Abstract
Objectives:
To investigate acupuncture-induced changes in targeted glutathione metabolites in breast cancer survivors with psychoneurological symptoms (PNS) and to examine associations between these metabolic changes and PNS improvements.
Methods:
This exploratory phase II single-arm study (N = 42) evaluated a 10-session, 5-week acupuncture intervention for PNS management in breast cancer survivors. Targeted glutathione metabolites (ie, reduced glutathione (GSH), oxidized glutathione (GSSG), cysteine-glutathione disulfide (CySSG), GSH/GSSG ratio) and reactive oxygen species (ROS) were measured pre- and post-treatment. Paired t-test and Wilcoxon signed-rank tests assessed changes in these biomarkers. The PNS were assessed using Patient-Reported Outcomes Measurement Information System (PROMIS)-29.
Results:
ROS levels significantly increased post-treatment (2.34 ± 1.02-2.83 ± 1.23 μmol/L, P = .017), while GSSG levels significantly decreased (0.19 ± 0.23-0.12 ± 0.23 µmol/L, P < .001). GSH/GSSG ratio significantly increased (2.73 ± 1.21-5.38 ± 1.99, P < .001), indicating a shift toward reduced oxidative stress. GSH and CySSG levels showed non-significant increases after acupuncture (GSH: 0.38 ± 0.14-0.42 ± 0.16 µmol/L, P = .07; CySSG: 0.00503 ± 0.00082-0.00532 ± 0.00076 µmol/L, P = .06). No significant correlations were found between changes in GSH metabolites and PNS composite scores or individual symptom scores.
Conclusion:
Acupuncture may modulate GSH metabolism, improve redox balance, and enhance antioxidant capacity in breast cancer survivors with PNS. However, these biochemical changes were not correlated with PNS improvement, suggesting that alternative pathways may mediate acupuncture’s therapeutic effects.
Trial registry information:
The study was registered in ClinicalTrials.gov (Identifier: NCT05417451).
Keywords
Introduction
Breast cancer survivors with hormone receptor-positive early-stage disease typically experience tumor growth driven by estrogen and are commonly prescribed 5 to 10 years of aromatase inhibitors after surgery to reduce the risk of recurrence. 1 These medications, which suppress estrogen production, are also used to prevent breast cancer in postmenopausal women at high risk and to treat hormone receptor-positive metastatic disease. 1 Despite their therapeutic benefits, approximately 50% of patients taking aromatase inhibitors reported experiencing joint pain or stiffness, with nearly half of those cases being severe enough to lead to discontinuation of treatment. 2 In addition to joint pain, many patients also reported concurrent psychoneurological symptoms (PNS) encompassing general pain, fatigue, and sleep disturbances that can persist beyond active treatment and significantly impact quality of life (QOL).3 -5 These lingering symptoms are thought to arise not only from the direct effects of cancer and its treatments but also from underlying immune dysregulation, highlighting a complex biological interplay that continues to affect survivors’ QOL.6,7
Given the long-lasting nature of side-effects of aromatase inhibitors in breast cancer survivors, effective management strategies are essential. Conventional pharmacological and behavioral interventions often have their limitations, necessitating the exploration of complementary and integrative health (CIH) approaches such as acupuncture to alleviate PNS and improve overall QOL. 8 Acupuncture is a modality of Traditional Chinese Medicine that works by stimulating specific points on the body, known as acupoints, to elicit beneficial physiological responses and promote recovery. The insertion and manipulation of thin needles at these points trigger a cascade of neurobiological events. 9 Emerging evidence from randomized controlled trials (RCTs) has reported that acupuncture can significantly reduce joint pain, cancer-related fatigue, and hot flashes.10,11 The updated Society for Integrative Oncology (SIO) and the American Society of Clinical Oncology (ASCO) guidelines also recommend that breast cancer patients experiencing aromatase inhibitor-induced joint pain receive acupuncture, based on strong evidence. 12
