Abstract
Colon cancer is one of the most prevalent cancers worldwide and the second leading cause of cancer-related deaths. The goal of the present study was to investigate the role of Prodigiosin (PG) in promoting programed cell death in irradiated Caco-2 colon cancer cells. We examined the extent to which PG disrupts the BCL-2/caspase-3 and PPAR-γ signaling pathways and affects apoptosis in these cancer cells. The inflammatory markers COX2, PGE2, NO, TNFα, and the inflammosome NLRP-3, MAPK in addition to prooxidant/antioxidant balance is also under investigation. Caco-2 cells were irradiated with gamma rays (6 Gy) either with or without PG and the results revealed that, PG established IC50 equivalent to 357.27 μgl/ml in Caco-2 cells. The flowcytometric analysis (Annexin V), BCL-2 and caspase-3 showed that PG induces apoptosis for Caco-2 cells. Furthermore, the PG + gamma irradiated (R) group of Caco-2 cells showed significant down regulation in proliferation and inflammatory cascade induction followed by changes in redox tone (expressed by increase in SOD and GSH activities and decrease in MDA concentration), resulted in reduction of tumor growth. It could be concluded that PG has an anti-proliferative action on Caco-2 cells because of its capability to enhance apoptosis in addition to its capability to enhance response of Caco-2 cells to gamma radiation. Based on our findings in the present study we were able to demonstrate that the oxidative status as well as inflammatory responses are grave for determining the longevity, life span and reactivity of Caco-2 colon cancer cells upon exposure to PG unaccompanied or accompanied by gamma radiation. Prodigiosin might represent a valuable key in contesting development of drug resistance of cancer cells and it could raise the radio-sensitivity of cells when PG delivered in combination with radiation exposures
Introduction
Colorectal cancer (CRC) ranks as the third most diagnosed cancer worldwide and is a leading cause of cancer-related mortality, underscoring an urgent public health challenge. 1 Despite significant advancements in medical therapy, CRC remains a critical concern due to its complex pathophysiology and tendency for metastasis, particularly to the liver. Research indicates that pro-inflammatory mechanisms play a pivotal role in initiating and accelerating the growth and dissemination of CRC at the molecular level. 2 The ability of cancer cells to detach from the primary tumor and metastasize via hematogenic or lymphogenic pathways primarily results in portal vein-type metastases, with an alarming 30% to 60% of CRC cases presenting with liver metastases at diagnosis. The unchecked progression of metastatic disease is a leading contributor to organ failure and mortality among cancer patients. 3
Moreover, lifestyle choices especially dietary habits are increasingly recognized as significant risk factors for CRC in contemporary populations. Unhealthy practices, including the consumption of fermented foods, alcohol, processed meats, smoking, sedentary behavior, and obesity, have been directly linked to the development of CRC. 4
Current therapeutic strategies for CRC encompass a range of interventions such as surgery, radiation therapy, and chemotherapy, tailored to the specific disease stage. 5 While immunomodulators enhance the body’s immune defenses against malignant cells, 6 traditional treatments frequently fall short, resulting in suboptimal outcomes, and undesired side effects on healthy tissues. Consequently, there is a critical need to identify novel, naturally occurring anti-tumor agents that demonstrate low toxicity and high efficacy for colorectal cancer treatment.
Notably, recent studies have shown that certain natural substances can positively influence immune responses against cancer. Prodigiosin (2-methyl-3-pentyl-6-methoxyprodigiosin), a red pigment secondary metabolite produced by the bacterium Serratia marcescens, is particularly promising due to its selective apoptotic effects on various human cancer cells while exhibiting minimal toxicity toward normal cells.7,8 This compound, recognized as a representative of tripyrrole red compounds, has demonstrated antibacterial, antifungal, antiprotozoal, antimalarial, and immunosuppressive properties in addition to its anticancer potential.8 -10 Prodigiosin’s ability to trigger proapoptotic responses in cancer cells regardless of their p53 status through the modulation of apoptotic and anti-apoptotic gene expression has been well-documented. 11
Radiotherapy offers the advantage of localized treatment through ionizing radiation, minimizing systemic damage. However, many malignancies develop radio-resistance, leading to treatment failures due to factors such as hyaluronic acid overexpression and tumor hypoxia.12,13 Tumor resistance to radiation and chemotherapy is a significant barrier to effective cancer treatment, often resulting from mechanisms such as DNA damage repair, dysregulated cell cycle progression, the resilience of cancer stem cells (CSCs), and epithelial-mesenchymal transition (EMT).14,15 Enhancing radiation therapy’s efficacy through the use of radiosensitizing agents can potentially amplify tumor damage while protecting healthy tissues, thereby reducing the need for high radiation doses. 16
In the realm of cancer treatment, a comprehensive understanding of the molecular pathways involved is essential for developing effective therapeutic strategies. This exploration focuses on the interactions between several critical pathways: the MAPK (mitogen-activated protein kinase) and Bcl-2 pathways, as well as the role of PPAR-γ (peroxisome proliferator-activated receptor-gamma), caspase-3, TNF-α (tumor necrosis factor-alpha), and NLRP3 (NOD-like receptor protein 3). Understanding these pathways will illuminate how combining pharmacological agents like PG with radiation therapy can enhance cancer treatment outcomes.
The MAPK pathway is crucial for mediating cellular responses to a variety of extracellular signals, including those involved in stress and inflammation. Its activation can lead to cell proliferation, differentiation, and survival, making it a significant player in cancer progression. 17 Concurrently, the Bcl-2 pathway is essential for regulating apoptosis, or programed cell death. It often acts as a guardian of cellular integrity, preventing apoptosis in cancer cells, thus contributing to tumor growth, and resilience against treatments. 18
PPAR-gamma is a nuclear receptor that plays a role in regulating genes involved in glucose and lipid metabolism, as well as inflammation. Its activation has been shown to influence tumor proliferation and survival, presenting a potential target for therapeutic intervention. 19 Additionally, caspase-3 is a key executor in the apoptotic process; its activation leads to the dismantling of cellular components, signifying the transition toward programed cell death. 20
The inflammatory mediator TNF-alpha is notably involved in the tumor microenvironment, promoting cancer cell survival, and proliferation while also activating apoptotic pathways under certain conditions. 21 The NLRP3 inflammasome, a multi-protein complex involved in the innate immune response, contributes to inflammation, and has been implicated in various cancer types. It is essential to understand how the interplay among these components can influence cancer therapy. 22
By examining the cross-talk between these pathways and their collective impact on cancer cell behavior, this investigation aims to elucidate how the combination of PG and radiation can target and manipulate these signaling networks. Our primary goal is to elucidate the anti-proliferative action of prodigiosin, particularly when used in conjunction with radiation therapy. Additionally, we will assess PG’s cytotoxic effects and its impact on apoptosis, alongside the effects of radiation. Key inflammatory markers including COX-2, PGE2, nitric oxide (NO), TNF-α, and the inflammasome NLRP-3 as well as the balance between prooxidants and antioxidants, will also be investigated. Ultimately, this study aims to establish a foundation for a novel therapeutic approach that combines prodigiosin with gamma radiation to effectively target and inhibit the growth of colon cancer cells in vivo.
