Abstract
Ovarian cancer, a frequently occurring gynecological malignancy with a poor prognosis and a 5-year survival rate below 45%, often progresses due to metastatic colonization. This review highlights the potential of traditional Chinese medicine (TCM) monomers as anticancer agents that inhibit the metastatic colonization of ovarian cancer cells. TCM monomers exhibit various mechanisms of action, including (1) inhibiting epithelial-to-mesenchymal transformation by modulating cell adhesion molecules; (2) reducing extracellular matrix damage through inhibition of degrading enzymes; (3) affecting cytoskeletal dynamics to alter cell movement; and (4) preventing angiogenesis by downregulating angiogenic factors. Additionally, TCM monomers can reshape the tumor microenvironment, enhance immune responses, and induce oxidative stress, resulting in reduced proliferation and survival of cancer cells. The comprehensive action of TCM monomers makes them promising candidates for individualized, multi-target therapies in drug-resistant cases. This paper reviews the current research on the mechanisms through which TCM monomers combat metastatic colonization, aiming to provide insights for future studies and clinical applications in ovarian cancer treatment, ultimately offering hope to affected patients.
Keywords
Introduction
Ovarian cancer, which presents as painless tumors growing on the ovaries, is the deadliest gynecological cancer and is characterized by a high rate of metastasis. 1 According to the 2022 statistics from the World Health Organization, ovarian cancer accounted for approximately 314 000 new cases and 207 000 deaths globally in 2020. Projections for 2040 indicate a significant rise in both new cases and mortality, particularly in countries with a low Human Development Index (HDI). In these regions, the number of cases and deaths is expected to increase by approximately 96% and 100%, respectively. This highlights the growing burden of ovarian cancer and underscores the need for targeted interventions to address disparities in healthcare access and outcomes. 2
Metastatic colonization is considered the last step in the progression of multistep primary tumors 3 and primary ovarian cancer. Metastatic colonization is the process by which cancer cells complete the initial stages of formation, growth, and proliferation.4,5 This process involves cancer cells leaving the primary site, infiltrating the bloodstream, and taking root and growing in distant organs. The process of metastasis is generally divided into 2 main stages 6 : (1) the physical translocation of cancer cells from primary tumors to the microenvironment of distant tissues, and (2) the survival and proliferation of cancer cells that metastasize to distant organs. In the process of metastatic colonization, cancer cells have to overcome various obstacles. These include the need to establish cytological conditions that support the metastasis and survival of isolated cancer cells in the tumor microenvironment (TME) and to initiate proliferation procedures to promote adaptive survival and growth upon reaching distant organs. 7 When cancer cells acclimatize to the new environment and multiply quickly, they form micrometastases, which may then develop into macroscopic metastatic tumors. 5 Thus, proliferation and metastasis are essential for metastatic cancer cells, and early interventions for the metastatic colonization of ovarian cancer cells are an important aspect of the treatment for ovarian cancer. Many studies have indicated that the active ingredients of Chinese herbal medicines can inhibit the metastasis and proliferation of ovarian cancer cells and influence the metastatic colonization of cancer cells, thereby reducing the risk of cancer cell multiplication and achieving an anti-ovarian cancer effect.
Traditional Chinese Medicine (TCM) is a holistic medical system that has been practiced for thousands of years in China and other parts of East Asia. Rooted in ancient philosophical concepts such as Yin-Yang and the 5 Elements, TCM aims to restore balance and harmony within the body. It encompasses a range of practices, including herbal medicine, acupuncture, moxibustion, cupping, and qigong, all of which are designed to promote health and treat various ailments by regulating the flow of Qi (vital energy) and blood. The TCM drugs mentioned in this article, such as ginseng and Tripterygium wilfordii, are indigenous plants of China. Their medicinal values have been documented in ancient Chinese medical texts, and these drugs are currently widely utilized in clinical treatments across the country. 8 Meanwhile, modern research has increasingly demonstrated the potential of TCM in the treatment of ovarian cancer, particularly through its ability to enhance immune function, inhibit tumor growth, and alleviate the adverse effects of conventional therapies such as chemotherapy. 9 In the field of modern medicine, the treatment of ovarian cancer is principally based on surgical reduction of cancer cell numbers followed by adjuvant chemotherapy. 10 However, the existing treatment approaches for ovarian cancer show limitations such as major adverse reactions, difficulties in achieving curative outcomes, high recurrence rates, and drug resistance. 11 In recent decades, a large number of clinical trials and studies have proved that in addition to showing anti-tumor, anti-inflammatory and immunomodulatory effects, TCM can also reduce the adverse reactions of chemotherapy drugs and improve the quality of life of patients. TCM may show therapeutic effects against ovarian cancer through multiple processes, including inhibiting the proliferation and migration of ovarian cancer cells, promoting apoptosis, and reversing drug resistance, and is safe and well-tolerated. 9 The main advantage of TCM formulations is that they contain many ingredients, such as celastrol and triptolide (TPL), which can produce comprehensive inhibitory effects on different targets of cancer cells. In addition, TCM treatment is characterized by syndrome differentiation. If TCM is used to treat ovarian cancer, doctors can tailor the treatment to the patient’s physique and disease characteristics, thereby improving the treatment outcomes. 12
Although previous studies have highlighted the potential of TCM in the treatment of ovarian cancer, a systematic review of the mechanisms underlying the inhibition of ovarian cancer metastasis by TCM monomers is still lacking. Therefore, from the perspective of cancer cell proliferation and metastasis, the mechanisms underlying the inhibition of metastatic colonization of ovarian cancer cells by TCM monomers have been discussed in detail in this paper, with the aim of providing ideas and inspiration for developing more TCM formulations suitable for ovarian cancer in the future.
Mechanisms Underlying the Inhibition of Invasion and Migration of Ovarian Cancer Cells by TCM Monomers
Inhibition of Epithelial-to-Mesenchymal Transition
Epithelial-to-mesenchymal transition (EMT) is a reversible process in which the relatively stable epithelial cells lose their polarity and intercellular adhesion and become mobile spindle stromal cells. During the process of EMT, tumor cells are affected by cell adhesion factors and transcription factors and lose some characteristics of epithelial cells while acquiring the characteristics of stromal cells. This process reduces adhesion between cells, greatly enhancing their invasion and migration abilities and providing impetus for ovarian cancer cell metastasis.13,14 Thus, these changes constitute the driving force for metastatic colonization of ovarian cancer cells.
Inhibition of the Expression of Cell Adhesion Molecules
E-cadherin is a calcium-dependent cell adhesion molecule that can maintain the epithelioid morphology of cancer cells, and decreased expression of this molecule is a major feature of EMT in cancer cells. 15 Metastasis of ovarian cancer first occurs through intraperitoneal metastasis. In this process, cancer cells detach from their primary mass into the peritoneal fluid, where they spread locally, adhere to the mesenchymal cells forming the inner surface of the peritoneum under the action of adhesion factors, and invade and adhere to the extracellular matrix and basement membrane components. Simultaneously, inflammatory cytokines released by tumors and immune cells damage the protective, anti-adhesion layer of mesothelial cells lining the peritoneal lumen, allowing exposed cancer cells to more easily adhere to the underlying extracellular matrix16,17 and thereby promoting cancer cell metastasis. Although the mechanisms by which tumor cells detangle from their primary mass is unknown, this process is widely believed to be the result of altered cell adhesion. 18 In addition, the process of cancer cell migration is also affected by the expression of many other related proteins. For example, integrin-β1, a transmembrane protein belonging to the integrin family, can interact with extracellular matrix proteins and participate in biological processes such as cell adhesion, migration, and proliferation. 19 The cell adhesion mechanism may also be related to the changes in the CDH1 and CDH2 genes. During EMT, CDH1 deletion or downregulation can cause tumor cells to lose intercellular adhesion, while interstitial CDH2 upregulation can cause tumor cells to migrate away from the primary tumor and invade surrounding tissues. 20 Many TCM monomers have been shown to inhibit cell metastasis by regulating the expression of genes related to the adhesion mechanism,5,21thereby blocking the metastatic colonization of ovarian cancer cells.
