Abstract
This study investigated the anti-cancer effects of the ethyl acetate fraction from Opuntia humifusa on human breast cancer MDA-MB-231 cells. O. humifusa, a cactus species capable of surviving under extreme environmental conditions, is currently being studied for its potential pharmacological properties. The MDA-MB-231 cell line is a highly invasive and metastatic model of triple-negative breast cancer (TNBC), which lacks estrogen receptor, progesterone receptor, and human epidermal growth factor receptor 2. TNBCs are often clinically challenging because they generally have a poor prognosis and limited treatment options. The ethyl acetate fraction was obtained from ethanol extracts of fruit powder of O. humifusa. Treating MDA-MB-231 cells with ethyl acetate fraction for 48 hours reduced cell viability by more than 50% and inhibited migration demonstrated by wound healing assays. DAPI staining proved nuclear condensation and fragmentation, and flow cytometry confirmed the induction and progression of apoptosis and arrest of cell cycle at the S and G2/M phases. Western blot analysis revealed that Akt, ERK1/2, integrin β1, HIF-1α, MMP-9, MMP-7, CDK2, cyclin A2, pro-caspase-9, pro-caspase-8, pro-caspase-3, and lamin A/C were decreased in a dose-dependent manner, whereas Smac/DIABLO, cleaved caspase-9, cleaved caspase-8, and cleaved caspase-3 were increased in a concentration-dependent manner, indicating downregulation of proliferation, activation of upstream signal transduction influencing cell cycle arrest and apoptosis, deactivation of metastasis, induction of cell cycle arrest at the S and G2/M phases, and activation of intrinsic and extrinsic apoptotic pathways. These findings suggest that the ethyl acetate fraction of O. humifusa induces apoptosis, cell cycle arrest, and anti-metastasis as well as contributing to upstream signal transduction of anti-proliferation, apoptosis, and cell cycle arrest in human triple-negative breast cancer cells.
Introduction
Triple-negative breast cancer (TNBC) is an aggressive subtype of breast cancer defined by the absence of estrogen receptor (ER), progesterone receptor (PR), and human epidermal growth factor receptor 2 (HER2). 1 Clinically, TNBC tends to grow and spread more rapidly than other breast cancer subtypes, contributing to a higher risk of early metastasis and relapse. As a result, TNBC patients often face a poor prognosis, with survival outcomes significantly worse than hormone receptor-positive or HER2-positive breast cancers. 2 Importantly, because TNBC tumors lack the targets for endocrine or anti-HER2 therapies, treatment options are limited primarily to surgery and chemotherapy. 3 However, TNBC frequently shows drug resistance and high relapse rates, so existing chemotherapy has only unsatisfactory success rates, leading to an urgent need for new treatment strategies.4 -7
Current research efforts are increasingly exploring alternative and adjunct treatments for TNBC, including those derived from natural products. Natural compounds have historically been a rich source of anti-cancer agents, and they offer the potential for multi-targeted therapy with lower systemic toxicity. In the context of TNBC, chemotherapy remains the mainstay, but the emergence of chemo-resistant cancer stem cell subpopulations often leads to tumor relapse. Notably, various plant-derived molecules (eg, sulforaphane, curcumin, resveratrol) have shown the ability to induce cell cycle arrest, trigger apoptosis, and inhibit metastatic pathways in TNBC models.8 -14 Such findings highlight the promise of phytochemicals as therapeutic leads. Therefore, there is a strong rationale for exploring medicinal plants and their bioactive constituents as novel treatments for TNBC, which may improve outcomes either as monotherapy or in combination with standard treatment.15 -17
One promising source of bioactive compounds is Opuntia humifusa, commonly known as the Eastern prickly pear or devil’s tongue cactus. Members of the Opuntia genus have been used for centuries in traditional medicine and as dietary supplements, reputed for beneficial effects against chronic ailments including diabetes, obesity, and even cancer.18 -21 O. humifusa in particular is native to North America and Northeast Asia (called Cheonnyuncho in Korea) and is remarkable for its stress tolerance, capable of surviving extreme cold (−20°C) by drastically reducing its water content. Phytochemical analyses reveal that O. humifusa contains a large amount of nutrients such as polyphenol compounds, dietary fiber, vitamins, calcium, minerals, amino acids, and complex polysaccharides, and in particular, the ethyl acetate (EtOAc) extract is found to contain flavonoids, trans taxifolin (dihydroquercetin), and dihydrokaempferol.22,23
Considering the rich polyphenol profile and notable biological activities of O. humifusa, the EtOAc fraction of this plant was selected as a source of anti-TNBC compounds. Fractionation using EtOAc is a common strategy in natural products research to enrich mid-polarity phytochemicals, which often include potent anti-oxidant, anti-inflammatory, and anti-cancer agents. 24 This fraction previously showed excellent anti-oxidant and anti-inflammatory effects compared to more polar or nonpolar fractions. 25 Based on these characteristics, it was hypothesized that the EtOAc fraction of O. humifusa could exert significant anti-cancer effects against TNBC cells. Therefore, the purpose of this study is to systematically elucidate whether the EtOAc fraction exhibits effective anti-cancer and anti-metastatic activities in TNBC MDA-MB-231 cells.