Materials and Methods
Chemicals
Prodigiosin a red pigment produced by Gram-negative bacteria (Serratia marcescens) was purchased from BIOTECH CONSORTIUM INDIA LIMITED, New Delhi, India.
The other chemicals were provided by Sigma Aldrich Comp.
Cell Lines and Cell Culture
The human colon cancer cells (Caco-2) utilized in this study was obtained from VACSERA’s Cell Culture Department in Cairo, Egypt. According to the distributor’s instructions, Caco-2 cancer cells were cultivated in RPMI media with 10% fetal bovine serum (ThermoFisher Scientific, USA), supplemented with 100 U/ml of penicillin and 100 mg/ml of streptomycin, and kept at 37°C in a humidified environment with 5% CO2.
Caco-2 (RRID: CVCL_0025), ATCC STR: ATB-37 Species of origin: Homo sapiens (Human) Disease, Colorectal adenocarcinoma.
Radiation Facility
The irradiation process used a 137Cs source (Gamma-cell 40 Exactor; NCRRT, AEA, Cairo, Egypt). At 85% confluency (a single exposure to 6 Gy), all Caco-2 cell groups were irradiated with γ-rays at a dosage rate of 0.012 Gy/s. To guarantee dose and dose rate homogeneity, a Fricke reference standard dosimeter (ISO/ASTM E 51,026) was used in all studies. 23
Cell Viability/Morphology Assay
The cytotoxic profile of prodigiosin was assessed by analyzing the cellular viability using the MTT test [3-(4,5-Dimethylthiazol-2yl)-2,5-diphenyl tetrazolium bromide (5 mg/ml in PBS; #M6494, BIO BASIC CANADA INC)]. The MTT assay is a colorimetric assay frequently used to evaluate cell viability, proliferation, and cytotoxicity, because it offers valuable data about the metabolic activities of cells. In living cells, MTT, a yellow tetrazole, is reduced and converted to purple formazan by mitochondrial electron transport chain. Different concentrations of PG were dissolved in culture media and applied to the cells for 24 hours; suspension in control media served as the treatment. The cells diluted with culture medium were kept as control. After adding the MTT, the absorbance was then measured at 560 nm and subtract background at 620 nm using an enzyme-linked immunosorbent assay (ELISA) plate reader (BioTeck, Bad Friedrichshall, Germany) in relevance to cellular density. 24 Using SPSS one-way ANOVA, the half-maximal inhibitory concentration (IC50) was calculated (IBM Inc., Chicago, IL, USA). The graphs were created with the Graph-Pad Prism software (v.8.0; Graph-Pad Prism Inc., USA). Using a phase-contrast inverted microscope equipped with a digital camera, cell morphology was captured (Nikon, Japan). The MTT experiment was performed in triplicate, meaning that 3 independent trials were conducted for each concentration tested.
Study Groups
Four Caco-2 groups were established in order to evaluate the anti-proliferative and radio-sensitizing effectiveness of PG: (1) Caco-2 Cells without treatment (vehicle treated; Control); (2) Cells treated with PG alone; (3) Cells exposed to a single dose of irradiation (6 Gy) the radiation exposure period was 18 minute (3 minute/gray); and (4) Cells treated with PG 24 hours prior to irradiation (PG + 6 Gy).
Determination of COX2, PGE2, NO, TNF-α, and IL-1β by ELISA Technique
The level of cyclooxygenase-2 COX2 (ab267646-Abcam, UK), PGE2 (KHL1701-Thermo Fisher Scientific), nitric oxide (ab65328), TNF alpha (ab181421) and IL-1β (ab214025) was estimated by ELISA kits according to the manufacturer’s instructions, for the enzyme-linked immunosorbent assays via ELISA microplate reader (DV990 BV 416; Gio.DE VITA and CO., Rome, Italy).
Western Blot Analysis
Primary antibodies to β-actin (ab8227) and NLRP-3 (ab263899) [EPR23094-1] RRID: AB_2841002 were obtained from Abcam (UK) and used in immunoblotting. In 6-well plates, 4 × 105 cells were planted in each well. Following the treatments, the cells were lysed for 30 minutes on ice in 10 µl of lysis buffer plus 100 mM PMSF (Solarbio, Beijing, China) in 1 ml of buffer. Using polyacrylamide and 10% sodium dodecyl sulfate electrophoresis gels, cell lysates were separated, blotted with polyvinylidenedifluoride membranes, and blocked with 5% skim milk in PBS plus 0.1% Tween 20 (TBST). Following that, primary antibodies were added and the membranes were incubated for 12 hours at 4°C. A secondary anti-rabbit antibody (1:4000; Protein-tech) combined with horseradish peroxidase was applied to the membranes for an hour at room temperature. The chemiluminescence detection was performed according to the manufacturer’s protocols of the Amersham detection kit, and then exposed to X-ray film. The protein level of each sample was quantified by densitometric analysis of the auto-radiograms through using a scanning laser densitometer (Biomed Instruments). The records obtained were assayed after normalization to β-actin protein expression (used as the housekeeping protein). 25
Detection of Proliferation MAPK, and Apoptotic Parameters BCL-2, PPAR-γ, and Caspase3 by ELISA
MAPK (MBS009637, MyBioSource, Inc., USA), BCL-2 (MBS701787, MyBiosource, Inc., USA), PPAR-γ (MBS263089, MyBioSource, Inc., USA), Caspase-3 (MBS260710, MyBiosource, Inc., USA), levels were measured using specific ELISA kit as directed by the product’s manufacturer.