One study 22 reported that the 48-h half-maximal inhibitory concentration (IC50) of sinomenine in ovarian cancer SKOV3 cells and IOSE80 cells was 2.12 mmol/L and 17.35 mmol/L, respectively. The findings also indicated that sinomenine can upregulate E-cadherin levels and downregulate matrix metalloproteinase (MMP)-9 levels in a dose-dependent manner to inhibit SKOV3 cell migration and invasion. Amentoflavone extracts at concentrations of 50 μmol·L−1 and 100 μmol·L−1 have also been shown to significantly promote the expression of E-cadherin and β-catenin in SKOV3 cells, 23 suggesting that amentoflavone can inhibit the migration and invasion of SKOV3 ovarian cancer cells by upregulating the expression of the E-cadherin/β-catenin complex. Treatment with Ganoderma lucidum spores at concentrations of 0, 0.5, 1 and 2 mg/mL for 3, 6, and 24 hours resulted in a gradual decrease in the number of adhesive cells with increasing spore concentrations, and the expression levels of E-cadherin in the treatment groups were higher than those in the control group, while the expression levels of N-cadherin and vimentin in the cells treated with the spores were lower than those in the control group. These results indicate that Ganoderma lucidum spores can inhibit the invasion and metastasis of human ovarian cancer cells by upregulating the expression of E-cadherin and downregulating the expression of N-cadherin and vimentin. SKOV3 cells treated with juglone at a concentration of 50 μmol/L for 24 hours showed a significantly increased expression level of caspase-3 and decreased adhesion rate. In addition, as the juglone concentration and stimulation time increased, the viability of SKOV3 cells gradually decreased and the expression levels of MMP-2 and MMP-9 proteins related to epithelial-mesenchymal transformation in SKOV3 cells also decreased, greatly weakening SKOV3 cell migration. 24 Paeoniflorin can significantly inhibit the expression of nuclear factor (NF)-κB and CDH2 proteins in a concentration-dependent manner and increase the expression of CDH1 protein, and the inhibitory effect on SKOV3/cisplatin (DDP) cell migration was the most obvious at a concentration of 100 μmol·L−1, indicating that paeoniflorin can downregulate the expression of nuclear factor (NF)-κB and CDH2. CDH1 expression is induced to inhibit the migration of cancer cells. 25 Brazilin (BR) is extracted from the common Chinese herb sapwood. After treatment with 20, 30, and 40 μmol·L−1 BR for 24 hours, the relative expression levels of Bcl-2, N-cadherin, and vimentin significantly decreased, while the relative expression levels of Bax and E-cadherin significantly increased. Cell migration and invasion ability in SKOV3 cells were gradually weakened, indicating that BR can promote the expression of E-cadherin and inhibit the EMT process in ovarian cancer cells by inhibiting the expression of N-cadherin and vimentin. 26 The combination of TPL (8 ng/mL) and DDP (10 μg/mL) has been shown to significantly downregulate the expression of integrin-β1, MMP-2, and MMP-9, and the inhibitory effect of this combination is greater than those of DDP and TPL individually. 27 These results indicate that TPL sensitized SKOV3/DDP cell adhesion, invasion, and metastasis against DDP.
TCM monomers can also inhibit the lymphatic metastasis of ovarian cancer cells. Downward invasion and cell metastasis of ovarian cancer epithelial cells refer to the process of deep invasion of ovarian cancer cells into surrounding tissues from the primary site and their eventual distant metastasis to other sites through the blood or lymphatic system. 28 Cell experiments 17 have shown that 48 hours after treatment with ginsenoside Rg3 at a concentration of 80 μg/mL, the number of SKOV3 cells invading the lower surface through the matrix gel and filter decreased. These results indicate that ginsenoside Rg3 could prevent the downward invasion of ovarian cancer cells, inhibit the occurrence of metastasis, and block the metastatic colonization of ovarian cancer cells.
Inhibition of Transcription Factor Expression
Transcription factors are a class of proteins that can regulate gene expression. These factors can inhibit the expression of E-cadherin and promote the expression of stromal cell markers, thereby promoting the epithelial-stromal transformation of cancer cells. TCM formulations may contain compounds or components that interfere with the activity of transcription factors and thus affect the transfer of SKOV3 cells. The combination of TPL with cycloheximide (CHX) has been shown to significantly accelerate the degradation rate of the transcription factors Gli1 and Gli2 in a dose- and time-dependent manner, and intraperitoneal administration of this combination at concentrations of 0.2 mg/kg/d and 0.4 mg/kg/d inhibited the growth of subcutaneous tumor grafts in A2780 tumor-bearing nude mice and significantly reduced tumor weight. TPL can also inhibit the proliferation and invasion of SKOV3 cells by regulating the Gli transcription factors. 29 Application of citrate synthase (CS) above a certain concentration to ovarian cancer SKOV3 cells has been shown to reduce the mRNA levels of the transcription factors Snail and Twist and significantly inhibit the invasion and migration ability of the cells. Moreover, CS expression has been shown to result in significantly upregulated E-cadherin levels, significantly downregulated vimentin and β-catenin levels, and inhibition of EMT, proving that CS can inhibit EMT of ovarian cancer SKOV3 cells by decreasing the expression of Snail and Twist and thereby inhibiting invasion and metastasis. 30 SKOV3 cells treated with different concentrations (0, 8, 16, and 32 μmol/L) of sophorins for 48 hours showed temporary dose-dependent inhibition of p-NF-κB expression, downregulation of COX-2 expression and the expression of the anti-apoptotic protein Bcl-2, and upregulation of the expression of the pro-apoptotic protein Bax, which inhibited the viability of SKOV3 cells. 31 Apigenin is extracted from celery and other vegetables, and SKOV3 cells treated with low, medium, and high doses of apigenin (40, 80, and 160 μmol·L−1, respectively) for 48 hours showed dose-dependent reductions in the relative expression rates of NF-κB and urokinase-type plasminogen activator (uPA) as well as reduced invasion and metastasis rates. 32 These experimental results demonstrated that apigenin can downregulate the expression of the transcription factors NF-κB and uPA in human ovarian cancer SKOV3 cells, inhibit SKOV3 metastasis, and thus prevent metastatic colonization of ovarian cancer cells.
Inhibition of Extracellular Matrix-Degrading Enzyme Activity
Ovarian tumor cells and the surrounding stromal cells can stimulate the synthesis and activity of a variety of MMPs, facilitating tumor growth, invasion, and eventual metastasis. Cysteine proteases, serine proteases, and MMPs are the major subtypes of proteinases that trigger cancer development and metastasis. 33 Among these, MMPs can degrade the proteases of the basement membrane and extracellular matrix, are responsible for protein catabolism, can regulate a variety of biological activities, and represent the most important proteolytic system to degrade the extracellular matrix and promote tumor invasion. This system plays important roles in DNA transcription, cell proliferation, and division, tissue morphogenesis and modification, and cell transfer. 34 For example, MMP-9 is the dominant MMP released by most endothelial cells. MMP-9 plays an important role in the decomposition of basal membrane glia and other matrix proteins, and the downregulation of MMP-9 expression can inhibit the invasion and metastasis of malignant tumors. 35 c-Jun N-terminal kinases (JNKs), a type of mitogen-activated protein kinase (MAPK), can be activated by stimulating factors such as oxidative stress, cytokines, ultraviolet light, and serum to increase the proliferation and metastasis ability of tumor cells, 36 which can promote metastatic colonization by cancer cells. TCM monomers have been shown to reduce the damage caused by cancer cells to the surrounding environment by inhibiting the activity of extracellular matrix-degrading enzymes, thus inhibiting the metastatic ability of cancer cells and reducing the metastatic colonization ability of ovarian cancer cells.
