Abstract
Papillary thyroid carcinoma (PTC) is the main type of malignant tumor of the thyroid gland in adults. Huaier, a traditional Chinese medicinal agent, has demonstrated antitumor efficacy across diverse malignant neoplasms. However, the specific impact of huaier in PTC remains insufficiently elucidated. In this study, we explored the function of huaier in PTC. We treated a PTC cell line (TPC-1) with varying concentration gradients of huaier and we observed that huaier effectively inhibited the proliferation of TPC-1 cells with an IC50 of 2.538 mg/ml. Huaier also modulated the cell cycle of the TPC-1 cell line, and induced apoptosis in TPC-1. Huaier treatment inhibited migration and invasion of TPC-1 cells. Further studies showed that in TPC-1 cells, huaier treatment inhibited the expression of the MAPK and HSP27/STAT3/AKT signaling pathway. We subsequently demonstrated in a nude mouse model that huaier treatment inhibited the progression of PTC tumors. In summary, our data suggest that huaier can inhibit the proliferation, migration, and invasion of PTC cells in vitro and the progression of PTC tumors in vivo, and that this effect may be related to the MAPK and HSP27/STAT3/AKT signaling pathway; therefore, huaier extract is expected to be a new direction for the treatment of PTC.
Introduction
Papillary thyroid carcinoma (PTC) is the main type of malignant tumor of the thyroid gland in adults and is most common in women aged 30 to 45 years. 1 It is characterized by multiple early lymph node metastases, yet the 5-year survival rate for patients with PTC exceeds 95%. 2 However, a small percentage of PTCs evolve to an aggressive type that cannot be fully cured by available therapies such as surgery and radioactive iodine. 3 In addition, 5% of patients with PTC will experience cancer recurrence and 1.7% to 15% will develop distant metastases.4,5 For these patients, reoperation is risky, and drug therapies such as tyrosine kinase inhibitors produce a large number of adverse effects, including fatigue, weight loss, hypertension, and proteinuria. 6 These greatly affect the quality of life of these patients. An effective and low-toxicity drug is needed in the treatment of PTC.
Studies have demonstrated that the Mitogen-Activated Protein Kinase (MAPK) signaling pathway plays an important role in the development of PTC. 7 MAPK-mediated yroid tumorigenesis upregulates numerous oncogenic proteins, which promote cancer progression by influencing cell proliferation, growth, migration, and survival, as well as tumor angiogenesis, invasion, and metastasis through various mechanisms. 8 Heat shock protein 27 (Hsp27), a 27-kDa molecular chaperone protein, which is induced and activated by phosphorylation in response to diverse cytotoxic stressors, including hormones, chemotherapy, and radiation. 9 HSP27 is recognized as a significant molecular target for inhibiting tumor proliferation and inducing apoptosis in cancer cells. Previous studies have demonstrated that the HSP/STAT3/AKT signaling pathway is involved in apoptosis of cervical cancer cells. 10 Phosphorylation ofHSP27 is also catalyzed by the MAPK superfamily, including ERK.11,12
The oral MEK1/2 inhibitor trametinib has not been promoted due to significant toxicity. 13 Chinese medicine has become a direction for the development of new anti-tumor drugs in cancer treatment because of its low toxicity, low side effects, low cost, and multi-targeting. 14 Among them, we note that huaier can exert direct anti-tumor effects through the MAPK signaling pathway.15,16
Huaier, a medicinal fungus, grows on ancient acacia trees and has been part of traditional Chinese medicine for over 1600 years. Huaier granules have been approved by the State Food and Drug Administration (SFDA) of China for the treatment of lung and breast cancer patients. 17 There is growing evidence that huaier has a wide range of antitumor effects, and its extracts can effectively inhibit the proliferation and invasion of tumors such as liver, 18 stomach, 19 colon, 20 and ovarian cancers. 21 However, the effect of huaier on papillary thyroid cancer has not been studied.
In this article, we analyzed the effects of huaier aqueous extract on human PTC cells through in vitro and in vivo experiments and explored whether the ERK/HSP27/STAT3 signaling pathway was involved in this effect.