Previous research suggests that acupuncture helps regulate multiple metabolic pathways. 13 A systematic review indicated that the most common metabolomic pathways impacted by acupuncture are glycine, serine, and threonine metabolism; and glutathione (GSH) metabolism. 14 Acupuncture has also been shown to reduce pro-inflammatory cytokines, alleviate oxidative stress, and restore neurotransmitter balance, factors that are closely associated with PNS experienced by cancer survivors.13,15 Oxidative stress, a key driver of PNS, results from an imbalance between the generation of reactive oxygen species (ROS) and the body’s antioxidant defenses. 16 ROS, such as hydrogen peroxide (H2O2), superoxide (O2−), and hydroxyl radical (OH−), are natural byproducts of cellular metabolism but can damage DNA, lipids, and proteins when produced in excess.17 -19 Antioxidants, including both enzymatic and non-enzymatic types, serve as the primary defense against ROS. Among them, GSH plays a central role in maintaining redox homeostasis by directly scavenging ROS or serving as a cofactor for detoxifying enzymes.20,21 The ratio of reduced GSH to oxidized glutathione (GSSG) is widely regarded as a sensitive indicator of oxidative stress, 20 with increased GSSG reflecting cellular damage and impaired redox balance. 22 23
Prior research has demonstrated alterations in GSH metabolism following acupuncture interventions, suggesting that acupuncture may modulate oxidative stress responses. 24 In animal studies, acupuncture appears to counteract oxidative stress by effectively increasing the GSH/GSSG ratio following cerebral ischemia.25,26 Both animal studies and clinical observations have shown that acupuncture significantly increases GSH levels in individuals with chronic inflammatory conditions—such as rheumatoid arthritis, depression, and chronic fatigue syndrome—compared to control.27 -29 In our systematic review examining the effects of acupuncture on human metabolomics, we also found a significant increase in GSH levels in several studies involving patients who underwent acupuncture treatments for PNS symptoms. 14 Our pilot study similarly indicated changes in targeted GSH metabolites after acupuncture treatment among 8 breast cancer survivors experiencing PNS. 24 GSH significantly increased after 10 sessions of acupuncture treatment. Pathway analysis also indicated that glutathione metabolism (raw P = .002, q = 0.071) was potentially involved in mechanisms of acupuncture among breast cancer survivors. This evidence emphasizes the potential therapeutic benefits of acupuncture in managing oxidative stress and enhancing overall health in both animals and humans.
Despite the growing body of acupuncture mechanism research, the precise biological mechanisms underlying acupuncture and its therapeutic effects on PNS among breast cancer survivors remain incompletely understood. In particular, it is unclear how acupuncture influences oxidative stress and related GSH metabolic pathways in this population. Therefore, studies are needed to elucidate these mechanisms, which can inform the development of targeted interventions and improve symptom management in breast cancer survivors. In the current study, we aim to investigate acupuncture-induced changes in targeted GSH metabolites. We measure and characterize the changes in targeted GSH metabolites and ROS following acupuncture in breast cancer survivors experiencing PNS. We also examine the correlation between changes of GSH metabolites and changes of PNS after acupuncture. Unlike more general markers of oxidative stress or inflammation (eg, cytokines, malondialdehyde), GSH, GSSG are central to maintaining cellular redox homeostasis, and reflect the glutathione redox system, while ROS represents the upstream driver of oxidative stress. This panel enables us to examine both oxidative stress burden and compensatory antioxidant capacity.
Methods
Study Design
This exploratory phase II study employed a single-arm design to evaluate acupuncture for PNS management in breast cancer survivors. The acupuncture protocol consisting of 10 sessions delivered over 5 weeks. Recruitment took place through the Breast Oncology Clinic at the University of Illinois Hospital & Health Sciences System (UI Health), the University of Illinois Cancer Center (UICC) Survivorship Clinic, and local breast cancer support groups. The study was registered with ClinicalTrials.gov (Identifier: NCT05417451) and received approval from the Institutional Review Board of the University of Illinois Chicago (IRB20210840).
