Abstract
Purpose:
Anti-epidermal growth factor receptor monoclonal antibody (anti-EGFR mAb) is the key drug for RAS/BRAF V600E wild-type metastatic colorectal cancer (mCRC). However, anti-EGFR mAb-induced skin fissures often affect a patient’s quality of life. Shiunko, a traditional Japanese topical herbal medicine, is used for burns and dermatitis and may potentially have wound-healing effects. Herein, we report cases of patients with mCRC who were treated with Shiunko for anti-EGFR mAb-induced skin fissure.
Methods:
We retrospectively reviewed consecutive patients with mCRC who received an anti-EGFR mAb-containing regimen and were treated with Shiunko twice a day for skin fissures at the National Cancer Center Hospital East between March 2022 and December 2022. Skin fissures were assessed at baseline and at every visit until 28 days after Shiunko initiation according to CTCAE v5.0.
Results:
Among the 11 patients, 5 patients were female; the median age was 61 (range, 43-79) years. The median treatment duration with anti-EGFR mAb before Shiunko initiation was 13.1 (range, 6-52) weeks. Skin moisturizer and topical steroids were applied for skin fissures in 11 and 5 patients, respectively. All patients had grade 2 skin fissures at baseline of Shiunko initiation. Two weeks after Shiunko initiation, complete recovery was noted in 4 patients and improvement to grade 1 was noted in 6 patients. There were no Shiunko-related adverse events. Ten patients continued anti-EGFR mAb treatment until disease progression, while 1 patient discontinued anti-EGFR mAb treatment due to severe eruptions.
Conclusion:
Shiunko could be a treatment option for anti-EGFR mAb-induced skin fissure. Further studies are warranted to investigate the efficacy and safety of Shiunko for anti-EGFR mAb-induced skin fissure.
Introduction
Anti-epidermal growth factor receptor (EGFR) monoclonal antibody (mAb) has anti-tumor activity against RAS/BRAF V600E wild-type metastatic colorectal cancer (mCRC).1 -4 Based on the results of a meta-analysis and a prospective phase 3 trial, clinical guidelines have recommended anti-EGFR mAb as first-line treatment for RAS/BRAF V600E wild-type mCRC, especially left-sided located tumors.5 -8 Additionally, the efficacy of anti-EGFR mAb as maintenance therapy has also been suggested. 9 Therefore, patients with RAS/BRAF V600E wild-type mCRC tend to receive long-term administration of anti-EGFR mAb. The management of skin toxicities, including early-onset acne-form rash or late-onset fissure and paronychia, is important for patients to continue long-term administration of anti-EGFR mAb. Supportive care, including antibiotics, moisturizers, and topical steroids, has been applied for acneiform rash10,11; however, appropriate supportive care for skin fissures has not been established. Skin fissures, which occur after 1 to 2 months or later from anti-EGFR mAb initiation, could affect a patient’s quality of life. Shiunko, a traditional Japanese herbal medicine, is composed of 5 active ingredients: lithospermum root, Japanese angelica root, yellow beeswax, sesame oil, and lard. 12 Shiunko is widely available and is used for burns, dermatitis, and hemorrhoid anal fissures; it may also have wound-healing effects. Most recently, Shiunko has showed efficacy in preventing radiation-induced dermatitis for breast cancer patients. 13 Herein, we present patients with mCRC who were treated with Shiunko for anti-EGFR mAb-induced skin fissure.
