Abstract
Concurrent chemoradiotherapy (CCRT) represents the established therapeutic modality for managing locally advanced non-small cell lung cancer (LA-NSCLC). However, its impact on improving the poor prognosis of LA-NSCLC patients is limited, and it can cause severe side effects. A 62-year-old Chinese female was diagnosed with unresectable stage IIIA lung adenocarcinoma. She refused CCRT. Enhanced computed tomography of the chest revealed a space-occupying lesion in her left pulmonary hilum, invading and encircling the pulmonary artery trunk. Due to the reported anti-tumor effects of basil, a stasis-removing Chinese herb, the patient received basil combined with cisplatin plus pemetrexed (CP) chemotherapy as first-line treatment. After 6 cycles of treatment, her condition achieved complete remission, and circulating tumor cells were reduced to zero. Regular follow-ups showed that the patient maintained progression-free survival for nearly 3 years. This case highlights the potential efficacy of basil combined with CP chemotherapy in treating LA-NSCLC. However, the curative effect of this regimen needs further validation through larger clinical trials.
Background
Lung cancer is the leading cause of cancer incidence and mortality worldwide, with non-small cell lung cancer (NSCLC) accounting for more than 80% of all lung cancer cases. 1 Approximately one-third of NSCLC patients are diagnosed at a locally advanced stage (LA-NSCLC), wherein the opportunity for curative resection is often forfeited. According to clinical practice guidelines such as those from the National Comprehensive Cancer Network (NCCN), concurrent chemoradiotherapy (CCRT) stands as the preferred therapeutic approach for unresectable LA-NSCLC patients.2,3 However, the prognosis for these patients remains unsatisfactory, with a 5-year overall survival rate of only 13% to 36%. 4 Presently, the integration of medicinal plants alongside radiotherapy or chemotherapy in cancer treatment has garnered increasing recognition. 5 The medicinal herb basil (Ocimum basilicum L.), has been shown to augment the sensitivity of tumor cells from various tissue origins to chemotherapy agents such as cisplatin. 6 Herein, we report the first case of complete remission in a locally advanced lung adenocarcinoma patient treated with a combination of cisplatin plus pemetrexed (CP) chemotherapy and basil.
Case Presentation
A 62-year-old woman was admitted to our hospital in January 2017 presenting with a persistent cough and shortness of breath lasting over 6 months. She had no history of smoking or alcohol consumption but had been managing hypertension for over a decade. Contrast-enhanced computed tomography (CT) of the chest (Figure 1A) revealed space-occupying lesions in the left pulmonary hilar region, indicative of central lung cancer, with consolidation and atelectasis of the left upper lobe, along with lesions encircling the left pulmonary artery trunk. Additional imaging modalities including head MR, abdominal CT, and bone emission computed tomography showed no evidence of metastasis. As per the eighth edition of the American Joint Committee on Cancer staging system, the patient was diagnosed with unresectable stage IIIa (cT4N0M0) lung cancer.
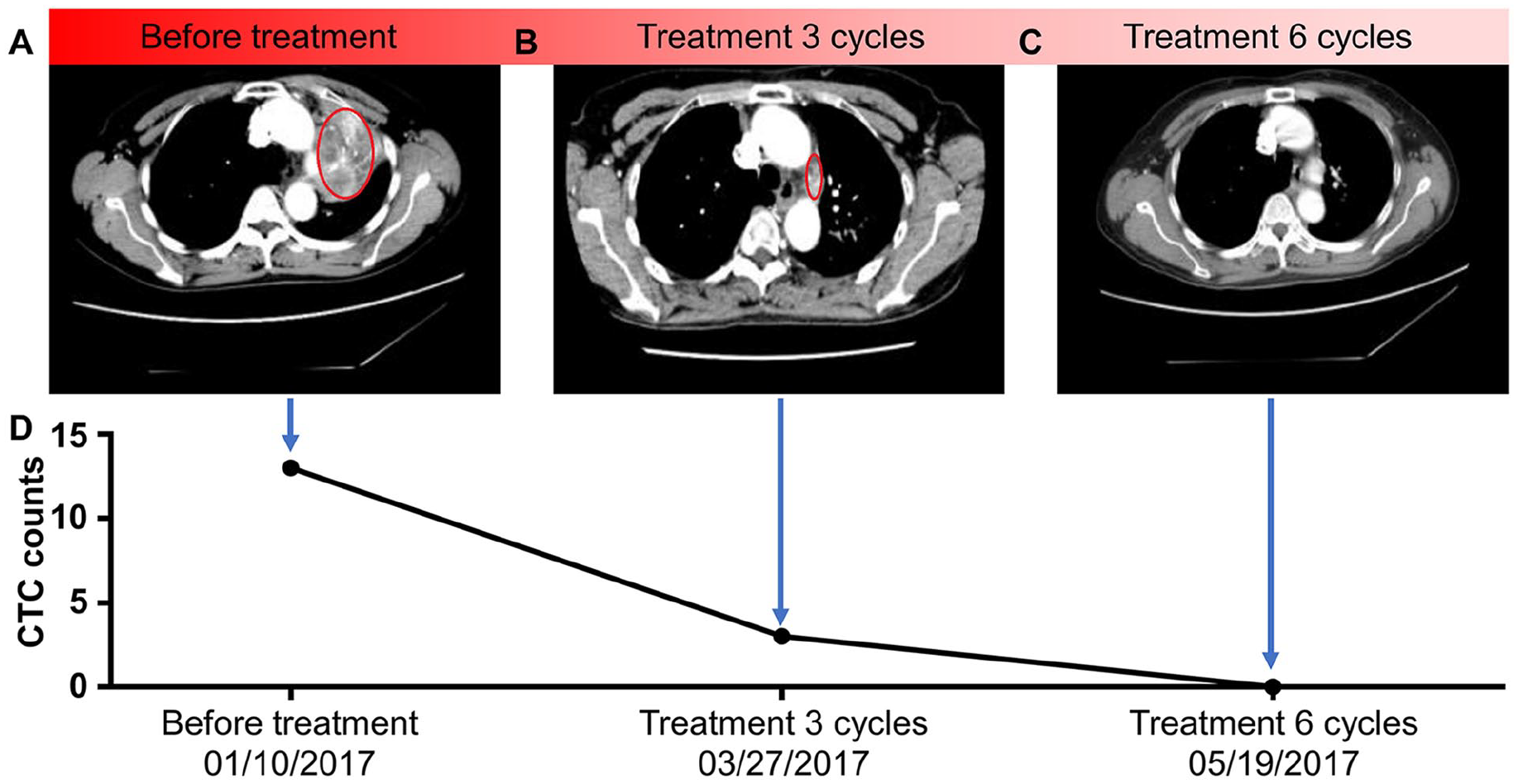
