Abstract
Background:
Liver compresses are commonly applied in complementary medicine during cancer treatment and are believed to have an energizing effect. We conducted a pilot study to investigate the influence of external application of yarrow liver compresses on the autonomous nervous system by analyzing the heart rate variability (HRV) in metastatic cancer patients undergoing radiation therapy and suffering from cancer-related fatigue (CRF).
Methods:
A randomized pilot trial was undertaken with patients suffering from CRF receiving palliative radiation therapy for bone or brain metastasis over 2 weeks. CRF patients were randomized into an intervention group receiving yarrow liver compresses and a control group receiving no external application. HRV was analyzed at the beginning (T1) and end (T2) period of the study during daytime (d) and nighttime (n) in both groups and quantified using time-, frequency-, and nonlinear dynamics domains.
Results:
A total of 39 patients were randomized between September 2017 and August 2019 and a total of 20 patients (10 per group) were available for analysis. For both groups, significant differences in HRV analyses were found in the comparison over the timeline between the first and second measurement (T1d→T2d) during daytime. Specifically, the intervention group showed decreased HRV and complexity of heart rate regulation, indicating increased sympathetic activity at time T2d, whereas the control group showed increased HRV and complexity of heart rate regulation, indicating increased vagal activity at time T2d. Furthermore, the observed HRV analyses showed almost no significant differences between the 2 groups in a direct comparison at the beginning and the end of the study (exception: T2d).
Conclusions:
Yarrow liver compresses led to increased sympathetic activity during daytime in the intervention group, whereas in the control group, which did not receive any external application, increased parasympathetic activity was observed.
Keywords
Introduction
Among all human diseases, cancer has the highest clinical, social, and economic burden and affects a large part of the worldwide population, knowing that the cumulative lifetime risk of developing cancer is 20.2%. 1 Nearly all oncology patients report cancer-related symptoms or side effects of cancer treatment such as pain, nausea, appetite loss, constipation and diarrhea, numbness, fatigue, sleeping disorders, exercise limitation, and decreased physical activity.2-4 Out of these symptoms, fatigue is one of the most prevalent and most challenging due to its multi-factorial pathogenesis and lacking gold standard for treatment.5,6
Fatigue and other cancer-associated symptoms mentioned above are likely to be related to impaired functioning of the autonomic nervous system (ANS) as cancer itself, metastasis, surgery, radiation therapy, and neurotoxic effects of chemotherapy can lead to direct and indirect structural impairment of the ANS.4,7 While large-scaled trials are lacking on this subject, various small studies and case reports have described cancer and its treatment as a cause for autonomic dysregulation.4,8
Heart rate variability (HRV) has been shown to be a reliable, instructive, and cost-effective method to assess the activity and function of the ANS. 9 HRV describes the fluctuation of time intervals between successive heartbeats due to constant modulation of heart activity by the ANS.10,11 A high HRV indicates a flexible, responsive ANS and therefore a healthy physiological functioning of self-regulation and adaptation within the body. Correspondingly, an imbalance of the ANS is reflected by decreased HRV.11,12 Traditionally HRV will be assessed by measuring in the linear time and frequency domain. However, these measures are often not sufficient to quantify the complex dynamics of physiological systems and their related time series. Therefore, various efforts have been made to apply nonlinear complexity measures to analyze HRV. 13 Methods from nonlinear dynamics have revealed new insights into alterations of the cardiovascular system under various physiological and pathological conditions, and thus providing additional prognostic information. 14
In recent decades, the use of complementary and alternative medicine (CAM) as supportive care during oncologic therapies has increased.15,16 Approximately 50% of cancer patients make use of CAM and high satisfaction rates have been reported with CAM use.17,18 This reflects the rising importance of conducting clinical trials in this area as clinical evidence is often fragmentary.
