Abstract
Chemoresistance is still an important factor affecting the efficacy of treatment in colorectal cancer (CRC) patients. Hypoxia is related to poor prognosis and treatment resistance in cancer. Relevant studies have shown that a hypoxic microenvironment can promote the polarization of M2 macrophages and thus promote tumor development. Previous research has found that bufalin has a wide range of antitumor effects, but whether bufalin can reverse tumor resistance by improving the hypoxic tumor microenvironment is still unclear. In present research, it was found that high expression of SRC-3 in CRC cells under hypoxic conditions promoted the polarization of M2 and caused chemotherapy resistance, while bufalin, a monomeric drug used in Chinese medicine, reduced the level of SRC-3 and HIF-1α, thereby reversing chemoresistance. In addition, overexpression of SRC-3 reduced the hypoxia-mitigating effect of bufalin on CRC cells to promote the polarization of M2. Bufalin also inhibits the polarization of M2 caused by hypoxic CRC cells. Therefore, bufalin has the potential to become a new adjuvant therapy that can be further explored in future studies on its treatment of CRC.
Introduction
CRC is a typical digestive system cancer in global, and is the main causes of malignant tumors in terms of death rate, which has increased in recent years.1,2 The chemoresistance and immune evasion of CRC cells promote the progression of cancer and contribute to chemotherapy failure. 3 M2 macrophages in the tumor microenvironment (TME) are mainly believed to promote abnormal tumor proliferation, angiogenesis, metastasis and drug resistance, thus promoting the progression of malignant tumors.4 -6 More importantly, M2 macrophage polarization is a key marker of chemotherapy resistance in tumor cells and is closely related to CRC prognosis.7,8
As the continuous proliferation and high metabolic demand of tumor cells limit the supply of oxygen, hypoxia is a common microenvironmental feature and affects chemotherapy resistance, radiation resistance, invasiveness and angiogenesis.9 -11 In previous studies, tumor hypoxia was believed to be a key factor in the phenotypic control of tumor associated macrophages (TAMs).12,13 Hypoxic tissues secrete high concentrations of chemokines, such as HIF-1α and endothelin-2, which subsequently attract macrophages and cause tumor growth, tumor immunosuppression, and angiogenesis through multiple signaling pathways.14 -16 A hypoxic microenvironment can promote TAM polarization, especially M2 phenotype differentiation, thus reversing the inhibitory effect of the proinflammatory microenvironment on malignant behavior, and is directly associated with CRC prognosis. 17
Relevant literature indicates that traditional Chinese medicine (TCM) can affect tumor progression by improving the hypoxic microenvironment. 18 TCM can regulate the TME and affect the polarization of M2, thus inhibiting the progression of tumors.19 -21 Cinobufacini is a typical drug in treating malignant tumors, including CRC.22,23 Studies have found that its main active monomer bufalin (BU) can obviously inhibit tumor metastasis, drug resistance and angiogenesis.20,24 -26 Moreover, BU inhibits the metastasis of ovarian cancer cells by targeting the mTOR/HIF-1α pathway. 27 Whether BU can reverse CRC resistance by inhibiting hypoxia-mediated polarization of M2 macrophages remains unclear.
In this study, considering the hypoxic tumor microenvironment, we analyzed the mechanism by which BU inhibits the polarization of M2 caused by hypoxic cancer cells to reverse colorectal cancer drug resistance .
Materials and Methods
Cell Lines and Culture
Hypoxic cells were cultured at 37°C and 1% O2 (21% O2 was used for normoxia). THP-1 (RRID:CVCL_0006), HCT116 (RRID:CVCL_0291), and CT26 (RRID:CVCL_7254) cells were obtained from the Cell Bank of Type Culture Collection (Shanghai, China) and were cultured in RPMI-1640 with 10% FBS and 1% double antibiotic.THP-1 cells were transformed into M0 macrophages after PMA (200 ng/ml) treatment, and then M0 macrophages were treated with tumor cell supernatants under different conditions.
CCK-8 Assay
CRC cells were plated in 96-well plates and treated with various conditioned media (CM) types. After 48 hours, cell viability was evaluated based on CCK-8 assay (Dojindo, MD, Japan).
