Abstract
Tumor resistance is typically blamed for the failure of radiotherapy and chemotherapy to treat cancer in clinic patients. To improve the cytotoxicity of tumor cells using radiation in conjunction with specific tumor-selective cytotoxic drugs is crucial. Pomegranate has received overwhelmingly positive feedback as a highly nutritious food for enhancing health and treating a variety of ailments. In the present study, we aimed to examine the effects as well as mechanism of action of pomegranate peel extract (PPE) and/or γ-radiation (6-Gy) on hepatocellular carcinoma (HCC) cell lines HepG2. The findings of this study showed that PPE treatment of HepG2 cells considerably slowed the proliferation of cancer cells, and its combination with γ-irradiation potentiated this action. As a key player in tumor proliferation, and inflammatory cascade induction, the down-regulation of STAT3 following treatment of irradiated and non-irradiated HepG2 cells with PPE as recorded in the present work resulted in reduction of tumor growth, via modulating inflammatory response manifested by (down-regulation of TLR4 expression and NFKB level), suppressing survival markers expressed by reduction of JAK, NOTCH1, β-catenin, SOCS3, and enhancing apoptosis (induction of tumor PPAR-γ and caspase-3) followed by changes in redox tone (expressed by increase in Nrf-2, SOD and catalase activities, and decrease in MDA concentration). In conclusion, PPE might possess a considerable therapeutic potential against HCC in addition to its capability to enhance response of HepG2 cells to gamma radiation,
Introduction
With a projected incidence of more than 1 million cases by 2025, liver cancer is considered a global health challenge. The major type of liver cancer is hepatocellular carcinoma (HCC), accounting for 90% of all occurrences. The principal risk factors for development of HCC include hepatitis B and C virus infection, non-alcoholic steatohepatitis, and diabetes mellitus.1-3 Hepatectomy, liver transplantation, and locoregional therapies including radiofrequency ablation, trans-arterial chemoembolization, and radiation therapy are curative treatments for individuals with early- or intermediate-stage HCC. For patients with advanced HCC, systemic therapies are the sole options for improving survival.3-6
The ability to use ionizing radiation locally, preventing systemic harm, is one advantage of radiotherapy. Ionizing radiation does not, like chemotherapeutic drugs, completely kill all target cells, which can have harmful side effects on the tissue around it. In addition, ionizing radiation has a weak effect on a significant portion of human malignant tumor cells. 7 Accordingly, by using radiosensitizing drugs, the effect of radiation on tumor tissues can be maximized to cause more tumor damage than would be predicted from radiation alone, reducing the need for high radiation doses while simultaneously sparing healthy tissues. 8 The majority of synthetic radiosensitizers that are often utilized are typically hazardous, non-selective, and non-tumor specific. Plant foods have attracted attention as a potential replacement for the very lethal radiosensitizers because of their minimal toxicity. 9
Punica granatum (pomegranate) has piqued researchers’ interest due to its significant potential for use in medicine and the food sector. These chemicals are found in large quantities not only found in the edible section of the fruit, but also in the non-edible fractions (i.e., leaves, buds, bark, seeds, peel, and blossoms). The peels, for example, account for nearly half of the entire fruit weight and are sometimes discarded as trash without being valorized. Prior to recent studies, the peel was viewed as a waste, but it is now recognized as a rich source of bioactive phytochemicals with potential biological functions by acting as antioxidants, physiological agents, antimicrobial agents, and stimulating agents for the immune system. Phytochemicals have the potential to affect diseases such as cancer, stroke, metabolic syndromes, and others. The commonly known phytochemicals include flavonoids such as quercetin, cardiac glycosides, alkaloids, phytate, hemagglutinin, saponins, tannins, oxalate, polysaccharides, and phenols.10-14 The peels recycling not only solves the bio-waste issues but also offers a source of beneficial compounds with a number of health advantages. 15 Pomegranate peel extract has the largest concentration of phytochemicals, primarily phenolic components like ellagitannins, tannins, and anthocyanins including ellagic acid and punicalagin. 14 These bioactive compounds have demonstrated antibacterial action against a variety of pathogens as well as antiproliferative activities in various cell lines.16-18
As an advanced cancer therapeutic convention, this study was commenced to gauge the antitumor activity of pomegranate peel extract together with γ-radiation against hepatocellular carcinoma in HepG2 cells in vitro. A diversity of molecular markers was analyzed in order to explore the primary mechanisms by which PPE repressed the growth of liver cancer or/and enhanced the radiosensitivity of cancer cells to γ-irradiation.
Materials and Methods
Chemicals
Pomegranate powder extracted from Punica granatum peels was purchased from Wuhan HengHeDa Pharm Co., Ltd., Shanghai, China (CAS: 476-66-4) and prepared using sterile saline.
Cell Lines and Cell Culture
Human liver cancer cells (HepG2) used in this investigation were purchased from the Cell Culture Department, VACSERA (Cairo, Egypt). Cancer cells HepG2 were cultured in RPMI media in addition to 10% fetal bovine serum (ThermoFisher Scientific, USA), supplemented with penicillin (100 U/mL) and streptomycin (100 mg/mL) and incubated at 37°C in a humidified environment of 5% CO2 according to distributor instructions.
Radiation Facility
Using a 137Cs source, cells were exposed to γ-rays (Gamma-cell-40 Exactor; NCRRT, AEA, Cairo, Egypt). There was a 0.012 Gy/s dosage rate. All of the experiments included dosimetry to guarantee dose uniformity, and a Fricke reference standard dosimeter was utilized to deliver dose rates.