A common CAM-based supportive care measure given during cancer treatment in Germany is the external application of liver compresses with Achillea millefolium (yarrow).19,20 In anthroposophic theories, yarrow and yarrow liver compresses are believed to have a tonic, energizing effect,21,22 but clinical studies are lacking on this subject.19,23 A previously published prospective pilot trial demonstrated that external application of yarrow liver compresses reduce cancer-related fatigue (CRF) in patients with metastatic cancer undergoing radiation therapy within a clinically relevant range. 19 Based on these findings, this second prospective pilot trial (preplanned secondary analysis) investigated the influence of external application of yarrow liver compresses on HRV in the same metastatic cancer patients undergoing radiation therapy and suffering from CRF. The aim of this investigation was to assess a basic characteristic of autonomic regulation processes in those patients and the previously observed reduction of CRF.
Patients and Methods
Patient Selection
This study was approved by the Ethical Committee of the Charité Universitätsmedizin Berlin, Germany (Reference number EA1/078/17). The trial was registered in the Cochrane Central Register of Controlled Trials (DRKS00012999).
The criteria for patient eligibility was age ≥18 years, with at least minor fatigue according to the general fatigue subscale of the multidimensional fatigue inventory (MFI-20), 24 estimated life expectancy >3 months, Karnofsky performance status scale ≥60%, and an indication for inpatient palliative whole brain radiation therapy (WBRT) or palliative analgesic RT of bone metastasis. Patients were required to be inpatients due to the foreseen logistical hurdles in preparing and applying liver compresses in an outpatient setting. Patients were required to give written informed consent. Patients were excluded in cases of presence of severe psychiatric disorders, allergies or fatigue-causing medical conditions such as severe hypothyroidism, sleep apnea, insomnia, anemia with hemoglobin levels <8 g/dL, cachexia with a body mass index <18.5, chronic kidney failure or acute depression. Patients participating in other clinical trials or with liver metastasis contraindicating warming compresses were also ineligible. Patients receiving both WBRT and bone RT were also ineligible.
Details of the conduction of the external application of yarrow liver compresses are described elsewhere. 19
Treatment
All patients included in the study received 2 weeks of palliative radiation therapy for brain or bone metastases as part of their oncological therapy. Potential study participants were informed about the study on-site. After inclusion in the study according to the inclusion and exclusion criteria, the subjects were randomized into an intervention and a control group.
Patients in the intervention group received an external application of yarrow liver compresses on a daily basis for a 2-week duration, mostly after lunch or in the afternoon. Over the entire period, a minimum of 7 external applications was conducted. Patients in the control group did not receive any external application.
The intervention (IN) and the control (CON) group were further divided into the 2 subgroups brain (brain, n = 6) radiation therapy (RT) and RT of bone (bone, n = 4) metastasis to determine if the RT has an effect on HRV measurements and ANS.
Data Recording and Data Preprocessing
At the beginning (T1) and at the end (T2) of the study period (interval about 2 weeks) from all patients a 24 hours Holter ECG (portable ECG devices Faros 180, sampling frequency: 500 Hz) was recorded under resting conditions in the patient room on the ward.