Materials and Methods
Preparation of Huaier Extractum
Huaier (Trametes robiniophila Murr) is a basidiomycete fungus that colonizes the trunks of locust trees and various other tree species. It falls under the Basidiomycota class within the Ascomycota phylum, and is mainly composed of proteoglycans. The extraction procedure was as follows: the dried fungus was ground to powder and boiled in water 3 times to remove the insoluble parts. 18 Sevag reagent removed the unconjugated protein, and dialysis removed other impurities. After ethanol precipitation and dehydrolysis, Huaier extract was obtained with approximately 1.5% to 1.8% proteoglycan. 22 In this study, Huaier extract was provided by Jiangsu Gaitianli Medicine Co., Ltd. Huaier extract was dissolved in DMEM medium (TransGen Cat# P10413) to obtain 100 mg/ml huaier. 23 The solution was sterilized with 0.22-µm filter and storage at −4°C.
Cell Culture
TPC-1 cells (Fuheng Cat# FH1039, RRID: CVCL_6298) were cultured in DMEM medium supplemented with 10% FBS (Gibco Cat# 20210108) and 1% penicillin/streptomycin at 37°C and 5% CO2.
CCK-8 Assay
TPC-1 cells were seeded in 96-well plates at 37°C at a density of 2 × 104/ml, respectively. The cell proliferation was tested at 24 hours using CCK-8 assay. To summarize, the cells were incubated with CCK-8 reagent (Beyotime Cat# C0037) at 37°C for 1 hours, followed by treatment with a series of concentration gradients of huaier for 24 hours. Six replicate wells were set up for each concentration, and the well without TPC-1 cells and only with culture medium was used as the blank well. We also set up a control group containing only TPC-1 cells and DMEM medium, without huaier. The absorbance of samples was measured at 450 nm using enzyme-labeled instrument (Thermo Fisher Scientific, Waltham, MA, USA). The survival rate of this experiment was calculated according to the following formula: Cell survival rate (%) = (absorbance of experimental well − absorbance of blank well)/(absorbance of control well − absorbance of blank well) × 100%. Subsequently, the survival of cells was assessed by CCK-8 assay to calculate the half maximal inhibitory concentration (IC50) of huaier on TPC-1 cells.
Effect of Huaier on Cell Morphology
Trypan blue staining assay (Beyotime Cat# C0011) to identify nonviable cells was performed as previously described. 24 TPC-1 cells were inoculated into 24-well plates at a density of 1 × 103/ml per well, 1 ml per well. Different concentrations of huaier aqueous extract (0, 1.56, and 6.25 mg/ml) were added to each well, and the cell growth was observed after 24 hours. The cells were stained with trypan blue at 24 and 48 hours after administration, observed under a microscope.
Identification of Apoptosis by Annexin V-FITC/PI Staining
TPC-1 cells were inoculated into 6-well plates at a density of 1 × 103/ml per well. When the cell confluence reached 80% to 90%, the medium was replaced with DMEM medium containing different concentrations of huaier aqueous extract (0, 1.56, and 6.25 mg/ml). The cells were stained using Annexin V-FITC (Yuanye Cat# J15N11H131088) and PI (Yuanye Cat# D11GR171092) staining solution (1:1) after 24 hours of co-culture. After staining, the cells were placed under red and blue excitation light to initially observe the apoptotic status. Next, TPC-1 cells were inoculated into 6-well plates at a density of 1 × 105/ml and co-cultured with the previously described concentrations of huaier aqueous extract for 24 hours. Cells were collected by centrifugation after digestion with 0.25% trypsin (NCM Biotech Cat# 2277233). Cells were labeled with Annexin V-FITC and PI according to the kit instructions (Beyotime Cat# C1052) and analyzed by flow cytometry.
Wound-Healing Assay
Wound-healing assay was conducted to assess cell migration of TPC-1 cells. TPC-1 cells were seeded in a 6-well plate at a density of 1 × 104/ml per well. Then, the cells were scratched with a 200-μL pipette tip. The cells were washed with phosphate buffered saline (PBS) for 3 times and co-cultured with different concentrations of huaier aqueous extract (0, 1.56, and 6.25 mg/ml). Images of the wound area are captured at initial (T = 0) and at 12 and 24 hours using a microscope. Wound areas are quantified using Image J software (RRID: SCR_003070) and the percentage of wound closure is calculated as [1 − (Area at Tx/Area at T0)] × 100%.