Cell Viability/Morphology Assay
Cell viability was assessed using the 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide test (MTT; Sigma). HepG2 cells were seeded in 96-well plates with 8000 and 6000 cells per well, respectively, to achieve this goal. Different quantities of pomegranate peel extract were dissolved in culture media and applied to the cells for 24 hours; suspension in control media served as the treatment. After 24 hours of treatments, 20 L of MTT (5 mg/mL) was added to each well, and the plates were then let to sit at 37°C in the dark for 3 to 4 hours. To dissolve formazan crystals, 150 L of DMSO was added to the media in each well. Finally, the absorbance was measured using a microplate reader operating at a 545 nm wavelength. For each tested concentration, experiments were carried out in triplicate. 8 Using SPSS one-way ANOVA, the half maximum inhibitory concentration (IC50) was determined (IBM Inc., Chicago, IL, USA). Graph-Pad Prism version 8.0 was used to create the graphs (Graph-Pad Prism Inc., San Diego, CA, USA). Using a phase-contrast inverted microscope equipped with a digital camera, cell morphology was captured (Nikon, Japan). There were 3 copies of each study.
Study Groups
Four HepG2 groups were established in order to evaluate the anti-proliferative and radio-sensitizing effectiveness of PPE: (1) HepG2 Cells without treatment (vehicle treated; control); (2) Cells treated with PPE alone; (3) Cells exposed to a single dose of irradiation (6 Gy) the radiation exposure period was 18 minutes (3 minutes/gray); and (4) Cells treated with PPE 24 hours prior to irradiation (PPE + 6 Gy).
Detection of Proliferation and Apoptotic Parameters
NF-κB (ab278120, Abcam, UK), MAPK (MBS009637, MyBioSource, Inc., USA), STAT 3 (orb562050, Biorbyt, USA), NOTCH-1 (MBS9501982, MyBioSource, Inc., USA), Beta Catenin (ab275100, Abcam, UK), SOCS 3 (OKCD07580, Aviva Systems Biology, USA), Nrf-2 (MBS744368, MyBioSource, Inc., USA), PPARɣ (MBS263089, MyBioSource, Inc., USA), Caspase-3 (MBS260710, MyBioSource, Inc., USA). Levels were measured using specific ELISA kits as directed by the product’s manufacturer.
Western blot Analysis
Immuno-blotting was conducted using primary antibodies to JAK-1 (ab125051), TLR-4 (ab13556), and β-actin (ab8227) acquired from Abcam (UK). In 6-well plates, 4 × 105 cells were planted in each well. After treatments, cells were lysed in 10 L of lysis buffer + 100 mM PMSF (Solarbio, Beijing, China) in 1 mL of buffer on ice for 30 minutes. Cell lysates were separated using electrophoresis gels made of 10% sodium dodecyl sulfate and polyacrylamide, blotted using membranes made of polyvinylidenedifluoride, and blocked with 5% skim milk in PBS plus 0.1% Tween 20 (TBST). After that, membranes were incubated with primary antibodies at 4°C for 12 hours. Rabbit polyclonal antibodies against JAK-1 and TLR-4 were utilized (1:1000). As a loading control for normalization, a rabbit polyclonal-actin antibody (1:4000, Proteintech, Rosemont, IL, USA) was applied. Membranes were treated with a secondary anti-rabbit antibody (1:4000; Protein-tech) coupled to horseradish peroxidase for 1 hour at room temperature. Using an improved chemiluminescent reagent, protein bands were seen (Thermo Fisher Scientific). Using the Protein Simple Digital imaging technology, band images were produced and quantified (FluorChem R, San Jose, CA, USA).
Apoptosis Assay by Flow Cytometry
The manufacturer’s recommendations were followed when using the “FITC Annexin V Apoptosis Detection Kit with PI” (BD Pharmingen™, BD Biosciences Co., USA; Number # 51-66121E) for flow cytometry apoptosis analysis. In a nutshell, PPE was incubated with liver cancer cells grown in 6-well plates at a density of 3 × 105 cells/well and at a concentration equal to its IC 50. The cells were collected after 24 hours, cleaned with PBS, and centrifuged at 130g for 5 minutes. The cells were centrifuged, resuspended in binding buffer, and then resuspended in cell staining buffer at 4°C. Flow cytometry tubes were then filled with 5 mL of FITC Annexin V and 10 mL of 20 mg/mL propidium iodide (PI), which were then incubated for 15 minutes at room temperature and in the dark. A flow cytometer (BD Accuri C6) was used to count the amount of apoptotic cells, and data were then processed on a FACSC-LSR (Becton and Dickinson Company) fitted with Cell Quest software.
Biochemical Assay
Malondialdehyde (MDA), which was detected in cell lysate using the Yoshioka et al method, was used to measure lipid peroxidation. This was done by using a thiobarbituric acid assay to create thiobarbituric acid reactive substances (TBARS), which were read at 532 nm. 19 Using Sinha’s approach, catalase (CAT) activity was measured in liver cells. 20 One way to think of the dichromate/acetic acid reagent is as a stop bath for catalase activity. Any hydrogen peroxide that has not been broken down by catalase will react with the dichromate to produce a blue precipitate of perchromic acid when acetic acid is added to the reaction media. The green solution was produced by heating this unstable precipitate, and the hue of this green solution was measured photometrically at 570 nm. Using the approach of, superoxide dismutase activity in cancer cells was assessed at 540 nm. 21
Statistical Analysis
The findings of each experiment were expressed as the mean ± standard error (SEM), with each experiment being performed at least in triplicate. Analysis was conducted using the statistical software program (SPSS Inc., Chicago, IL, USA). Using the P .05 statistic, statistical significance between all groups was examined. Graphs for statistical analyses were created using Prism, version 8. (Graph Pad Software, La Jolla, CA, USA).