Flow Cytometry Test for Apoptosis
The “FITC Annexin V Apoptosis Detection Kit with PI” (BD Pharmingin TM, BD Biosciences Co., USA; Number # 51-66121E) was used for flow cytometry apoptosis analysis in accordance with the manufacturer’s instructions. In addition to cell morphology, apoptosis can also be found by flow cytometric examination of heterogeneous cell populations that retain cell viability. This technique can differentiate cells that are intact, apoptotic, or dead from either necrosis or apoptosis all at once. A calcium-dependent phospholipid-binding protein with a strong affinity for phosphatidylserine (PS) is found on the inner side of the plasma membrane. Translocation of PS from the inner to the outer layer of the plasma membrane, which exposes it at the external cell surface, is one of the difficult-to-detect alterations at the cell surface that characterize early apoptotic stages. In addition to apoptosis, necrosis also exhibits this tendency. The distinction between these 2 forms of cell death lies in the fact that, while the cell membrane remains intact in the early stages of apoptosis, during necrosis the cell membrane breaks down, allowing propidium iodide (PI) entrance, which can be used to identify necrotic cells.
In summary, colon cancer cells were cultured in 6-well plates at a density of 3 × 105 cells per well, exposed to prodigiosin (PG) at a concentration equivalent to its IC50. Following a 24-hour incubation period, Caco-2 cells were extracted by trypsinization from both treated and untreated groups. They were then rinsed with precold PBS. After centrifuging, the cells were again suspended in 100 µl binding buffer and, at 4°C, in cell staining buffer. Following that, 10 ml of 20 mg/ml propidium iodide (PI) and 5 ml of FITC Annexin V and were added to flow cytometry tubes, and the tubes were incubated for 15 minutes at room temperature and in the dark. The number of apoptotic cells was measured using a flow cytometer (BD Accuri C6), and the data were subsequently analyzed using Cell Quest software on a FACSC-LSR (Becton & Dickinson Company).
Biochemical Assay
Determination of Malondialdehyde Level
The level of malondialdehyde (MDA), a key indicator of oxidative stress, was quantified calorimetrically, following the method of Yoshioka et al. 26 This assay enables the measurement of lipid peroxidation products, which are critical in understanding cellular damage in various pathological conditions.
Malondialdehyde (MDA) is a reactive aldehyde formed from the breakdown of polyunsaturated fatty acids. The assay is based on the principle that MDA reacts with thiobarbituric acid (TBA) in an acidic environment to form a colored complex, which can be measured spectrophotometrically. Specifically, 1 molecule of MDA reacts with 2 molecules of TBA, producing a pink-colored compound that absorbs light at 535 nm. The intensity of the color is directly proportional to the concentration of MDA in the sample, making it a useful biomarker for oxidative stress.
Procedure
Cells were homogenized in an appropriate buffer to create a lysate, followed by centrifugation to separate the supernatant, which contains the soluble components necessary for the assay. A volume of 0.5 ml of the supernatant was pipetted into a centrifuge tube. To this, 2.5 ml of 20% trichloroacetic acid (TCA) was added. The mixture was gently shaken and allowed to incubate for 10 minutes at room temperature to precipitate proteins. Next, 1 ml of TBA solution was added to the tube. The mixture was shaken again and then incubated in a boiling water bath for 30 minutes. This step facilitates the formation of the TBA-MDA complex. After boiling, the tubes were rapidly cooled. Following incubation, 4 ml of n-butyl alcohol was added to the tube, and the solution was shaken vigorously. Centrifugation at 3000 rpm for 10 minutes was then performed to separate the butanol upper layer, which contains the TBA-MDA complex. The absorbance of this layer was measured at 535 nm against n-butyl alcohol as the blank.
Determination of Superoxide Dismutase Activity (SOD)
Principle
Superoxide dismutase activity is measured according to the method of Minami and Yoshikawa. 27 Superoxide dismutase (SOD) catalyzes the dismutation of the superoxide radical (O2−) into hydrogen peroxide (H2O2) and elemental oxygen (O2).
Superoxide ions, generated from auto-oxidation of pyrogallol, convert the nitro blue tetrazolium chloride (NBT) to NBT-diformazan which absorbs light at 550 nm.
Superoxide dismutase reduces the superoxide ion concentration thereby lowering the rate of NBT-diformazan formation. The extent of reduction in the appearance of NBT-diformazan is a measure of SOD activity present in samples.
Practice
In covered polyethylene tubes, the following reagents were applied to the 0.1 ml sample: 0.4 ml NaCl, 0.75 ml ethanol, chloroform
Mixture was vigorously mixed then, centrifuged at 10 000 r.p.m at 4°C for 60 minutes.
The clear supernatant was transferred to other test tubes then, the following reagents were added. 0.25 ml supernatant, 0.5 ml cacodylic buffer, 0.1 ml triton, and 0.1 ml NBT
The mixture was then incubated at 37°C for 5 minutes.
About 0.01 ml pyrogallol was applied and left 5 minutes then 0.3 ml stopper solution was added.
Absorbance was measured against buffer at λ 540 nm.
Determination of Reduced Glutathione
Reduced glutathione (GSH) concentration was estimated by the method of Beutler et al. 28
Principle
The method depends on the fact that both protein and non-protein SH-groups react with Ellman’s reagent [5,5-dithiobis-(2-nitrobenzoic acid)] to form a stable yellow color of 5-thio-2-nitrobenzoic acid, which can be measured at 412 nm. In order to determine glutathione level, precipitation of protein SH-groups was necessary before the addition of Ellman’s reagent.
Procedure
A volume of 100 µl 10% supernatent was pipetted in a centrifuge tube containing 100 µl 20% TCA. The tube was thoroughly mixed, left to stand at room temperature for 5 minutes, then centrifuged at 5000 rpm for 10 minutes.
Exactly 100 µl supernatant, standards and distilled water were transferred to test tubes labeled as T, S and B, respectively. A volume of 2 ml 0.3 M phosphate buffer was added to all tubes. The volume in all tubes was completed to 2.5 ml with distilled water, then 0.5 ml of Ellman’s reagent was added. The tubes were vortexed and incubated for 10 minutes at room temperature, then the absorbance of T and S were read against B at 412 nm.
Statistical Analysis
Every experiment was conducted at least 3 times, and the results were reported as the mean ± standard error (SEM; n = 3). One-way analysis of variance (ANOVA) was employed for the statistical analysis, followed by Tukey’s post hoc test for multiple comparisons between treatment means. Graph Pad Software, La Jolla, CA, USA, produced Prism, version 8, and was used to create the graphs for statistical analysis. Statistical significance was determined with P-values less than .05.
Results
Inhibitory effect of prodigiosin on Caco-2 cells proliferation.