After treatment with 100 μg/mL curcumin, the mRNA and protein expression levels of MMP-9 decreased significantly. 37 These results suggest that curcumin and basil polysaccharide can inhibit the invasion of SKOV3 cells by significantly downregulating the expression of osteopontin (OPN), CD44, and MMP-9. Lan conducted a cell control test by adding allicin to the medium (concentrations of 5, 10, 20, and 40 μg/mL, respectively) for 24, 48, and 72 hours, and found that it significantly increased the expression of the epithelial signature protein E-cadherin, obviously decreased the expression levels of the interstitial marker proteins N-cadherin, vimentin, and β-catenin, and obviously increased the expression levels of MMP-2, MMP-7, MMP-9, and glycogen synthase kinase (GSK)-3β in comparison with the control group. In addition, the number of SKOV3 cells showing transmembrane migration decreased significantly after allicin treatment. Thus, allicin may inhibit EMT by regulating the GSK-3β/β-catenin pathway and metalloproteinase expression, thereby inhibiting the invasion and metastasis of ovarian cancer cells. 38 Treatment of SKOV3 cells with different concentrations of germacrone, the active substance extracted from the TCM formulation Zedoary, downregulated the expression of phosphorylated Janus kinase 2 (p-JAK2) and phosphorylated signal transducer and activator of transcription 3 (p-STAT3) in a concentration-dependent manner and reduced the activity of p-JAK2/JAK2 and p-STAT3/STAT3 and the expression of MMP-2 and MMP-9. This experiment confirmed that germacrone could inhibit the proliferation, migration, and invasion of SKOV3 ovarian cancer cells by downregulating the activity of the JAK2/STAT3 signaling pathway and the expression of MMP-2 and MMP-9. 39 Artesunate is a sesquiterpene lactone compound extracted from Artemisia annua. The expression levels of MMP-2 and MMP-9 mRNAs and those of protease-inhibiting molecules in SKOV3 ovarian cancer cells were significantly decreased by treatment with mesartesunate at a concentration of 100 µg/mL. These results suggest that artesunate can inhibit the invasion and metastasis of ovarian cancer cells by blocking protease hydrolysis of the extracellular matrix. 40 Fengiie treated SKOV3 cells with dihydromyricetin at different concentrations and found that dihydromyricetin can dose-dependently reduce the viability of cancer cells, inhibit cell migration and invasion ability, and induce cell apoptosis. Their findings also suggested that dihydromyricetin could activate the MAPK, caspase-3, JNK, and extracellular signal-regulated kinase (ERK) pathways. Consequently, the discovery of Golgi stacking protein (GRASP65) is reduced, causing substantial changes in the matrix water content. This results in the rupture and transformation of ovarian cancer cells and effectively reduces the movement and invasion ability of ovarian cancer SKOV3 cells. 41 Sinsinine, the active ingredient extracted from the Chinese herb Sinophyllum sinensis, can upregulate E-cadherin levels and downregulate MMP-9 levels in a dose-dependent manner, and act on SKOV3 cells showing EMT to prevent them from invading the surrounding area. 22 Treatment of SKOV3 cells with 5 and 10 μmol/L protodioscin significantly reduced the number of invasive cells and the expression of the invasion and migration-related proteins vascular endothelial growth factor (VEGF), MMP-2, and MMP-9. These results indicated that protodioscin could reduce the survival and motility of ovarian cancer SKOV3 cells by inhibiting the expression of metalloproteinases 42 and thereby effectively inhibit metastatic colonization of ovarian cancer cells.
A new peptide prolyl isomerase, Pin1, has been shown to specifically catalyze the cis-trans isomerization of phosphorylated Ser/Thr-Pro amide bonds in proteins. This process can change the conformation of the protein, thereby regulating the function of the protein, and can be used to regulate SKOV3 cell invasion. 43 The results showed that the expression and activity of Pin1 decreased in a dose-dependent manner with different concentrations of juglone. This process can inhibit the proliferation and metastasis of tumor cells, 44 thereby preventing the metastatic colonization of ovarian cancer cells.
Influence on Cell Motility: Inhibition of Cytoskeletal Recombination
The cytoskeleton is mainly composed of actin microfilaments, microtubules, and intermediate filaments. Cell migration is a highly dynamic process driven by the cytoskeleton. 45 Cells are connected to the external environment through the cytoskeleton. The cytoskeleton receives external signals that guide complex cellular behaviors such as the formation, invasion, and migration of lamellar pseudopods. During the process of cancer cell metastasis, the high flexibility of the cytoskeleton of tumor cells can promote the escape of cancer cells from the primary tumor and the formation of new cell colonies at distant sites. Actin stress fibers play an important role in this process. Actin regulates cell morphology and movement by interacting with microtubules and microfilaments. 46 In the process of cell metastasis, actin stress fibers can help cancer cells change their shape, increase the migration ability of cells, and promote the penetration of cancer cells in the basement membrane and interstitial tissues to finally reach the target organ and complete distant metastasis. 47 In malignant ovarian cancer cells, the form of actin stress fibers is significantly reduced, and F-actin accumulates in the cortical region, leading to more extensive invasion and metastasis. 48 Studies have analyzed the correlation between extended survival of patients with ovarian cancer and the expression levels of EMT-related proteins and found that a total of 7 cytoskeletal proteins are involved in the progression of cancer. Among them, 4 proteins were significantly associated with survival in patients with ovarian cancer, 49 indicating the importance of blocking cytoskeletal casting in inhibiting cancer cell metastasis. TCM monomers can affect the morphological changes and migration ability of cells by influencing the recombination of cytoskeleton, such as the polymerization and depolymerization of microfilaments. 50 This can help block the metastatic colonization of ovarian cancer cells and play an anticancer role.
Methyl vanillate in the TCM herb Elsholtzia has been shown to significantly inhibit the expression of transcription factors (Snail and ZEB2) in SKOV3 cells at a concentration of 100 μmol/L. Reduced expression of these transcription factors can inhibit the assembly of the cytoskeletal F-actin, weaken the formation of cell filaments, and ultimately delay cell motility. This effect plays an important role in inhibiting EMT and ovarian cancer migration. 51 53 Rhein is derived from the Chinese medicine rhubarb. Lihong et al 52 treated SKOV3 cells with rhein at concentrations of 8.8, 17.60, and 26.40 μmol·L−1 for 24 hours. Their results showed that treatment with rhein significantly decreased the cellular pseudopodia, caused breakage and disordered arrangement of microfilaments in the cell, an uneven plasma membrane, widened intercellular spaces, and a series of ultrastructural changes. The cell migration and invasion ability in vitro also decreased significantly. Modern studies have shown that cytoskeletal recombination is closely related to cell colony formation, and that both of these are important processes for cell outward migration. After treatment with 100, 200, and 300 μmol/L of lotus leaf for 48 hours, the migration ability of ovarian cancer cells decreased in a concentration-dependent manner, which significantly inhibited the viability and colony-forming ability of the SKOV3 cell line and inhibited the growth of ovarian epithelial cancer cells and its invasion and migration. 53 The KRT18P55 gene may be involved in the intermediate filament cytoskeletal and exogenous apoptosis signaling pathway. Studies have shown that ginsenoside 20(S)-Rg3 can reduce the expression level of KRT18P55 in a concentration-dependent manner. In this process, 20(S)-Rg3 inhibits the proliferation and migration of cancer cells by inhibiting the composition of the intermediate filaments of the cytoskeleton in ovarian cancer, 54 and ultimately plays a role in blocking the metastatic colonization of ovarian cancer cells to distant organs.
Inhibition of Angiogenic Factor Expression
Angiogenic factors, including VEGF and basic fibroblast growth factor (bFGF), are a class of proteins that can promote the formation of new blood vessels. The formation of many blood vessels at the tumor site can increase the chance of tumor cells entering the circulation. Angiogenic factors play both direct and indirect roles in tumor metastasis. 55 First, endothelial cells under the regulation of VEGF directly participate in endoscopy, which can translocate invasive cancer cells to the vascular lumen; thus, inhibiting angiogenesis can reduce the metastasis of malignant tumors. Second, endothelial cells are one of the main sources of cancer-associated fibroblasts (CAFs). The heterogeneous group of CAFs is the main inducer of cancer cell migration and invasion, and many fibroblasts can indirectly promote the metastasis of cancer cells. 56 The presence and activity of CAFs in the TME may also be influenced by bFGF, which can affect the functioning of CAFs by regulating the signaling pathway of cell proliferation and migration and thus affect the growth and invasion ability of tumors. 57 TCM monomers can inhibit the metastasis of cancer cells through the regulation of angiogenic factors, and then block the metastatic colonization of ovarian cancer cells and play a role in preventing the deterioration of ovarian cancer.
In one study, injection of a dose of ginsenoside Rg3 into the peritoneum caused a reduction in the number of blood vessels toward the tumor mass. These results suggest that ginsenoside Rg3 may inhibit ovarian tumor-induced angiogenesis, thereby reducing ovarian cancer metastasis. In addition, a research team found that scorpiotoxin polypeptide from scorpion at a concentration of 12.5 to 200 μg/mL can upregulate the expression of thrombospondin-1 (TSP-1) and inhibit the expression of VEGF, affecting tumor angiogenesis and inhibiting the migration of ovarian cancer cells. 58 At-EE is an effective component of the Chinese herb Amomum tsaoko. After adding 10 μg/mL of grass ethanol into SKOV3 cells, the expression levels of VEGF and interleukin (IL)-6 mRNA in SKOV3 cells decreased significantly in comparison with those in the control group, proving that grass ethanol can inhibit angiogenesis in vitro by affecting the secretion of IL-6 and VEGF by SKOV3 cells and thus inhibit the migration and metastatic colonization of ovarian cancer cells. 59 Sphingosine-1-phosphate (S1P) promotes cell migration, proliferation, survival, and differentiation. S1P and S1P receptor subtype (sphingosine 1-phosphate receptor [S1PR] 1-3) are widely expressed in vascular endothelial cells, have a regulatory effect on vascular function, and are highly expressed in ovarian cancer cells. 60 Berberine is an effective ingredient extracted from Coptis, a TCM drug. Studies have shown that after treatment with berberine at concentrations of 5, 10, 25, 50, and 100 mmol/mL, the relative expression levels of the cell pathway proteins S1P and S1PR1, VEGF, IL-8, MMP-9, and MMP-2 in SKOV3 cells were significantly inhibited. The effectiveness of berberine was concentration-dependent. Thus, the results implied that berberine could inhibit the invasion and migration of ovarian cancer cells by inhibiting angiogenesis. 61
In addition to inhibiting vascular and abdominal metastases, some TCM monomers can also play a role in inhibiting lymphatic metastasis. Russo Spena et al found that rhein, as a potential Rac1 small-molecule inhibitor, can regulate the Rac1/LIMK1/cofilin signaling pathway, inhibit the movement and invasion of ovarian cancer lymph node metastatic cells in a dose-dependent manner, 53 and block the metastatic colonization of ovarian cancer cells.Our institute has compiled the following Traditional Chinese Medicine monomers that inhibit ovarian cancer cell invasion, as shown in the table below (Table 1). Meanwhile, we illustrated the process of inhibition of ovarian cancer cell metastasis by traditional Chinese medicine herbs in a graphical format (Figure 1).