Quantitative RT-PCR (RT-qPCR)
Total RNA was extracted with TRIzol (Invitrogen), and an RT-qPCR kit (TaKaRa) was applied for reverse transcription to detect the relevant mRNA levels. The primer sequences were as follows:
H-TGF-β, 5′-GGTGGGTGAGAAGCTGACAAGA-3′ and 5′-CCTCCTGGGTGAGAAGCTGAG-3′;
H-IL10, 5′-TGCTGGGTGAGAAGCTGAAGA-3′ and 5′-GGTTGGGTGAGAAGCTGAGG-3′;
H-β-actin, 5′-ATTGCTGGGTGAGAAGCTGAA-3′ and 5′-GCTGTGGGTGAGAAGCTGATGGAA-3′;
H-SRC-3, 5′-GTGGGTGAGAAGCTGAGTGTTT-3′ and 5′-TTTTGGGTGAGAAGCTGATGAG-3′;
M-TGF-β, 5′-ACCGCAACAACGCCATCTATGAG-3′ and 5′-GGCACTGCTTCCCGAAT GTCTG-3′;
M-IL10, 5′-TCCCTGGGTGAGAAGCTGAAGAC-3′ and 5′- CACCTGCTCCACTGCCTTGC-3′;
M-β-actin, 5′-TATGCTCTCCCTCACGCCATCC-3′ and 5′-GTCACGCACGATTTCCCTCTCAG-3′;
M-SRC-3, 5′-CGACGATTTACGATTTCCCTCTC-3′ and 5′-ACAACGATTTCACGATTTCCCTCGTAG-3′.
ELISA Experiment
The Human and Mouse IL10 and TGF-β levels were detected via relevant ELISA Kit (BOSTER, China) according to the instructions.
Western Blotting (WB)
Cultured cells were lysed with RIPA and the protein level was detected based on BCA kit (Beyotime) . In the WB experiment, an enhanced fluorescence substrate (Bio-Rad) was applied. The spectral band was measured via the chemiluminescence detection system (Bio-Rad). The primary antibodies required for the experiments were specific for SRC-3 (2126T), HIF-1α (48085T), and β-actin (4967S) and bought from Cell Signaling.
Flow Cytometry Analysis
After the polarization, different groups of cells were collected and washed via PBS. CD11b is a marker of macrophages, and CD206 is a marker of M2-type macrophages. We evaluated the polarization status of M2-type macrophages by observing the changes of CD111b+CD206+. Subsequently, 1 × 106 cells were stained with anti-CD11b-PE (BD, 555388,USA) and CD206-FITC (BD, 551135,USA) at 4°C for half an hour. The results were analyzed by flow cytometry.
In Vivo Xenograft Model
Oxaliplatin (OXA) is one of the commonly used chemotherapy drugs in patients with colorectal cancer. In the present research, 5 × 106 hypoxic CRC cells (CT26) were inoculated subcutaneously into the right hind thigh of BALB/c mice (age, 5-6 weeks; weight, 18-20 g; Shanghai SLAC Laboratory Animal Co., Ltd.) to establish a colorectal tumor xenotransplantation model. The mice were placed in a pathogen-free environment at 22°C, with 30% relative humidity and a 12-hour light and dark cycle, giving them free access to food and water. To evaluate the anti-tumor effects of drugs in vivo, 1 week later, when mice had a tumor size of about 50 mm3, they were randomly divided into various groups (the Vehicle, OXA (3 mg/kg), BU (0.5 mg/kg), BU (0.5 mg/kg) + OXA (3 mg/kg) group), with 6 mice in each group (24 mice in all). The drugs were administered by intraperitoneal injection 5 days/week for 3 weeks. The tumor volumes were detected before and after treatment every 2 days. The estimated tumor volume (V) was determined based on formula V = W2 × L × 0.5, in which W refers to the largest tumor diameter in centimeters and L refers to the next largest tumor diameter. After the treatment, mice were sacrificed to excise the tumors, which wee weighed and photographed, and the tumor tissue and serum obtained were stored in a −80ºC refrigerator for subsequent experiments. All animal experiments were approved by the institutional animal care and use committee of Putuo Hospital, Shanghai University of Traditional Chinese Medicine.