Transwell Invasion Assay
Transwell invasion assay was performed using Transwell insert system (Labselect Cat# 3460). Matrigel (Nova Cat# 0827065) was covered on the basolateral Transwell chambers. A cell suspension (0.2 ml, 5 × 104 cells/ml) was introduced into the upper chamber at 37°C with 5% CO2. DMEM medium containing 20% FBS was added into the lower chamber. After 24 hours of incubation, the invading cells on the bottom surface of the chamber were fixed with 4% paraformaldehyde (Labgic Cat# BL539A) and stained with 0.1% crystal violet. The number of invasive cells was counted and photographed under an inverted microscope (XD-202, Jiangnan Yongxin, Jiangsu, China). The data was analyzed using Image J software.
Western Blot Analysis
The TPC-1 cells were treated with huaier aqueous extract in different concentrations (0, 1.56, and 6.25 mg/ml) for 24 hours. The quantified protein samples were added to the gel wells, separated by SDS-PAGE (Beyotime Cat# P0012A), and transferred onto PVDF membranes. The membranes were incubated with primary antibody anti-ERK (Proteintech Cat# A4782, RRID: AB_2863347), anti-AKT (Proteintech Cat# A18675RRID: AB_2862411), anti-P38 MAPK (Proteintech Cat# A5049), anti-STAT3 (Proteintech Cat# A22434), and anti-HSP27 (Proteintech Cat# A11156, RRID: AB_2861510) or anti-β-actin (Proteintech Cat# AC038, RRID: AB_2863784) overnight at 4°C. After washed with PBST, the membranes were incubated with secondary antibody (Proteintech Cat# AS014, RRID: AB_2769854). The intensity of each band was quantified by Image J software and normalized to that of β-actin.
Animal Experiments
All animal protocols were approved by the Animal Care and Use Committee of Nanjing Drum Tower Hospital, Nanjing, China (Approval No. 2021AE01045). All animal experiments complied with the humane care of the animals during implementation to ensure maximum protection of their welfare and rights. Xenograft model was constructed by using the 4-5 weeks old male BALB/c nude mice (GemPharmatech Cat# SCXK2018-0008, RRID:IMSR_APB:4790; body weight, 17 ± 1 g) and was maintained at specific pathogen-free (SPF) conditions in the Laboratory Animal Center of China Pharmaceutical University, Nanjing, China. After mice were fed for a week, the TPC-1 human PTC cells were inoculated subcutaneously into the armpit of nude mice (5 × 106/ml, 125 μL). The longest diameter (a) and shortest diameter (b) of subcutaneous grafted tumors were measured by vernier calipers. The tumor volume was calculated as V = (a × b2)/2. When the tumor volume reached 50 mm3, the model was considered successful.
To investigate the effect of the Huaier extract, a total of 24 mice were evenly divided into 3 groups: the control group, the low-dose huaier group, and the high-dose huaier group. The control group was intragastric with normal saline, the low-dose huaier group was intragastric with 4 g/kg huaier extract, and the high-dose huaier group was intragastric with 8 g/kg huaier extract. All treatments were performed once a day for 21 days. During the experiment, the mice were provided with standard laboratory food and water, and the feeding conditions of the mice were consistent with the adaptation period. The survival quality of nude mice was observed. The body weight of mice was measured every 3 days, and the tumor volume was observed. Mice were sacrificed by cervical dislocation the day after the last dose. The tumor of each mouse was dissected under aseptic conditions. Tumor tissues were rapidly removed, rinsed, photographed and weighed. Tumors were paraffin-embedded after formaldehyde fixation. Sections were stained with HE and observed microscopically. Three regions of interest (ROI) were selected for each section by an experienced pathologist who did not know the experimental grouping. The criteria were located in the center of the tumor and the area size was 4 mm2 (2 mm × 2 mm) . The tumor cells were manually calculated within the ROI, and the tumor cell density = the number of tumor cells/the ROI area.
Statistical Analysis
Measurement data are presented as the mean ± SD. Data was normally distributed. GraphPad Prism 10.0 software (RRID:SCR_002798) was used for statistical analyses. The data between the experimental group and the control group were analyzed using t-test, with P < .05 being considered statistically significant.