Traditional Chinese Medicine Monomers for Inhibiting the Invasion and Migration of Ovarian Cancer Cells.


Process of inhibition of ovarian cancer cell metastasis by traditional Chinese medicine herbs.
Mechanisms by Which TCM Monomers Inhibit the Proliferation of Ovarian Cancer Cells
The proliferative ability of cancer cells is closely related to metastatic colonization. Proliferating cancer cells are more likely to detach from the primary site. Moreover, cancer cells may be partially “squeezed” by excessive proliferation, causing protrusion of the basal layer and entry into the blood or lymphatic system and thus leading to the possibility of metastatic colonization. 64 On the other hand, once cancer cells have successfully metastasized to distant organs or tissues, they need to adapt and colonize in their new environment. During this process, the cancer cells need to maintain their proliferation capacity in order to continue to grow and spread in their new environment. Therefore, metastatically colonized cancer cells usually show a higher ability to proliferate in order to adapt to the new living environment. 8 TCM monomers have been shown to inhibit the proliferation of ovarian cancer cells, block the metastatic colonization of ovarian cancer cells, and exert anticancer effects. In this section, the mechanisms by which TCM monomers inhibit the proliferation of ovarian cancer cells are described in detail from the perspectives of regulating the TME, inducing oxidative stress, and blocking the cell cycle.
Regulation of the TME
The TME plays a crucial role in tumor metastatic colonization and treatment response. The TME mainly contains malignant cells that constitute tumors, immune cells that participate in immune responses, and blood vessels that supply oxygen and nutrients to tumor cells. It also includes fiber cells that support growth as well as the extracellular matrix and intercellular substances. 65 The TME is also affected by many factors, such as metabolic remodeling and the expression of platelet-activating factors. Immunotherapy aims to recruit immunosuppressive cells by regulating the expression of immune factors to activate, enhance, or improve the functioning of the body’s immune system. This treatment helps encourage immune cells to recognize, attack and eliminate cancer cells more effectively. 66 Anti-angiogenic therapy can destroy tumor angiogenesis, 67 thereby reshaping the TME and creating harsh conditions of low oxygen and low nutrition and consequently inhibiting the proliferation of cancer cells. Simultaneously, however, these harsh conditions promote the metabolic remodeling of tumor cells to adapt to the environment and maintain survival. Metabolites produced by metabolic remodeling, such as lactic acid and acetone, change the acid-base balance and redox state of the TME,68,69 thus exerting an important influence on the proliferation and invasion ability of tumor cells.
Platelet-activating factors can also affect the TME. Previous studies have shown that (1) platelet activators can regulate neovascularization in the TME and that (2) platelet activators can regulate the inflammatory state of the TME by regulating the release of inflammatory factors and activation of immune cells. These effects can inhibit the proliferation of tumor cells and thus prevent metastatic colonization of tumors.70,71 The use of TCM monomers can help regulate the TME, affect the angiogenesis and immune response of tumor cells, regulate metabolic recombination, platelet-activating factors, and other factors affecting the TME, inhibit the process of metastatic colonization, and thereby improve the therapeutic effects. In this section, we will discuss the mechanisms by which TCM monomers regulate the TME, focusing on the activation of the expression of immune factors, inhibition of the expression of angiogenic factors, and inhibition of metabolic remodeling and platelet-activating factors.
Activation of the Expression of Immune Factors
The immune system is a key component of the TME in ovarian cancer, and immunotherapy represents one of the important applications of TCM in the treatment of ovarian cancer. Ovarian cancer cells may escape detection and attack by the immune system in a number of ways, protecting themselves and continuing to proliferate. Immune escape mainly involves the failure of immune cells to recognize ovarian cancer-related antigens, inhibition of antigen-presenting cells (macrophages, dendritic cells and B lymphocytes), inhibition of tumor-killing immune cells (natural killer cells), and activation of immunosuppressive cells (regulatory T cells [Treg cells], tumor-related macrophages, and bone marrow-derived suppressor cells). 72 Recruitment of immunosuppressive cells can suppress the anti-tumor immune response and accurately identify cancer cells in the process of inhibiting metastatic colonization. These include Treg cells, macrophages, or myelogenic suppressor cells (MDSCs). These treatments can influence the anti-tumor immune response of the body and thereby inhibit the metastatic colonization of ovarian cancer. 67
TCM has shown certain efficacy in activating the expression of immune factors and preventing immune escape against ovarian cancer. Ginsenoside Rg1 is an active component in ginseng. Zanotelli et al treated nude mice with ovarian cancer with different doses of ginsenoside Rg1 for 14days and observed the immunomodulatory effects and mechanisms by collecting whole blood and preparing serum samples 24hours after the last administration. They measured the levels of the immune factors IL-2 and interferon (INF)-γ secreted by tumor immune cells. The increased expression of IL-12P40 indicated that ginsenoside Rg1 could enhance the immune response and immune regulation by promoting the expression of immune factors in anti-tumor immune cells, thereby inhibiting the proliferation of ovarian cancer cells and achieving an anti-ovarian cancer effect. 73 The CD4+/CD8+ ratio of T lymphocytes is commonly used to assess the functional status of the immune system. 74 One study 75 showed that intraperitoneal or oral administration of 4 mg/kg/day DDP, 0.15 mg/kg/day TPL (the active ingredient of the TCM drug Tripterygium wilfordii), or a combination of 4 mg/kg/day DDP and 0.15 mg/kg/day TPL in mice greatly enhanced the serum levels of the inflammatory cytokines IL-2 and tumor necrosis factor (TNF)-α and increased the CD4+/CD8+ ratio, and that these effects were maximized by the combination of DDP and TPL. These findings confirmed that TPL could regulate the balance between immune cell subsets and the expression of inflammatory factors, thereby exerting anti-tumor proliferative activity. Mature dendritic cells facilitate antigen presentation by cancer cells and stimulate an immune response. 76 Shikonin extracted from the Chinese herb comfrey has been shown to promote the maturation of dendritic cells, increase the dendritic cell recognition signal on the surface of SKOV3 cells, enhance the immune capacity of the body, and inhibit the proliferation of ovarian cancer SKOV3 cells. 77 TCM monomers have been also shown to regulate the signaling pathways related to immune activity to activate immune activity and prevent immune escape of cancer cells. For example, after treatment with Ophiopogon saponins, rats with ovarian cancer showed significantly reduced expression of IGF2, IGFR1R, and IRS1 proteins related to immune function, significantly prolonged survival, and significant reductions in tumor volume and weight, implying that Ophiopogonin saponins can inhibit the IGF2, IGFR1R, and IRS1 pathways, improve immune function, and thereby inhibit the growth of ovarian cancer transplantation tumors. 78 TCM monomers have also been shown to be effective in immunotherapy of ovarian cancer after chemotherapy. The combination of 10 μmol/L berberine with 1 μmol/L cisplatin has been shown to upregulate the expression of anti-cisplatin-induced RAD51, enhance the role of cisplatin in upregulating Bax expression in HEY cells and SKOV3 cells, downregulate Bcl-2 expression, and induce apoptosis. These results demonstrated that berberine could improve the immune function and the sensitivity of SKOV3 cells to chemotherapy and radiotherapy. 79 In one study, nude mice injected with SKOV3 cells received intraperitoneal administration of 100 mg/kg matrine from the Chinese herb Sophora flavescens Aiton, 3 mg/kg ip cisplatin, or a combination of both drugs. After 3 weeks of treatment, the cancer inhibition rate was 57.56% in the matrine treatment group, 50.93% in the cisplatin group, and 70.52% in combination treatment group. These results indicate that matrine can improve the sensitivity of cancer cells to radiotherapy and chemotherapy. The findings of the study also suggested that in addition to enhancing the body’s immune regulatory function, matrine can counteract the toxic side effects of chemoradiotherapy to suppress the immune system. 80 In another study, mice with ovarian cancer received low and high doses of ginsenoside Rg1 (200 and 400 mg/kg, respectively), cisplatin (2 mg/kg), or a combination of both (Rg1 200 mg/kg + cisplatin 2 mg/kg). Assessment of serum IL-2, INF-γ, and IL-12P40 levels showed that the combined treatment had the most significant effect. These results confirm that ginsenoside Rg1 can be used as an immune adjuvant with cisplatin in the treatment of ovarian cancer to reverse the body’s tolerance to chemotherapy and enhance the effect of chemotherapy. 75
Inhibition of the Expression of Vascular Growth Factors
Angiogenesis, a key step in tumor metastasis and growth, occurs primarily through the budding of new blood vessels from existing microvascular beds. These newly formed blood vessels provide nutrients and oxygen for tumor growth and are also pathways for distant metastasis.81,82 TCM monomers can inhibit angiogenesis and progression in ovarian cancer by inhibiting the expression of pro-angiogenic factors, upregulating anti-angiogenic factors, inhibiting the proliferation of vascular endothelial cells, reducing the density of capillaries, and regulating the related signaling pathways. 83 Targeted inhibition of angiogenesis is one of the clinical treatment methods for cancer. 15 Hypoxia is a driving factor for tumor growth and metastasis. By activating hypoxia-inducing factors, cells can sense hypoxia signals and regulate the expression of angiogenic genes and angiogenic pathways. 84 Inhibition of angiogenesis can reduce the proliferation ability of metastatic cancer cells, hinder the process of metastatic colonization, and induce anticancer effects.