Co-Culture and CM Acquisition
M0 macrophages were treated with serum-free supernatants of normoxic and hypoxic CRC cells for 4 days, and the changes of M2 polarization phenotype of macrophages were observed. This method was adopted in the subsequent co-culture experiments in this paper. Serum-free supernatant was collected after CRC cells were treated under different conditions, and then M0 macrophages were treated to observe the changes in M2 type polarization phenotype of macrophages.
CT26N-Mφ/CT26H-Mφ: M0 macrophages were treated with the supernatant of normoxic or hypoxic CRC cells.
CT26HSRC-3 KD-Mφ: M0 macrophages were treated with cell supernatant collected after SRC-3 knockdown by hypoxic CRC cells.
(CT26H + BU)-Mφ: After treating hypoxic CRC cells with BU for 48 hours, the cell supernatant was collected to treat M0 macrophages.
(CT26HSRC-3 OE + BU)-Mφ: After overexpression of SRC-3, hypoxic CRC cells were co-cultured with BU for 48 hours, and the supernatant was collected and treated with M0 macrophages.
CM(CT26H-Mφ): M0 macrophages were treated with hypoxic CRC cell supernatant for 4 days and the cell supernatant was collected as conditioned medium.
CM(CT26HSRC-3 KD-Mφ): M0 macrophages were treated with CT26HSRC-3 KD supernatant for 4 days and the cell supernatant was collected as conditioned medium.
CM(CT26H + BU-Mφ): After treating hypoxic CRC cells with BU for 48 hours, the cell supernatant was collected to treat M0 macrophages. The cell supernatant was then collected as a conditioned medium.
The conditioned medium for HCT116 cells was the same as the above method.
Cell Transfection
Hypoxic CRC cells were digested and cell suspensions were spread evenly in a 6-well plate in a cell incubator. After reaching 70% to 90% confluence, plasmid trans fection was performed. Briefly, 125 µl Opti-MEM (Invitrogen; Thermo Fisher Scientific, Inc.) and 7.5 µl Lipofectamine® 2000 reagent (Invitrogen; Thermo Fisher Scientific, Inc.) were added to an Eppendorf tube and mixed well in a biological safety cabinet. Firstly, 2.5 μg plasmid and 5 μl volume Lipofectamine2000 were gently mixed with the gun head, left for 5 minutes, then mixed with 125 μl opti medium, placed for 5 minutes, then mixed with 1 ml pure medium, and added into the 6-well plate holes. The cells were returned to the cell incubator and transfected 48 hours later.
Statistical Analysis
The GraphPad Prism 6.02 package was applied to carry out the statistical analyses. All data were expressed as mean ± SD. The statistical differences of different groups were analyzed based on t test. Spearman’s analysis and the Mann-Whitney test were applied to determine the significance level. *P < .05 was used as the statistical judgment criterion.
Results
Hypoxic CRC Cells Promote M2 Macrophage Polarization
Differentiation of monocytes into M2 is considered to be a marker of increased tumor chemotherapy resistance, and hypoxia is a key factor in the regulation of the M2 macrophage phenotype. We collected the supernatant of normoxic and hypoxic CRC cells to observe its effect on the polarization of M2. The results of RT-qPCR showed that the levels of M2 markers IL10 and TGF-β increased by more than two-fold, and the differences in ELISA results were more significant (Figure 1A-D, P < .05 or P < .01). In line with the above results, flow cytometry showed that the proportion of CD11b+CD206+ cells in M0 treated with the supernatant of hypoxic CRC cells was obviously enhanced compared with that in M0 treated with the supernatant of normoxic CRC cells (Figures 1E and S1A-B, P < .05 or P < .01). More interestingly, M2 polarization induced by hypoxic CRC cells showed greater resistance (Figure 1F and G, P < .01). In conclusion, these results suggest that hypoxic CRC cells can induce the polarization of M2 and promote chemotherapy resistance in CRC.