Methods
We retrospectively reviewed consecutive patients with mCRC (ICD-11: C18-C20) who received an anti-EGFR mAb-containing regimen and were treated with Shiunko for anti-EGFR mAb-induced skin fissure. The eligibility criteria were as follows: age, ≥18 years; Eastern Cooperative Oncology Group (ECOG) performance status (PS) score, 0 to 2; and treatment with Shiunko for grade 2 anti-EGFR mAb-induced skin fissures at the National Cancer Center Hospital East between March 2022 and December 2022. The exclusion criteria were as follows: previous malignancies; and prior treatment of Shiunko for anti-EGFR mAb-induced skin fissures. The study protocol was approved by the Institutional Review Board of the National Cancer Center Hospital East (2022-106). The requirement for informed consent was waived due to the retrospective study design, with opt-out opportunity provided on the institutional website. The patients received panitumumab (6 mg/kg every 2 weeks) or cetuximab (initial 400 mg/m2 and subsequent 250 mg/m2 weekly) in combination with 5-FU, leucovorin, and oxaliplatin (FOLFOX) or irinotecan. Shiunko (TSUMURA Shiunko Ointment®, TSUMURA Inc., Shiunko of 100 g consists of 27.0 g of JP White Beeswax, 1.8 g of JP Lard and 71.2 g of the oil extract prepared by mixing the ingredients in the following ratio (JP Sesame Oil: JP Japanese Angelica Root: JP Lithospermum Root = 100.0 g: 10.0 g: 10.0 g)) was applied twice a day to the skin fissure for patients with grade 2 anti-EGFR mAb-induced skin fissures. Skin fissure and Shiunko-related adverse events were assessed according to CTCAE v5.0 and a previous study protocol about xerosis 14 at baseline and at every visit until 28 days after Shiunko initiation by each investigator. We selected the CTCAE term for skin fissure “Skin and subcutaneous tissue disorders – Other, specify” (CTCAE v5.0 MedDRA v20.1 Code; 10040785).
Results
Table 1 presents the baseline characteristics of the 11 patients who were treated with topical Shiunko for grade 2 anti-EGFR mAb-induced skin fissures. All patients were diagnosed with RAS/BRAF V600E wild-type mCRC without previous history of other malignancies. Panitumumab and cetuximab were administered in 10 patients and one patient, respectively. Four patients and 5 patients had received surgery and prior chemotherapy, respectively. None of the patients had received radiotherapy. At the baseline of topical Shiunko initiation, the median duration of anti-EGFR mAb