Participants were eligible if they met the following criteria: (1) female, age ≥ 18 years; (2) diagnosed histologically with stage 0 to III ER/PR-positive, HER2-negative breast cancer; (3) actively receiving adjuvant endocrine therapy; (4) completed primary cancer treatment (such as surgery, chemotherapy, or radiation therapy) at least 3 months and no more than 5 years prior; (5) experienced at least 2 of 3 psychoneurological symptoms—pain, fatigue, or sleep disturbance—with an average severity rating of 3 or higher on a numeric rating scale (range 0-10) within the previous month; and (6) able to communicate effectively in English. Exclusion criteria included having a bleeding disorder, pregnancy, or an inability (physical or cognitive) to participate fully in study activities.
Procedures
Initial screening was conducted through reviewing participants’ electronic health records (EHRs) to confirm whether they met the first 3 inclusion criteria. Potential participants were subsequently contacted via email and text messages containing study details. For individuals expressing interest, the research coordinator either approached them directly in the clinic waiting area or reached out by telephone to provide further information about the study and conduct a comprehensive eligibility screening. Eligibility determination included consultation with healthcare providers, further review of the EHR, and direct inquiry regarding symptom severity, specifically asking participants to rate their pain, fatigue, and sleep disturbances over the previous month on a scale of 0 to 10.
Upon confirming eligibility, participants were scheduled for a baseline study visit. At the baseline visit (week 0), participants provided informed consent and completed self-administered questionnaires regarding demographic information, clinical history, and current symptom status. Prior to their initial acupuncture session, a fasting blood sample (10 mL) was collected from each participant between 8:00 a.m. and 11:00 a.m. Blood samples were drawn using red-top, clot-activator silicone-coated Vacutainer® tubes that were allowed to clot at room temperature for approximately 30 minutes, and then centrifuged at 2000g for 15 minutes to obtain serum. The serum samples were carefully aliquoted into sterile polypropylene tubes using a Pasteur pipette and stored at −80°C for future analyses.
Participants then commenced the acupuncture intervention (5 weeks, 10 sessions) within 24 hours of blood collection. Within 1 week after completion of the 10th acupuncture session, participants completed the self-report questionnaires, and a second fasting blood sample (10 mL) was collected following the same procedures used at baseline.
Acupuncture Intervention
The acupuncture intervention was structured according to the Standards for Reporting Interventions in Controlled Trials of Acupuncture (STRICTA) guidelines (see Supplemental Material 1). 30 The treatment included 10 acupuncture sessions delivered twice weekly over a 5-week period, with a minimum interval of 1 day between sessions. All participants received a semi-standardized acupuncture protocol consisting of 2 components: (1) a core set of acupuncture points targeting common symptoms such as generalized pain, fatigue, and sleep disturbance, and (2) supplemental points individualized to address up to 3 of the participant’s most painful body regions (eg, breast/chest, fingers, shoulders, lower back, knees, hips, wrists, or feet/toes). A comprehensive description of the acupuncture points and treatment procedures is detailed in our previously published protocol. 31
Acupuncture needles remained inserted for 30 minutes per session, with gentle rotations performed at 10-minute intervals (10, 20, and 30 minutes) to enhance the therapeutic movement of qi and blood. A standardized Korean DBC® acupuncture needle, measuring 0.25 × 40 mm, constructed of stainless steel with a wound stainless-steel handle, was used across all sessions. The intervention was delivered by a licensed acupuncturist certified by the National Certification Commission for Acupuncture and Oriental Medicine (NCCAOM), who continuously monitored participants for any potential adverse reactions, including needle shock, bruising, or dizziness.
For fidelity check, study acupuncturists completed an acupuncture protocol checklist in REDCap after each acupuncture session. In this checklist, we evaluated treatment delivery (needle depth, stimulation times, adherence to standardized point selection), participant engagement, and the implementation of treatment-related skills. In addition, a study team member monitored intervention fidelity for each acupuncturist every 6 months.