Time series of 4 hours length during the daytime (d) and nighttime (n) for each intervention time point (at the beginning during daytime [T1d] and nighttime [T1n]; at the end during daytime [T2d] and nighttime [T2n]) were extracted from the ECG raw data, using the “EDF browser” (European Data Format browser). The EDF browser was used to scan ECGs visually and to pre-select 4 hours segments, with a minimum of artifacts (ie, caused by movement, detachment of electrodes, etc.). Afterward, heart rate time series consisting of successive beat-to-beat intervals (BBI) were extracted automatically from the 4 hours data recording segments using in-house software (programming environment MATLAB® R2011b). From these 4 hours BBI segments, those segments of 30 minutes length that best approximated steady-state behavior were selected (HRV analyses in the frequency domain analysis requires stationary conditions). All BBI times series were subsequently filtered by an adaptive filter algorithm to remove and interpolate ventricular premature beats and artifacts to obtain normal-to-normal beat interval (NN). 25
Methods of Heart Rate Variability Analysis
Heart rate variability (HRV) was quantified by well-established methods of time- and frequency domains 26 and nonlinear dynamics 13 to assess autonomic modulation (sympathetic, vagal). 26 These methods have shown relevance in clinical research and have proven their suitability to explore dynamic and structural features of cardiovascular regulation. They are based on various concepts, thereby revealing multiple aspects of cardiovascular variability. 13
Time- and frequency domain
Heart rate variability was quantified by calculating indices from the time- and frequency domains (TD, FD) according to the recommendations of the Task Force of the European Society of Cardiology. 27 The following standard HRV indices from the TD were calculated:
In the time domain (TD), the following indices were applied: meanNN: The mean value of the NN-intervals of BBI [ms]; sdNN: Standard deviation of the NN-intervals of BBI [ms]; cvNN: Ratio of sdNN divided by meanNN as a coefficient of variation of BBI; rmssd: Square root of the mean squared differences of successive NN intervals [ms]; renyi4: Rényi entropy as a complexity measure of the BBI estimated from α-weighted probabilities distributions (α=4).
In the frequency domain (FD), the following standard indices were applied: LFn: Normalized low-frequency power (0.04–0.15Hz) of BBI [a.u.]; HFn: Normalized high-frequency power (0.15–0.4Hz) of BBI; ULF: Power in the ultra-low frequency band (<0.003Hz) of BBI [ms
2
]; P: Total power of the density spectra [ms
2
].
The power spectra of equidistant linear interpolated (10Hz) NN interval time series (resampled to 2 Hz) was obtained by applying the fast Fourier transform. To avoid leakage effects a Blackman Harris window function was applied.
Nonlinear domain: Symbolic dynamics
The analysis of symbolic dynamics (SD) has proven to be sufficient for the investigation of complex systems and describes nonlinear aspects within time series (BBI).13,28 Therefore, at first the time series (BBI) were transformed into a symbol sequence of 4 symbols with a given alphabet A = {0,1,2,3} to classify dynamic changes. From this new alphabet (symbol string) words were created consisting of 3 successive symbols (64 different word types). The following SD indices from the probability distribution of each word type within the symbol sequence were estimated: wpsum02: Relative portion (sum/total) of words consisting only of the symbols ‘0’ and ‘2’, a measure for reduced HRV [arbitrary unit; a.u.]; wpsum13: Relative portion (sum/total) of words consisting only of the symbols ‘1’ and ‘3’, a measure for increased HRV [a.u.]; wsdvar: Standard deviation of the word sequence [a.u.]; pWxxx: Single word type probabilities [a.u., ‰]; Shannon: Shannon entropy of word type distribution [bit] as a complexity measure.
Statistical Analyses
The trial was designed as a randomized, non-blinded prospective clinical pilot trial. Block randomization was performed with stratification according to the radiotherapy site brain vs. bone with an allocation ratio of 1:1 and a block size of 4 TN. The researcher in charge of study coordination (WS) randomized and enrolled the participants and assigned the intervention. As soon as the study staff at the ward found an eligible patient through the screening process, it informed the researcher in charge of study coordination by telephone, who first enrolled and then assigned the patient to either the intervention or the control group by revealing the next line of the previously covered randomization list and communicated the group assignment of the patient. ECG analyses (HRV) were then performed with the previously defined groups.
A subsequent sample size recalculation was planned after a pilot phase. We based the power calculation for the pilot phase on a 3-point reduction on the 25 general fatigue subscale of the MFI-20 in the intervention group vs. control group at the end of treatment compared to baseline as the reduction of fatigue was the primary outcome of the previously published prospective pilot trial. 19 The requirement for an unpaired t-test with a common standard deviation of 3 points, alpha .05 and a power of 80% was a sample size of 34 patients (17 per group). Estimating a drop-out rate of 10%, 38 patients should be included for the pilot phase. As the trial had an unblinded and non-confirmatory design, P-values were regarded as exploratory, and no alpha-adjustment was planned. We used G*Power Version 3.1.3 to perform the sample size calculation.