For the assessment of the possible antitumor efficacy of PG on colon cancer; Caco-2 cell line was subjected to increasing concentrations of PG to evaluate PG cytotoxic activity utilizing MTT assay. 100% viable Caco-2 cells cultured in RPMI complete medium are regarded as a positive control. As shown in Table 1 and Figure 1A and B, inhibition in Caco-2 cells was observed 24 hours of PG treatment resulted in Caco-2 cells proliferation inhibition in a dose dependent manner and the half maximal inhibitory concentration (IC50) was approximately estimated to be 357.27 μg/ml.
Effect of different concentrations of prodigiosin on Caco-2 proliferation .
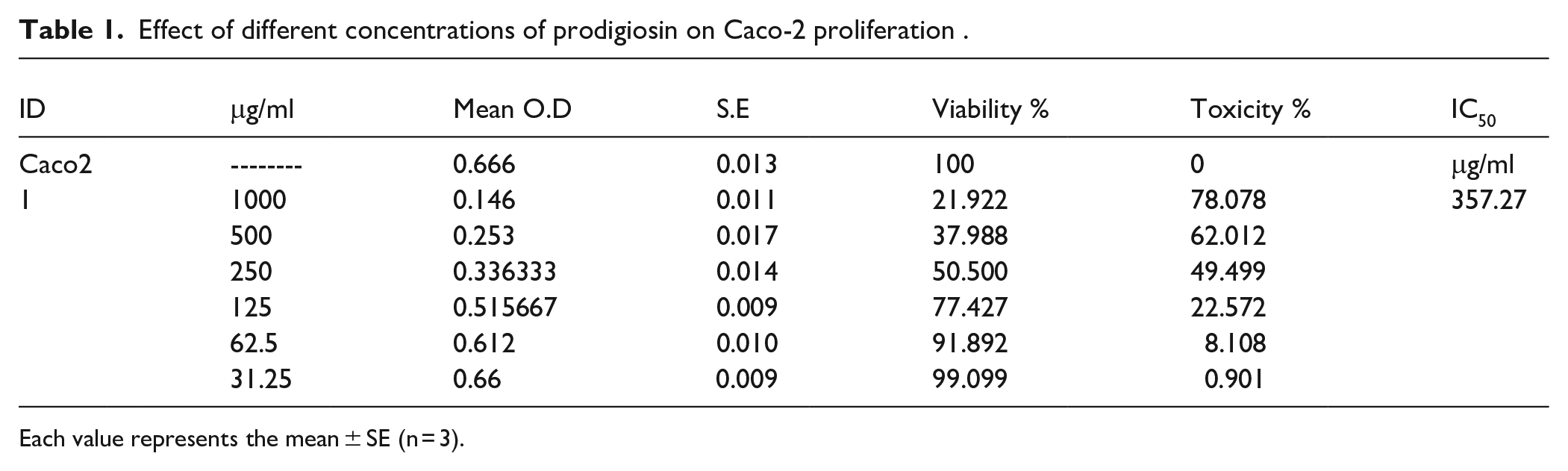
Each value represents the mean ± SE (n = 3).

(A) Antitumor efficacy of PG on Caco-2 colon cancer cells and (B) effects of PG on Caco-2 proliferation under microscope.
Cell morphological alterations were observed in phase-contrast images in a PG dose-dependent way (Figure 1B). Cells were checked for any physical signs of toxicity, for example, partial or complete loss of the monolayer, rounding, shrinkage, or cell granulation compared to control group. There were signs of apoptosis, membrane blebbing, and cells detachment accompanied by decrease in cell counts concurrently with rising PG concentrations.
Effect of Prodigiosin on Inflammatory Determinants (COX2, PGE2, and NO)
In order to elucidate the anti-inflammatory efficacy of PG on colon cancer we conducted further analysis on inflammatory determinants (COX2, PGE2, and nitric oxide) that is illustrated in Figure 2. Figure 2A to C demonstrated the results obtained for C, PG, R, PG + R groups of Caco-2 cell. In PG-treated Caco-2 colon cancer cells, the data pointed to significant decreases in COX-2 by 30.82% and PGE2 activities by 67.92% as well as significant decreases in NO concentration by 77%, when compared to control Caco-2 cells. Also, the 6 Gy gamma irradiation of Caco-2 cells resulted in remarkable decreases (P < .05) in COX-2 as well as PGE2 activities by 44.7% and 20.3% respectively, associated with a noticeable down regulation in the NO concentration by 30.3% comparing with control Caco-2. Besides, the coupling of PG exposure and 6 Gy irradiation of Caco-2 cell induced significant diminution (P < .05) in COX-2, PGE-2 activities by 63.95%, 42.8% respectively, and nitric oxide concentration by 59% comparing with their equivalent values in control Caco-2 cells. It seems that the changes in the 3 parameters are more pronounced in group of PG + R Caco-2 cells compared with PG or R cells groups.

Effect of prodigiosin PG and/or 6 Gy gamma irradiation on inflammatory determinants (A: COX2, B: PGE-2 activities, and C: NO) in human Caco-2 colon cancer cells. Values are stated as mean values ± SEM (n = 3 independent values). Caco-2 + prodigiosin + R: Caco-2 cancer cells treated with prodigiosin and exposed to 6 Gy. The columns that have (a) significant from Caco-2, (b) significant from Caco-2 + PG, (c) significant from Caco-2 + R, and (d) significant from Caco-2 + PG + R are indicated at (P < .05). Statistical analysis was performed utilizing one way analysis of variance (ANOVA), followed by Tukey’s post hoc test.
Impact of PG and/or γ-Irradiation Exposure on TNF-α, NLRP-3, and IL-1β
To find out the role of inflammatory response in cancer we further determined TNF-α/NLRP-3. The NLRP3 inflammasome is a critical component of the innate immune system that mediates caspase-1 activation and the secretion of proinflammatory cytokine IL-1β in response to microbial infection and cellular damage. TNF-α is a positive regulator of NLRP3 inflammasome priming. 29 The TNF-α/NLRP-3 pathway is a series of cell proteins interactions that plays a vital role in the development of cancer.30,31 The data exemplified in Figure 3 displayed that there were substantial increases in TNF-α level (Figure 3A), and NLRP-3 protein expression as well as IL1-β level in Caco-2 group (Figure 2B and C). Current study results demonstrated significant alterations in inflammatory response parameters upon treatment of Caco-2 cells with PG where TNF-α level was declined by 55.5% and NLRP-3 protein expression was significantly decreased (P < .05) by 41% associated with decline in IL1-β level by 27% with the respective Caco-2 group. Just about the same issue, our experimental results upon irradiation of Caco-2 cancer cells identify a notable decrease in TNF-α level by 40% together with NLRP-3 protein expression by 59.7% and IL1-β level by 50.7% with values equivalent to cancer group. Nevertheless, both treatment modalities in combination (Caco-2 + PG + R) revealed the most observable decrease in TNF-α level by 47% followed by a noticeable decline in NLRP-3 protein expression by 73.44% and IL1-β level by 65.67% compared to Caco-2 cancer group, Figure 3A to C.