In one previous study, 0.4 mg/mL of cuttlefish sulfate polysaccharides was shown to significantly inhibit the neovascularization of the allantoic membrane of chicken embryo and reduce the expression of VEGF and bFGF in endothelial cells. These results indicated that Sepiella sulfate polysaccharides could play a significant anti-angiogenesis role and inhibit metastatic colonization of ovarian cancer. 85 Scorpion venom polypeptides have been shown to significantly inhibit the expression of VEGF, promote the expression of the angiogenesis-related gene TSP-1, and thereby inhibit angiogenesis to hinder SKOV3 cell proliferation. The inhibition of cell proliferation was most significant when the mass concentration was greater than 50 μg/mL. 64 In one study, SKOV3 cells and matrigel were injected subcutaneously into the right posterior abdomen of mice (5 mice in each group), and after 3 weeks of treatment with Phyllanthus emblica L extract (100 mg/kg body weight/day, 10% sucrose), miR-375 was activated in mice. The treatment with Phyllanthus emblica L extract also targeted the IGF1R and SNAIL1 genes in SKOV3 cells to downregulate the expression of pro-angiogenesis factors. The results showed that Phyllanthus emblica extract could inhibit the proliferation of SKOV3 cells by inhibiting angiogenesis. 86 In addition, another study found that administration of a certain dose of cardamom extract in a chicken embryo allantoic membrane model can reduce the angiogenesis of SKOV3 cell culture medium. In addition, the oxygen content of the SKOV3 cell medium was significantly reduced after the addition of cardamom, simulating a hypoxic microenvironment to inhibit the protein expression of hypoxia-inducible factor (HIF)-α and VEGF. 87 These results indicated that cardamom could inhibit the proliferation of ovarian cancer cells and block the metastatic colonization process by inhibiting angiogenesis.
Inhibition of Metabolic Remodeling
Metabolic remodeling refers to the reshaping of mitochondrial function in cancer cells to adapt to the needs of rapid proliferation and growth. Glycolysis and lipid metabolism are the 2 major metabolic processes for energy production and anabolic growth of cancer cells, and these processes are mainly mediated by mitochondria in cells. 88 Aerobic glycolysis refers to the selection of cells to produce energy through glycolysis metabolism when oxygen is sufficient. 89 This metabolic mode offers certain advantages in tumor cells. 90 Studies have shown that TCM monomers can effectively inhibit the expression of glycolytic-related enzymes in tumor cells by regulating the related signaling pathways and gene expression, which can slow down tumor growth, increase the sensitivity to radiotherapy and chemotherapy, inhibit the metastatic colonization process, and improve the therapeutic effect.
In one study, 2 ovarian cancer cell lines SKOV3 and HO8910 were treated with carvacrol, the active component of the Chinese herb parsley, at different concentrations for 48 h, and at the IC50 value of 120 mg/L, the mRNA and protein levels of the aerobic glycolysis-related molecules HIF-1α, hexokinase 2 (HK2), and lactate dehydrogenase A (LDHA) in ovarian cancer cells were decreased. Carvacrol can inhibit glucose uptake and lactic acid production in ovarian cancer cells by downregulating HIF-1α expression, and it can inhibit aerobic glycolysis of ovarian cancer cells, thus inhibiting cancer cell proliferation. 91 The exosome miR-21-5p derived from ovarian cancer cells can inhibit the sensitivity of ovarian cancer cells to DDP by regulating the E1 subunit α1 of pyruvate dehydrogenase (PDHA1). Moreover, this exosome can promote cell viability and glycolysis of cancer cells and inhibit their chemical sensitivity. 92 After treatment with 0.1, 0.25, and 0.5 μg/mL for 48 hours, the expression levels of glycolytic enzymes in ovarian cancer cells was decreased, and glucose intake and lactic acid production were significantly decreased. These results indicate that the glycolytic metabolism in SKOV3 ovarian cancer cells was decreased by glycolytic ethanol through reduced glucose intake and lactic acid production, effectively inhibiting the rapid proliferation of ovarian cancer cells. 93 Shikonin has also shown the ability to inhibit the glycolysis of ovarian cancer cells, which can inhibit their proliferation, migration, and colony formation. The combination of shikonin and paclitaxel has been shown to affect the metabolic capacity of SKOV3 cells by inhibiting the expression of PKM2. 78 The pentose phosphate pathway (PPP) is another intracellular metabolic pathway associated with aerobic glycolysis. This pathway converts glucose-6-phosphate to ribo-5-phosphate and carbon monoxide molecules, while producing the antioxidant gene NADPH and riboside. 94 In one study, treatment of SKOV3 cells with arctigenin (ARG), isolated from the TCM drug burdock fruit, at doses of 1, 5, 10, and 20 mg/L for 72 hours resulted in significantly decreased expression levels of G6PD, GLUT3, PFK, PKM2, and TKT, which are associated with the PPP. This study confirmed that arctigenin can inhibit the proliferation of human ovarian cancer SKOV3 cells by targeting the PPP. 95
Studies on the inhibitory effects of TCM monomers on the proliferation of ovarian cancer cells through metabolic remodeling and metastatic proliferation have mainly focused on the regulation of aerobic glycolytic inhibition, while studies on the inhibition of proliferation of ovarian cancer cells through the regulation of lipid metabolism are limited. Thus, more mechanisms are expected to be studied and discussed from this perspective in the future.
Inhibition of Platelet-Activating Factor Expression
Platelets can significantly stimulate the proliferation of ovarian cancer cells. When platelet-activating factors are activated, they can also activate epidermal growth factor receptor (EGFR). This process leads to phosphorylation of the key protein Src, which activates focal adhesion kinase (FAK) and Paxillin, 2 proteins involved in cell signaling and extracellular matrix interactions, and further activates downstream proteins to promote cell proliferation.96,97 Platelet-activating factor receptor (PAFR) is overexpressed in ovarian cancer cells, and platelet-activating factors can promote ovarian cancer progression through a platelet-activating factor/PAFR-mediated inflammatory signaling pathway. Platelets have been identified as potential new drug targets for cancer treatment. 98
TCM monomers can play specific roles in inhibiting platelet activation against cancer. Both ginkgolide compounds and ginkgolide B derivatives, the active components of ginkgolide, have been shown to inhibit platelet aggregation induced by platelet-activating factors in rabbits. 99 Moreover, after treating mice with ovarian cancer with different concentrations of ginkgolide B for 6 hours, the PAFR signaling pathway was found to be significantly inhibited. Further, ginkgolide B has been shown to inhibit the proliferation of SKOV3 cells by inhibiting the expression of the PAFR signaling pathway 100 and hindering metastatic colonization. In addition, platelet-activating factors can also affect angiogenesis in the TME, hindering the metastatic colonization process of ovarian cancer cells and achieving anticancer effects.