Materials and Methods
Plant Material and Preparation of EtOAc Fraction
Freeze-dried powder of O. humifusa fruit was provided by Upo Wetland (Changnyeong, South Korea). For extraction, 250 g of the powder was subjected to reflux extraction with 50% ethanol (EtOH) for 6 hours, repeated 4 times to ensure complete extraction. The EtOH extracts were concentrated under reduced pressure using rotary evaporator (EYELA rotary evaporator N-1000, Tokyo, Japan) at 55°C. The concentrate was suspended in water and then fractioned with the following sequence of organic solvents: n-hexane (hexane), dichloromethane (DCM), EtOAc, n-butanol (BuOH), and water (H2O). Each solvent fraction was concentrated by rotary evaporation at 45°C and stored at −20°C until use. For in vitro experiments, the concentrated EtOAc fraction was dissolved in dimethyl sulfoxide (DMSO), and DMSO was used as a vehicle control in all assays performed (Figure 1).

Schematic diagram of the extraction and fractionation procedure of Opuntia humifusa fruit.
Cell Line and Reagents
MDA-MB-231 human breast cancer cells (KCLB Cat# 30026, RRID: CVCL_0062) and RAW 264.7 murine macrophages (KCLB Cat# 40071, RRID: CVCL_0493) were obtained from the Korean Cell Line Bank (KCLB, Seoul, South Korea). Roswell Park Memorial Institute medium (RPMI 1640, 1X, containing 2.05 mM L-glutamine), Dulbecco’s Modified Eagle Medium (DMEM), fetal bovine serum (FBS), and penicillin/streptomycin were purchased from HyClone (Logan, UT, USA). Phosphate-buffered saline (PBS, pH 7.4) was obtained from Welgene (Gyeongbuk, South Korea).
Annexin V Binding Buffer, FITC Annexin V, and Propidium Iodide Solution were purchased from BioLegend (San Diego, CA, USA). Cell Cycle Phase Determination Fixative, RNase A Solution, and Propidium Iodide Reagent were obtained from Cayman Chemical Company (Ann Arbor, MI, USA). All other reagents used were of analytical grade or of the highest possible purity.
In Vitro Experiment
Culture of MDA-MB-231 and RAW 264.7 Cells
MDA-MB-231 cells and RAW 264.7 cells were cultured at 37°C in a humidified incubator with 5% CO₂. MDA-MB-231 cells were maintained in RPMI 1640 medium supplemented with 10% fetal bovine serum (FBS) and 100 units/mL penicillin, and subcultured every 2 to 3 days. RAW 264.7 cells were maintained in DMEM with the same supplementation and subcultured every 1 to 2 days. In all experiments, cells were seeded and allowed to stabilize for 24 hours before the medium was replaced with serum-free medium containing the EtOAc fraction of O. humifusa or DMSO. All controls used 1% DMSO as the solvent.
Cell Viability Assay
Cell viability was assessed using the CellTiter 96® AQueous One Solution Cell Proliferation Assay Kit (Promega, Madison, WI, USA) following the manufacturer’s instructions. Cells were seeded in 96-well plates and incubated at 37°C for 24 hours. After incubation, the cells were treated with various concentrations of O. humifusa EtOAc fractions for 24 hours, 36 hours, or 48 hours. Subsequently, MTS reagent [3-(4,5-dimethylthiazol-2-yl)-5-(3-carboxymethoxyphenyl)-2-(4-sulfophenyl)-2H-tetrazolium] was added and the plates were incubated for 2 hours at 37°C in a humidified atmosphere with 5% CO₂. Absorbance was measured at 490 nm using a Cell Imaging Multi-Mode Reader (Agilent BioTek Cytation5, VT, USA).
Fluorescent Labeling of Nuclei Using 4′,6-Diamidino-2-Phenylindole (DAPI)
MDA-MB-231 cells were seeded into 96-well plates and incubated at 37°C in a humidified atmosphere containing 5% CO₂. After stabilization for 24 hours, cells were treated with 250 μg/mL of O. humifusa EtOAc fraction for 48 hours, which was selected based on cell viability experiments as the most effective concentration to induce apoptosis without excessive cytotoxicity, as shown in the results. The DAPI staining solution (ThermoFisher, Waltham, MA, USA) was prepared by dissolving the entire contents of a commercial DAPI vial in 2 mL of dimethylformamide to obtain a 14.3 mM (5 mg/mL) stock solution. An intermediate dilution of 300 μM was prepared by adding 2.1 μL of the stock to 100 μL of PBS, and a final working solution of 300 nM was obtained by diluting 10 μL of the intermediate solution in 10 mL of PBS. Following treatment, cells were washed 2 times with PBS. Subsequently, 100 μL of the 300 nM DAPI staining solution was added to each well and incubated for 3 minutes at room temperature in the dark. The stain was then removed, and the cells were washed twice with PBS. Nuclear morphology was examined using a Cytation 5 Cell Imaging Multi-Mode Reader (Agilent BioTek, VT, USA) equipped with a DAPI filter.
Apoptosis Assay
Apoptosis was assessed using the FITC Annexin V Apoptosis Detection Kit with PI (BioLegend, San Diego, CA, USA) according to the manufacturer’s protocol. MDA-MB-231 cells (1.5 × 10⁵ cells/mL) were seeded into 6-well plates and treated with various concentrations of O. humifusa EtOAc fractions for 48 hours. Cells were harvested by centrifugation at 300g, washed twice with cold staining buffer, and resuspended in 200 μL of Annexin V Binding Buffer. Annexin V-FITC (5 μL) and propidium iodide (5 μL) were added, and the mixture was gently vortexed and incubated for 15 minutes at room temperature in the dark. After adding 400 μL of Binding Buffer, samples were analyzed immediately using a flow cytometer (Agilent Novocyte Advanteon, CA, USA) equipped with NovoExpress™ software (Agilent Technologies, RRID: SCR_024676) for data acquisition and analysis. A minimum of 10 000 events were acquired per sample to ensure statistical reliability.