The nonparametric Wilcoxon test was applied (SPSS 21) for the statistical evaluation of significant univariate differences in linear and nonlinear indices of HRV between the time points (T1d, T1n, T2d, T2n) for CON and IN, and the nonparametric Mann-Whitney U-test was applied for the statistical evaluation of significant univariate differences between CON and IN. Univariate significance was considered for values of P < .05. Descriptive statistics were used to describe the data’s basic features in terms of mean values and standard deviations.
Results
Between September 2017 and August 2019, a total of 79 preselected patients were screened, 39 eligible were randomized, and after exclusion of dropouts a total of 20 patients (10 per group) were available for analysis (Figure 1). The pilot trial was terminated in September 2019 due to ineligibility of many patients and withdrawal of several patients from the trial after random allocation (Figure 1). The baseline patients’ characteristics are shown in Table 1. There were no significant differences between the 2 groups. The median number of yarrow liver compresses applied in the intervention group was 10.5 (range 8-14).
Patient Characteristics.
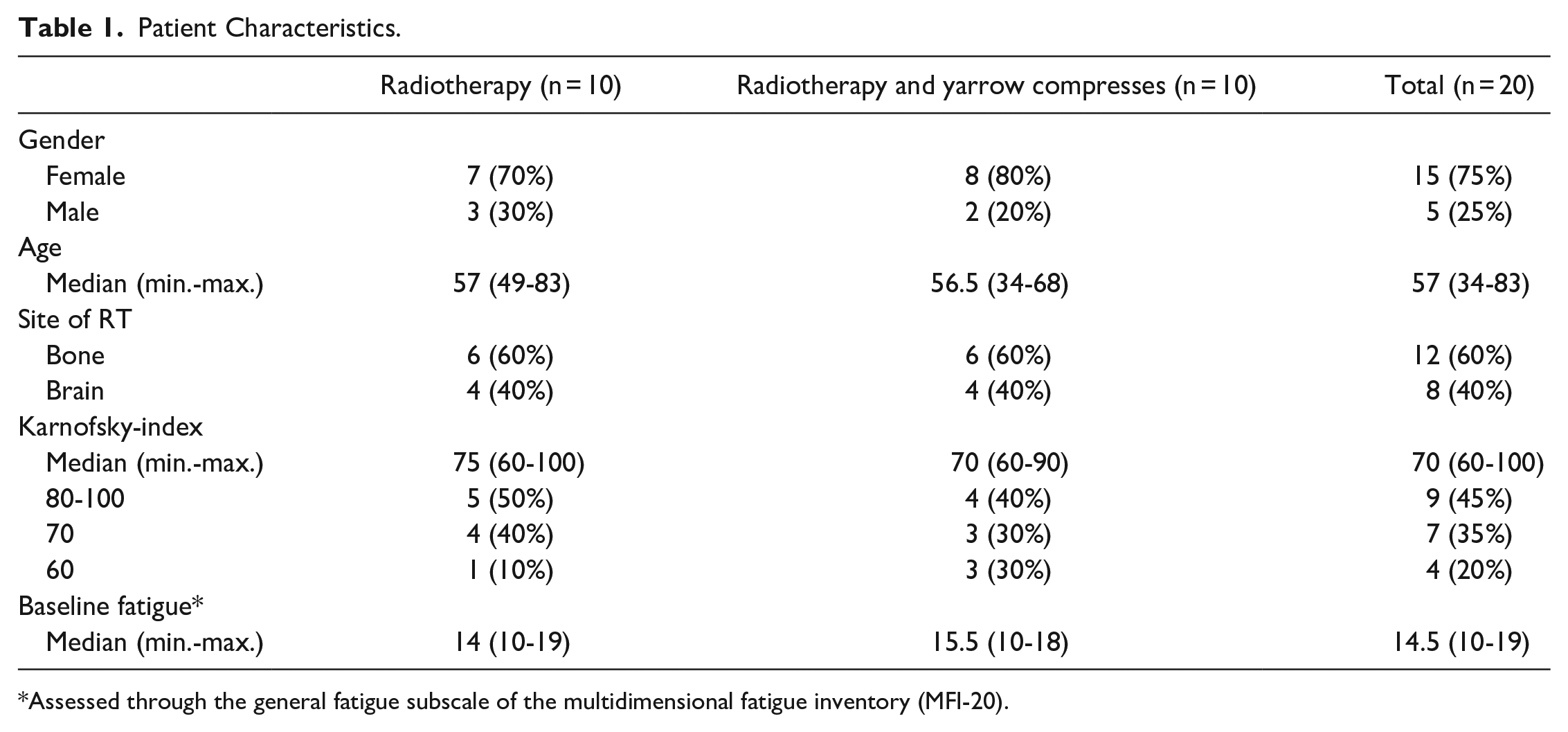
Assessed through the general fatigue subscale of the multidimensional fatigue inventory (MFI-20).

Flow diagram of the trial.
Comparisons of CON With IN for All Intervention Time Points (T1d, T1n, T2d, T2n)
T1d, T1n, T2d, and T2n for CON Versus IN
HRV analysis revealed no significant differences between IN and CON in TD, FD and SD at any intervention time point with the exception of SD at T2d, which showed significant differences (pW300, pW302, pW303; P < .05) between IN (pW300: 0.2 ± 0.6; pW302: 0.1 ± 0.2, pW303: 0.04 ± 0.1) and CON (pW300: 1.6 ± 1.8; pW302: 1.7 ± 2.6, pW303: 1.3 ± 2.4).
The subgroups of CON and IN (skull, bone) showed no significant differences between the subgroups of IN and CON in TD, FD and SD.
Comparisons of the Time Course (T1d→T2d and T1n→T2n) for CON and IN
T1d→T2d for CON