Induction of Oxidative Stress
During cell proliferation, some intracellular signaling pathways are regulated by reactive oxygen species (ROS), which affect cell proliferation. ROS are a class of highly active oxidizing molecules in the cell. Excessive accumulation of ROS in cells can lead to mitochondrial function impairment through oxidation of mitochondrial proteins and lipids, resulting in mitochondrial dysfunction, mitochondrial membrane potential loss, and changes in mitochondrial permeability causing eventual apoptosis or necrosis. ROS inhibit the proliferation of cancer cells through this series of processes. 101 Oxidative stress refers to the presence of too many ROS and free radicals in the environment inside and outside the cell. Oxidative stress impairs the redox balance in the cell, causing damage to biological macromolecules such as proteins, lipids, and nucleic acids, and eventually resulting in abnormal cell function or even cell death. 102 During the treatment of ovarian cancer, ROS accumulation in cells can exceed the normal level as a result of the drugs, leading to the production of oxidative stress. 32 Consequently, the activity of cancer cells can be damaged and their proliferation can be blocked, inhibiting metastatic colonization.
TCM components can achieve anticancer effects by inducing oxidative stress. Four kinds of Lilium polysaccharides have shown dose-dependent free radical-scavenging effects in SKOV3 cells. The authors concluded that Longya Lilium polysaccharides possess antioxidant activity that can promote apoptosis and inhibit the proliferation of SKOV3 cells. 103 Moreover, treatment of SKOV3 cells with 50 μmol/L juglone for 24 hours significantly increased ROS levels and upregulated the expression levels of Cyt C and activated caspase-3 in cancer cells. These results confirmed that juglone could induce apoptosis and inhibit cell proliferation by promoting ROS accumulation in SKOV3 ovarian cancer cells. 24 The IC50 values of baicalin treatment for 72 hours in SKOV3 and TOV-21G cells have been reported to be 54 and 91 μM, respectively, and baicalin was shown to greatly increase the intracellular ROS content and cause G2/M cell cycle arrest, indicating that it can inhibit the proliferation of SKOV3 ovarian cancer cells. This effect has been suggested to be mainly attributable to ROS-mediated DNA breakage, which leads to cell cycle arrest. 104 Treatment with high and low concentrations of cardamomine (5, 20 μM), with BAY11-7082 (10 μM) as a positive control, has been shown to increase ROS levels in ovarian cancer cells and significantly inhibit cell viability, implying that cardamomine could induce oxidative stress and inhibit the proliferation and migration of ovarian cancer cells. 105 Yu et al treated A2780 and SKOV3 cells with different concentrations of zionoside Ⅶ, an active component of zionoside, for 24 hours, and found that the ROS levels of ovarian cancer cells increased significantly, suggesting that zionoside Ⅶ may induce oxidative stress in ovarian cancer cells, decrease mitochondrial membrane potential, and promote cell damage in the mitochondrial pathway of ovarian cancer cells. These effects may inhibit the proliferation of ovarian cancer SKOV3 cells 106 and prevent metastatic colonization of ovarian cancer.
Cell Cycle Arrest
The series of cellular activities that lead to cell division and replication is called the cell cycle. It can be divided into 2 main stages: interphase and mitosis (M). The interphase is further divided into G1, S, and G2 phases; thus, the GI, S, G2, and M phases are the 4 phases of the cell cycle.98,107 Cells develop in the G1 phase and prepare for DNA replication. During the S phase, DNA replication occurs and chromosomes are duplicated. The G2 phase is the period of growth and preparation for cell division. Finally, during the M phase, the cell divides into 2 daughter cells. The whole process is controlled by genes that regulate the cell cycle. 108 Mutations in genes can cause uncontrolled cell division and dysregulation of the cell cycle, leading to the onset and progression of cancer. 109 Studies have shown that many TCM monomers can negatively regulate cell division and growth through the regulation of tumor-suppressor genes, prevent the cell cycle from getting out of control into unlimited proliferation, block tumor cells during the cell cycle, and thus inhibit the proliferation of tumor cells, 110 thereby achieving the effect of blocking metastatic colonization. Obliquin is a triterpenoid compound extracted from Tribulus terristris L. SKOV3 cells treated with obliquin at concentrations of 25, 50, and 100 μg/mL for 48 hours showed reduced Ki-67-positive rates. The Ki-67 antigen is a human proliferative cell-specific nuclear protein, and its antibody can recognize the cell nuclei and chromosomes in the late G1 phase, S phase, G2 phase, and M phase of the cell cycle. Inotodiol can inhibit the positive expression of Ki-67 in SKOV3 cells, resulting in cell cycle arrest and inhibiting the proliferation of ovarian cancer cells. 111 Bcl-2 protein expression was significantly downregulated, while Bax protein expression was significantly upregulated after the application of 100 mg/L Tribulus terrestris saposide to ovarian cancer cells for 24, 48, and 72 hours. Simultaneously, the proportion of S phase cells decreased and the proportion of G1 phase cells increased after treatment with Tribulus terrestris saponins. Thus, the findings indicated that Tribulus terrestris saponins could induce apoptosis and inhibit the proliferation of ovarian cancer cells by blocking the cell cycle. 110 Flavanones (FA) are active ingredients derived from rhododendrons. One study showed that SKOV3 cells treated with FA at concentrations of 40, 80, and 160 μM for 24 and 48 hours showed decreased expression of ERK/MAPK, a signaling pathway associated with cell proliferation, and significant arrest of the G2/M cell cycle, proving that FA could inhibit the proliferation of ovarian cancer cells by inducing G2/M cell cycle arrest.112,113 Moreover, ovarian cancer cells treated with 19.5 μg/mL icariin for 48 hours showed significantly reduced cell proliferation, migration, and invasion abilities as well as significantly increased cell apoptosis rate. Thus, the findings indicated that icariin may inhibit the proliferation of ovarian cancer cells by inducing cell apoptosis and affecting cell cycle distribution. 114 In one study, after treatment with baicalin at 80 μM for 48 hours, cyclin A mRNA and protein expression in SKOV3 cells increased; McL-1, Bcl-x1, Bcl-2, and survivin mRNA expression decreased; the proportion of cells in the G1 and G2 stages decreased significantly; and the proportion of cells in the S stage increased significantly. These findings demonstrated that baicalin could inhibit the STAT3 pathway by upregulating cyclin A expression and thereby reducing the expression of the downstream STAT3 genes McL-1, Bcl-xl, Bcl-2, and survivin to induce S phase arrest and inhibiting the proliferation of SKOV3 cells. 115
In the drug-tolerance phase of chemotherapy, cancer cell resistance weakens the blocking effects of chemotherapeutic drugs on the tumor cell cycle. In this phase, the addition of TCM monomers can reduce drug resistance and improve the effects of radiotherapy. 115 Hogg 77 suggested that shikonin could reduce the blocking effect of radiation on the ovarian cancer cell cycle, thereby improving the effect of radiotherapy. Furthermore, studies have also revealed that genistein, 112 a phytoestrogen derived from the common plant soybean and a type of flavonoid, is similarly effective in inhibiting the growth of ovarian cancer cells. When ovarian cancer cells were treated with 3 and 9 μg/mL genistein for 24 hours after X-ray irradiation, genistein was found to increase DNA double-strand breaks in the ovarian cancer cells, inhibit the process of DNA damage repair caused by radiation, and block cells in the G2/M phase. These findings implied that flavonoids have certain growth-inhibiting and radiosensitizing effects on hypoxic ovarian cancer cells, which may exert anti-tumor effects by regulating the cell signaling pathway and increasing the cell apoptosis rate and can also block irradiated hypoxic ovarian cancer cells with the highest radiosensitivity in the G2/M stage, 113 thereby blocking the metastatic colonization of ovarian cancer and achieving an anticancer effect. This study enumerates the inhibitory effects of traditional Chinese medicine monomers on the proliferation of ovarian cancer cells (Table 2). And the inhibitory effects of Chinese herbs on the proliferation of metastatic ovarian cancer cells were demonstrated through images (Figure 2).
Inhibitory Effects of Traditional Chinese Medicine Monomers on the Proliferation of Ovarian Cancer Cells.