Results
Effects of Pomegranate Peel Extract on Anti-Proliferation of HepG2 Cells
For the assessment of the possible antitumor efficacy of PPE on liver cancer, the HepG2 cell line was subjected to increasing concentrations of PPE to evaluate PPE cytotoxic activity utilizing the MTT assay. One hundred percent viable HepG2 cells cultured in RPMI complete medium are regarded as a positive control. As shown in Table 1 and Figure 1A and B, inhibition in HepG2 cells was observed. Twenty-four hours of PPE treatment resulted in HepG2 cell proliferation inhibition in a dose dependent manner and the half maximal inhibitory concentration (IC50) was estimated to be approximately 83.9 μg/mL.
Effect of Different Concentrations of PPE on HepG2 Proliferation.
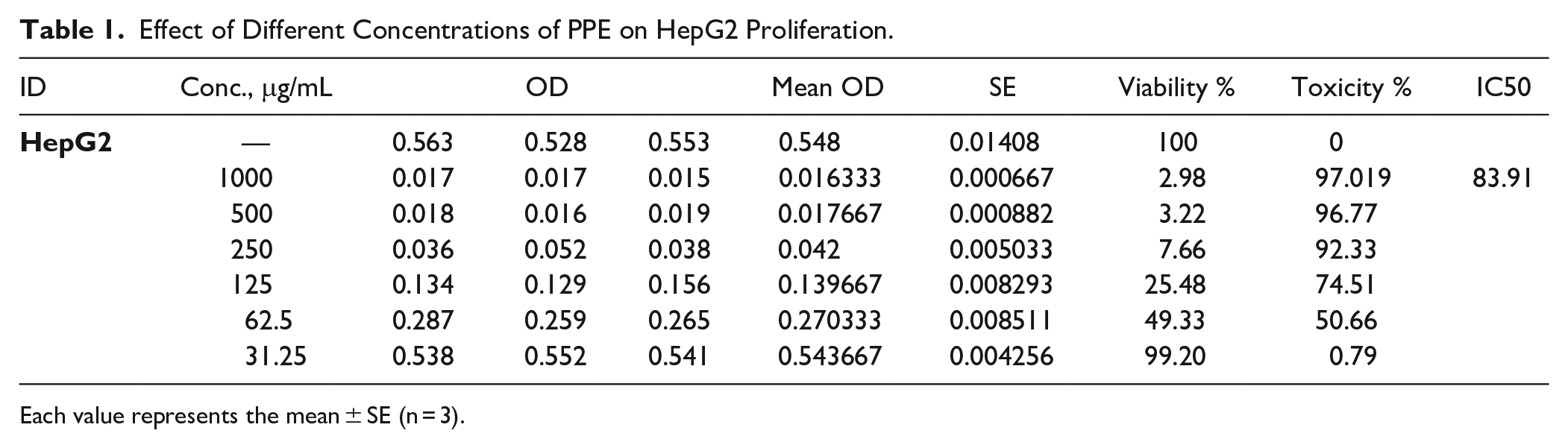
Each value represents the mean ± SE (n = 3).

Antiproliferative effect of PPE on HepG2 cells. (A) Anti-proliferative effect of PPE on HepG2 cells. (B) Effects of PPE on HepG2 proliferation under microscope.
Cell morphological alterations were observed in phase-contrast images in a PPE dose-dependent way (Figure 1B). There were signs of apoptosis in addition to cell disintegration, membrane blebbing, and cell detachment accompanied by decrease in cell counts concurrently with rising PPE concentrations.
PPE Abates Cancer-Triggered Inflammatory Response via Hampering TLR4/NF-κB Signaling and Stimulating Nrf2 Level
To ascertain the role of inflammatory response, further determinations revealed PPE-induced TLR4/NF
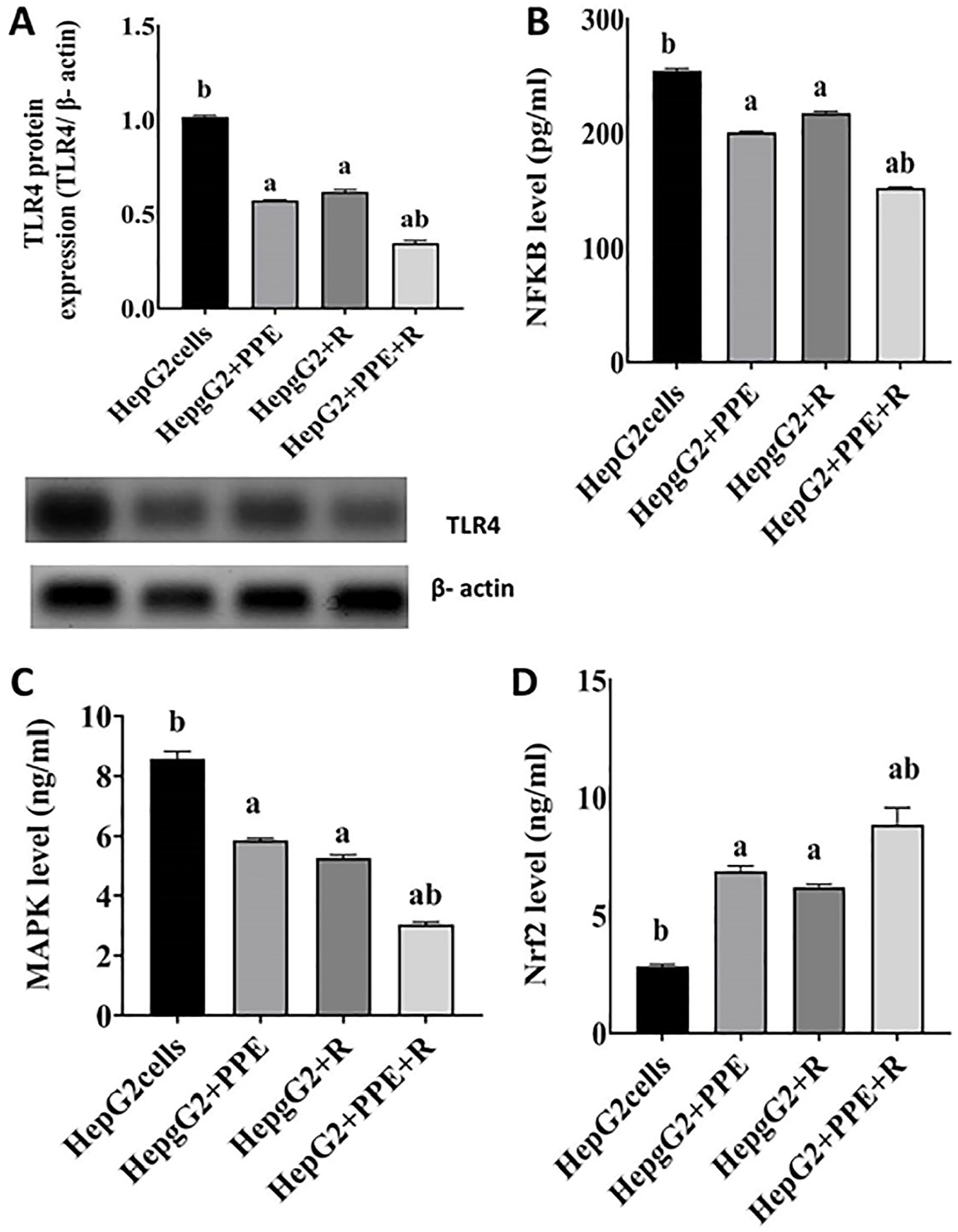
(A-D) PPE and/or IR restrains inflammatory response evoked by the expression of cancer TLR4 protein. SDS-PAGE of TLR4 (95 kDa) standardized to β-actin protein (43 kDa) expression. Results are stated as mean values ± SEM (n = 3 independent values). Columns denoted with “a” significantly different from HepG2 cancer cells; “b” significantly different from HepG2 + R, at P < .05. HepG2 cancer cells were treated with PPE and exposed to 6 Gy. Statistical analysis was performed utilizing one-way analysis of variance (ANOVA).
Impact of PPE and/or γ-Irradiation Exposure on of Janus Kinase (JAK) and Signal Transducer and Activator of Transcription-3 (STAT3)
To find out the role of inflammatory response in cancer we further determined JAK-STAT3. The JAK-STAT pathway is a series of cell protein interactions that plays a vital role in the development of cancer. The data exemplified in Figure 3 displayed that there were substantial increases in JAK protein expression (Figure 3A), and STAT3 level in HepG2 group (Figure 3B). Current study results demonstrated significant alterations in inflammatory response parameters upon treatment of HepG2 cells with PPE where JAK protein expression was significantly decreased (P < .05) by 60% and STAT3 level was declined by 50% with the respective HepG2 group. Just about the same issue, our experimental results upon irradiation of HepG2 cancer cells identify a notable decrease in JAK protein expression by 51% together with STAT3 level by 64% with values equivalent to the cancer group. Nevertheless, both treatment modalities combined (HepG2 + PPE + R) revealed the most observable decrease in JAK protein expression by 133%, followed by a noticeable decline in STAT3 level by 121% compared to the HepG2 cancer group (Figure 3A and B).