Effect of PG on TNF-α/NLRP-2/IL-1β signaling. Values are presented as mean values ± SEM (n = 3 independent values. Caco-2 cancer cells treated with PG and exposed to 6 Gy. The columns that have (a) significant from Caco-2, (b) significant from Caco-2 + PG, (c) significant from Caco-2 + R, and (d) significant from Caco-2 + PG + R are indicated at (P < .05). Statistical analysis was performed utilizing one way analysis of variance (ANOVA), followed by Tukey’s post hoc test. (A) and (C) by ELISA and (B) by western blot.
Prodigiosin Decreases Proliferation and Induces Apoptosis in Human Caco-2 Colon Cells Through Targeting MAPK, BCL-2, PPAR-γ, and Caspase3 Protein Levels
In the current work, the imbalance between proliferation and apoptosis was further illustrated by increasing MAPK, BCL-2 level and diminishing the level of PPAR-γ and the proapoptotic caspase-3 in Caco-2 group.32,33 Apoptosis enhancement was observed due to treatment of Caco-2 colon cancer cells with PG as presented by the remarkable MAPK and BCL-2 down regulation in protein levels by 21%, 43% respectively (Figure 4A and B), and elevation (P < .05) in PPAR-γ level by 19% (Figure 4C), and cleaved caspase-3 level by 363% (Figure 4D), compared to untreated Caco-2 cancer cells. It is important to note that irradiation of Caco-2 cells reveals a significant reduction in MAPK by 64%, BCL-2 levels by 58% and increase in PPAR-γ by 91% and cleaved caspase-3 levels by 503% relative to untreated colon cancer cells. Further, exposure of Caco-2 cells in combination with PG treatment to γ-irradiation induces significant (P < .05) decline in MAPK by 82%, BCL-2 levels by 77% accompanied with noteworthy increase in PPAR-γ by 159% and cleaved caspase-3 level by 838% compared to its equivalent value in Caco-2 group, Figure 4A to D.
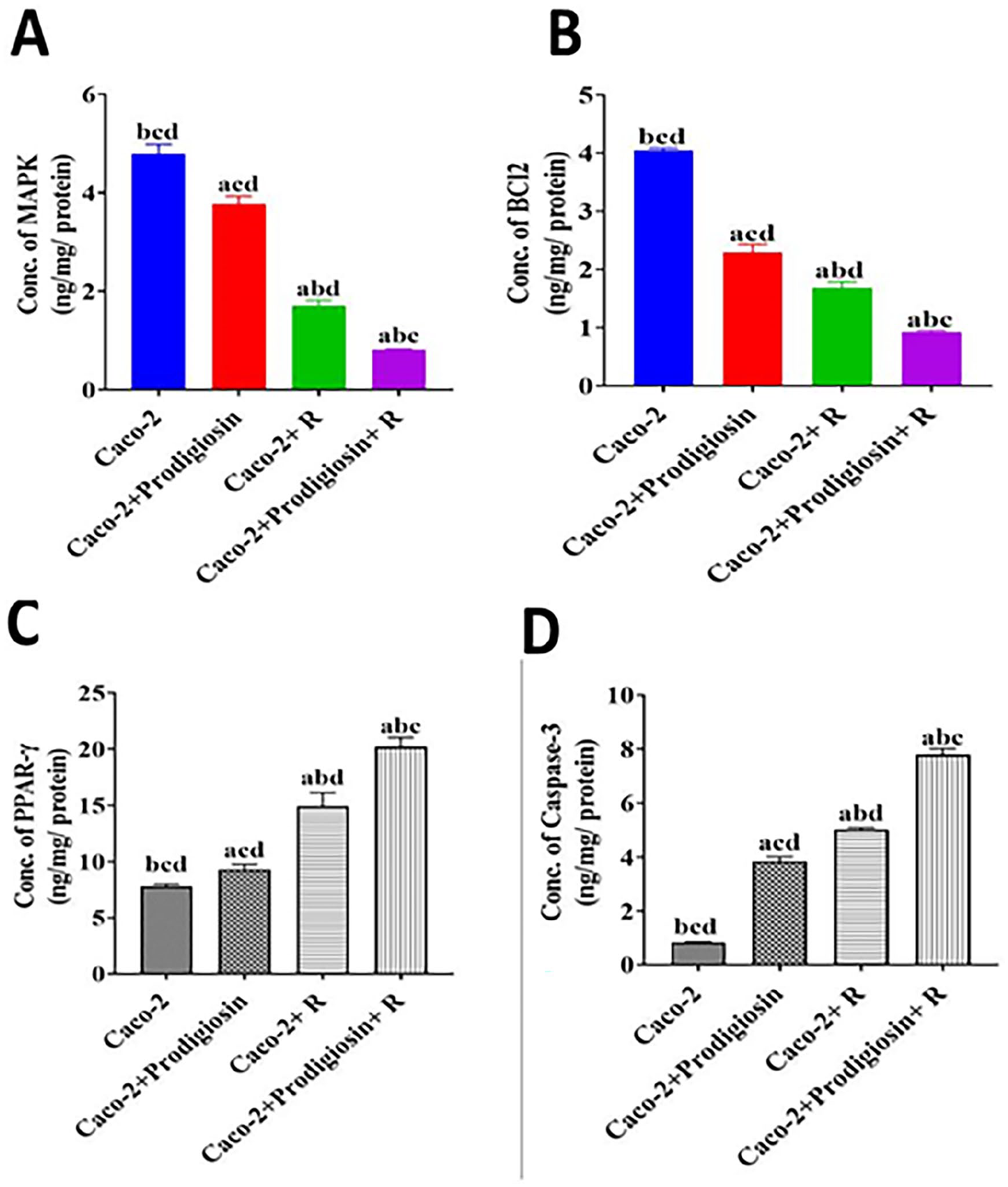
Effect of PG on proliferation and apoptosis of human Caco-2 colon cells. (A) Concentration of MAPK, (B) concentration of BCL2, (C) for PPAR-γ concentration, and (D) for caspase-3 concentration. Values are stated as mean values ± SEM (n = 3 independent values). Caco-2 colon cancer cells treated with PG and exposed to 6 Gy. The columns that have (a) significant from Caco-2, (b) significant from Caco-2 + PG, (c) significant from Caco-2 + R, and (d) significant from Caco-2 + PG + R are indicated at (P < .05). Statistical analysis was performed utilizing one way analysis of variance (ANOVA), followed by Tukey’s post hoc test Apoptosis evaluation by flow cytometry.