Results
Huaier Inhibits Cell Viability and Disrupts Cell Morphology in TPC-1 Cells
To assess the impact of huaier extract on TPC-1 papillary thyroid cancer cells, we measured the survival of cells after 24 hours of dose-dependent effect of huaier aqueous extract by CCK-8 assay. Figure 1A shows that huaier significantly inhibited viability of TPC-1 in a dose-dependent manner. At a concentration of 0.10 mg/ml, the cell viability was 84.65%, and at the highest concentration of 12.50 mg/ml, the cell viability was 24.70%. The IC50 of huaier on growth of TPC-1 cells were 2.538 mg/ml.
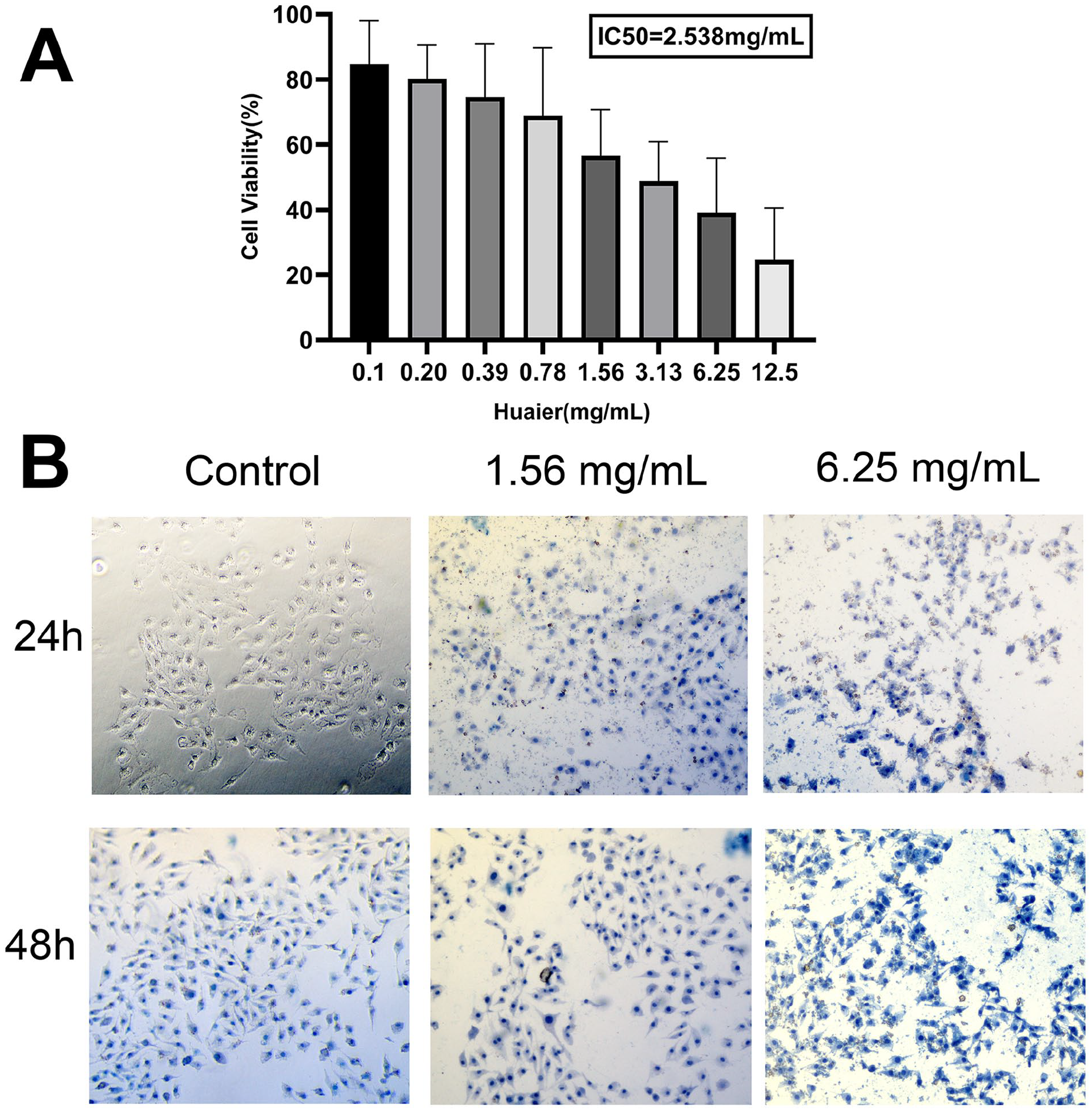
Huaier inhibits cell viability and disrupts cell morphology in TPC-1 cells. (A) The effect of huaier on the proliferation of TPC-1 cells. (B) The effect of huaier on the morphology of TPC-1 cells.