Cell Cycle Analysis
Cell cycle distribution was determined using the Cell Cycle Phase Determination Kit (Cayman Chemical, Ann Arbor, MI, USA). MDA-MB-231 cells (1.5 × 10⁵ cells/mL) were treated with different concentrations of O. humifusa EtOAc fractions for 48 hours, then collected and washed. The cells were fixed in ice-cold fixative at −20°C for 2 hours. After centrifugation, the fixative was removed, and the cells were resuspended in 500 μL of staining solution containing 10 μL of RNase A and 10 μL of PI. Samples were incubated for 30 minutes at room temperature in the dark, and then analyzed by flow cytometry. A minimum of 10 000 events were acquired per sample to ensure statistical reliability.
Wound Healing Assay
MDA-MB-231 cells were seeded at 4 × 10⁵ cells/mL in 6-well plates and cultured for 24 hours. A scratch was made through the monolayer using a sterile 100 μL pipette tip. The medium was replaced with serum-free medium containing either 1% DMSO or O. humifusa EtOAc fraction (250 μg/mL). Wound closure was monitored at 0 and 48 hours using a phase-contrast microscope (BioTek Cytation5, VT, USA) at 40× magnification. Cell migration was quantified by measuring the wound width from captured images.
Western Blot Analysis
MDA-MB-231 cells were treated with 250 μg/mL of O. humifusa EtOAc fraction, washed with PBS, and harvested using a cell scraper. Cell pellets were lysed in buffer on ice for 1 hours, followed by centrifugation at 10 000g for 10 minutes. Protein concentration was determined using a BCA assay (Pierce, Rockford, IL, USA). Equal amounts of protein were mixed with Laemmli sample buffer, heated at 95°C for 5 minutes, and separated by SDS-PAGE (10%-12%). Proteins were transferred to PVDF membranes using a wet transfer system (Hoefer, MA, USA). Membranes were blocked with 5% non-fat milk in TBST and incubated overnight at 4°C with primary antibodies. After washing, membranes were incubated with HRP-conjugated secondary antibodies (1:5000 dilution, Enzo Life Sciences (Farmingdale, NY, USA). for 1 hours. Protein bands were detected using WesternBright ECL (Advansta, San Jose, CA, USA).
Primary antibodies used were as follows (all 1:1000 dilution): GAPDH (Cell Signaling Technology Cat# 2118, RRID: AB_561053), cleaved caspase-3 (Cat# 9664, RRID: AB_2070042), cleaved caspase-8 (Cat# 9496, RRID:AB_561381), cleaved caspase-9 (Cat# 7237, RRID: AB_10895832), pro-caspase-3 (Cat# 9662, RRID: AB_331439), pro-caspase-8 (Cat# 4790, RRID: AB_10545768), pro-caspase-9 (Cat# 9502, RRID:AB_2068621), ERK1/2 (Cat# 9102, RRID: AB_330744), Akt (Cat# 9272, RRID: AB_329827), lamin A/C (Cat# 2032, RRID: AB_2136278), HIF-1α (Cat# 3716, RRID: AB_2116962), integrin β1 (Cat# 4706, RRID: AB_823544), MMP-7 (Cat# 71031, RRID: AB_2799796), MMP-9 (Cat# 3852, RRID: AB_2144868), Smac/DIABLO (Cat# 15108, RRID: AB_2798711), cyclin A2 (Cat# 67955, RRID: AB_2909603), CDK4 (Cat# 12790, RRID: AB_2631166), and CDK2 (Cat# 2546, RRID: AB_2276129), all from Cell Signaling Technology (Danvers, MA, USA). The antibody against pro-caspase-12 (ABclonal Cat# A22864, RRID: not available) was obtained from ABclonal (Woburn, MA, USA), and the β-actin antibody (Enzo Life Sciences Cat# ADI-905-733, RRID: AB_10616090) was from Enzo Life Sciences (Farmingdale, NY, USA).
Statistical Analysis
All statistical analyses were performed using GraphPad Prism (RRID: SCR_002798) version 8.0.2 (GraphPad Software, San Diego, CA, USA). Data were presented as mean ± standard error of the mean (SEM) from at least 3 independent experiments. To evaluate statistical significance, two-way ANOVA followed by Dunnett’s multiple comparisons test was conducted to compare treatment groups with the control or vehicle control group. A P-value of less than .05 (P < .05) was considered statistically significant.
Results
Preparation of EtOAc Fraction from O. humifusa
The EtOH extract and EtOAc fraction of O. humifusa fruit powder were successfully prepared. The fruit powder was extracted with 50% EtOH and then fractionated with EtOAc. The EtOAc fraction was concentrated under reduced pressure, and it was in the form of mucilage rather than a dry powder. A total of 46.11 g of EtOAc fraction (including the weight of the glass vial) was obtained from 250 g of O. humifusa fruit powder. Excluding the vial, the net weight of the EtOAc fraction was 9.3 g (Figure 2).