Accordingly, in relation to the divergent apoptotic cascade stages, the way cells attach to annexin V (x axis) and/or PI (y axis) in a flow cytometric dot plot chart aids in differentiating between cells that underwent necrosis (positive PI, negative annexin v staining/upper left quadrant) and cells that underwent apoptosis (annexin V positive cells/lower right quadrant).
Depending on such strategy, Caco-2 group apoptosis was distinguishably lowered opposed to all the other groups (P < .05) as presented in Figure 5. Figure 5A to D shows the results of 1 representative from each group, while Figure 5E shows the results of means of 3 samples from each group. Two distinct populations appeared following the treatment with IC50 value of prodigiosin: 1 was identified as an early apoptotic cell since it only displayed a single positive annexin V expression, and late apoptotic cells the displayed double positive Annexin V and PI staining (Figure 5/upper right quadrant). The early apoptotic cells % increased from 6.1% in the Caco-2 group to 13.8% in the Caco-2 groups treated with PG, respectively. Concomitant treatment with PG + radiation elevated that percentage to 29.1%. It is noteworthy that the highest percentage of apoptotic cells 37.2% (early and late, the sum of the 2 quadrant) and the lowest percentage of viable cells (45.5%) were all reported in the group treated with PG + radiation. In Figure 5, it is feasible to observe that such populations were concurrent with a gradual and appreciable elevation in the number of necrotic cells, which increased from 17.3% in control Caco-2 group to 29.5% in Caco-2 group treated with PG. Moreover irradiation of Caco-2 cells increased the number of necrotic cells to 44.8%. Then again, the apoptosis of gamma irradiated Caco-2 treated with PG was significantly higher than their corresponding non- radiated group (P < .05).

(A) Flow cytometric analysis of apoptosis using Annexin V-FITC and PI Staining. Caco-2 control cells, Caco-2 + PG, Caco-2 + Radiation, Caco-2 + PG + R. Annexin V/PI staining indicates viable (Q1-LL), early apoptotic (Q1-LR), late apoptotic (Q1-UR), necrotic (Q1-UL) cells. R3: Transitional population with low Annexin V/PI. B: Percent of cells in different group. The columns that have (a) significant from Caco-2, (b) significant from Caco-2 + PG, (c) significant from Caco-2 + R, and (d) significant from Caco-2 + PG + R are indicated at (P < .05). Mean % (±SD, n = 3) of cell populations.
Defensive Effects of Prodigiosin Against Oxidative and Antioxidant Status Induced in γ-Irradiated Caco-2 Cells
Oxidative stress in colon cancer cells was evaluated based on enzymatic (SOD) and GSH antioxidants and MDA (lipid peroxidation end-product) measurement. The data illustrated in Figure 6; showed a remarkable restoration of the regular redox tone. The levels of MDA were considerably raised in Caco-2 colon cancer cells (Figure 6A), associated with a marked decrease in SOD activity (Figure 6B), and GSH concentration (Figure 6C), This effect was significantly improved with concurrent PG treatment, showed by significant decrease (P < .05) in MDA by percent change decrease 64% with significant increase (P < .05) in GSH and SOD activities by 385%, 353% respectively compared to untreated Caco-2 group. More pronounced amelioration was observed after irradiation of Caco-2 cells, where MDA decreased by 16% and significant increase (P < .05) in GSH and SOD activities by 82%, 128%. What is more, exposure of Caco-2 cells to γ-irradiation in combination with PG produce a significant recovery in redox tone where MDA noteworthy decreased by 31% concomitant with significant increase in GSH and SOD activities by 263%, 245% respectively with respect to Caco-2 group (Figure 6A to C).

Protective effect of PG against oxidative stress in Caco-2 colon cancer. (A) for MDA level, (B) for SOD activity, and (C) for GSH concentration. Each value represents Mean ± SEM (n = 3). Caco-2 cancer cells treated with PG and exposed to 6 Gy. The columns that have (a) significant from Caco-2, (b) significant from Caco-2 + PG, (c) significant from Caco-2 + R, and (d) significant from Caco-2 + PG + R are indicated at (P < .05). Statistical analysis was performed utilizing one way analysis of variance (ANOVA), followed by Tukey’s post hoc test.
Discussion
Colon cancer is one of the most prevalent cancers globally and the second leading cause of cancer-related mortality. It typically begins with inflammation that can progress to adenocarcinoma, highlighting the critical role of the tumor microenvironment in cancer development. 34 Our study aimed to explore the anticancer effects of prodigiosin (PG) in combination with radiation therapy on Caco-2 colon cancer cells, addressing the urgent need for innovative therapeutic strategies to overcome radiation resistance, a significant barrier to effective treatment.
The findings of our study reveal that both prodigiosin and radiation significantly reduce the activity of COX-2, a key inflammatory marker associated with cancer progression, apoptosis, and metastasis. 35 Elevated COX-2 levels are frequently observed in adenomas and adenocarcinomas, and the use of COX-2 inhibitors has been shown to lower colorectal cancer incidence.36,37 Our results indicate that the combination of PG and radiation is particularly effective in reducing COX-2 activity, suggesting a synergistic effect that enhances the anti-inflammatory properties of both treatments. This aligns with previous studies, such as those by Li et al, 38 which demonstrated that COX-2 inhibitors can enhance tumor apoptosis and inhibit angiogenesis, thereby improving therapeutic outcomes.
Furthermore, our investigation into the inflammatory microenvironment revealed that the combination therapy significantly reduced the expression of TNF-α and NLRP-3 inflammasome components in Caco-2 cells compared to controls. This is consistent with the literature indicating that inflammation plays a pivotal role in colorectal carcinogenesis. 39 The activation of the NLRP3 inflammasome has been linked to promoting tumor growth and metastasis.40,41 By inhibiting both COX-2 and NLRP3, prodigiosin may disrupt the inflammatory signaling pathways that facilitate tumor progression, providing a potential mechanism for enhancing radiosensitivity in colorectal cancer.