treatment was 13.1 (range, 6-52) weeks; dose interruption and reduction of anti-EGFR mAb was required in 2 and 1 patients due to panitumumab-induced skin fissure, respectively. Topical medications applied prior to Shiunko were skin moisturizers (n = 11), topical steroids (n = 5), and hydrocolloid dressings (n = 3) (Table 1, Figure 1). Skin moisturizers (n = 11) and topical steroids (n = 2) were applied concomitant with Shiunko. After Shiunko initiation, the severity of skin fissures improved in all patients within 4 weeks. At 2 weeks after Shiunko initiation, skin fissures resolved in 4 patients (grade 0), improved to grade 1 in 6 patients, and were maintained in one patient. Furthermore, among the 8 patients who were evaluated 4 weeks after Shiunko initiation, skin fissures improved to grades 0 and 1 in 5 and 3 patients, respectively (Supplemental Figure 1). No Shiunko-related adverse events were observed. Below are the representative cases (Table 1, Supplemental Figure 1).
Patient Characteristics.

Abbreviations: ECOG PS, Eastern Cooperative Oncology Group performance status; EGFR, epidermal growth factor receptor; CTCAE, Common Terminology Criteria for Adverse Events.
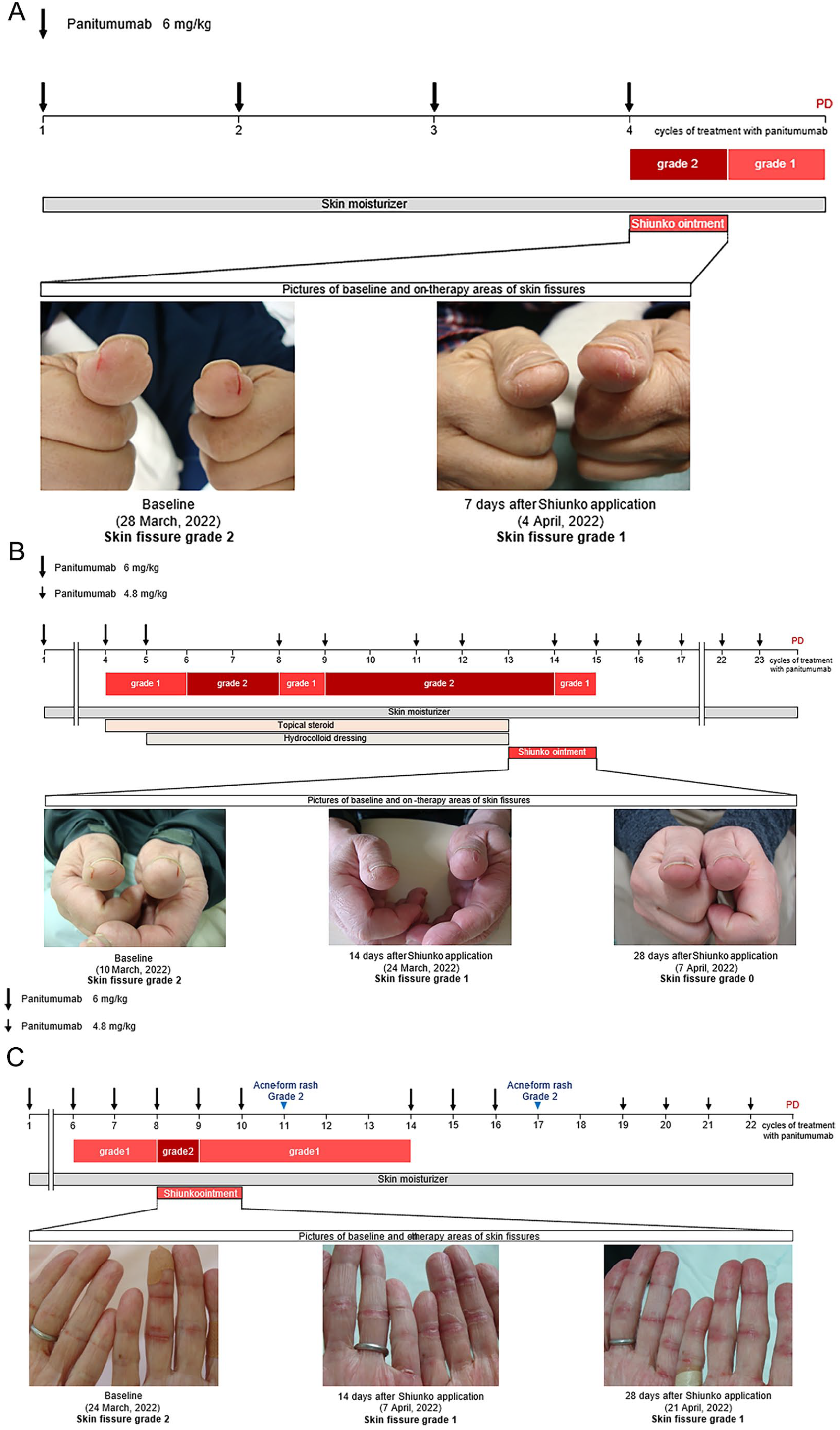
Progress chart of patients treated with Shiunko: (A) Case 1, (B) Case 2, and (C) Case 3.
Case 1
A 45-year-old man who was diagnosed with rectal cancer with peritoneal metastasis received chemotherapy with panitumumab plus irinotecan as third-line treatment. He then developed skin fissures with bleeding and pain on his thumb, and topical Shiunko was started. Seven days after Shiunko application, skin fissures improved, and bleeding and pain resolved (Figure 1A).
Case 2
A 66-year-old man who was diagnosed with sigmoid colon cancer with liver metastasis received chemotherapy with panitumumab plus FOLFOX as first-line treatment. He developed skin fissures with bleeding and pain on his thumb that was not relieved by dose reduction of panitumumab (4.8 mg/kg); hence, topical Shiunko was initiated. Signs of wound-healing were observed 7 days after initiation, and bleeding and pain resolved 14 days after (Figure 1B).
Case 3
A 68-year-old woman who was diagnosed with sigmoid colon cancer with lymph node and bone metastases received chemotherapy with panitumumab plus FOLFOX as first-line treatment. She developed skin fissures with bleeding and pain on her finger. Pain ultimately resolved 14 days after Shiunko initiation (Figure 1C).