Then we observed the effect of huaier aqueous extract on the morphology of TPC-1 cells by trypan blue staining experiment. We chose 1.56 and 6.25 mg/ml as the experimental group and set 0 mg/ml as the control group. As shown in Figure 1B, the control group had intact cell membranes and almost no trypan blue staining after 24 hours. After 48 hours, the nuclei of a few cells had been stained, although the color was still light. With the increase of concentration and time, more TPC cells are nonviable, and the nucleus and cytoplasm were stained with trypan blue.
Huaier Regulates the Cell Cycle and Induces Apoptosis in TPC-1 Cells
To investigate the possible mechanism of huaier aqueous extract on the inhibitory effects of PTC cell proliferation, we investigated the effect of huaier aqueous extract on cell cycle regulation by flow cytometry in TPC-1 lines. As shown in Figure 2A and B, the cell population decreased at G1 but increased at G2 phases in TPC-1 cell lines treated with huaier compared with control group.

Huaier regulates the cell cycle and induces apoptosis in TPC-1 cells. (A and B) The effect of huaier on the cell circle of TPC-1 cells. (C) The representative images of PI-annexin V staining assay of TPC-1 cells. (D) The effect of huaier on the apoptosis rate of TPC-1 cells. Quadrant 1 (Q1) represents mechanically damaged/necrotic cells; Q2 corresponds to late-stage apoptotic cells; Q3 indicates early-stage apoptotic cells; and Q4 denotes viable cells.
We further explored whether this inhibitory effect was achieved through the induction of apoptosis. The PI-annexin V staining assay demonstrated that, as the concentration of huaier aqueous extract increased, the cell membrane became incomplete, resulting in the nucleus being stained by PI. At the concentration of 6.25 mg/ml, there was more cellular debris and most of the cells were in late apoptosis (Figure 2C). Then we used flow cytometry to calculate apoptosis rates. As shown in Figure 2D, the late-apoptosis or cell death rate (Q2) and the early-apoptosis rate (Q3) exhibit a dose-dependent increase.
Huaier Inhibits the Migratory and Invasive Abilities of TPC-1 Cells
The migratory ability of TPC-1 cells was determined by wound healing assay following treatment with huaier aqueous extract. Figure 3A shows representative images of the wound healing results. Figure 3B shows the wound healing rate. At 12 hours, the percentage of wound healing in the control group, 1.56 mg/ml huaier group and 6.25 mg/ml huaier group was 23.0%, 12.1% (P < .01), and 5.8% (P < .01), respectively, and at 24 hours, the percentage of wound healing was 30.7%, 16.32% (P < .01), and 7.3% (P < .01), respectively. Compared with the control group, the percentage of wound healing decreased in the experimental group, and the higher the concentration of huaier, the lower the percentage of wound healing. The invasive ability of TPC-1 cells was determined by transwell assay following treatment with huaier aqueous extract for 24 hours. Figure 3C shows representative images of the results of transwell assay. Figure 3D shows the relative ratio of the number of migrated cells. With the cell invasion rate of the control group set at 100%, the relative cell invasion rate was 46.4% (P < .01) for the 1.56 mg/ml huaier group and 24.3% (P < .01) for the 6.25 mg/ml huaier group. The increase in the concentration of huaier reduced the invasion rate of PTC cells.

Huaier inhibits the migratory and invasive abilities of TPC-1 cells. (A) A wound-healing experiment was used to detect the effects of different concentrations of huaier on TPC-1 cell migration. (B) Quantification of (A). (C) The Transwell migration assay was used to determine the effects of different concentrations of huaier on the migration of TPC-1 cells. (D) Quantification of (C). All data are expressed as the mean ± standard deviation (SD) of 3 experiments. **P < .01 compared with the untreated control.