Hypoxic CRC cells promote M2 macrophage polarization. (A-D) RT-qPCR and ELISA were used to detect the expression of polarization markers in M2 macrophages after the treatment of M0 macrophages with the supernatant of normoxic or hypoxic CRC cells. (E) Flow cytometry was used to observe the expression of CD11b+CD206+ in macrophages treated with different types of media. (F and G) The effect of M2 macrophages with different degrees of polarization on the drug resistance of CRC cells was evaluated by CCK-8 assay. The results are presented as the means ± SDs.
Knockdown of SRC-3 in Hypoxic CRC Cells Inhibits M2 Macrophage Polarization
We further considered what might account for the differences in the above results. ELISA, RT-qPCR, and WB results showed increased expression of SRC-3 in hypoxic CRC cells than the normoxic cells (Figure 2A and B, P < .01). SRC-3 is associated with chemotherapy resistance and poor clinical prognosis.28,29 Therefore, SRC-3 is a key target in the regulation of M2 polarization. We knocked down SRC-3 in hypoxic CRC cells and validated it (Figure S2A, P < .05 or P < .01). Furthermore, knockdown of SRC-3 attenuated the promotion of M2 polarization by hypoxic CRC cells, thereby reducing their resistance to OXA (Figure 2C-I, P < .05 or P < .01). In total, these results indicated that SRC-3 derived from hypoxic CRC cells is a key factor in regulating the M2 polarization and promoting drug resistance.

Knockdown of SRC-3 in hypoxic CRC cells inhibits M2 macrophage polarization. (A and B) WB and RT-qPCR were used to observe the expression level of SRC-3 in normoxic and hypoxic CRC cells. (C-F) RT-qPCR and ELISA were used to detect the expression of polarization markers in M2 macrophages after SRC-3 knockdown in hypoxic CRC cells. (G) Flow cytometry was used to observe the expression of CD11b+CD206+ in macrophages after SRC-3 knockdown in hypoxic CRC cells. (H and I) The effect of M2 macrophages with different degrees of polarization on the drug resistance of CRC cells was evaluated by the CCK-8 method. The results are presented as the means ± SDs.
BU Can Inhibit the Polarization of M2 Caused by Hypoxic CRC Cells
BU is a main active monomer, and previous research found that BU has obvious antitumor effects. To study the effect of BU on M2 polarization induced by hypoxic CRC cells, low dose BU was selected for subsequent experiments, shown in Figure S3A-B. RT-qPCR test result showed that when M0 were stimulated with (HCT116H + BU)-CM or (CT26H + BU)-CM, the levels of IL10 and TGF-β were significantly reduced, and the differences in ELISA results were more significant (Figure 3A-D, P < .05 or P < .01). In line with the above results, the flow cytometry result indicated that when M0 were stimulated with (HCT116H + BU)-CM or (CT26H + BU)-CM, the proportion of CD11b+CD206+ cells was significantly reduced (Figure 3E, P < .05). Moreover, BU reversed OXA resistance in CRC cells by inhibiting M2 polarization induced by hypoxic CRC cells (Figure 3F and G, P < .05). In summary, these results suggest that BU reduces M2 polarization caused by hypoxic CRC cells.

BU can inhibit the polarization of M2 macrophages induced by hypoxic CRC cells. (A-D) RT-qPCR and ELISA were used to observe the expression levels of IL-10 and TGF-β after BU treatment of hypoxic CRC cells. (E) The effect of BU on the polarization of M2 macrophages induced by hypoxic CRC cells was observed by flow cytometry. (F and G) The effect of M2 macrophages with different degrees of polarization on the drug resistance of CRC cells was evaluated by the CCK-8 method. The results are presented as the means ± SDs.
BU Reduces HIF-1α Expression by Targeting SRC-3 in Hypoxic CRC Cells
The HIF-1α can obviously resist tumor cell death and is a key factor in regulating cell apoptosis and cell cycle.30,31 Some studies have found that HIF-1α level can be regulated by SRC-3, which can directly induce the transcription of HIF-1α. 32 Therefore, we speculated that BU could decrease HIF-1α level based on targeting SRC-3. The WB results suggested that BU reduced the protein level of SRC-3 and HIF-1α in hypoxic CRC cells (Figure 4A). Moreover, BU lowered the mRNA levels of SRC-3 and HIF-1α, as shown by RT-qPCR (Figure 4B and C, P < .05 or P < .01). WB and RT-qPCR result indicated that HIF-1α level was increased in hypoxic CRC cells after overexpression of SRC-3 (Figure 4D-F, P < .05 or P < .01). In addition, when SRC-3 was overexpressed in such CRC cells, the reduction in HIF-1α expression by BU was mitigated (Figure 4G and H, P < .05). In summary, BU can inhibit HIF-1α transcription by targeting SRC-3, thereby reducing HIF-1α level in hypoxic CRC cells.