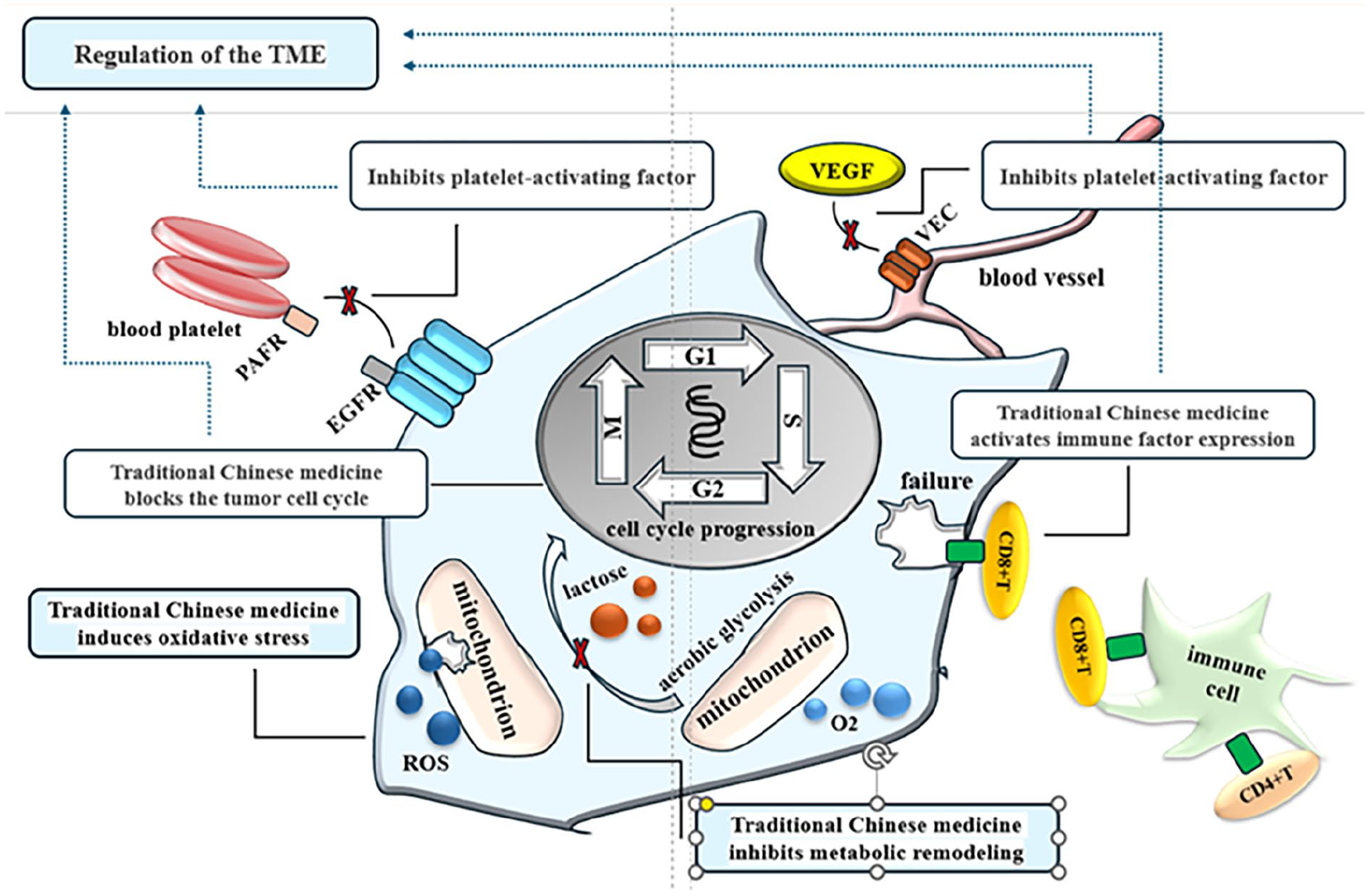
Inhibitory effects of Chinese herbs on the proliferation of metastatic cells of ovarian cancer.
Discussion
Ovarian cancer is a very deadly disease. Due to the hidden early onset, rapid disease progression, drug resistance in the course of treatment, and substantial adverse effects of chemoradiotherapy and other factors, the current treatment methods are still flawed and insufficient, thus affecting the treatment outcomes of patients with ovarian cancer. A large number of recent studies have shown that TCM monomers can significantly inhibit metastatic colonization of ovarian cancer while alleviating adverse reactions and anticancer cell resistance. These studies have shown the potential benefits of TCM monomers and their substantial potential for future development. This review discusses the advantages of TCM monomers in the treatment of ovarian cancer from the perspectives of multi-component synergism, individualized treatment, and enhancing the effects of radiotherapy and chemotherapy.
Synergistic Effects of Multiple Components
The TCM formulations used in clinical practice are often composed of a variety of Chinese medicines, which can produce comprehensive inhibitory effects on different targets of tumor cells through synergistic effects, thus inhibiting cell proliferation and metastasis. 116 Studies have shown that hirudin can regulate MAPK1, AKT1, CASP3, CASP8, MMP1, MMP2, MMP9 and other major target genes, thereby affecting the energy metabolism of tumor cells, interfering with the TME to affect the growth and invasion of cancer cells, and playing an anti-ovarian cancer role. The IC50 of hirudin has been reported to be 15.53 mmol/L.117,118 Similarly, berberine has been used to treat ovarian cancer cells at concentrations of 5, 10, 25, 50, and 100 mmol/mL, and it has been shown to inhibit the expression of VEGF, regulatory cancer cell adhesion factor, and S1P/S1PR1 pathway-related proteins in a dose-dependent manner. Thus, berberine can inhibit the metastasis and proliferation of ovarian cancer by acting on ovarian cancer cells from multiple angles and targets, thereby preventing the metastatic colonization of ovarian cancer cells. 61
Individualized Treatment
TCM focuses on treatment based on syndrome differentiation and can be tailored according to the patient’s constitution and disease characteristics, thereby improving the therapeutic effect. According to traditional TCM theory, individualized therapy based on syndrome types is advantageous for individual patients. Balancing qi, blood, Yin, and Yang and removing phlegm and dampness are the effects of TCM compounds on cancer patients. 119 Fuzi, which is used in TCM, is strongly toxic, but in moderate amounts, it can play an unexpected role in the treatment of cancer, although the effects of Fuzi on drug metabolizing enzymes (DMEs) and transporters (ETs) suggest that the use of Fuzi in combination with other drugs may lead to interactions that cause toxic reactions and harm the human body. Polymorphisms of DMEs and ETs in individual patients can lead to differences in the efficacy and toxicity of Fuzi. 120 Consistent with these findings, TCM practice involves adjusting the dosage on the basis of the patient’s physique, allowing personalized treatment and improving the effectiveness of the treatment.