Targeted GSH Analysis and ROS
After thawing on ice, 20 µL serum sample was mixed with 10 µL of an internal standard solution containing GSH and GSSG and 70 µL of acetonitrile. After vortex-mixing for 3 minutes on a MM 400 mill mixer, the samples were centrifuged at 21,000g and 5°C for 10 minutes. The supernatants were diluted with water fivefold. A standard solution containing standard substances was prepared in the IS solution in a similar way. This solution was serially diluted to have 9 calibration solutions. About 10 µL of each of the resultant sample solutions and calibration solutions was injected into a C18 column to run liquid chromatography–multiple reaction monitoring mass spectrometry (LC-MRM/MS) on an Agilent 1290 Ultra-High-Performance Liquid Chromatography (UHPLC) system coupled to an Agilent 6495C QQQ mass spectrometer with positive-ion detection. A heptafluorobutyric acid (HFBA) buffer solution and acetonitrile were used as the binary-solvent mobile phase for gradient elution under optimized conditions. The levels of ROS in the serum sample were detected by plate-based colorimetric ELISA kit (Cayman Chemicals, Ann Arbor, MI).
Measures
Demographics and Clinical Characteristics
Data on sociodemographic factors—including participants’ age, race, educational background, income level, and marital status—were collected using self-reported questionnaires. Additionally, clinical characteristics such as participants’ height, weight, cancer stage, and previous cancer treatments were recorded at baseline.
PROMIS Measures
The psychoneurological symptoms were assessed using the Patient-Reported Outcomes Measurement Information System (PROMIS)-29 (version 1.0) questionnaire. 32 PROMIS has demonstrated strong psychometric properties—including excellent internal consistency and convergent validity—in measuring cancer-related symptoms (Cronbach’s α range: .86-.96).33,34 Specifically, the PROMIS Pain Interference subscale, consisting of 4 items, evaluates how pain affects participants’ engagement in daily physical, psychological, and social activities. 35 The PROMIS Fatigue subscale, also comprised of 4 items, measures fatigue levels ranging from mild subjective tiredness to profound and persistent exhaustion. 36 The PROMIS Sleep Disturbance subscale, containing 4 items, captures participants’ perceptions regarding their sleep quality, depth, and restorative effects, as well as difficulties initiating or maintaining sleep, and overall satisfaction with sleep. 32
Statistical Analysis Plan
Sample size calculations were based on the paired Wilcoxon signed-rank test comparing participants’ metabolite levels before and after acupuncture. Using the median absolute deviation of pre–post differences in GSH from our feasibility study, we estimated an effect size of 0.5. With 35 participants, a two-sided paired Wilcoxon signed-rank test would provide 80% power to detect this effect size at an alpha of .05. To account for an anticipated 20% attrition rate during the acupuncture intervention, we plan to recruit 42 participants.
Sociodemographic and clinical characteristics of participants were summarized using means and standard deviations (SD) for continuous variables, and frequencies with percentages for categorical variables. To evaluate changes in metabolites and ROS concentrations before and after 10 sessions of acupuncture, paired t-test and Wilcoxon signed-rank test were performed. Since 58% of participants provided blood samples immediately after the final session and 42% provided samples within 1 week, we conducted sensitivity analyses to evaluate whether sampling timing influenced the results. We performed linear regression models with post-treatment metabolite levels as the outcome, adjusting for baseline metabolite levels and the blood-collection timing group. The PNS cluster score was calculated by averaging the scores of the PROMIS Pain Interference, Fatigue, and Sleep Disturbance measures. Correlation analysis was conducted to examine associations between the changes in metabolite levels and the changes in PNS. All statistical analyses were performed using IBM SPSS Statistics (Version 29.0).