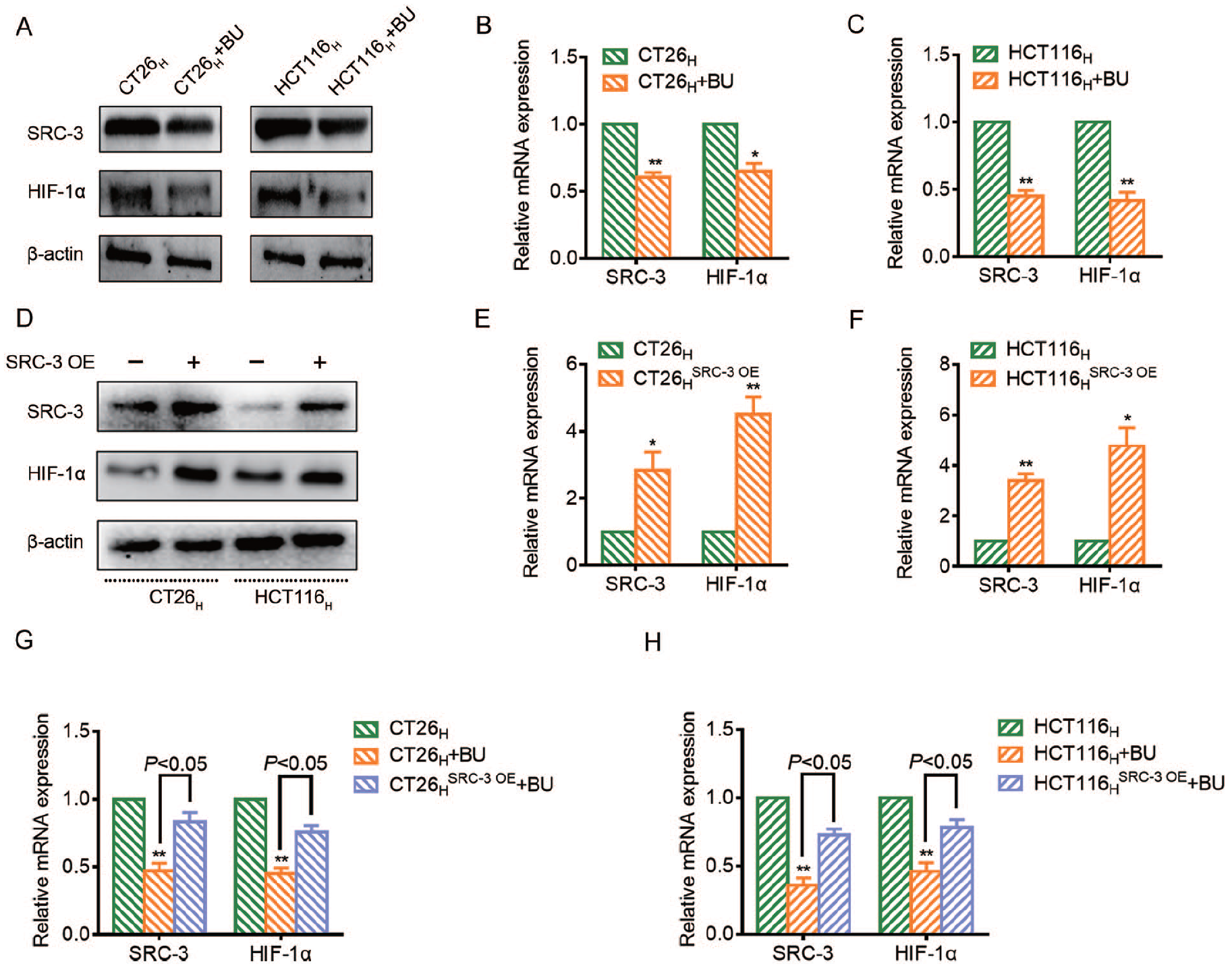
BU reduces HIF-1α expression by targeting SRC-3 in hypoxic CRC cells. (A-C) The effects of BU on the expression of SRC-3 and HIF-1α in hypoxic CRC cells were observed by WB and RT-qPCR. (D-F) The expression of HIF-1α in hypoxic CRC cells after overexpression of SRC-3 was observed by WB and RT-qPCR. (G and H) The effect of BU on HIF-1α expression in hypoxic CRC cells when SRC-3 was overexpressed was observed by RT-qPCR. The results are presented as the means ± SDs.