Schematic representation of the extraction and fractionation of the ethyl acetate layer from O. humifusa fruit powder. The fruit powder was extracted with 50% ethanol, concentrated, and partitioned with ethyl acetate and distilled water (1:1, v/v). The ethyl acetate fraction was collected and concentrated for further experiments.
Effects of EtOAc Fraction from O. humifusa on Cell Viability
To evaluate the cytotoxic effect of the EtOAc fraction derived from O. humifusa, an MTS assay was performed on MDA-MB-231 breast cancer cells and RAW 264.7 macrophages. In MDA-MB-231 cells, treatment with the EtOAc fraction for 24 to 48 hours led to a clear, concentration- and time-dependent decrease in cell viability. Notably, cell viability was reduced by more than 50% at concentrations of 200 μg/mL or higher, indicating potent anti-cancer activity of the fraction.
In contrast, RAW 264.7 cells exposed to the same concentrations for 24 hours did not exhibit significant cytotoxicity. Moreover, a modest yet reproducible increase in cell viability was observed at the concentration of 100 μg/mL (Figure 3).

Effects of ethyl acetate fraction of O. humifusa on cell viability. MDA-MB-231 cells (5 × 103 cells/well) were treated with the indicated concentrations of the ethyl acetate fraction (Control [untreated], DMSO, 100, 150, 200, and 250 μg/mL). Cell viability was assessed using the MTS assay after 24, 36, and 48 hours. RAW 264.7 cells (1 × 104 cells/well) were treated with the indicated concentrations (100, 150, 200, and 250 µg/mL) of the ethyl acetate fraction. Cell viability was assessed at 24 hours using the MTS assay. The amount of control DMSO is equal to the amount of DMSO used to dissolve the 250 μg/mL ethyl acetate fraction. Data are presented as mean ± SD (n = 3). ***P < .001 versus DMSO-treated group; ###P < .001 versus control.
Induction of Apoptosis
Treatment of MDA-MB-231 cells with the EtOAc fraction of O. humifusa induced apoptosis, as evidenced by DAPI nuclear staining, flow cytometry, and western blot analysis. DAPI staining revealed distinct morphological hallmarks of apoptosis in EtOAc-treated MDA-MB-231 cells, including nuclear condensation, chromatin fragmentation, and the formation of apoptotic bodies (Figure 4C, white arrows). In contrast, nuclei in control (Figure 4A) and DMSO-treated cells (Figure 4B) maintained intact, round morphology with homogeneous fluorescence intensity, showing no evidence of apoptotic changes (Figure 4).
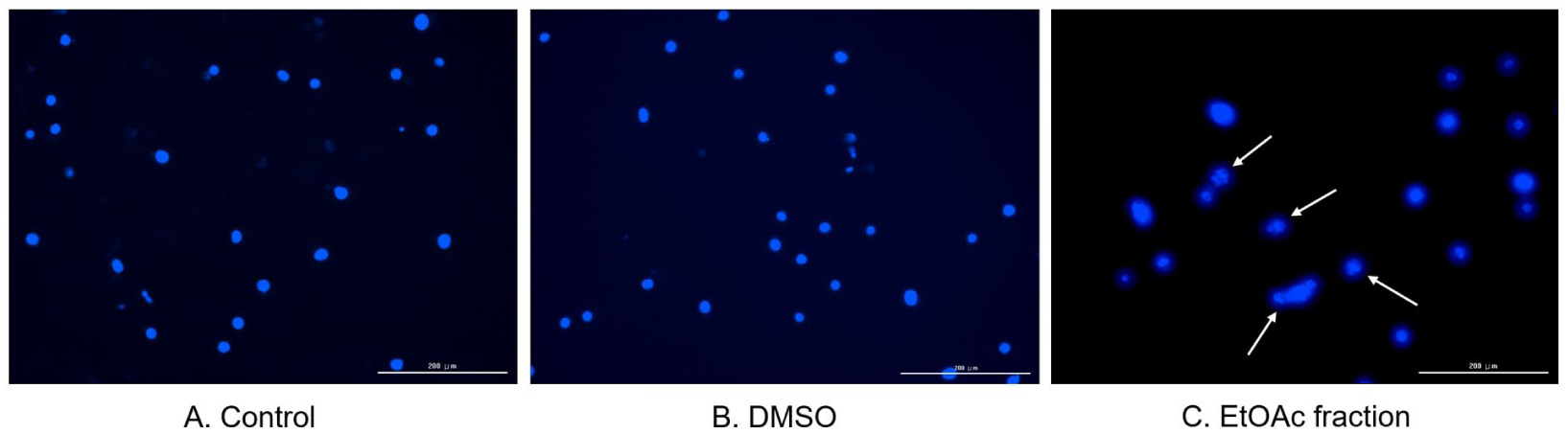
Nuclear morphological changes induced by the ethyl acetate fraction of O. humifusa in MDA-MB-231 cells. MDA-MB-231 cells were treated for 48 hours with (A) control, (B) DMSO, or (C) 250 µg/mL of the ethyl acetate fraction. After fixation, cells were stained with DAPI and imaged using a fluorescence microscope. Characteristic features of apoptosis, such as chromatin condensation and nuclear fragmentation, are indicated by white arrows. Scale bar: 200 µm.