Results
Demographic and Clinical Characteristics
Baseline demographic and clinical characteristics of the 42 participants are presented in Table 1. The study included 42 women with a mean age of 52.0 ± 10.5 years and an average body mass index (BMI) of 30.0 ± 6.0. The sample was composed of 50.0% White, 40.5% African American, and 2.4% Asian; additionally, 21.4% identified as Hispanic. Regarding education, 64.3% of participants completed at least a bachelor’s degree, while 2.4% had less than a high school education. In terms of marital status, 35.7% were married or living with a partner, 33.3% had never married, 26.2% were divorced or separated, and 4.8% were widowed. Participants’ household income varied, with 12 (28.6%) earning less than $35 000; 10 (23.8%) between $35 000 and $55 000; 12 (28.6%) between $55 000 and $100 000; and 8 (19.0%) exceeding $100 000. Among the participants, 19 (45.3%) had been diagnosed with Stage 0 or 1 breast cancer, 17 (40.5%) with Stage 2, and 6 participants (14.3%) with Stage 3. All participants were undergoing endocrine therapy, while 78.6% had received radiation therapy, and 64.3% had undergone chemotherapy. Additionally, 10 participants (23.8%) reported antidepressant use.
Baseline Characteristics of the Participants (N = 42).

Data are provided as n (%) for categorical variables and as mean ± standard deviation for continuous variables.
Abbreviations: BMI, body mass index; N, number; SD, standard deviation.
Concentration of GSH Metabolites and ROS Before and After Acupuncture
We examined changes in GSH metabolites and ROS levels following the acupuncture intervention. The concentrations of targeted GSH metabolites—including GSH, GSSG, and cysteine glutathione (CySSG), along with the GSH/GSSG ratio—were measured at baseline (pre-treatment) and immediately after the completion of the 5-week, 10-session intervention.
Figure 1 displays paired metabolite values measured before and after acupuncture treatment. ROS levels increased significantly after acupuncture treatment (baseline 2.34 ± 1.02 μmol/L vs post-treatment 2.83 ± 1.23 μmol/L, P = .017). In contrast, GSSG levels decreased significantly post-treatment compared to baseline (baseline: 0.19 ± 0.23 µmol/L vs post-treatment: 0.12 ± 0.23 µmol/L, P < .001), indicating a reduction in oxidized glutathione. Although GSH and CySSG showed non-significant increases following acupuncture (GSH baseline: 0.38 ± 0.14 µmol/L vs post-treatment: 0.42 ± 0.16 µmol/L, P = .07; CySSG baseline: 0.00503 ± 0.00082 µmol/L vs post-treatment: 0.00532 ± 0.00076 µmol/L, P = .06), these changes were directionally consistent with improved redox status. Together, these metabolite shifts result in a significant increase in the GSH/GSSG ratio (baseline: 2.73 ± 1.21 vs post-treatment: 5.38 ± 1.99, P < .001) after acupuncture, suggesting a shift toward reduced oxidative stress and overall enhancement in antioxidant capacity.

Changes in GSH metabolites and ROS pre and post 10 sessions of acupuncture (n = 42).
For the sensitivity analyses, ROS, GSH, GSH/GSSG ratio, CySSG changes were consistent between participants who provided samples immediately after the final session and those who provided samples within 1 week. A modest trend (P = .08) was noted for GSSG, with the 1-week group (change: −0.08 ± 0.08) showing a slightly greater decrease than the immediate group (change: −0.04 ± 0.05); however, this difference did not reach statistical significance.
Correlation Between Alterations of GSH Metabolites and Alterations of PNS Symptoms
All PNS symptoms showed significant improvement after 10 sessions of acupuncture sessions, as reported in our previous publication. 42 We further explored the associations between changes in GSH metabolites and alterations of PNS symptoms after acupuncture treatment. Correlation analyses were performed to investigate relationships between the magnitude of changes in GSH, GSSG, CySSG, the GSH/GSSG ratio, and the changes in patient-reported symptom severity scores.