BU Inhibits M2 Polarization Caused by Hypoxic CRC Cells Through the SRC-3/HIF-1α Pathway
We subsequently investigated the function of SRC-3/HIF-1α in regulating M2 macrophages induced by hypoxic CRC cells by BU. The results showed that in hypoxic CRC cells overexpressing SRC-3, the inverse influence of BU on the polarization of M2 was weakened (Figure 5A-D P < .05 or P < .01). As in Figure 5, the ratio of CD11b+CD206+ was enhanced (Figure 5E and F, P < .05). Moreover, these results suggest that SRC-3 is a critical target in the regulation of BU-mediated M2 polarization. Furthermore, HIF-1 in hypoxic CRC cells was knocked down and verified (Figure S4A-B, P < .05 or P < .01). Knockdown of HIF-1α attenuated the promotion of M2 macrophage polarization by hypoxic CRC cells, as shown in the figure. RT-qPCR showed a decrease in mRNA levels of IL10 and TGF-β, which was also observed in ELISA experiments (Figure 5G and H, P < .05 or P < .01). Hence, the SRC-3/HIF-1α axis is involved in a key mechanism that BU inhibit M2 polarization caused by hypoxic CRC cells.

BU inhibits M2 macrophage polarization induced by hypoxic CRC cells through the SRC-3/HIF-1α pathway. (A and B) The levels of IL-10 and TGF-β in M0 macrophages treated with supernatant after co-culture of BU and HCT116HSRC-3 OE/CT26HSRC-3 OE were analyzed by RT-qPCR. (C and D) Evaluation of macrophage polarization. Different CM types were added to M0 macrophages for 4 days, and IL10 or TGF-β production was determined by ELISA. (E and F) Frequencies of CD11b+CD206+ cells among PBLs treated for 4 days with different CM types as evaluated by FACS (left); the graph (right) reflects the FACS data. (G and H). The effect of hypoxic CRC cells on the polarization of M2 macrophages was observed after HIF-1α knockdown. The results are presented as the means ± SDs.
BU Inhibits the Polarization of M2 Caused by Hypoxic CRC Cells In Vivo
To explore whether treatment of hypoxic CRC cells with BU can regulate the antitumor effects of the TME, this study constructed a xenograft mouse model of hypoxic CRC cells. This model contained every kind of cell in the TME. In line with the results of former studies, BU inhibited tumor growth in the hypoxic group (Figure 6A and B, P < .05 or P < .01). The results shown in Figure S5A indicate that toxicity was minimal for all 6 test treatments. Next, we evaluated whether BU inhibits hypoxic tumor growth by the regulation of the M2 macrophages polarization in vivo. According to the result, BU decreased the level of the M2 biomarkers, as shown in the experimental result of ELISA and RT-qPCR (Figure 6C-E, P < .05 or P < .01). We further found that BU inhibited the mRNA expression of HIF-1α in tumor tissues (Figure 6F, P < .05 or P < .01). In total, the above results indicated that BU inhibits hypoxic CRC-caused M2 polarization in vivo via the SRC-3/HIF-1α pathway.

BU inhibits the polarization of M2 macrophages induced by hypoxic CRC cells in vivo. (A) Xenograft tumor growth curves. (B) Photographs of tumors and graph of tumor weights. (C-E) RT-qPCR and ELISA for the serum levels of IL10 and TGF-β. (F) mRNA expression levels of SRC-3 and HIF-1α in tumor tissues of different groups were detected by RT-qPCR assay. The results are presented as the means ± SDs.