Huaier Inhibits MAPK and HSP27/STAT3/AKT Signaling Pathways in TPC-1
To ascertain whether the MAPK and HSP27/STAT3/AKT signaling pathway is involved in the anti-oncogenic effects of huaier on PTC cells, we analyzed the expression levels of relevant proteins in the control and huaier-treated cells. As shown in Figure 4, compared to the control group, the expression of ERK1/2 decreased by 12.10% (P < .05) in the 1.56 mg/ml huaier group and by 26.59% (P < .01) in the 6.25 mg/ml huaier group. In contrast, the expression of P38 MAPK increased by 109.90% (P < .05) and 128.29% (P < .01), respectively, compared to the control group. Meanwhile, the expressions of HSP27, STAT3 and AKT in the experimental group containing huaier were down-regulated compared with the control group, and the higher the concentration of huaier, the lower the expression levels. These results indicated that huaier targets the MAPK and HSP27/STAT3/AKT signaling pathway in human PTC cells.
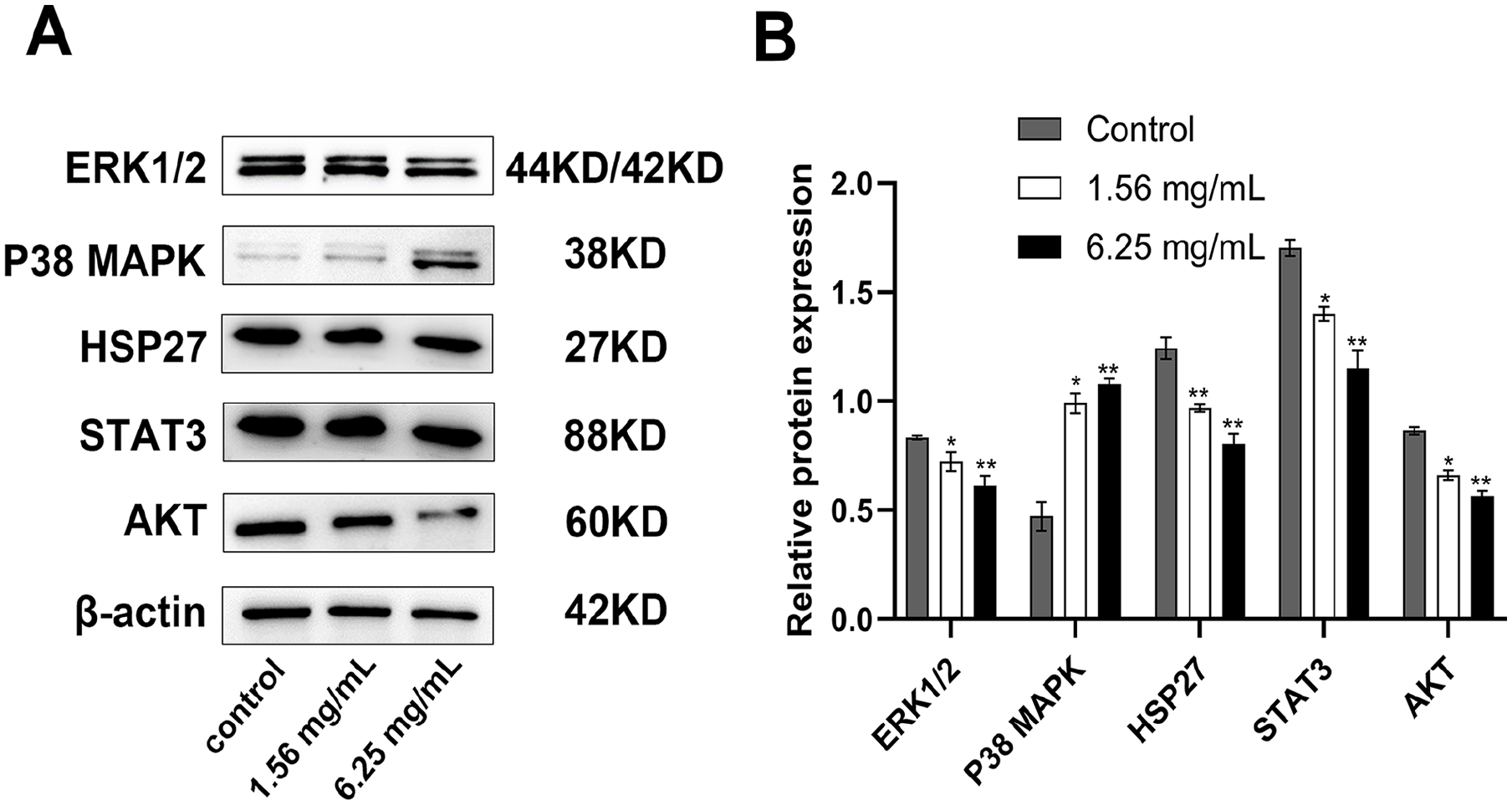
Huaier inhibits MAPK and HSP27/STAT3/AKT signaling pathways in TPC-1. (A and B) Immunoblot showing expression levels of ERK1/2, P38 MAPK, HSP27, STAT3, and AKT proteins in TPC-1 cells after 24 hours treatment with huaier. The grayscale ratio of the target protein/β-actin was calculated. The values are presented as the mean ± SD of 3 independent experiments; *P < .05 and **P < .01 compared with the untreated control.