Our analysis did not reveal any significant correlations between changes in GSH metabolites and changes in PNS composite score, or each individual PNS (see Table 2).
Correlation Between Alterations of GSH Metabolites and Alterations of PNS (N = 42).

Pearson correlation coefficient with 95% confidence interval, and P-value were reported in this table. PNS composite score was calculated based on the average PROMIS T score of pain interference, fatigue, and sleep disturbance.
Abbreviations: CySSG, Cysteine glutathione; GSH, reduced glutathione; GSSG, oxidized glutathione; PNS, psychoneurological symptoms.
Discussion
Our study revealed a significant increase in the GSH/GSSG ratio and ROS, and a significant decrease in GSSG following acupuncture treatment, suggesting overall enhancement of the body’s antioxidant defense system. These shifts align with previous research indicating that acupuncture can regulate GSH metabolism, modulate redox homeostasis, and play a crucial role in mitigating oxidative stress. Similarly, Li et al 28 found that electroacupuncture significantly increased glutamine (P = .02) and GSH (P < .011) concentrations, indicating its involvement in GSH metabolism among 60 patients with moderate depression. In another study of acupuncture in 40 overweight and obese individuals, Ghaemi et al 16 also observed increased levels of GSH and glutathione peroxidase (ie, an enzyme using GSH to neutralize hydrogen and lipid peroxides in order to prevent cell damage from oxidation).
Breast cancer survivors with PNS have higher levels of underlying inflammation and oxidative stress compared to healthy individuals.37,38 Over time, their bodies may adapt to this chronic inflammatory state, leading to blunted or dysregulated antioxidant response. Acupuncture, through its localized and controlled microtrauma, induces a transient increase in ROS (see Figure 2). While transient ROS elevation may indicate an adaptive redox response that promotes cellular signaling and homeostasis, the absence of temporal data limits our ability to determine whether this response is transient or sustained. We speculate that this short-lived burst of ROS may act as a hormetic trigger, a “good stress (oxidative eustress)” stimulating the body to strengthen its antioxidant defenses. 39 As a result, levels of endogenous antioxidants such as GSH increase, aiding in neutralization of excess ROS and protecting surrounding tissues from oxidative damage. 40 During this redox process, GSH is oxidized to form GSSG. 40 A higher GSH/GSSG ratio reflects a more favorable antioxidant capacity and a lower oxidative burden, whereas a lower ratio is indicative of heightened oxidative stress. 40 These findings suggest that acupuncture may contribute to the restoration of redox homeostasis by increasing GSH levels, reducing GSSG accumulation, and ultimately raising the GSH/GSSG ratio, thereby mitigating chronic oxidative stress. In the sensitivity analysis examining the timing of post-intervention blood collection, only a small and non-significant trend was observed for GSSG: participants who provided samples 1 week after the final session exhibited a slightly greater reduction in GSSG compared with those who provided samples immediately. This pattern is biologically plausible: immediate post-treatment samples may capture both the cumulative effects of previous repeated acupuncture sessions and the short-lived acute oxidative response triggered by the final needling. In contrast, samples collected within 1 week likely reflect a more complete recovery phase in which transient increases in GSSG have been reduced or recycled back to GSH. Although this trend did not reach statistical significance, it highlights the greater time sensitivity of GSSG compared with other GSH-related markers. Future studies may benefit from standardizing post-treatment blood-collection timing or recording exact time-to-collection to allow for more precise modeling. Given the limited human evidence on short- and long-term oxidative stress responses to acupuncture, longitudinal RCTs are warranted to clarify the temporal patterns and clinical relevance of these changes. Future studies should incorporate more frequent and standardized post-treatment sampling (eg, immediate, 1-week, 6-week time points) to better characterize the temporal dynamics of redox changes and to identify which biomarkers best capture the early versus delayed phases of acupuncture’s biological effects.