Discussion
CRC is a malignant tumor with high mortality rates, and it is mainly treated with surgery along with radiotherapy, chemotherapy, immunotherapy and traditional Chinese medicine. However, chemotherapy often leads to drug resistance in patients, which leads to a poor prognosis. Relevant studies have reported that M2 in the tumor microenvironment can promote tumor angiogenesis, metastasis, drug resistance and immunosuppression.33,34 In addition, due to the rapid proliferation of tumors, hypoxia is an important TME factor of solid tumors, interacting with chemotherapy resistance, radiation resistance, aggressiveness, and angiogenesis.35,36 Hypoxic microenvironments contribute to tumor plasticity and heterogeneity, promoting the development of more aggressive and metastatic phenotypes. In the present research, it was found that hypoxic CRC cells were more able to promote the polarization of M2 than normoxic CRC cells, thus promoting CRC chemotherapy resistance.
Targeting factors that influence the polarization of macrophages caused by chemotherapy may be a key approach to improve the efficacy of chemotherapy. SRC-3, also known as NCOA3 or AIB1, is 1 of the 3 homologous members in the p160 SRC family, and it plays a critical role in a variety of intracellular signaling pathways that are critical for cancer proliferation and metastasis. 37 We further found that SRC-3 was highly expressed in hypoxic CRC cells. The SRC-3 is a steroid receptor coactivator that is overexpressed in a variety of cancers and is defined as a true oncogene. 38 Studies have found that SRC-3 overexpression is related to chemotherapy resistance.39 -41 Overexpression of SRC-3 can be detected in CRC specimens, and the SRC-3 level is positively related to TNM staging. 42 In our study, we found that hypoxic CRC cells have a reduced effect on M2 macrophage polarization after SRC-3 knockdown, suggesting that SRC-3 plays a key role in the process of inducing M2 macrophage polarization in hypoxic CRC cells to promote chemotherapy resistance.
TCM can play an antitumor role by regulating the TME and can target HIF-1α to improve tumor efficacy. 43 The present research found that BU reduced the polarization of M2 caused by hypoxic CRC cells, thereby reversing CRC resistance. Furthermore, WB and RT-qPCR experiments suggested that BU reduced the level of SRC-3 and HIF-1α. Some studies have found that BU can prevent the proliferation of BC cells by directly targeting SRC-3 protein, 44 and former studies also confirmed that BU can target SRC-3. 45 Moreover, some scholars found that HIF-1α can promote the polarization of M2 macrophages. As a coactivator, SRC-3 is recruited into the promoter of a macrophage migration inhibitor and promotes its expression through interaction with the transcription factor HIF-1α, thus reducing chemical sensitivity. 46 SRC-3 has been shown to interact directly with HIF-1α via the N-terminal bHLH/PAS domain in vitro to enhance the transcriptional activity of HIF-1α in cancer cells. 32 Therefore, we conducted rescue experiments and found that after overexpression of SRC-3 in hypoxic CRC cells, BU was less effective in inhibiting the polarization of M2 caused by hypoxic CRC cells, and we further found that the effect of BU on SRC-3 and HIF-1α expression was decreased. In addition, BU reduced M2 polarization caused by hypoxic CRC cells and enhanced the antitumor effect of OXA. The results suggest that the SRC-3/HIF-1α pathway is a critical factor in BU-mediated inhibition of the M2 polarization caused by hypoxic CRC cells.
Conclusion
In summary, BU affects the activation of HIF-1α by inhibiting SRC-3 expression under hypoxic conditions, thereby regulating the M2 polarization induced by hypoxic CRC cells to reverse CRC resistance.