Huaier Extract Inhibited PTC Growth in Xenograft Models
We further conducted the in vivo impacts of huaier extract on PTC cells. TPC-1 cells were injected subcutaneously in male BALB/c athymic nude mice to generate tumors. Huaier extract was administrated daily to the animals via gavage. The application of huaier extract demonstrated a suppressive effect on tumor growth, leading to reduced tumor weights and volumes (Figure 5A-D). Additionally, there was no significant disparity observed in the body weights of mice between the treated and control groups, suggesting that the administered dose of huaier in this study did not induce notable toxicity. (Figure 5E). To further investigate the effect of huaier on PTC, we performed hematoxylin and eosin staining of tumor sections to observe the tumor tissue.

Huaier extract inhibited PTC growth in xenograft models. (A) Establishment of macroscopic subcutaneous tumor models through tumor cell injection in different groups. (B-D) Compared with the control group, the mice in the Huaier group had smaller tumor volume and lighter tumor weight. (E) Tumor tissue in the experimental group showed tumor cell regression and a significant reduction in tumor cell density compared to the control group. (F) Tumor cell density in ROIs. For (C-F), the values are presented as the mean ± SD; *P < .05 and **P < .01 compared with the untreated control.
The average tumor cell density in ROIs in the control group was 68.3 cells/mm2, while in the 4 g/kg Huaier group, the average tumor cell density in the ROIs was 56.3 cells/mm2 (P < .05). In the 8 g/kg Huaier group, the average tumor cell density was 50.0 cells/mm2 (P < .01). Compared to the control group, tumor tissue in the experimental group showed tumor cell regression and a significant reduction in tumor cell density (Figure 5E and F).
Discussion
Traditional Chinese Medicine (TCM) has been employed in China for a long time to treat tumors. The increasing research and high-quality clinical trials in TCM have garnered growing international recognition for the utilization of TCM in the treatment of cancer. Many studies have shown that huaier extracts exert anti-tumor properties in a range of tumors.10,21,23,24 Currently, huaier granules is approved by the State Food and Drug Administration (SFDA) of China for the treatment of breast cancer, lung cancer, rectal cancer, liver cancer, stomach cancer, colon cancer, and so on. Recently, huaier aqueous extract has been proved to inhibit the development of pancreatic cancer, 25 prostate cancer, 26 neuroblastoma, and other tumors. 27 This suggests that the anti-tumor effect of huaier has a broad spectrum. However, there are no studies about huaier on PTC until now. Our study provides preliminary evidence that huaier aqueous extract inhibits the proliferation, invasion and metastasis of human papillary thyroid carcinoma TPC-1 cells in a dose-dependent manner in vitro, and also exerts antitumor effects in vivo.
The MAPK pathway plays a pivotal role in regulating cell proliferation, survival, and human tumorigenesis, with well-established significance in thyroid tumorigenesis, particularly in PTC. 28 The classical MAPK pathway involves extracellular mitogenic stimuli activating a receptor tyrosine kinase (RTK) in the cell membrane, subsequently signaling through RAS, RAF (depicted as BRAF-V600E), MEK, and ERK. ERK undergoes phosphorylation (P) and enters to the nucleus, where it upregulates tumor-promoting genes and downregulates tumor suppressor genes and thyroid iodide-handling genes. 29 Previous studies have demonstrated that the mechanism of anti-tumor effects of huaier focuses on direct anti-tumor effects and modulation of immunity to suppress tumors. 30 There is evidence that huaier can play a direct anti-tumor effect in breast cancer, 31 lung cancer, 16 and liver cancer cells 15 through MAPK signaling pathway.