HRV analysis revealed significant differences in all domains (TD, FD, SD) between T1d and T2d for CON (Table 2). The subgroups of CON (brain, bone) revealed no significant differences in total of all domains (TD, FD, SD) over time (T1d→T2d).
Results of Heart Rate Variability Analyses for the Control Group (CON) and the Intervention Group (IN) for the Intervention Time Points (T1d, T1n, T2d, T2n) Represented as Mean ± Standard Deviation.

p < .05, **p < .01, n.s. = not significant.
T1n→T2n for CON
HRV analysis revealed no significant differences in TD, FD, and SD over time for CON, nor for the subgroups (Table 2).
T1d→T2d for IN
HRV analysis revealed significant differences only in SD between T1d and T2d for IN (Table 2) and for the subgroup bone (Shannon, pW010).
T1n→T2n for IN
HRV analysis revealed significant differences only in SD over time (T1n→T2n) for IN (Table 2), but not for the subgroups (brain, bone).
Discussion
In this study, we demonstrated the presence of an altered autonomic function caused by the therapeutic application over time in the metastatic cancer patient intervention group. We could show that HRV analyses did not reveal significant differences in direct comparison of the intervention times between either group (exception: T2d). Moreover, the subgroup analyses for both intervention and control group (radiation therapy: brain, bone) revealed no significant differences between the respective subgroups. The altered autonomic function was most clearly visible in the temporal progression of the therapy (external application). Especially at T2d, the intervention group exhibited an increased sympathetic activity, exhibited by a decreased HRV, and the control group an increased parasympathetic activity, exhibited by an increased HRV. The strongest differences were found between T1 and T2 for the control group, and weaker but also evident in the intervention group. At T1, the control group showed decreased HRV values in comparison to T2, whereas, the intervention group showed at T1 increased HRV values in comparison to T2.