The effect of the EtOAc fraction on apoptosis of MDA-MB-231 cells was reconfirmed by Annexin V/PI double staining followed by flow cytometry (Figure 5). In the control and DMSO-treated groups, the majority of cells were Annexin V⁻/PI⁻ (88.48% and 90.90%, respectively), indicating viable cells. However, treating MDA-MB-231 cells with the EtOAc fraction significantly decreased the proportion of viable cells to 29.20%. In contrast, the percentage of early apoptotic cells (Annexin V⁺/PI⁻) increased dramatically from 2.64% (control) and 2.68% (DMSO) to 46.97% in the EtOAc-treated group. Late apoptotic cells (Annexin V⁺/PI⁺) were also elevated from 8.33% and 6.02% to 22.79%, respectively. The necrotic population (Annexin V⁻/PI⁺) remained low and comparable across all groups (Control: 0.56%, DMSO: 0.40%, EtOAc: 1.04%). Quantitative analysis (Figure 5B) confirmed that the total percentage of apoptotic cells (early + late) was significantly increased in the EtOAc-treated group compared to the DMSO control group.

Flow cytometry analysis of apoptotic cell death induced by the ethyl acetate fraction of O. humifusa in MDA-MB-231 cells. MDA-MB-231 cells were treated with control, DMSO, or 250 μg/mL of the ethyl acetate fraction for 48 hours. Following treatment, cells were stained with Annexin V-FITC and propidium iodide (PI), and analyzed by flow cytometry using the PE channel for PI detection. (A) Representative dot plots showing 4 distinct cell populations: viable cells (Annexin V⁻/PI⁻, lower left quadrant), early apoptotic cells (Annexin V⁺/PI⁻, lower right), late apoptotic or necrotic cells (Annexin V⁺/PI⁺, upper right), and necrotic cells (Annexin V⁻/PI⁺, upper left). (B) Quantification of the percentage of cells in each quadrant presented as a stacked bar graph. Data are presented as mean ± SD (n = 3). ***P < .001 versus DMSO-treated group.
To further elucidate the molecular mechanisms involved in apoptosis of MDA-MB-231 cells, western blot analysis was performed (Figure 6). Treatment of MDA-MB-231 cells with the EtOAc fraction dose-dependently increased the levels of cleaved caspase-3, cleaved caspase-8, cleaved caspase-9, and Smac/DIABLO, whereas it dose-dependently decreased the levels of pro-caspase-3, pro-caspase-8, pro-caspase-9, and pro-caspase-12. These results suggest that the EtOAc fraction induces apoptosis via extrinsic and intrinsic pathways and may also involve endoplasmic reticulum (ER) stress-related signaling, while simultaneously inhibiting the inhibitor of apoptosis (IAP).

Western blot analysis of apoptosis-related proteins in MDA-MB-231 cells treated with the ethyl acetate fraction of O. humifusa. (A) MDA-MB-231 cells were treated with control, DMSO, or 150 and 250 μg/mL of the ethyl acetate fraction of O. humifusa for 48 hours. Protein levels of cleaved and pro-forms of caspase-3, caspase-8, caspase-9, pro-caspase-12, and Smac/DIABLO were analyzed. (B) Protein relative levels of cleaved and pro-forms of caspase-3, caspase-8, caspase-9, pro-caspase-12, and Smac/DIABLO. Data are presented as mean ± SD (n = 3). **P < .01, ***P < .001 versus DMSO-treated group.
Induction of Cell Cycle Arrest
To determine whether the EtOAc fraction of O. humifusa affects cell cycle progression, MDA-MB-231 cells were treated with the fraction for 48 hours and analyzed by flow cytometry. When treated with 250 μg/mL of the EtOAc fraction, the percentage of cells in the G1 phase significantly decreased from 63.08% to 32.72%, whereas the percentages of cells in S and G2/M phases significantly increased from 5.61% and 16.52% to 13.40% and 36.93%, respectively (Figure 7). These results suggest that the EtOAc fraction induces cell cycle arrest in S and G2/M phases.

Cell cycle distribution of MDA-MB-231 cells following treatment with the ethyl acetate fraction of O. humifusa. MDA-MB-231 cells were treated with control, DMSO vehicle, or 250 μg/mL of the ethyl acetate fraction of O. humifusa for 48 hours. (A) Flow cytometric histograms show the distribution of cells in sub-G1, G1, S, and G2/M phases, expressed as a percentage of total gated cell populations. (B) Quantitative analysis of the proportion of cells in each cell cycle phase based on histogram data. Data are presented as mean ± SD (n = 3). ***P < .001 versus DMSO-treated group.
To elucidate the molecular mechanism of this cell cycle arrest, the levels of key regulatory proteins involved in this pathway were analyzed by western blot analysis. Cyclin A2 and CDK2 were significantly decreased in a dose-dependent manner, while CDK4 showed little change (Figure 8). Since cyclin A2 and CDK2 are essential for both S phase progression and G2/M phase transition, their downregulation is consistent with the previously confirmed flow cytometric data. These results suggest that the EtOAc fraction effectively induces cell cycle arrest in S and G2/M phases by inhibiting cyclin A2 and CDK2-mediated signaling pathways.

Western blot analysis of cell cycle regulatory proteins in MDA-MB-231 cells treated with the ethyl acetate fraction of O. humifusa. (A) MDA-MB-231 cells were treated with control, DMSO, or 150 and 250 μg/mL of the ethyl acetate fraction of O. humifusa for 48 hours. Protein levels of CDK2, CDK4, and cyclin A2 were assessed. (B) Protein relative expression of CDK2, CDK4, and cyclin A2. Data are presented as mean ± SD (n = 3). ***P < .001 versus DMSO-treated group.