Discussion
To our knowledge, this is the first report on the case regarding the potential efficacy and safety of topical Shiunko for anti-EGFR mAb-induced skin fissures. Currently, there are no randomized clinical data regarding the prevention or treatment of anti-EGFR mAb-induced skin fissures. Treatments, including urea cream, salicylic acid, zinc oxide, and medium- to high-potency steroid creams, are often recommended based on case studies and expert opinion, 15 but data on their efficacy for anti-EGFR mAb-induced skin fissures are limited. The most important components of the lithospermum root in Shiunko are derivatives of shikonin such as deoxyshikonin, acethylshikonin, and isobutylshikonin. 16 There are several mechanisms responsible for the wound-healing effects of shikonin, including antioxidant and anti-inflammatory activity caused via the reduction of reactive oxygen species that induce secondary damage in wounds, 17 keratinocyte proliferation and epithelial wound closure activity caused by upregulation of transforming growth factor-β, and collagen secretion.18 -20 In fact, application of Shiunko showed a significant wound-healing effect 17 compared to Japanese angelica root or prednisolone in a mouse model. 21 Furthermore, shikonin showed antibacterial activity against methicillin-resistant Staphylococcus aureus, which is the most common causative agent of skin infections. 22 Based on these findings, topical Shiunko therapy could be a treatment option for anti-EGFR mAb-induced skin fissures, leading to decreased pain in patients receiving anti-EGFR mAb for RAS/BRAF wild-type mCRC.
The major limitation of our study was its sample size at a single institution retrospective design without statistical analysis. We were not able to examine the impact of risk factors for skin fissure, including combined chemotherapy, age, history of eczema, and prior treatment with cytotoxic drugs,15,23 and to assess the frequency of grade 2 skin fissure in all patients who received anti-EGFR mAb due to nature of the retrospective study. Considering these limitations, further studies are warranted to investigate the efficacy and safety of Shiunko for anti-EGFR mAb-induced skin fissures.
Supplemental Material
sj-pptx-1-ict-10.1177_15347354231225962 – Supplemental material for Potential Efficacy of Shiunko for Anti-Epidermal Growth Factor Receptor (EGFR) Monoclonal Antibody-Induced Skin Fissure: A Single Institutional Case Series
Supplemental material, sj-pptx-1-ict-10.1177_15347354231225962 for Potential Efficacy of Shiunko for Anti-Epidermal Growth Factor Receptor (EGFR) Monoclonal Antibody-Induced Skin Fissure: A Single Institutional Case Series by Mashiro Okunaka, Daisuke Kotani, Saori Mishima, Maho Nakamura, Akihito Kawazoe, Hideaki Bando, Takayuki Yoshino and Kohei Shitara in Integrative Cancer Therapies
Footnotes
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: MO has nothing to disclose.
DK reports honoraria from Takeda, Chugai, Lilly, MSD, Ono, Eisai, Taiho, Bristol Myers Squibb, Daiichi-Sankyo, Pfizer, Merckbiopharma, and Sysmex: research funding from Ono, MSD, Novartis, Servier, Janssen, IQVIA, Syneoshealth, CIMIC, and Cimicshiftzero.
SM has reports honoraria from Merck Seronobiopharma: research funding from Roche Diagnostics. MN has nothing to disclose. AK reports personal fees from Daiichi Sankyo, Lilly, Ono, Taiho, BristolMyers Squibb, Merck Serono Biopharma, Sumitomo Dainippon, zymeworksand AstraZeneca outside the submitted work. HB reports honoraria from Bayer, Ono Pharmaceutical, Eli Lilly Japan, and Taiho pharmaceutical: research funding from Ono Pharmaceutical. TY reports honoraria from Bayer, Chugai, Merck Biopharma, MSD, Ono Pharmaceutical, and Takeda: research funding from Taiho, Ono Pharmaceutical, Chugai, Amgen, MSD, Daiichi-Sankyo, Eisai, FALCO biosystems, Genomedia, Molecular Health, Nippon Boehringer Ingelheim, Pfizer, Roche Diagnostics, Sysmex, and Sanofi. KS reports receiving personal fees for advisory roles from Lilly, Bristol Myers Squibb, Takeda, Pfizer, Ono Pharmaceutical, Merck Pharmaceutical, Taiho Pharmaceutical, Astellas, Novartis, AbbVie, GlaxoSmithKline, Daiichi Sankyo, Amgen, Boehringer Ingelheim, Guardant Health Japan,and Janssen; receiving honoraria (lecture fee) from Takeda, Bristol-Myers Squibb and Janssen; and receiving research funding from Astellas, Ono Pharmaceutical, Daiichi Sankyo, Taiho Pharmaceutical, Chugai, Merck Pharmaceutical, Medi Science, Eisai and Amgen, outside the submitted work.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