(A, B) Effect of PPE on JAK
PPE Down-Regulates Tumor Proliferation Through Targeting β-Catenin and NOTCH1 Signal
Wnt signaling is activated because of β-catenin nucleus translocation that results in cell proliferation; NOTCH1 is involved in the Wnt signaling pathway. The data illustrated in Figure 4 demonstrate that the level of β-catenin and NOTCH1 were significantly increased in the HepG2 cancer group. On the other hand, the HepG2 cell line treated with PPE resulted in a noticeable (P < .05) decline in β-catenin level by 50% (Figure 4A), and NOTCH1 level by 64% compared to its corresponding cancer group (Figure 4B). Upon irradiation of HepG2 cancer cells our experimental results identify a significant decrease in β-catenin level by 36% associated with a pronounced decrease in NOTCH1 level by 73% paralleled to human HepG2 hepatoma group. However, exposure of HepG2 to γ-irradiation in combination with PPE produced a significant down-regulation in β-catenin by 89.5% and NOTCH1 level by 104% compared to the untreated liver cancer group (Figure 4A and B).

(A, B) Effect of PPE on β-catenin (Figure 4A) and NOTCH1 level (Figure 4B). Values are stated as mean values ± SEM (n = 3 independent values). Columns denoted with “a” significantly different from HepG2 cancer cells, “b” significantly different from HepG2 + R, at P < .05. HepG2 cancer cells treated with PPE and exposed to 6 Gy. Statistical analysis was performed utilizing one-way analysis of variance (ANOVA).
Pomegranate Peels Extract Decrease Proliferation and Induce Apoptosis in Human HepG2 Hepatoma Cells Through Targeting SOCS3, PPAR-γ, and Caspase3
In the current work, the imbalance between proliferation and apoptosis was further illustrated by increasing SOCS3 level and diminishing the level of PPAR-γ and the proapoptotic caspase-3 in HepG2 group. Apoptosis enhancement was observed due to treatment of HepG2 liver cancer cells with PPE as presented by the remarkable SOCS3 down-regulation by 66% (Figure 5A), elevation (P < .05) in PPAR-γ by 2.2-fold (Figure 5B), and cleaved caspase-3 level by 2.7-fold (Figure 5C), compared to untreated HepG2 cancer cells. It is worth mentioning that irradiation of HepG2 cells reveals a significant reduction in SOCS3 level by 46% and increase in PPAR-γ level by 2-fold and cleaved caspase-3 by 6.3-fold change relative to untreated liver cancer cells. Further, exposure of HepG2 cells in combination with PPE treatment to γ-irradiation induces a significant (P < .05) decline in SOCS3 level by 115% accompanied with noteworthy increase in PPAR-γ level by 2.9 and cleaved caspase-3 level by 8.2-fold change compared to its equivalent value in HepG2 group, Figure 5A to C.