Inhibition of Migration and Metastasis-Associated Markers
After treating MDA-MB-231 cells with EtOAc fraction, the migration ability of these cells was evaluated through wound healing assay. After 48 hours of incubation, the untreated control and DMSO-treated groups exhibited significant closure of the scratch wound area, indicating active cell migration. In contrast, cells treated with 150 and 250 μg/mL of the EtOAc fraction demonstrated markedly suppressed wound closure, with wider gaps remaining compared to the control groups. This inhibitory effect on cell migration was more pronounced at 250 μg/mL than at 150 μg/mL, suggesting a concentration-dependent decrease in migration activity (Figure 9). These results suggest that the EtOAc fraction inhibits the migration of MDA-MB-231 cells in a concentration-dependent manner.

Wound healing assay of MDA-MB-231 cells treated with the ethyl acetate fraction of O. humifusa. MDA-MB-231 cell monolayers were scratched and then treated with control, DMSO, or the ethyl acetate fraction at 150 or 250 μg/mL. At 0 hours, scratch wounds were freshly made and initial wound widths were recorded. At 48 hours post-treatment, wound closure was assessed by imaging. Yellow dashed lines indicate the wound margins. Cells treated with the ethyl acetate fraction exhibited markedly reduced migration compared to control and DMSO-treated groups, as evidenced by decreased wound closure. Scale bar: 1000 µm.
To elucidate the mechanism of these anti-migration effects, the levels of migration-, invasion-, and angiogenesis-related mediators were analyzed by western blot. The EtOAc fraction decreased the expression levels of MMP-7, MMP-9, HIF-1α, and integrin β1 in a concentration-dependent manner (Figure 10). Since these mediators are directly or indirectly related to extracellular matrix degradation, cell adhesion, hypoxic adaptation, cell migration, invasion, proliferation, and differentiation, it is suggested that the EtOAc fraction cooperatively exhibits anti-metastatic activity by suppressing the expression levels of these mediators.
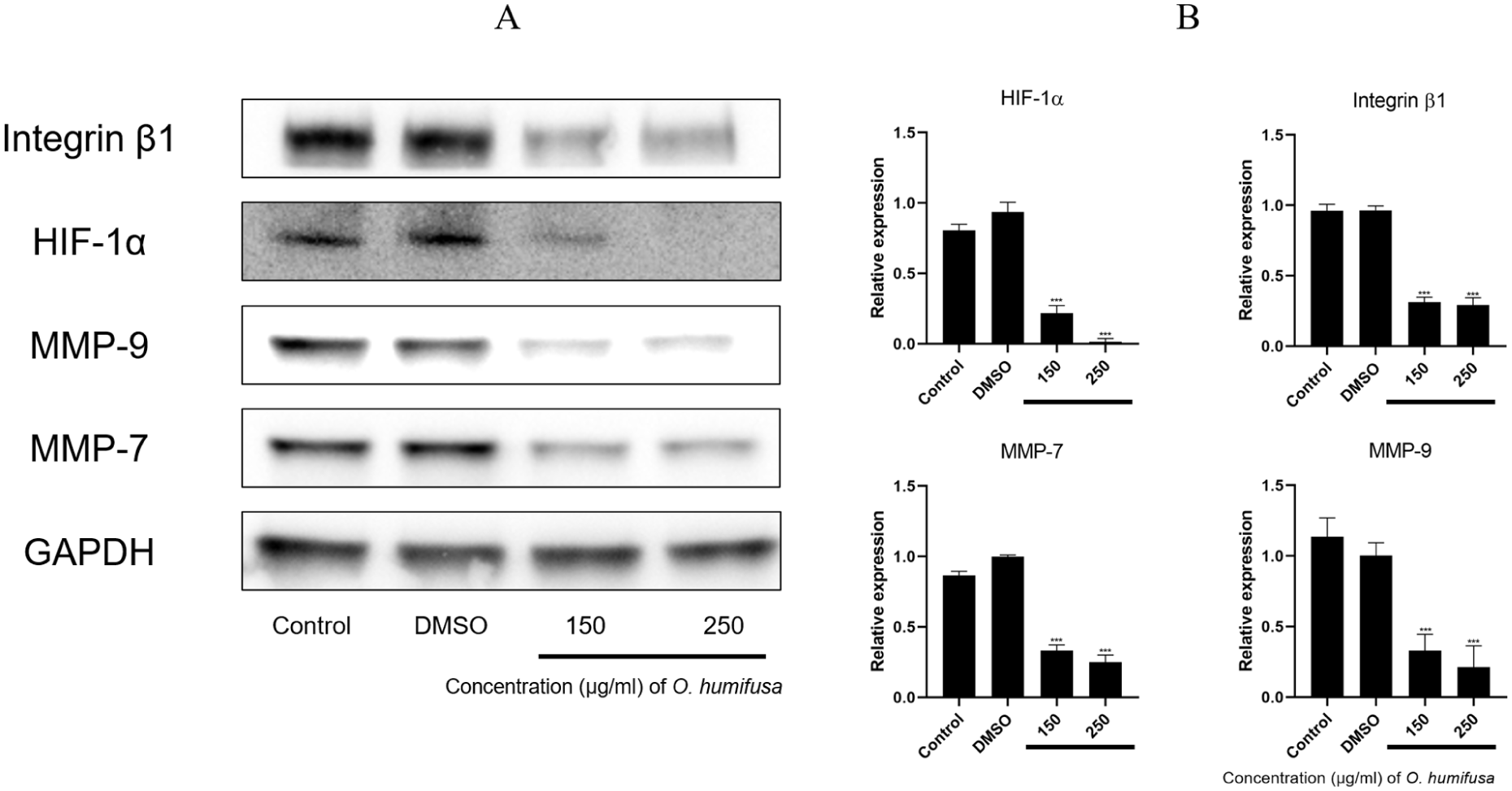
Effects of the ethyl acetate fraction of O. humifusa on metastasis- and hypoxia-associated proteins in MDA-MB-231 cells. (A) MDA-MB-231 cells were treated with control, DMSO, or the ethyl acetate fraction of O. humifusa (150 and 250 μg/mL) for 48 hours. Protein expression levels of MMP-7, MMP-9, HIF-1α, and integrin β1 were assessed. (B) Protein relative expression of MMP-7, MMP-9, HIF-1α, and integrin β1. Data are presented as mean ± SD (n = 3). ***P < .001 versus DMSO-treated group.