HSP27 is identified as one of the downstream targets of ERK, and high levels of HSP27 enhance the malignant behavior of tumor cells, including over-proliferation, metastasis and less apoptosis. 12 In human papillary thyroid cancer cells, upregulation of HSP27 facilitates proliferation and confers resistance to apoptosis. 32 Recent studies have demonstrated the interaction between HSP27 and STAT3 as well as AKT, suggesting enhancement of cellular proliferation and tumorigenic in human cervical cancers. 10 In addition, previous studies have proved that STAT3/AKT signaling is related to the proliferation and invasion of papillary thyroid carcinoma. 33 Interestingly, Huaier was found to inhibit the proliferation and metastasis of lung cancer cells by down-regulating the STAT3-mediated signaling pathway, whereas the mechanism in thyroid cancer has not yet been investigated. 16 Therefore, our study also explored whether the anti-tumor mechanism of huaier is related to this pathway. The results showed that huaier regulated the expression of ERK, HSP27, STAT3 and AKT protein in papillary thyroid carcinoma cells, which tentatively suggests that the antitumor effect of huaier may be achieved by inhibiting ERK/HSP27/STAT3/AKT signaling pathway.
In the current study, huaier is often combined with chemotherapy or targeted drugs, which can reduce the dose of standard chemotherapy or targeted drugs, decrease their adverse effects, and enhance their anti-tumor effects. In the study of huaier combined with sorafenib for the treatment of hepatocellular carcinoma, the combination treatment regimen not only reduced the dosage of sorafenib, but also inhibited the growth of hepatocellular carcinoma more effectively. 34 The same is also true in the study of huaier combined with gemcitabine in the treatment of cholangiocarcinoma and pancreatic cancer.35,36 Huaier enhanced the efficacy of imatinib in the treatment of Ik6-positive Ph-positive acute lymphoblastic leukemia. 37 Huaier enhanced the therapeutic efficacy efficacy of paclitaxel in breast cancer 38 and the sensitivity of oxaliplatin in hepatocellular carcinoma. 39
We compared huaier with doxorubicin and lenvatinib, which are commonly used in the standard treatment of iodine refractory-PTC. The IC50 of huaier for TPC-1 was 2.538 mg/ml, while the IC50 of doxorubicin and lenvatinib for the same cell line was 0.143 and 0.191 mg/ml,40,41 respectively. It can be seen that huaier has a relatively weak inhibitory effect on thyroid cancer in vitro compared with the above drugs. Considering that previous studies have proved that huaier can be used as an adjunct to standard chemotherapy or targeted therapy for other malignant tumors to improve the sensitivity of these standard therapies and reduce adverse reactions, we speculated that huaier can also play a similar role in iodine-refractory thyroid cancer.
There are certain limitations in our study that ought to be considered. Firstly, our study was more cursory: we verified that huaier inhibited the proliferation, invasion and metastasis of TPC-1 cells in vitro, and in vivo only that huaier inhibited the proliferation of TPC-1 cells. Secondly, the inhibitory mechanism of Huaier on thyroid cancer needs to be further explored and verified.
In summary, our findings provide preliminary evidence supporting the proposition that huaier aqueous extract manifests antitumor effects on papillary thyroid cancer cells through inhibition of cell growth, induction of apoptosis and suppression of cell migration, and this effect may be realized through ERK/HSP27/STAT3/AKT pathway. Moreover, huaier has a favorable inhibitory effect on thyroid tumor growth in vivo. The present study may provide a promising therapeutic drug for thyroid cancer.
Footnotes
Acknowledgements
We acknowledge Prof. Liang Feng of China Pharmaceutical University for his guidance on the experimental design of this study.
Author Contributions
The manuscript presented here was carried out in collaboration between all the authors. Fa-Zhan Xu, Lu-Lu Zheng and Jia-Bo Qin conceived and designed the study. Yi-Xuan Li, Ke-Hao Chen and Ling-Hui Dai searched the literature, carried out experiments and analyzed the data. Fa-Zhan Xu and Lu-Lu Zheng wrote the manuscript. Jian-Feng Sang and Qiao-Ling Zhu revised the manuscript. All authors read and approved the final version of the manuscript.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by General Project of Social Development in Jiangsu Province, China (Grant No. BE2023657) and the National Natural Science Foundation of China (Grant No. 81701810).
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