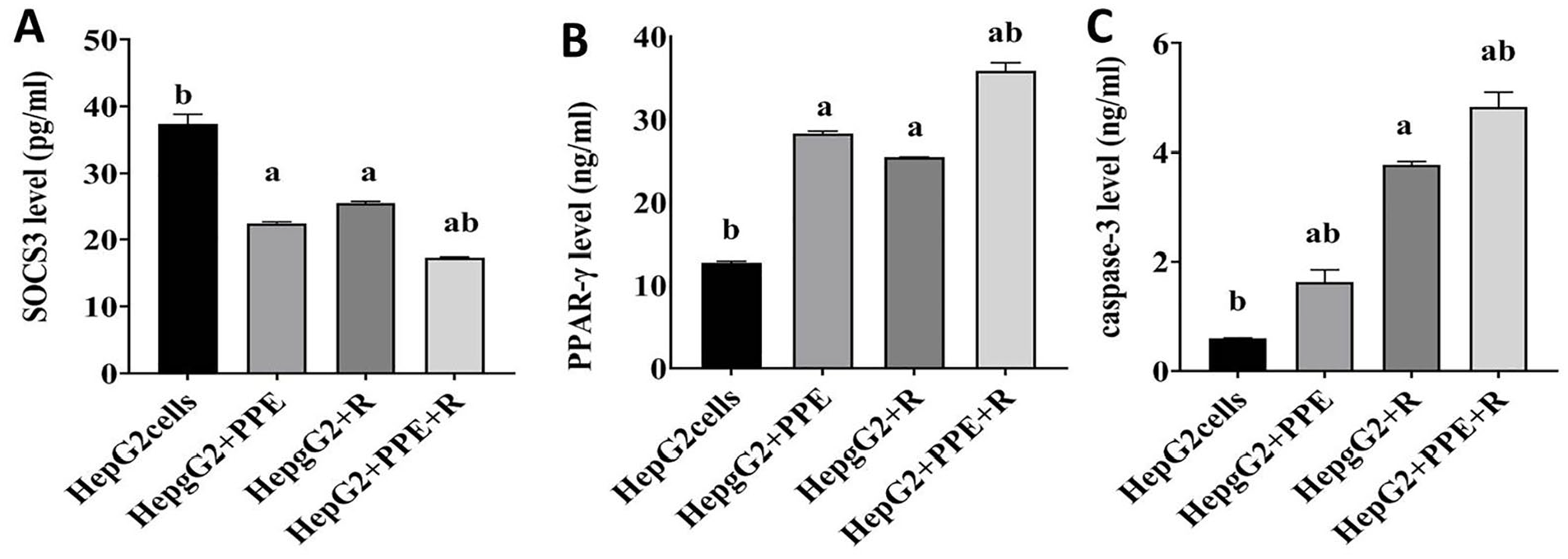
(A-C) Effect of PPE on proliferation and apoptosis of human HepG2 hepatoma cells. Values are stated as mean values ± SEM (n = 3 independent values). Columns denoted with “a” significantly different from HepG2 cancer cells, “b” significantly different from HepG2 + R, at P < .05. HepG2 cancer cells treated with PPE and exposed to 6 Gy. Statistical analysis was performed utilizing one-way analysis of variance (ANOVA).
Apoptosis evaluation by flow cytometry
Besides cell morphology, flow cytometric analysis of heterogeneous cell populations, in which cell viability is maintained, can also be used to detect apoptosis. This method allows for the disclosure of sample intact cells, apoptotic cells, and dead cells due to apoptosis and/or necrosis simultaneously. The inner side of the plasma membrane contains a calcium dependent phospholipid-binding protein that possesses a great affinity for phosphatidylserine (PS). Early apoptotic stages are characterized by changes at the cell surface that are challenging to detect, such as translocation of PS from the inner to the outer layer of the plasma membrane, therefore becoming exposed at the external cell surface. This behavior also happens during necrosis in addition to apoptosis. These 2 types of cell death differ in that the cell membrane is still intact during the early stages of apoptosis, however during necrosis; the cell becomes leaky due to the loss of cell membrane integrity that enables propidium iodide (PI) entry that can be utilized for necrotic cell designation. Thus, the way cells attach to annexin V (x axis) and/or PI (y axis) in a flow cytometric dot plot chart helps distinguish intact viable cells (double negative staining/lower left quadrant) from cells that experienced apoptosis (annexin V positive cells/lower right quadrant) and cells undergoing necrosis (double positive staining/upper left quadrant), in respect to the divergent apoptotic cascade stages.
Depending on such strategy, HepG2 group apoptosis was distinguishably lowered as opposed to all the other groups (P < .05), as presented in Figure 6. Figure 6A shows the results of one representative from each group, while Figure 6B shows the results of means of 3 samples from each group. Two distinct populations appeared following the treatment with IC50 value of pomegranate peel extract: one was identified as an early apoptotic cell since it only displayed a single positive annexin V expression, and late apoptotic cells the displayed double positive Annexin V and PI staining (Figure 6 upper right quadrant). The early apoptotic cells% increased from 0.4% in the HepG2 group to 2.5 or 3.3% in the HepG2 groups treated with PPE or radiation, respectively. Concomitant treatment with PPE + radiation elevated that percentage to 7%. The late apoptotic cells% increased from 1.2% in the HepG2 group to 3.5 or 11% upon treatment with PPE or radiation, respectively. Concomitant treatment with PPE + radiation elevated the late apoptotic cells percentage to 15% as shown in Figure 6A. It is noteworthy that the highest percentage of late apoptotic cells and the lowest percentage of viable cells were all reported in the group treated with PPE + radiation.