Enhancing the Effects of Radiotherapy and Chemotherapy
TCM can also play an auxiliary role in the process of chemotherapy. TCM monomers can inhibit the proliferation of cells and reduce the number of cancer cells, allowing chemotherapeutic drugs and radiotherapy to kill cancer cells more easily. In addition, these monomers can enhance the effects of radiotherapy and chemotherapy by improving sensitivity to chemotherapy and reducing drug resistance and side effects. Different concentrations of TPL have been shown to enhance the sensitivity of cisplatin in SKOV3 cells by inducing autophagy of SKOV3 cells, thereby inhibiting tumor growth in a concentration- and time-dependent manner. 121 Jiaxin et al reported that cells cultured in a combination of 20 µmol/L acacillin with 6.25 µmol/L FITC-labeled cisplatin for 1 hour showed more significant inhibition of ABCA3 expression than cells cultured in cisplatin alone. Moreover, molecular docking analyses showed that acacillin had good binding ability with the targets of ABCA3, confirming that acacillin could inhibit the expression of ABCA3 and thereby increase the accumulation of DDP in cells to improve the sensitivity of ovarian cancer cells to cisplatin. 122 Berberine has been also shown to inhibit the repair of cancer cells by upregulating the expression of the tumor-suppressant gene programed cell death factor-4 (PDCD4) and downregulating the expression of miR-21, thereby improving the sensitivity of cancer cells to cisplatin. 123
The anticancer effects of TCM are also reflected in the ability to reduce the drug resistance of cancer cells during chemotherapy. Chunlei et al treated ovarian cancer cells with 10 mmol/L burdock aglyin, and their results showed a significant improvement in the apoptosis of SKOV3/DDP cells. This experiment showed that in addition to increasing the expression of the apoptotic proteins caspase-3/9 and cleaved caspase-3/9, arctigenin also decreased the IC50 of cisplatin in cells and inhibited the mRNA and protein expression of survivin in cells. Thus, arctigenin may reduce the SKOV3/DDP resistance of ovarian cancer-resistant cells by inhibiting the survivin gene. 124 SKOV3/DDP cells treated with 4 μmol/L shikonin showed significantly increased sensitivity to cisplatin, and the IC50 of cisplatin in these cells decreased from 22.51 ± 3.15 μmol/L to 13.43 ± 3.24 μmol/L. Thus, shikonin can reverse SKOV3/DDP resistance in ovarian cancer cells. 125
TCM monomers can also play an important role in reducing the side effects of chemotherapy and improving the survival rate of patients. Patients with middle and advanced tumors often experience cancer-related pain, and paeoniolide itself has an analgesic effect. As an adjuvant drug used in chemotherapy, paeoniolide can be expected to reduce the pain of patients while reversing tumor resistance and reducing the dose of chemotherapy drugs. 36 As an adjuvant to radiotherapy, shikonin, another TCM monomer, may alleviate the G2/M blockade induced by X-rays and inhibit the PI3K/AKT signaling pathway, thus improving the sensitivity of SKOV3 cells to radiation. 126
Future Research Directions
Future studies should consider further exploring the mechanisms underlying the ability of TCM monomers to inhibit the metastatic colonization of ovarian cancer, especially in terms of regulating the TME, blocking the tumor cell cycle, and inducing oxidative stress. Moreover, using modern biotechnological approaches, further analysis of the molecular mechanisms underlying the effects of TCM monomers in blocking metastatic colonization by inhibiting the proliferation and migration of ovarian cancer cells can provide a theoretical basis and supporting data for further optimization and promotion of TCM in clinical practice. Most of the existing studies on the inhibitory mechanisms of TCM monomers for ovarian cancer cells involved animal experiments, cell experiments, and other in vitro experiments, and clinical studies are relatively few. More clinical trials may help further clarify these findings and highlight the mechanisms in more detail. The TCM formulations mentioned in this article are commonly used and frequently employed in clinical treatments within China. For instance, Kang-Ai injection, which is primarily derived from ginseng, has shown significant effectiveness as an adjuvant therapy in the chemotherapy of advanced gynecological malignant tumors. Clinical research data indicate that, in comparison with conventional chemotherapy alone, the combination of Kang-Ai injection with chemotherapy can significantly improve objective response rates, disease control rates, and quality of life improvement rates. 127
Currently, the application of TCM in inhibiting metastatic colonization of ovarian cancer faces 2 major challenges: (1) Complexity of mechanisms: TCM typically exerts therapeutic effects through multi-target and multi-pathway synergies. For instance, certain herbal compounds may simultaneously modulate the PI3K/AKT and Wnt/β-catenin signaling pathways, yet their key bioactive components and dynamic mechanisms require comprehensive elucidation. (2) Standardization and quality control: Variations in cultivation environments and processing techniques lead to significant batch-to-batch fluctuations in active ingredients (eg, flavonoids and saponins), potentially compromising therapeutic consistency.
We plan to further investigate the molecular mechanisms by which TCM regulates the post-migration proliferation of tumor cells, particularly its modulation of pro-proliferative and pro-apoptotic signaling pathways. While TCM has demonstrated preliminary potential in suppressing ovarian cancer metastasis, it must transcend the limitations of empirical medicine through interdisciplinary integration (eg, systems biology and bioinformatics) to achieve data-driven modernization. Future breakthroughs in mechanistic understanding, clinical validation, and standardized production could position TCM as a distinctive therapeutic modality in oncological practice, offering personalized treatment options for patients. This transformation hinges on resolving critical bottlenecks to ensure both scientific rigor and reproducible efficacy.
Conclusions
TCM formulations can play an important role in inhibiting the metastatic colonization of ovarian cancer. This study focused on ovarian cancer cell proliferation and metastasis and attempted to explore the mechanisms by which TCM monomers inhibited the metastatic colonization of ovarian cancer and reveal the potential ability of TCM monomers in regulating the TME, blocking the tumor cell cycle, and inducing oxidative stress. TCM monomers can inhibit the metastasis of ovarian cancer cells through various mechanisms, mainly by inhibiting the expression of cell adhesion molecules, angiogenic factors, and transcription factors, EMT, and the activity of extracellular matrix-degrading enzymes, and by affecting cell movement. TCM monomers have also shown notable effects in inhibiting cell proliferation, including regulation of the TME, blocking the tumor cell cycle, and inducing oxidative stress. Specifically, they can effectively reduce the proliferation ability of ovarian cancer cells by inhibiting angiogenic factors, metabolic remodeling, activating immune factor expression, and inhibiting platelet-activating factors. They can also inhibit the metastatic colonization of ovarian cancer cells and achieve anticancer effects.
In addition to these factors, TCM also offers obvious advantages in the treatment of ovarian cancer. In comparison with traditional treatment methods, TCM formulations cause fewer adverse reactions and toxicity, resulting in relatively little harm to patients, and can improve the sensitivity and efficacy of chemotherapeutic drugs. In addition, through their comprehensive conditioning effects, TCM formulations can inhibit the metastatic colonization of tumor cells while improving patients’ immunity and overall physical condition. Therefore, TCM has broad application prospects in the treatment of ovarian cancer. This article lists some common TCM monomers that have been shown to inhibit the proliferation and metastasis of ovarian cancer. Details can be found in the attached table at the end of the article.
In summary, studies on the mechanisms by which TCM monomers inhibit metastatic colonization of ovarian cancer are of great importance and will provide new ideas and methods for the treatment of ovarian cancer.
Glossary
OC: ovarian cancer; TCM: traditional Chinese medicine; TPL:triptolide;EMT: epithelial-mesenchymal transformation; Bax: Bcl-2-associated X protein; PARP: poly ADP-ribose polymerase; STAT3: signal transducer and activator of transcription-3; JAK: Janus kinase; IL-6: interleukin-6; PI3K: phosphatidylinositide 3-kinases; MMP-7: matrix metalloproteinase-7; MMP-9: matrix metalloproteinase-9; PDK1: phosphatidylinositol-dependent protein kinase 1; mTOR: mammalian target of rapamycin; MAPK: mitogen-activated protein kinase; JNK: C-Jun N-terminal kinase; ROS: reactive oxygen species; VEGF: vascular endothelial growth factor; CDH2: cadherin 2;CDH1:cadherin 1
BR:Brazilin;CS:citrate synthase;mRNA:Messenger RNA; OX-2:Cyclooxygenase-2; uPA:urokinase-type plasminogen activator; MMPs: Matrix Metalloproteinases; PN: osteopontin;
CD44: Cluster of Differentiation 44;GSK: glycogen synthase kinase; ERK: extracellular signal-regulated kinase;RASP65: Golgi stacking protein;KRT18P55: Keratin 18 Pseudogene 55;bFGF: basic fibroblast growth factor; CAFs: cancer-associated fibroblasts;TSP-1: thrombospondin-1;S1P: Sphingosine-1-phosphate;MDSCs: myelogenic suppressor cells;IL-2: Interleukin-2;INF: interferon; TNF: Tumor Necrosis Factor:Insulin Receptor Substrate 1;EY cells: Human Embryonic Kidney cells; ARG: arctigenin; PDCD4:the tumor-suppressant gene programed cell death factor-4;ABCA3:ATP-Binding Cassette Subfamily A Member 3;PPP: pentose phosphate pathway;G6PD:Glucose-6-Phosphate Dehydrogenase;PFK: Phosphofructokinase;PKM2: Pyruvate Kinase M2;TKT: Pyruvate Kinase M2;ROS: reactive oxygen species; Cyt C:Cytochrome C;STAT3: Signal Transducer and Activator of Transcription 3;McL-1:Myeloid Cell Leukemia Sequence 1;Bcl-xl: B-cell Lymphoma-extra Large;Bcl-2: B-cell Lymphoma 2; TME: tumor microenvironment; CD4+/CD8+: cluster of differentiation 4 receptors/cluster of differentiation 8 receptors; DDP: Delivered Duty Paid; IGFR1R: insulin-like growth factor 1 recepto; IGF2: insulin-like growth factor 2; IRS1: insulin receptor substrate-1; bFGF: basic fibroblast growth factor; IC50: half-maximal inhibitory concentration; PAFR: platelet-derived growth factor receptors; EGFR: epithelial growth factor receptor.
Footnotes
Acknowledgements
The investigators extend their heartfelt thanks to Professor Han Fengjuan and all members of her medical research team who contributed to the review and calibration of the manuscript.
Author Contributions
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The study was supported by the National Natural Science Foundation of China (82074484), the National Natural Science Foundation of China (82274566), and the Traditional Chinese Medicine Research Project of Heilongjiang Provincial Administration of Traditional Chinese Medicine, China (ZHY2024-234).
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
Data sharing not applicable to this article as no datasets were generated or analyzed during the current study.