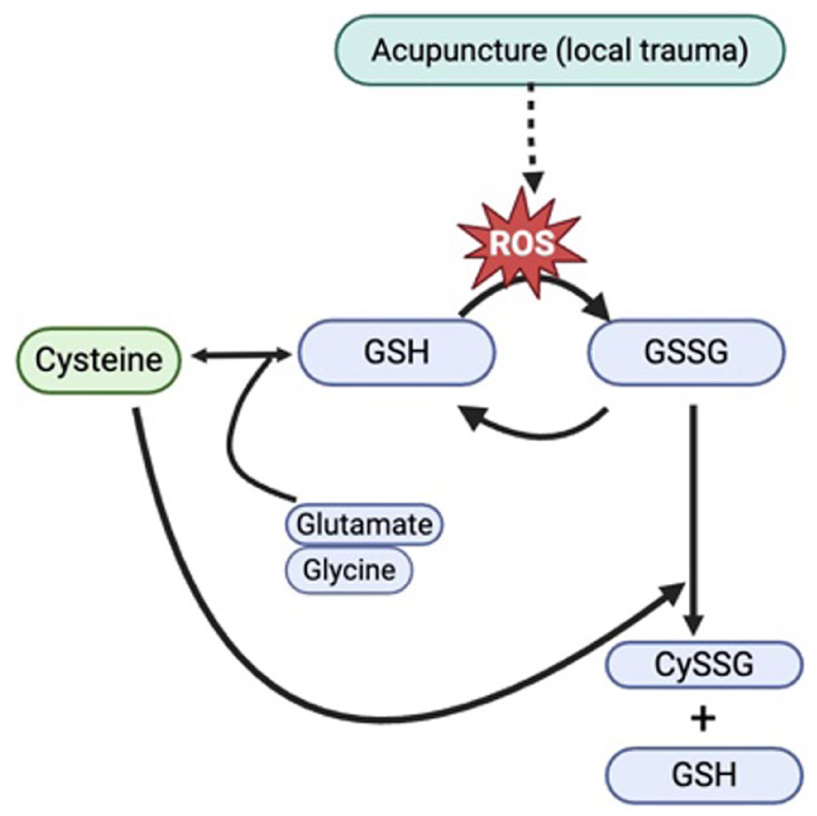
Proposed mechanism of acupuncture in modulating glutathione metabolism.
In this study, the GSH metabolites changes were not significantly associated with improvements in PNS. This finding suggests that while acupuncture modulates oxidative stress pathways, its therapeutic effects on PNS may be mediated by other mechanisms. One possible explanation is that rather than directly modulating symptom relief, GSH metabolism plays a broader role in redox homeostasis. While a review of prior studies has linked oxidative stress to chemotherapy-induced fatigue, depression, and cognitive impairment in cancer survivors, 41 our results suggest that specific changes in GSH metabolites alone may not be a primary driver of PNS improvement following acupuncture. Instead, other metabolic pathways such as sulfur metabolism may play a more direct role, as suggested by our previous findings. 42 Additionally, individual variability in oxidative stress responses may contribute to the lack of a clear association between GSH changes and PNS relief. Acupuncture likely exerts its effects through multiple biological pathways, including inflammatory modulation, neurotransmitter regulation, and gut microbiome interactions, rather than solely through redox balance. Future studies should explore whether specific subgroups of breast cancer survivors with distinct baseline oxidative stress profiles exhibit differential responses to acupuncture for PNS relief. Given that ROS are primarily produced by mitochondria and are tightly linked to cellular metabolism, 43 investigating mitochondrial function and metabolic pathways alongside oxidative stress markers could provide deeper mechanistic insights into acupuncture’s therapeutic effects. Moreover, employing a more comprehensive network metabolomics analysis may help to identify potential interaction effects between GSH metabolism and other metabolic pathways involved in acupuncture’s therapeutic effects. In summary, although our findings indicate that acupuncture significantly altered GSH metabolism in breast cancer survivors experiencing PNS, the therapeutic effects of acupuncture on PNS appear to be independent of alterations in GSH metabolism, suggesting that alternative biological pathways may be involved.