Comparisons of the control with the intervention group at T2d indicate a higher complexity of heart rate dynamics in the control group than in the intervention group.
The comparisons of the time course of the therapy (T1d→T2d and T1n→T2n) for both groups indicated the strongest differences for the control group between the first (T1d) and second measurement time point (T2d) at daytime. Particularly, at T2d an increased HRV (sdNN, cvNN, ULF, P, wpsum02↓, wsdvar) and complexity (renyi4) of heart rate regulation were present in the control group, pointing to increased vagal activity. For the intervention group, these significant differences were much less pronounced but indicated a decreased HRV (not significant but trending, eg, rmssd↓) and complexity (Shannon↓, pWxxx↓) of heart rate regulation at T2d, which indicates to increased short-term sympathetic activity during ANS regulation. at T2d for the intervention group. This corresponds with a reduction in complexity and variability of ANS modulation as shown for the intervention group. Viola et al 29 already published the assumption that a shift toward sympathetic dominance and to a simplification of cardiovascular regulation is associated with reduced complexity of the ANS. This qualitatively-proven, increased sympathetic activity in the intervention group by HRV analysis could then possibly be the result of external application of yarrow liver compresses. However, whether the observed sympathetic dominance was caused by the yarrow itself, or by the application of the liver compresses remains unclear. Nevertheless, external application appears to trigger the sympathetic nervous system (SNS) toward more activity.
Constantly adapting to environmental influences, the ANS has the most important regulatory function on the homeostatic systems of the body. 7 The SNS has an ergotropic effect on the organism as it mobilizes energy and resources, whereas the parasympathetic nervous system (PNS) regulates regenerative functions of the body.10,30,31 An imbalance between these systems leads to autonomic dysregulation which is associated with a reduced state of health 32 as has been described with various diseases, 9 such as cancer. To assess the activity and balance of the ANS, HRV analyses have been shown to be a reliable, instructive, and cost-effective method. 9
In the existing literature regarding HRV and cancer-related fatigue, the exact pathophysiology of CRF remains unclear. However, various studies demonstrate certain correlations and discuss alterations of the HRV as an important factor in the development of CRF. Freeman and Komaroff 33 showed that patients with fatigue revealed alterations in SNS and PNS activity and speculated that these findings may be explained by cardiovascular deconditioning, postviral idiopathic autonomic neuropathy, or both. Likewise, Fagundes et al 34 assumed that lower HRV and elevated norepinephrine could be associated with several adverse health outcomes; and fatigue could signal the need for increased vigilance of other health threats. Crosswell et al 35 reported lower parasympathetic activity, as measured by resting HRV, was associated with higher levels of fatigue. They could show that especially, breast cancer survivors who had lower levels of RMSSD reported higher levels of average fatigue and that HRV indices (RMSSD, HF) were negatively correlated with circulating concentrations of IL-6 and C-reactive protein. They concluded that HRV represented a potential contributor to cancer-related fatigue but suggest that inflammation does not mediate this association in younger, healthy breast cancer survivors.
There are no generally admitted reference values of HRV and in our study, we had no healthy controls to compare the HRV parameters of our patients to. However, Dantas et al 36 investigated the HRV of a large number of healthy, nonmedicated adults intending to provide reference values of HRV. Compared to their published findings, our patients in both the intervention and control group seem to show decreased HRV at all measurement periods. Additionally, the systematic review of Kloter et al 37 could show that a majority of the included studies indicated that a decreased HRV is common in cancer patients, likely reflecting autonomic dysfunction associated with the disease. They further reasoned that HRV may be a useful non-invasive tool to evaluate the prognosis of cancer patients as they demonstrated that higher HRV correlated positively with patients’ survival outcome. They concluded that individuals with higher HRV and advanced coping mechanisms seem to have a better prognosis in cancer progression.