(A, B) Flow cytometric detection of apoptosis employing Annexin V-FITC conjugated with PI staining. Figure 6A shows a representative from each group, PI + cells first and second quadrants (upper right and upper left) represented late apoptotic and necrotic cells; meanwhile the lower left third quadrant for the PI-/AV-cells illustrated normal cells. Lower right quadrant numbers indicate early apoptotic cells (AV−/PI+) in percent of the whole gated cells. Figure 6B shows percent of apoptosis detected by flow cytometry, (mean ± SEM, n = 3). Three similar iterations of an exemplary experiment were shown.
In Figure 6, it is feasible to observe that such populations were concurrent with a gradual and appreciable elevation in the number of necrotic cells (N). Then again, the apoptosis of gamma irradiated HepG2 treated with PPE was significantly higher than their corresponding non- radiated group (P < .05).
Defensive Effects of Pomegranate Peel Extract Against Oxidative and Antioxidant Status Induced in γ-Irradiated HepG2 Cells
Oxidative stress in liver cancer cells was evaluated based on enzymatic (CAT and SOD) antioxidants and MDA (lipid peroxidation end-product) measurement. The data illustrated in Figure 7 showed a remarkable restoration of the regular redox tone. The levels of MDA were considerably raised in HepG2 liver cancer cells (Figure 7A), associated with a marked decrease in SOD (Figure 7C) and CAT (Figure 7B) activities. This effect was significantly improved with concurrent PPE treatment, shown by significant decrease (P < .05) in MDA (by 48.7%), with a significant increase (P < .05) in CAT and SOD activities by 1.5-fold change and 2.13-fold change respectively, compared to the untreated HepG2 group. More pronounced amelioration was observed after irradiation of HepG2 cells. What is more, exposure of HepG2 cells to γ-irradiation in combination with PPE produced a significant recovery in redox tone where MDA showed a noteworthy decrease of (51%) concomitant with a significant increase in CAT and SOD activities by 1.9-fold change and 2-fold change respectively with respect to the HepG2 group Figure 7A to C.

(A-C) Protective effect of PPE against oxidative stress in HepG2 liver cancer. Each value represents mean ± SEM (n = 3). Columns denoted with “a” significantly different from HepG2 cancer cells, “b” significantly different from HepG2 + R, at P < .05. HepG2 cancer cells treated with PPE and exposed to 6 Gy. Statistical analysis was performed utilizing one-way analysis of variance (ANOVA).
Discussion
One of the most common tumors in the world is hepatocellular carcinoma (HCC), which still exhibits high levels of conventional treatment resistance. In order to achieve new levels of effectiveness in the ongoing fight against cancer, it was necessary to delve thoroughly into relatively subtle aspects and develop novel approaches to cancer treatment. Radiation resistance is regarded as one of the primary factors of radiotherapy failure and consequent tumor reversal, despite the fact that radiation therapy is promoted as a viable technique for curing cancer. Radiation therapy’s efficacy may be improved by implementing radiosensitizing drugs to make tumors more sensitive to radiation. 9 This study was started to evaluate the anticancer effect in vitro of pomegranate peel extract in combination with radiation as an advanced cancer therapeutic approach against HepG2 cancerous cells.
The results of the current investigation showed that the HepG2 group had notably lower levels of Nrf2 and remarkably higher levels of TLR4, NF-B, and MAPK (P < .05) protein expression (Figure 2). TLR4 expression is remarkably low under normal circumstances, but it is upregulated when the liver is injured due to pro-inflammatory conditions. TLR4 expression has been linked to HCC’s poor prognosis and increased capacity for invasion and metastasis. 22 TLR activation results in downstream nuclear factor kappa B (NF-kB) signaling activation and the creation of inflammatory mediators, which then promotes the survival and development of HCC cells. 23 The importance of NF-kB in numerous stages of cancer onset and promotion is becoming more widely acknowledged. Hepatic cell NF-kB sustains hepatic inflammatory processes, which aids in the growth of HCC. These studies have established that the NF-kB signaling cascade is continuously active and causes ongoing inflammation to promote carcinogenesis. Interestingly, complex connections exist between the NF-kB signal transduction pathway and many biochemical pathways. 24
Janus kinase/signal transducer activator of transcription factor (JAK/STAT), as well as the mitogen-activated protein kinase/extracellular signal-regulated kinase (MAPK/ERK), Wnt/β-catenin are a number of molecular cellular pathways that are essential for the development, growth, and metastasis of HCC tumors. 25 Liver cancer, typically activates the MAPK/ERK signaling cascade. 26 MAPK/ERK signal transduction system has been linked to the development of hepatic tumors in a number of studies, both in humans and in experimental animal models.27,28 Recent findings from multiple investigations suggested a connection between ERK phosphorylation and nuclear factor-erythroid 2-related factor 2 (Nrf2) stimulation.29,30 It was discovered that Nrf2 regulates the production of cytoprotective genes via antioxidant response elements (ARE). Therefore, in response to various types of stress, Nrf2 is capable of maintaining the stable state of the cell internal environment. 31
Referring to the above-mentioned outcomes, the JAK protein expression and STAT3 concentrations in the HepG2 group both noticeably increased (P < .05) (Figure 3). Overexpression of STAT3 is crucial for cancerous cell proliferation, persistence, immunosuppression, and invasion. STAT3 is a serious oncogene that has a role in HCC development. 32 STAT3 stimulation as a transcription factor causes the production of numerous genes that contribute to the distinct characteristics of malignancy. He and Karin proposed that cytokines and/or growth factors like IL-6 produced in the cancer microenvironment are responsible for activating STAT3 in cancerous cells. STAT3 moves from the cytoplasm to the nucleus after being activated by JAK to promote the transcription of target genes like CyclinD1. 33 The JAK/STAT cascade is frequently dysregulated in cancer, particularly HCC, resulting in increased oxidative stress, growth factor stimulation, and inflammatory response.33,34