(A) Contrast-enhanced chest CT demonstrated a left hilar mass lesion, suggesting central lung cancer with left upper lobe consolidation and atelectasis, as well as lesions invading and surrounding the left pulmonary artery trunk. (B) The enhanced chest CT showed that 3 cycles of treatment led to a significant reduction in the left hilar mass, re-opening in the left upper lobe, and a curative effect classified as partial response (PR). (C) The enhanced chest CT revealed that the left hilar mass disappeared, and the curative effect reached complete remission (CR) following 6 cycles of treatment. (D) Number of circulating tumor cells (CTCs) in peripheral blood detected using the NanoVelcro CTC capture system before treatment, after 3 cycles, and after 6 cycles of treatment.
Biopsy of the left upper lung tumor confirmed lung adenocarcinoma (Figure 2A). Immunohistochemical staining (Figure 2B-D) revealed the following characteristics: TTF-1 (+), NapsinA (−), P63 (+) (minimal), CK5/6 (+) (sparse), CK7 (+), CD56 (−), Syn (−), and CgA (−). PD-L1 staining exhibited 3+ positivity in tumor cells (70% +), while immune cells showed minimal positivity (<1%), with no PD-1 expression. ARMS PCR testing indicated the following genetic profile: EGFR gene mutation testing for exons 18, 19, 20, and 21 was negative; ALK gene fusion was negative; and ROS1 gene fusion was positive. Genetic testing indicated intact mismatch repair (MMR) genes (MLH1 +, MSH2 +, MSH6 +, PMS2 +) and an AG genotype in ERCC1 (rs11615).

Biopsy of the left upper lung mass revealed lung adenocarcinoma by (A) HE staining (200×) and (B-D) immunohistochemical staining (TTF-1, 100×; CK7, 100×; PD-L1(SP142), 200×).
The patient declined CCRT and opted for 3 cycles of chemotherapy with CP regimen, comprising 500 mg/m2 (800 mg) of pemetrexed and 75 mg/m2 (100 mg) of cisplatin. Concurrently, she received a decoction of 30 g/day of the aboveground parts of basil (Ocimum basilicum L.), divided into 2 oral doses. Each cycle lasted for 3 weeks. Symptomatic relief was observed post-chemotherapy, with resolution of cough and dyspnea. Subsequent chest CT scans demonstrated a reduction in tumor size, categorized as a partial response (PR) according to Response Evaluation Criteria in Solid Tumors (RECIST) guidelines version 1.1 (Figure 1B). Circulating tumor cells (CTCs) in peripheral blood were quantified using a temperature-controlled NanoVelcro CTC capture system,7 -9 which revealed a significant decrease in CTC count from 13 to 3 per 2 ml of blood after 3 cycles of chemotherapy (Figure 1D). Following initial treatment, the patient underwent an additional 3 cycles of CP chemotherapy along with basil. Repeat chest CT scans showed complete resolution (CR) of the tumor after a total of 6 cycles of chemotherapy (Figure 1C). Concurrently, CTC count dropped further to 0 per 2 ml of blood (Figure 1D), and genetic analysis continued to show an AG genotype in ERCC1 (rs11615). Given the achieved CR with chemotherapy, the patient declined further chest radiotherapy. She was followed up regularly during the 3-year remission period. The patient continued to use basil regularly. Disease progression was noted in January 2020. Due to the outbreak of COVID-19, the patient did not come to our hospital for treatment. Follow-up reports indicate that the patient underwent additional chemotherapy and targeted therapy at another institution (details unspecified) and passed away in June 2020.