In the current literature, there are only a few relevant publications on the effects of warming compresses. Huber et al 38 found that the application of hot abdominal compresses did not increase the hepatic blood flow in healthy subjects but state that it could nevertheless have a beneficial effect in patients with liver disease. They report that the volunteers of their study experienced the compresses as pleasant and most of them fell asleep during the application. It is clear from the literature that sleep disturbances are present in fatigue patients. A common trend in these studies was that objective sleep measurements did not correlate with subjective reports from fatigue patients. It was suggested that this difference was due to misperception. 39 Findings indicate that nocturnal parasympathetic activity, as reflected by HRV,40-42 is decreased in individuals with fatigue, suggesting hypervigilant sleep.43. Other findings indicate that there is a correlation between fatigue severity and daily sympathetic activity. 44 The study by Meeus et al 43 reported differences in HRV at night in patients with fatigue compared to control subjects, indicating increased sympathetic activity. Daily sympathetic hyperactivity may cause fatigue, and decreased vagal tone during sleep may lead to nonrestorative sleep. 45
Nagashima et al 46 observed an increased PNS-activity during the one-time application of heat- and steam-generating sheets to the lumbar and abdominal region in women with moderate constipation. 25 minutes after removal of the compresses there was no significant difference in PNS activity. External application of liver compresses was found by Ghadjar et al 19 to reduce fatigue within a clinically relevant range in patients with metastatic cancer undergoing radiation therapy. Interestingly, we could demonstrate a significantly higher SNS activity at T2 during daytime over time. These changes in the HRV could explain the previously published reduction of CRF 19 as a possible result of the observed increase in SNS activity. It would also fit the assumption that yarrow has a tonic and activating effect on the body and mind.21,22 The increase in SNS activity seems contrary to the findings of Nagashima et al, 47 however, the measuring points of their study differ from our pilot trial as well as the number of applications and the study period. The resting conditions and the warmth of hot abdominal compresses could indeed have led to an increase in PNS-activity during the application in our study population and this daily regenerating relaxation over 2 weeks could then have led to less fatigue and an increase in activity, therefore increased SNS activity, during the rest of the day. To assess this hypothesis in further studies, ECG recording should take place during the applications additionally to our measurement periods.
Furthermore, other studies showed that physical exercise can have a beneficial effect on CRF 47 and that physical exercise and therapeutic eurythmy can exert a long-term increase in HRV (positive change in autonomic regulation toward a more relaxed pattern and an improvement of complexity in the region of high-frequency oscillations),48,49 which has been a suggested goal in cancer treatment due to the association of higher HRV variables with prolonged survival in cancer patients. 50 Physical exercise leads to a short-term increase in SNS activity, 51 as did the application of yarrow-liver compresses as the measurement T2d took place after the last application. This supports the hypothesis that activation of the SNS during daytime leads to fewer CRF-related symptoms (as the higher SNS-activity could be a short-term effect of the application aligning with the believed activating, tonic effect of yarrow-liver compresses). Long-term, physical exercise leads to higher HRV, 52 to observe such a long-term increase in HRV in our study population the study period was possibly too short. Even though statistically non-significant, we could observe an increase in HF as a correlate for PNS activity and an increase in P as a correlate for complexity during T2n. This indicates that the ability of the PNS to perform its regenerative functions could have been activated by the yarrow-liver compresses.
Since various therapeutic approaches to autonomic nervous system dysfunction have shown a rather limited effect on fatigue symptoms, 53 the external application of yarrow liver compresses could be a relevant additional therapeutic option for CRF patients in the future, supported by HRV analysis as a diagnostic option.
The current literature mostly interprets increased SNS activity as an indicator for stress, but it is important to keep in mind that this conclusion is mostly drawn from trials with healthy participants and that patients suffering from CRF usually show altered HRV parameters. 54 Therefore, findings are comparable only to a certain extent. Being aware of our small study population, further studies are necessary to investigate the possible role of SNS activation to reduce CRF.