Additionally, the findings showed a notable increase in the levels of NOTCH1 and β-catenin in the HepG2 group (Figure 4). Patients with HCC usually have the Wnt/β-catenin pathway induced, which leads to a more virulent HCC subtype with a poor prognosis. 35 The Wnt/β-catenin signaling cascade is one such important mechanism that determines a cell’s destiny by prioritizing tumor growth over apoptosis. 36 A promising method for blocking oncogenic Wnt signals is the protein β-catenin, which functions as a canonical Wnt signaling cascade transcriptional co-activator. 37 Remarkably, abnormal Notch activity has been found in various cancers. Notch1 stimulation promotes HCC proliferation and expansion and has been linked to a poor prognosis. 38 Also, it is important to note that a prior report demonstrated that Wnt/β-catenin could increase Notch pathway activity. 39
In the current investigation, the disparity between proliferation and apoptosis was illustrated by raising the level of SOCS3 and diminishing the level of PPAR-γ and the proapoptotic caspase-3 in HepG2 group (Figure 5). As reported in the study of Dai et al, 40 SOCS3 is an immune-oncogenic molecule that is predominantly implicated in the genesis of tumor mainly via the JAK/STAT signaling transduction pathway and cytokine receptor activation. These findings suggest that SOCS3 is important for the genesis and progression of several malignancies and may serve as an important objective for cancer detection and treatment.41,42 Our findings demonstrated a correlation between the rise in SOCS3 and the decline in PPARγ and caspase3. According to our findings, the disruption of the moderators of proliferation and apoptosis causes tumors growth and expansion. It has been reported that PPARγ may enhance intercellular adhesion, trigger cell cycle termination, decrease cell proliferation, induce apoptosis of various cancer cells, and impair the tumor microenvironment inflammatory state at both the transcriptional and the protein levels.43-45 Apoptosis is a type of controlled cell death that eliminates unstable cells and genetic alterations to preserve the integrity of the internal environment. Cancer, on the other hand, inhibits this process, which causes a buildup of numerous genetically defective cells. Our findings showed that the level of the apoptotic protease caspase-3 in the HepG2 group had significantly decreased. Caspase-3-stimulated apoptosis is a prominent focus in the field of inhibiting tumor progression. 8 Oxidative stress is a crucial factor in the multi-step, complex process that influences how quickly HCC develops (Figure 7). Overproduction of reactive oxygen species (ROS) affects cell adhesion, cell cycle, gene expression, cell metabolism, and cell death, which results in oxidative damage to DNA and a surge in chromosomal abnormalities linked to tumorigenesis. 46 In addition, ROS can activate various signal transduction pathways within cells, including those controlled by NF-kB, Wnt/β-catenin, PI3K, p53, and MAPK which can augment angiogenesis, promote proliferation and halt apoptosis. 47
Cancer cell treatment by γ-irradiation resulted in a considerable delay in tumor formation via down-regulating inflammatory moderators, suppressing proliferative cell signals and inducing apoptosis. Numerous signaling mechanisms are activated when radiation damages DNA; however, the intrinsic apoptotic pathway is thought to be the main mechanism for ionizing radiation-stimulated apoptosis, which prevents malignant cells from proliferating and growing. 7 Radiation exposure results in several malignant cells going through apoptosis.7,48,49 Despite the fact that radiosensitization of malignant cells has been shown to be an effective candidate for cancer eradication, it is also crucial to lessen the damaging effects of RT on the normal tissues that surround cancerous tissue. Thus, for even radioresistant liver tumors, implementing drug/RT regimens could enable enhanced tumor control. Nevertheless, due to hyaluronic acid upregulation and a lack of oxygen (hypoxia) in the tumor-microenvironment, a variety of malignancies develop a radioresistance that is responsible for the failure of RT and the relapse of tumor or metastasis.50,51 Tumor resistance is frequently cited as the reason for radiotherapy and chemotherapy treatment failure. Chemotherapy often shows a low response rate due to various drug resistance mechanisms. Patients who develop resistance to chemotherapy drugs cannot benefit from the cytotoxicity induced by the prescribed drug and will likely have a poor outcome with these treatments. Radioresistance in cancer is often caused by the repair response to radiation-induced DNA damage, cell cycle dysregulation, cancer stem cell (CSCs) resilience, and epithelial-mesenchymal transition (EMT). 14,52 As a result, it is critical to discover methods to improve radiation cytotoxicity impact in conjunction with certain tumor-selective cytotoxic drugs. Prevailing dietary recommendations for preventing chronic diseases such as cancer supports high intake of fruit and vegetables high in antioxidants. The findings of this study showed that PPE treatment of HepG2 cells considerably slowed the proliferation of malignant cells, and that PPE therapy combined with ɣ-irradiation had a synergistic impact. The PPE administration period relative to RT was significant in determining effects, according to radio-modifying implications of PPE in liver cancer cells; reinforcement is often stronger after exposure for 24 hours prior to R. The 6 Gy-R was chosen for additional research since Askar et al 53 found that most of the stimulated damage response systems operate efficiently for a few hours after RT.
The inhibition of STAT3 after PPE treatment for both non-irradiated and radiated HepG2 cells suppressed cancer development via a variety of pathways, including the generation of pro-inflammatory cytokines (down-regulation of TLR4 expression and NFKB level), which are critical mediators of malignant proliferation and inflammatory pathway induction, suppression of survival markers level (reduction of JAk, β-catenin NOTCH1, SOCS3), and enhancing apoptosis (induction of tumor Nrf-2, PPAR-γ, and caspase-3), followed by enhancement of redox tone of the cells. These results could highlight the effective role of PPE in cancer treatment.