Discussion
Guidelines from the NCCN and others recommend CCRT as the standard treatment for patients with LA-NSCLC.2,3 However, most patients with unresectable LA-NSCLC experience disease progression after CCRT and may suffer from severe side effects. 10 In this report, the patient refused CCRT due to adverse effects and opted for a low-toxicity chemotherapy regimen, adopting the first-line chemotherapy protocol of CP for advanced lung adenocarcinoma. 11 Compared to other platinum-based doublet chemotherapies, CP demonstrates slightly better efficacy, lower toxicity, and can be combined with other treatments. 12
Traditional Chinese medicine (TCM) is an important adjuvant therapy in the treatment of NSCLC. Our research group has discovered that Chinese medicine Xiaoji decoction (XJD) and cisplatin exhibit synergistic effects. 13 When combined with Western medical treatments such as chemotherapy, TCM has been shown to enhance the anti-tumor efficacy of these treatments and reduce their adverse reactions. 14 A systematic analysis of 14 randomized controlled trials (1451 patients) by Wang et al 15 observed that, compared to chemotherapy alone, the addition of TCM significantly increased the objective response rate (ORR) and disease control rate (DCR). Similarly, Yang et al 16 noted significant improvements in clinical outcomes in the TCM treatment group in another meta-analysis of 20 randomized controlled trials (1669 patients), with a combined odds ratio (OR) of 2.99. These findings consistently indicate that the efficacy of TCM combined with chemotherapy is significantly better than chemotherapy alone. Additionally, numerous studies have explored the efficacy and safety of using TCM formulations containing astragalus in combination with platinum-based chemotherapy for treating cancers such as NSCLC and gastric cancer.17,18 These studies have demonstrated that astragalus can enhance the sensitivity to chemotherapeutic agents like cisplatin and reduce associated side effects. 17 Compared to platinum-based chemotherapy alone, the combination with astragalus-containing TCM has shown improved survival rates, ORR, and DCR. In this report, astragalus was not used, and only basil was administered. There are no known contraindications between the 2 herbs.
Basil is a medicinal plant that has been shown to be used as anti-cancer and anti-metastatic agents. The anti-cancer mechanisms of basil include inhibiting cancer cell migration and invasion through immunoregulation, angiogenesis, apoptosis, and cell adhesion.5,19 Several studies have reported that basil modulates tumor cell signaling and exhibits therapeutic effects in various xenograft tumor models, including the Walker-256 rat transplantable liver cancer model, and the Lewis lung carcinoma mouse model.20,21 Furthermore, basil has been granted a national invention patent (Patent No.: CN02135772.2) and has received approval from the National Medical Products Administration for clinical drug trials (Approval No.: 2006L03339). It is currently undergoing Phase II clinical trials. In this report, the patient achieved complete remission after 6 cycles of basil and CP chemotherapy, with CTC counts consistently declining to 0/2 ml, and progression-free survival was observed for up to 3 years. Additionally, we noted that the expression of ERCC1 before and after chemotherapy remained AG, suggesting that the patient retained sensitivity to platinum-based treatments. To the best of our knowledge, this is the first reported case of locally advanced lung adenocarcinoma showing complete response to a regimen of basil and CP chemotherapy.
Conclusion
This report presents the first case of a patient with locally advanced lung adenocarcinoma achieving a complete response to combined chemotherapy with CP and basil. The CTC count declined to 0/2 ml, indicating the effectiveness of this treatment approach. While this study opens new avenues for treating patients with locally advanced lung adenocarcinoma, further studies involving more patients are necessary to evaluate the general effectiveness of this combined chemotherapy regimen.
Footnotes
Availability of Data and Material
The data used to support the findings of this study are available from the corresponding author upon request.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by Guangdong Hospital of Traditional Chinese Medicine Special Fund for Scientific and Technological Research in Chinese Medicine (No: YN2018MJ01), Guangzhou Science and Technology Program (No: 202201010863) and Guangdong Provincial Natural Science Foundation - Key (No: 2018B030311061).
Ethics Approval and Permission for Publication
This study was approved by the Ethics Committee of Guangdong Provincial Hospital of Chinese Medicine (Z2017-159.2-01). Written consent was obtained from the patient’s legally authorized representative for publication of this case report.