Despite the merits of these findings, this study has some limitations. First, the relatively small sample size with 100% female breast cancer survivors, limits the generalizability of these findings. Sex-related differences in oxidative stress pathways, hormonal regulation, and neuroimmune responses may influence biomarker dynamics and treatment effects. Future research should include both male and female participants, to determine whether these biological responses to acupuncture are consistent across populations and to enhance the external validity of the findings. Second, the absence of a placebo-controlled design restricts our ability to distinguish acupuncture’s effects from potential placebo responses. Future RCTs with large control groups and extended follow-up periods are needed to confirm our results and to further elucidate the underlying mechanisms through which acupuncture exerts its therapeutic effects. Third, this study was exploratory in nature; therefore, no adjustments for multiple comparisons (eg, Bonferroni or FDR) were applied to the biomarker analyses (GSH, GSSG, CySSG, ROS). Accordingly, the results should be viewed as exploratory and hypothesis-generating. Future studies are needed to validate these findings in larger RCTs. Lastly, another limitation involves the timing of post-treatment blood collection. Although all samples were obtained within 1 week of the 10th acupuncture session, 58% of participants provided samples immediately after the final treatment. Certain metabolites, such as GSSG, may be sensitive to acute physiological shifts following acupuncture, which could introduce variability. Future studies should standardize the timing of post-intervention blood draws and consider incorporating both immediate and delayed sampling to differentiate short-term responses from more enduring biochemical adaptations.
In conclusion, this exploratory study suggests that acupuncture may modulates GSH metabolism, enhancing antioxidant capacity, and reducing oxidative stress in breast cancer survivors with PNS. While these findings provide preliminary insights into potential biological mechanisms, causal inference cannot be made given the absence of a control group. Large-scale, placebo-controlled trials incorporating comprehensive metabolomic profiling and targeted analysis of the GSH pathway are warranted to further clarify the mechanisms of acupuncture and the clinical significance of changes in GSH metabolites and ROS.
Supplemental Material
sj-docx-1-ict-10.1177_15347354251411913 – Supplemental material for Changes in Glutathione Metabolites and Reactive Oxygen Species from Baseline to Post-Treatment in Breast Cancer Survivors with Psychoneurological Symptoms Receiving Acupuncture
Supplemental material, sj-docx-1-ict-10.1177_15347354251411913 for Changes in Glutathione Metabolites and Reactive Oxygen Species from Baseline to Post-Treatment in Breast Cancer Survivors with Psychoneurological Symptoms Receiving Acupuncture by Hongjin Li, Shuang Gao, Judith M. Schlaeger, Hannah Choi, Yinglin Xia, Jun Sun and Ardith Z. Doorenbos in Integrative Cancer Therapies
Footnotes
Acknowledgements
We would like to thank all the participants who greatly contributed to the findings of this study.
Ethical considerations
The study was registered with ClinicalTrials.gov (Identifier: NCT05417451) and received approval from the Institutional Review Board of the University of Illinois Chicago (IRB20210840).
Consent to participate
We confirm that all participants provided written informed consent.
Author contributions
All authors read and approved the final version of the manuscript. Conceptualization: Hongjin Li, Ardith Z. Doorenbos, Judith M. Schlaeger, Jun Sun, Yinglin Xia; Methodology: Hongjin Li, Yinglin Xia; Data analysis: Hongjin Li, Shuang Gao; Writing - original draft preparation: Hongjin Li, Shuang Gao, Hannah Choi; Writing - review and editing: Hongjin Li, Shuang Gao, Hannah Choi, Judith M. Schlaeger, Jun Sun, Yinglin Xia, Ardith Z. Doorenbos.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Cancer Institute (R21 CA277153) and National Centre for Complementary and Integrative Health (K24AT011995).
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability statement
The data supporting the findings of this study are available in the manuscript and can be obtained from the corresponding author upon request.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