Our findings also suggest that the interplay between PG and the COX-2/PGE2 pathway could be crucial in modulating immune responses. Previous research has shown that PGE2 can inhibit T-cell function and promote an immunosuppressive tumor microenvironment.42,43 By potentially reducing PGE2 levels through COX-2 inhibition, prodigiosin may enhance the efficacy of the immune response against tumor cells. This is particularly relevant given the emerging focus on immunotherapy in cancer treatment, where the modulation of immune checkpoints like PD-L1 is critical. 44
In addition to the inflammatory markers, our study highlights the role of the NLRP3 inflammasome in mediating the effects of PG and radiation. The significant reduction in IL-1β and NLRP-3 expression in treated Caco-2 cells underscores the importance of targeting inflammasome pathways in colorectal cancer therapy. This finding is supported by previous studies that have demonstrated the role of NLRP3 in promoting CRC progression and its potential as a therapeutic target.34,45 The interaction between NLRP3 and the gut microbiota, as well as its influence on the tumor microenvironment, opens new avenues for research into the complex mechanisms underlying colorectal cancer.
Overall, our study contributes to the growing body of evidence supporting the use of natural compounds like prodigiosin in combination with conventional therapies to enhance treatment efficacy in colorectal cancer. By demonstrating the synergistic effects of PG and radiation on inflammatory markers and signaling pathways, we provide a foundation for future research aimed at developing effective combination therapies that target the tumor microenvironment. As we continue to unravel the complexities of cancer biology, our findings underscore the importance of integrating anti-inflammatory strategies with traditional treatment modalities to improve patient outcomes in colorectal cancer.
Our study reveals that prodigiosin (PG) may mitigate tumor-associated macrophage (TAM)-mediated immunosuppression by inhibiting pro-inflammatory cytokines such as TNF-α, IL-2, and interferon-gamma (IFN-γ). This modulation of the immune response aligns with findings by Cuevas et al, 46 who demonstrated that PG can effectively inhibit circulating levels of these cytokines in vivo, thereby stabilizing atherosclerotic lesions, and suggesting a broader role in immune modulation.
The current results indicate that Caco-2 cells exhibited significantly elevated levels of mitogen-activated protein kinases (MAPK) (P < .05; Figure 4A). The MAPK signaling pathway, particularly the RAS–RAF–MAPK kinase (MEK)–extracellular signal-regulated kinase (ERK) cascade, is crucial in regulating various cellular processes, including proliferation, differentiation, and survival. 17 Dysregulation of this pathway is frequently implicated in oncogenesis and treatment resistance, particularly in colorectal cancer, where overexpression, and activation of ERK MAPK are common. 47 Our findings suggest that PG may alter the dynamics within this pathway, potentially enhancing apoptosis while inhibiting proliferation.
The balance between apoptosis and proliferation is critical in cancer progression. Our data show a decrease in pro-apoptotic caspase-3 levels and an increase in anti-apoptotic BCL-2 levels in Caco-2 cells, indicating a disruption in the regulatory mechanisms governing these processes. PPAR-γ, known for its role in promoting apoptosis and reducing cell proliferation, was also affected; further supporting the notion that PG influences the tumor microenvironment by modulating these pathways.48 -50 The observed decrease in caspase-3 levels, a key player in apoptosis, highlights the challenge posed by cancer cells in evading programed cell death, a hallmark of malignancy. 16
Oxidative stress, characterized by the overproduction of reactive oxygen species (ROS), plays a vital role in the progression of colorectal cancer by causing DNA damage and chromosomal aberrations. 51 Our study demonstrates that PG treatment leads to an increase in caspase-3 levels and a decrease in BCL-2, consistent with findings by Hassankhani et al, 5 who noted that prodigiosin-induced apoptosis is associated with the inhibition of BCL-2, and survivin in HT-29 cells. These results suggest that PG may effectively promote apoptosis in colorectal cancer cells by targeting these anti-apoptotic pathways.
Moreover, the flow cytometric analysis corroborated our findings, confirming that PG induces apoptosis in Caco-2 cells. The tripyrrole structure of prodigiosin is significant, as it has been shown to trigger autophagy and apoptosis in various cell lines, including K562, while also increasing ROS levels. 52 This aligns with the broader literature indicating that PG’s anticancer effects are mediated through multiple mechanisms, including the activation of MAPK and Akt pathways, suppression of cell cycle progression, and enhancement of intracellular acidification and DNA cleavage. 53
Moreover, PG has demonstrated significant anti-tumor efficacy in vivo, notably against Lewis lung carcinoma in BALB/c mice, where tumor volumes decreased by 34.18% after 28 days of treatment. 54 This finding underscores PG’s potential as a therapeutic agent in cancer treatment. In human gastric cancer cell lines (HGT-1), PG has been shown to induce apoptosis, evidenced by a continuous decline in cell viability and morphological changes such as cell shrinkage and chromatin condensation. 55
In breast cancer studies, PG has exhibited cytotoxicity through the mitochondrial pathway in both estrogen receptor-positive (MCF-7) and negative (MDA-MB-231) cell lines, as well as in multidrug-resistant (MDR) MCF-7 cells, demonstrating a dose- and time-dependent effect. 56 These findings suggest that PG’s mechanism of action involves promoting apoptosis, potentially through mitochondrial dysfunction and DNA cleavage. 57 Notably, PG has been identified as a proton sequestering agent that disrupts the intracellular pH gradient, leading to ATP depletion and subsequent apoptotic cell death in neuroblastoma cells. 58 Additionally, PG has been reported to promote cytochrome c release, further implicating the mitochondrial pathway in its apoptotic effects. 59
Our study also revealed that Caco-2 colon cancer cells exhibited significantly higher malondialdehyde (MDA) concentrations and lower activities of superoxide dismutase (SOD) and glutathione (GSH; Figure 6). This pattern suggests the presence of oxidative stress within the cancer cells, which, while typically associated with cell death and growth inhibition, can also function as signaling molecules that regulate various pathways, including cell cycle, apoptosis, and energy metabolism. 60 Prasad et al 61 and Moloney and Cottor 62 highlight that the production of reactive oxygen species (ROS) is often elevated in cancer cells due to increased metabolic activity and oncogenic activation, further contributing to the complexity of ROS’s role in cancer biology.
Prodigiosin (PG), a red pigment produced by various bacterial species, has garnered attention for its potential anti-cancer properties through inhibition of the MAPK signaling pathway. MAPK signaling, particularly the ERK sub-pathway, is critically involved in regulating cell proliferation, differentiation, and survival. Dysregulation of this pathway is frequently implicated in various cancers, including colon cancer, where mutations in RAS, and RAF gene families play pivotal roles in tumorigenesis. Our findings demonstrate that PG effectively inhibits the activation of the MAPK pathway, which may contribute to its anticancer effects. The significance of MAPK signaling in colorectal cancer has been well-documented. For instance, mutations in the KRAS gene, a member of the RAS family, occur in approximately 40% of colorectal cancer cases and are associated with poor prognosis and resistance to targeted therapies.63,64 These mutations lead to persistent activation of the MAPK pathway, promoting uncontrolled cellular proliferation and survival.