Supplemental Material
sj-tif-1-ict-10.1177_15347354251325806 – Supplemental material for Mechanism Study of Bufalin Reversal of Drug Resistance by Inhibiting Hypoxic Colon Cancer Cell-Induced Polarization of M2 Macrophages
Supplemental material, sj-tif-1-ict-10.1177_15347354251325806 for Mechanism Study of Bufalin Reversal of Drug Resistance by Inhibiting Hypoxic Colon Cancer Cell-Induced Polarization of M2 Macrophages by Haijing Wang, Linlin Jia, Hongjie Yu, Hui Tang, Huabowen Chi, Wei Zhang and Jinbao Chen in Integrative Cancer Therapies
Supplemental Material
sj-tif-2-ict-10.1177_15347354251325806 – Supplemental material for Mechanism Study of Bufalin Reversal of Drug Resistance by Inhibiting Hypoxic Colon Cancer Cell-Induced Polarization of M2 Macrophages
Supplemental material, sj-tif-2-ict-10.1177_15347354251325806 for Mechanism Study of Bufalin Reversal of Drug Resistance by Inhibiting Hypoxic Colon Cancer Cell-Induced Polarization of M2 Macrophages by Haijing Wang, Linlin Jia, Hongjie Yu, Hui Tang, Huabowen Chi, Wei Zhang and Jinbao Chen in Integrative Cancer Therapies
Supplemental Material
sj-tif-3-ict-10.1177_15347354251325806 – Supplemental material for Mechanism Study of Bufalin Reversal of Drug Resistance by Inhibiting Hypoxic Colon Cancer Cell-Induced Polarization of M2 Macrophages
Supplemental material, sj-tif-3-ict-10.1177_15347354251325806 for Mechanism Study of Bufalin Reversal of Drug Resistance by Inhibiting Hypoxic Colon Cancer Cell-Induced Polarization of M2 Macrophages by Haijing Wang, Linlin Jia, Hongjie Yu, Hui Tang, Huabowen Chi, Wei Zhang and Jinbao Chen in Integrative Cancer Therapies
Supplemental Material
sj-tif-4-ict-10.1177_15347354251325806 – Supplemental material for Mechanism Study of Bufalin Reversal of Drug Resistance by Inhibiting Hypoxic Colon Cancer Cell-Induced Polarization of M2 Macrophages
Supplemental material, sj-tif-4-ict-10.1177_15347354251325806 for Mechanism Study of Bufalin Reversal of Drug Resistance by Inhibiting Hypoxic Colon Cancer Cell-Induced Polarization of M2 Macrophages by Haijing Wang, Linlin Jia, Hongjie Yu, Hui Tang, Huabowen Chi, Wei Zhang and Jinbao Chen in Integrative Cancer Therapies
Supplemental Material
sj-tif-5-ict-10.1177_15347354251325806 – Supplemental material for Mechanism Study of Bufalin Reversal of Drug Resistance by Inhibiting Hypoxic Colon Cancer Cell-Induced Polarization of M2 Macrophages
Supplemental material, sj-tif-5-ict-10.1177_15347354251325806 for Mechanism Study of Bufalin Reversal of Drug Resistance by Inhibiting Hypoxic Colon Cancer Cell-Induced Polarization of M2 Macrophages by Haijing Wang, Linlin Jia, Hongjie Yu, Hui Tang, Huabowen Chi, Wei Zhang and Jinbao Chen in Integrative Cancer Therapies
Footnotes
Acknowledgements
Not applicable.
Author Contributions
JC, WZ and HC: Conceived or designed the study. LJ, HW and HY: Collected the data, analyzed and interpreted the data. LJ and HT: drafted the article. HW and LJ: Did part of experiments and aided in the construction of tumor model. JC, WZ and HC: Critically revised the article. All authors approved the final version to be published.
Data Availability
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This project was sponsored by the Shanghai Rising-Star Program (Sailing special Project, 22YF1441400), Science and Technology Innovation Project of Shanghai Putuo District Health System (ptkwws202409), Shanghai Sixth People’s Hospital jointly established scientific research fund (23-LY-03), Shanghai Famous Traditional Chinese Medicine Putuo Inheritance Studio Construction Project (ptzygzs2408) and TCM Scientific Research Project of Hubei Provincial Administration of Traditional Chinese Medicine Item (No. ZY2023M038).
Ethics Statement
All animal experiments were conducted in accordance with guidelines and protocol approved by the institutional animal care and use committee of Putuo Hospital, Shanghai University of Traditional Chinese Medicine, China.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