Downregulation of Upstream or Downstream Key Signaling Molecules Associated with Cell Proliferation, Survival, and Anti-apoptosis
To further investigate the upstream or downstream regulatory pathways that may be affected by the EtOAc fraction, the levels of Akt, ERK1/2, and lamin A/C were assessed by western blot analysis. Both Akt and ERK1/2 were decreased in a dose-dependent manner, which are key mediators of the upstream regulatory pathways of cell proliferation and survival. In addition, lamin A/C was also decreased in a dose-dependent manner, which is a key mediator of the downstream regulatory pathways involved in cell survival, anti-apoptosis, nuclear integrity, chromatin organization, and DNA replication (Figure 11). These results suggest that the EtOAc fraction of O. humifusa cooperatively regulates the upstream or downstream signaling pathways involved in cell proliferation, survival, and apoptosis.

Effects of the ethyl acetate fraction of O. humifusa on Akt, ERK1/2, and Lamin A/C expression in MDA-MB-231 cells. (A) Cells were treated with control, DMSO, or the ethyl acetate fraction of O. humifusa (150 and 250 μg/mL) for 48 hours. Western blotting was conducted to assess the levels of total Akt, ERK1/2, and Lamin A/C. (B) Protein relative expression of Akt, ERK1/2, and Lamin A/C. Data are presented as mean ± SD (n = 3). **P < .01, ***P < .001 versus DMSO-treated group.
Discussion
The EtOAc fraction of O. humifusa was successfully prepared through the EtOH extraction and EtOAc fractionation processes. The EtOAc fraction used in this study is known to contain dehydroquercetin, quercetin, isorhamnetin, kaempferol and their derivatives.26,27 The EtOAc fraction significantly decreased the viability of MDA-MB-231 TNBC in a concentration- and time-dependent manner, but did not exhibit cytotoxic effects on RAW 264.7 macrophages at the same concentration, indicating selective cytotoxicity against TNBC. On the other hand, the DMSO-treated RAW 264.7 cells showed a decrease in cell viability compared to the control DMSO-untreated RAW 264.7 cells, and these results have been similarly reported in other studies.28 -30 Therefore, it is suggested that RAW 264.7 cells may be sensitive to the DMSO solvent. Although RAW 264.7 macrophages are not epithelial in origin, they were selected as a non-cancerous reference model in this study. These cells are widely used in natural compound screening studies to evaluate the cytotoxicity of non-malignant cells due to their stable growth, reproducibility, and sensitivity to cytotoxicity. The purpose of this experimental design was to evaluate whether the EtOAc fraction induces cytotoxicity in general normal cells, rather than directly comparing the cytotoxicity differences observed between breast normal cells and breast cancer cells.31,32
Interestingly, treatment with 100 μg/mL of the EtOAc fraction consistently resulted in a slight but significant increase in RAW 264.7 cell viability, as confirmed by direct microscopic observation showing increased cell density. Although the underlying mechanism is still unclear, it is suggested that the EtOAc fraction may have cell proliferation and survival-supporting effects at low concentrations rather than at high concentrations.33,34
EtOAc fraction exhibited anti-cancer activity by inducing apoptosis through various pathways in MDA-MB-231 cells. DAPI staining showed typical apoptotic nuclear changes including chromatin condensation and nuclear fragmentation. Annexin V/PI staining showed a significant increase in the early and late apoptotic cell populations. Western blot analysis showed that the levels of cleaved caspase-3, -8, -9, and Smac/DIABLO were increased in a dose-dependent manner, whereas the levels of pro-caspase-3, pro-caspase-8, pro-caspase-9, and pro-caspase-12 were decreased in a dose-dependent manner. These data suggest that the EtOAc fraction induces apoptosis through extrinsic and intrinsic pathways, along with the IAP inhibition pathway via Smac/DIABLO. Meanwhile, the involvement of ER stress-mediated apoptosis is also presumed, but further verification is needed. Although a band corresponding to pro-caspase-12 was detected in the western blot analysis, it is widely acknowledged that CASP12 functions as a non-functional pseudogene in most human populations, particularly those of Caucasian descent, due to a premature stop codon. In this context, the observed band in MDA-MB-231 cells may represent a caspase-12-like isoform or result from antibody cross-reactivity with other caspases, such as caspase-4, which has been suggested to serve as a functional homolog of caspase-12 in humans. Similar results have been reported in previous studies using MDA-MB-231 and other human cancer cell lines.35 -37 Interestingly, functional CASP12 expression has been reported in a subset of individuals of African ancestry, indicating potential population-dependent variability.38,39 These results are consistent with previous reports that plant-derived polyphenol compounds can induce apoptosis in cancer cells through various caspase cascades. Simultaneous activation of multiple apoptosis pathways suggests that it may be useful in the treatment of highly migratable, invasive, and treatment-resistant cancers such as MDA-MB-231 (Figure 11).40 -45