Additionally, mutations in the BRAF gene also play a critical role in colorectal cancer progression. Approximately 10% of colorectal cancers harbor BRAF mutations, which often correlate with more aggressive disease and reduced survival rates.65,66 The persistent activation of the RAF-MEK-ERK signaling cascade due to these mutations highlights a critical interplay between genetic alterations and MAPK pathway dysregulation. By inhibiting this pathway, PG may offer a therapeutic strategy to counteract the effects of these oncogenic mutations. For example, several studies have shown that targeting the MAPK pathway can restore sensitivity to other therapeutic agents in KRAS and BRAF-mutant cancers.67,68 Thus, the context provided by existing literature not only highlights the relevance of PG’s action but also positions it as a potential adjuvant treatment that could enhance the efficacy of current therapies for colon cancer. In summary, our study’s findings regarding PG’s inhibition of the MAPK pathway align with existing literature that underscores the importance of targeting this pathway in colorectal cancer, especially in the context of RAS/RAF mutations. Future studies should focus on elucidating the precise mechanisms of PG action and its potential combination with existing treatments to improve therapeutic outcomes for patients with colorectal cancer.
The interplay between ROS and apoptotic signaling is crucial, as increased ROS levels can lead to apoptotic body formation through the downregulation of MAPK pathways. This suggests that PG’s ability to modulate ROS levels may be a key factor in its anticancer effects. Prodigiosin’s multifaceted biological activities, including its antioxidant properties and minimal toxicity to normal cells, position it as a promising candidate for cancer therapy. 69 Its structural features, including a tripyrrole backbone that produces red fluorescence, enhance its appeal for therapeutic applications. 70 Moreover, PG’s antioxidant properties may extend its utility beyond cancer treatment to applications in food preservation and as a sunscreen agent, protecting against UV damage. 71 Recent studies also suggest that PG can alleviate oxidative stress in hepatoma cells by activating the nuclear factor erythroid 2-related factor 2 (Nrf2), which is integral to the cellular stress response.70,72
The combination of PG with γ-irradiation has shown a significant synergistic effect in inhibiting tumor growth. Radiation induces DNA damage, triggering various signaling pathways, primarily the intrinsic apoptotic pathway, which is crucial for preventing cancer cell proliferation. 73 Our findings indicate that PG treatment prior to radiation enhances apoptosis in Caco-2 cells, supporting the notion that natural compounds can sensitize cancer cells to ionizing radiation.74,75 The timing of PG administration relative to radiation exposure is critical, as enhanced effects were observed when PG was administered 24 hours before radiation treatment.
In summary, our findings underscore the potential of prodigiosin as a promising therapeutic agent in colorectal cancer treatment. By modulating inflammatory responses and influencing key signaling pathways related to apoptosis and proliferation, prodigiosin may enhance the efficacy of existing therapies and offer a novel approach to overcoming treatment resistance. Future studies should further elucidate the molecular mechanisms underlying these effects and explore the clinical applicability of prodigiosin in combination with conventional cancer therapies.
Future Directions
To enhance the translational relevance of prodigiosin’s (PG) radiosensitizing effects, several future research directions are proposed.
In Vivo Animal Studies
Conducting in vivo studies using appropriate animal models will be crucial to validate the radiosensitizing effects of PG in a physiological environment. These studies can help elucidate the pharmacokinetics, optimal dosing strategies, and potential side effects of PG, allowing us to assess its efficacy in a more complex biological context. Mechanistic Investigations: Further studies are needed to explore the underlying molecular mechanisms through which PG exerts its radiosensitizing effects. Utilizing techniques such as proteomics and genomic analyses could provide insights into the pathways affected by PG and its interactions with radiation therapy at the cellular level.
Combination Therapies
Investigating the potential synergy of PG with other chemotherapeutic agents and radiation therapies may yield promising results. Future studies should assess various combinations and treatment regimens to identify the most effective strategies for enhancing therapeutic outcomes in cancer treatment.
Clinical Trials
Once in vivo efficacy and safety are established, initiating clinical trials will be the next critical step. These trials should aim to evaluate the safety, optimal dosing, and effectiveness of PG as an adjunct to radiation therapy in cancer patients.
Patient-Derived Models
Utilizing patient-derived xenografts or organoids may provide a platform to evaluate the effectiveness of PG in a more personalized manner. This approach could facilitate the tailoring of treatment strategies based on specific tumor characteristics and patient responses.
By addressing these future directions, we can better understand the full therapeutic potential of prodigiosin and establish a solid foundation for its application in enhancing the effectiveness of cancer treatment.
Conclusion
Based on our preclinical findings, we have demonstrated that the apoptotic and survival pathways of colon cancer cells are significantly influenced by changes in inflammatory responses, specifically COX-2, PGE2, nitric oxide (NO), TNF-α, and the inflammasome NLRP3, as well as by the prooxidant/antioxidant balance. The imbalance between proliferation and apoptosis was further illustrated by an increase in MAPK and BCL-2 levels, alongside a decrease in PPAR-γ and the pro-apoptotic marker caspase-3 in Caco-2 colon cancer cells. The natural red pigment prodigiosin has shown considerable promise in sensitizing these cancer cells to ionizing radiation. Our results indicate that prodigiosin effectively induces apoptosis, decreases proliferation, and reduces key inflammatory markers, including COX-2, PGE2, and NO, while also disrupting the MAPK/TNF-α/NLRP3 signaling pathway. These findings highlight prodigiosin’s potential as a therapeutic strategy in cancer treatment. By enhancing radiosensitivity when administered alongside radiation exposure, prodigiosin could pave the way for more effective treatment strategies in the fight against colon cancer.
Footnotes
Acknowledgements
Not applicable.
Ethical Considerations
Not applicable.
Consent to Participate
Not applicable.
Consent for Publication
Not applicable.
Author Contributions
N.M.E designed the study. N.M.E and N.M.M carried out the practical work. L.M.A, A.S.S performed the data analysis and wrote the draft of the manuscript. N.M.E. critically read and revised the manuscript. All authors approved the manuscript.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
All data presented in this manuscript are reported in the manuscript.