Furthermore, small doses of stress, more specifically SNS activation, could create a particular immuno response in the body, which might be explained through hormesis. 55 Hormesis is a dose-response concept which describes that substances or conditions having toxic effects in high doses can inversely in small doses have a beneficial effect through triggering adaptive, regenerating processes in the exposed organism. Therefore, hormesis could explain the positive effect of low doses of stress (SNS activation through yarrow-liver applications) on the CRF, which decreased, as previously published. 19
In contrast to increased SNS activity, reduced HRV, in the intervention group, we observed a significantly increased HRV and complexity, corresponding to a higher level of PNS activity in the control group at the end of the study (T2). As the control group was more fatigued than the intervention group at the end of the study period, 19 the higher PNS activity could play an important role in the experience of CRF. PNS activity and HRV (increased) have been associated with immune dysfunction and inflammation, which have been implicated in a wide range of conditions including different types of cancer. 10 Alterations, particularly in the PNS, are a plausible but understudied factor in cancer-related fatigue as some studies suggest that vagal afferent overactivity plays an important role in the pathophysiology of CRF.56-58 Therefore, especially because HRV analysis has shown to be a promising prognostic tool for patients with oncologic diseases59,60 further investigations could be interesting to assess the causality between PNS overactivity and CRF.
Limitations are that the pilot trial was non-blinded and there were no placebo compresses, thus it remains unclear if the observed effects are due to the application, the yarrow itself, a placebo effect, or the increased personal attention the patients experienced during the application of the compresses. Therefore, the conduction of a placebo-controlled trial could bring interesting findings regarding which component plays the most important role in the modulations of the ANS. Furthermore, since the compositions of the subgroups (skull, bone) are too small to make a valid statistical statement, our main results refer only to the total groups IN and CON. Since sleep behavior and fatigue, as well as sleep and HRV, are related, it would be very useful to include sleep quality as an outcome parameter in ongoing studies to get more information about sleep behavior and HRV during external liver compress applications.
In sum, our HRV analyses showed that the application of yarrow liver compresses in metastatic cancer patients who received an external application during radiation therapy led to increased sympathetic activity during daytime, whereas in the control group without an external application we observed a significant increase in parasympathetic activity during daytime. Given the previous finding that the external application of liver compresses reduces fatigue in CRF patients with metastatic cancer undergoing radiation therapy, it seems that patients with CRF could benefit from the increased SNS activation of warming yarrow liver compresses.
Footnotes
Authors Contributions
Clara Foucré: Collected and interpreted the data, Writing—original draft; Writing—review and editing. Steffen Schulz: Analyzed and interpreted the data, Writing— original draft; Writing—review and editing. Wiebke Stritter: Conceptualization; Data curation; Funding acquisition; Project administration. Irina von Mackensen: Collected the data; Writing— original draft; Writing—review and editing. Jacqueline Luchte: Collected the data; Writing—original draft; Writing—review and editing. Pune Ivaki: Analyzed and interpreted the data, Writing—original draft; Writing—review and editing. Andreas Voss: Analyzed and interpreted the data, Writing—original draft; Writing—review and editing. Pirus Ghadjar: Conceptualization; Funding acquisition; Methodology; Roles/Writing—original draft; Writing—review and editing. Georg Seifert: Conceptualization; Funding acquisition; Methodology; Roles/Writing—original draft; Writing— review and editing.
Availability of Data and Materials
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The Christophorus foundation supported this work with a grant (grant number 277CST). Christophorus Stiftung, GLS Treuhand, Kernerplatz 2, 70182 Stuttgart. The Helixor foundation supported this work with a grant (grant number 57/16 and 45/17). Helixor Heilmittel GmbH, Fischermühle 1, 72348 Rosenfeld.
Ethics Approval
This study was approved by the Ethics Committee of the Charité Universitätsmedizin Berlin, Germany.
