40
The utility of the cerebellum as a reference region remains a topic of debate,22,24,48 although its suitability has been supported by studies in rats 33 and baboons. 41 Further studies could explore whether arterial blood sampling provides greater sensitivity than reference region-based approaches. However, the invasive nature of arterial sampling presents challenges, particularly in longitudinal studies assessing stress responses or behavior, as the procedure itself may introduce confounders.
Conclusions
This study proposes [11C]GR103545 PET in titi monkeys as an assessment tool with translational capability for further studying the role of KOR in clinically relevant stress-related behaviors resulting from social isolation, representing an advancement in our understanding of KOR dynamics in this setting. By demonstrating the feasibility of [11C]GR103545 PET and its sensitivity to pharmacological modulation via CERC-501, we provide critical evidence supporting the utility of the radiotracer in titi monkeys. The findings underscore the potential of titi monkeys to bridge the gap between preclinical and human studies in understanding the neurobiological basis of social behaviors. Future investigations leveraging this monkey model could unravel the complex interplay between KOR and other neurochemical systems, such as OT, that shape social attachment and resilience to stress. This includes investigations in a longitudinal setting, offering pathways for therapeutic intervention in stress-related disorders.
Supplemental Material
sj-doc-1-mix-10.1177_15353508251341082 - Supplemental material for Mapping Kappa Opioid Receptor Binding in Titi Monkeys with [11C]GR103545 PET
Supplemental material, sj-doc-1-mix-10.1177_15353508251341082 for Mapping Kappa Opioid Receptor Binding in Titi Monkeys with [11C]GR103545 PET by Alita Jesal D Almeida, Brad A. Hobson, Logan E. Savidge, Claudia Manca, John P. Paulus, Karen L. Bales and Abhijit J. Chaudhari in Molecular Imaging
Footnotes
Acknowledgements
The authors would like to thank Joshua Waltenburg of the Center for Molecular and Genomic Imaging (CMGI) for synthesizing the radiotracer for this study and Charles Smith and Sarah Tam of CMGI for conducting the PET acquisitions.
Ethical considerations
All experiments with animals were approved by the UC Davis Institutional Animal Care and Use Committee (IACUC protocol number: 23483) and used facilities fully accredited by AAALAC International.
Author Contributions
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Institutes of Health, (grant number P51 OD011107, R01 MH125411, S10 OD021715, U54 NS127758).
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