In addition to inducing apoptosis, the EtOAc fraction also induced cell cycle arrest. Flow cytometry analysis revealed a significant decrease in the G1 population and a concurrent increase in the S and G2/M populations after EtOAc fraction treatment. These results suggest that the EtOAc fraction induces arrest at both DNA synthesis and mitotic entry checkpoints. Western blot analysis showed a dose-dependent decrease in the expression of CDK2 and cyclin A2, key regulators of S and G2/M phases, which is consistent with the flow cytometry data. CDK2 is involved in both G1 to S phase progression and S to G2 phase progression, while cyclin A2 is involved in both DNA replication and the G2/M phase transition. In contrast, CDK4 levels were relatively unchanged, suggesting that the EtOAc fraction has little effect on CDK4-mediated G1/S phase progression. Taken together, these results indicate that cell cycle arrest is induced through inhibition of CDK2 and cyclin A2-dependent pathways rather than CDK4-dependent pathways. This is consistent with other findings and suggests that inhibition of CDK2-cyclin complex formation may be a promising strategy for the treatment of TNBC resistant to hormone-based therapy.46,47
In a study of MDA-MB-231 cells treated with the EtOAc fraction of Orostachys japonicus, both extrinsic and intrinsic apoptotic pathways were activated and the cell cycle was arrested in sub-G1, but in this study, similarities and differences were found in that both extrinsic and intrinsic apoptotic pathways were activated but the cell cycle was arrested in S and G2/M phases. 8
The mechanisms of anti-cancer activity of EtOAc fractions from other plants rich in polyphenols have also been studied. For example, the EtOAc fraction of Scutellaria baicalensis induced apoptosis and arrested the cell cycle at the S and G2/M phases in MCF-7 breast cancer cells. In addition, the EtOAc fraction of Phoenix dactylifera L. (Ajwa dates) promoted apoptosis and S phase arrest in prostate cancer cells. And the EtOAc fraction of Camellia sinensis was reported to induce caspase-3, -8, and -9-mediated apoptosis in colon cancer cells.48 -50 Our results share some similarities and differences with previous studies in that they activate both the extrinsic and intrinsic apoptotic pathways and arrest the cell cycle at the S and G2/M phases.
Finally, the EtOAc fraction showed inhibitory effects on cell migration. First, in the wound healing test, the EtOAc fraction showed visible inhibition of active migration of MDA-MB-231 cells in a concentration-dependent manner. Of course, since the rate of apoptosis is high in the high-concentration range of the EtOAc fraction, it cannot be ruled out that this may have affected the results of cell migration inhibition. In the western blot analysis, the EtOAc fraction decreased the levels of MMP-7, MMP-9, HIF-1α, and integrin β1 in a concentration-dependent manner. These mediators are involved in extracellular matrix degradation, cell adhesion, hypoxic tumor microenvironment adaptation, cell migration, invasion, proliferation, and differentiation.51,52 Therefore, it can be assumed that the inhibition of cell migration can be sufficiently caused by metastasis suppressors, although apoptosis may have some effect. Meanwhile, the levels of Akt and ERK1/2, which are upstream regulators of signaling involved in cell migration as well as apoptosis and cell cycle arrest, were decreased in a concentration-dependent manner. In addition, the levels of lamin A/C, a core component of the nuclear envelope and a downstream mediator governed by cleaved caspase-3, an executioner caspase, were decreased in a concentration-dependent manner. Collectively, these results suggest that when the EtOAc fraction effectively inhibits cell adhesion, extracellular matrix remodeling, and migration, it directly or indirectly affects not only key mediators of these pathways, but also upstream regulators and downstream mediators of signal transduction.
Conclusion
The EtOAc fraction from O. humifusa selectively decreased the viability of TNBC cells, induced apoptosis via the extrinsic and intrinsic pathways, and induced cell cycle arrest at the S and G2/M phases. In addition, the EtOAc fraction inhibited cell migration by downregulating adhesion and migration-related proteins. Furthermore, the EtOAc fraction modulated upstream and downstream components of signaling pathways involved in apoptosis, proliferation, and migration. These findings provide a basis for further investigation into the therapeutic potential of the EtOAc fraction in TNBC, particularly its multifaceted anti-cancer activities.
Footnotes
Ethical Considerations
This study was approved by the Korea National Institute for Bioethics Policy Public Institutional Review Board (INJE IRB 2024-11-037-001).
Author Contributions
Myung Chul Cha: Conceptualization, Investigation, Methodology, Resources, Validation, Visualization, Writing - original draft. Eonji Yeo: Methodology, Writing - review & editing. Dong Seok Lee: Conceptualization, Formal analysis, Funding acquisition, Project administration, Resources, Supervision, Validation, Writing - review & editing.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was supported by the BK21 FOUR (Fostering Outstanding Universities for Research, No.5199990614110) funded by the Ministry of Education (MOE, Republic of Korea) and National Research Foundation of Korea (NRF).
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
The data used to support the findings of this study are included within the article.