It has been established that punicalagin, ellagic acid (EA), and other polyphenols are present in considerable quantities in the syrup and peel of the pomegranate fruit. 54 EA is a polyphenolic compound that belongs to the class of ellagitannins (ETs). It has been demonstrated that this substance, besides triggering apoptosis, can halt angiogenesis, tumor cell invasion, and metastasis in a variety of cancers. 55 Additionally, it is found that EA can enhance the impacts of chemotherapy and radiation. 56 According to Cheshomi et al 57 the key signal transduction networks likely used by EA to assert its anticancer actions in human tumors are the P53, WNT/β-catenin, and apoptosis signaling mechanisms. Pomegranate extract enriched in EA can inhibit NF-κB and down-regulate the production of iNOS, COX-2, and TNF-α in colon cancer.58,59 This is consistent with the findings of both the current investigation and the study of Bishayee et al. 46
According to the aforementioned observations, PPE treatment of HCC cells resulted in a remarkable decrease in JAK protein expression and STAT3 level along with a reduction in SOCS3 (Figures 3 and 5). Pomegranate most likely controlled oxidative stress and NF-κB to achieve this effect. 60 Numerous evidence points suggest that NF-κB controlled the transcription of cytokines and inductive enzymes such as IL-6, TNF-α, IL-2, COX-2, and iNOS. In line with this, one of the key mechanisms for the decrease in STAT3 activity is the down-expression of IL-6 levels, which may be connected to the pomegranate’s anti-inflammatory properties. Additionally, the administration of pomegranate components to the HCC group dramatically reduces SOCS3 levels. IL-6 is a crucial component of the inflammation acute-phase response, and SOCS3 functions as a response regulator by regulating the IL-6–STAT3 cascade. 61 According to Chao et al ellagic acid, a key component of PPE, effectively reduced the production of IL-6, TNF-α, and NF-κB. As a result, one of the important mechanisms for reducing HCC is the lowering IL-6 and NF-κB expression in HCC, which may be related to the anti-inflammatory effect of pomegranate. 62
Conjointly, it was demonstrated that EA, which inhibits carcinogenesis by blocking the constitutive induction of the Wnt pathway resulting in the destruction of β-catenin, could explain the decline of β-catenin level by pomegranate.39,47 It has been suggested that pomegranate may have a broad-spectrum antitumor effect, especially in NOTCH-driven malignancies. 63 Besides, a combinational approach would produce stronger anticancer effectiveness. Therefore, it is conceivable that pomegranate may be used in conjunction with radiation therapy to eradicate cancer cells by inhibiting a variety of signal transduction pathways. Pomegranate seed oil observably raised the expression of PPAR-ɣ and reduced the number of colonic tumors. 64 Furthermore, TLR4-dependent MAPK cascade inhibition resulted in decreased malignant cells multiplication and death via PPAR-ɣ activation due to PPE effect. 65
According to the available data, HepG2 treated with PPE had notably increased amounts of Nrf2 (Figure 2). We have demonstrated that PPE concurrently targets the NF-kB-mediated inflammatory mechanism and Nrf2-regulated redox signals to achieve HCC cancer chemoprevention, hence verifying this hypothesis. 36 These data suggest that PE-mediated tumorigenesis prevention may entail anti-inflammatory processes via contemporaneous yet divergent control of 2 linked molecular pathways, the NF-B and Nrf2 cascades. 66 Wei et al observed that these potential effects might be attributable to lower levels of MDA, and enhanced Nrf2 and SOD activity, confirming the findings we have made. This suggests that antioxidative behavior may be a fundamental way of pomegranate protection. Because pomegranate contains many phenolic compounds (punicalin, EA, and punicalagin) rather than just one pure polyphenol, it possesses potent antioxidative properties. 67 Pomegranate is considered a good natural source of antioxidants due to its exceptional effectiveness in neutralizing free radicals. 68
Combination treatment (PPE + R) down-regulated TLR4, NF-κB, MAPK, β-catenin, and the NOTCH and SOCS3 axis, preventing the irradiation-induced up-regulation of the cell survival process. In their study, Das et al 69 demonstrated that EA therapy reduced the concentration of p-Akt in HepG2 cells, which in turn prevented the induction of proliferative signals. The western blot findings demonstrated that the combination treatment inhibited the stimulation of the proliferative pathway by reducing JAK transcription. Thus, by inducing the expression of caspase 3, PPAR-ɣ, and the Nrf-2 axis while simultaneously hindering the NF-κB, TLR4, MAPK, NOTCH, SOCS3, and Wnt/β-catenin signals, we were capable of demonstrating how PPE combined with radiation can generate a disparity between both the cell growth and apoptosis mechanisms, shifting the malignant cells toward cell death. As a result, the present study is successful in demonstrating how HepG2 cell treatment by PPE, IR, and their combination affects both the cellular proliferation and apoptotic processes.
Conclusions
In order to achieve a new level of efficacy in the ongoing combat against cancer, it was necessary to delve deeply into relatively fine details due to the urgent medical needs. The relationship among PPE and gamma irradiation in hepatocellular carcinoma represents one of the intriguing challenging topics. This pairing has potential. Our findings demonstrate PPE’s capacity to inhibit proliferation and trigger apoptotic cancer cell death, both of which are critical drug development considerations. Another significant result is that PPE can make malignant cells more sensitive to gamma irradiation. Via investigating modern cellular mechanisms, we verified the assumption and provided scientific concept evidence. Considering these investigations, we are optimistic that PPE and gamma irradiation in combination will serve as a milestone in the fight against cancer. Future in vivo research and clinical trials applying this combination shall confirm the efficacy of the current strategy.
Footnotes
Author contributions
NE designed the study. NE and EH carried out the practical work. ME performed the data analysis and wrote the draft of the manuscript. NE critically read and revised the manuscript. All authors approved the manuscript.
Availability of Data and Material
All data presented in this manuscript are reported in the manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
