Abstract
Co-therapy with herbal extracts along with current clinical drugs is being increasingly recognized as a useful complementary treatment for cancer. The anti-cancer property of the phyto-derivative acetyl-11 keto β boswellic acid (AKBA) has been studied in many cancers, including prostate cancer. However, the whole extract of the gum resin Boswellia serrata (BS) and anti-androgen enzalutamide has not been explored in prostate cancer to date. We hypothesized that the BS extract containing 30% (AKBA) with enzalutamide acted synergistically in the early phase of cancer, especially in LNCaP cells, by inhibiting androgen receptor (AR) and by reducing cell proliferation, and further, that the extract would be superior to the action of the active ingredient AKBA when used alone or in combination with enzalutamide. To test our hypothesis, we treated LNCaP cells with BS extract or AKBA and enzalutamide both individually and in combination to analyze cell viability under different levels of dihydrotestosterone (DHT). The inhibition of androgen receptor (AR) followed by the expression of prostate-specific antigen (PSA) and the efflux mechanism of the cells were analyzed to determine the effect of the combination on the cellular mechanism. Cells derived from prostate cancer patients were also tested with the combination. Only 6 µM enzalutamide along with BS in the range of 4.1 µg/ml to 16.4 µg/ml gave the best synergistic results with nearly 50% cell killing even though standard enzalutamide doses were as high as 48 µM. Cell killing was most effective at intermediate DHT concentrations of approximately 1 nM, which corresponds to normal physiological serum levels of DHT. The Pgp expression level and the androgen receptor expression levels were reduced under the combination treatment; the former helping to minimize drug efflux and the latter by reducing the sensitivity to hormonal changes. Furthermore, the combination reduced the PSA level secreted by the cells. In contrast, AKBA could not achieve the needed synergism for adequate cell killing at equivalent concentrations. The combination of enzalutamide and BS extract containing 30% AKBA because of their synergistic interaction is an attractive therapeutic option for treating early stage (hormone-dependent) prostate cancer and is superior to the use of AKBA alone.
Introduction
Prostate cancer is the fifth most common cancer, representing 6.6% of all male cancer mortality. 1 It is well known that prostate cancer depends on testosterone for its growth and progression. 2 Hence, androgen deprivation therapy (ADT) is used in which testosterone is ablated either by surgical or pharmacological methods, in an attempt to limit or control the growth of cancer, became a standard of care for metastatic carcinoma of prostate. 3 The idea of hormone ablation was introduced by Huggins in 1940 by inducing castration and reducing testosterone levels. 4 Since then, ADT has become the mainstay of treatment for prostate cancer patients until resistance with time and fatal diseases reported. 5 New approaches have been developed, including luteinizing hormone releasing hormone (LHRH) antagonists that directly bind to the LHRH receptor, 6 chemotherapy and anti-androgens. 7
Clinically, combined androgen blockage is not encouraged as a meta-analysis of bicalutamide showed the risk of adverse event occurrence. 8 However, enzalutamide used with standard first-line therapy in metastatic hormone-sensitive patients (1125 men) was associated with significantly longer progression-free and overall survival than standard care. 9 Similarly, in another study, enzalutamide with ADT reduced the risk of metastatic progression with a consistent safety profile. 10 Any medicine that can have a beneficial effect on enzalutamide can have a favorable outcome.
Our aim is to boost the action of enzalutamide as a first line therapy for hormone sensitive prostate cancer with a combination of an herbal extract, Boswellia serrata. In vitro studies showed that acetyl-11 keto β boswellic acid (AKBA), an active ingredient in the extract, is a STAT 3 and Akt inhibitor in prostate cancer cells 11 and hence can delay transition to androgen independence. AKBA is also found to inhibit or suppress AR expression levels in LNCaP cells 12 and hence this could work well in the androgen-dependent phase by increasing the efficacy of both enzalutamide and ADT. The scope here is to provide additional treatment options by adding AKBA or its extract to the treatment protocol and to analyze their efficacies in the androgen dependent phase along with enzalutamide. Use of the extract if found efficacious can certainly reduce the cost of treatment with enzalutamide and potentially reduce its adverse effects. The combination of BS extract with 30% AKBA and enzalutamide in relation to the effects of active ingredient AKBA has not been studied before and would provide valuable data for the future with more efficacious and affordable treatment options. Since LNCaP cells are hormone-dependent, we have also tried to investigate the potential synergism of the above drugs as a function of testosterone level.
There are 2 specific objectives of this study: (a) to understand the effectiveness of boswellic acid (AKBA), when used in combination with enzalutamide, the standard of care to understand its potential for synergism and (b) to examine the efficacy of the extract of Boswellia serrata, both singly and in combination with enzalutamide, as compared to the use of the active ingredient AKBA alone. The use of active ingredients has been promoted in modern medicine because of the homogeneity and predictability of the composition; however, there is evidence that in some cases, natural extracts have superior efficacy to the active ingredient because of the synergism between various constituents of the extract. 13 Further, it is not always practical to isolate an active ingredient because of issues concerning ingredient degradation and stability. Another aspect to be kept in mind is that the pharmacokinetics in vivo can be substantially altered, sometimes favorably, due to the presence of other ingredients in the extract. 14
In general, we hypothesized that the BS extract containing 30% (AKBA) acted synergistically with enzalutamide in the early phase of cancer, especially in LNCaP cells, by inhibiting androgen receptor (AR) and by reducing cell proliferation, and that the extract would be superior in action to the active ingredient, AKBA, used alone.
Materials
Cell Culture
The androgen-dependent LNCaP cells and HEK293 cells were obtained from the National Centre for Cell Science (NCCS), Pune. LNCaP cells were cultured in RPMI-1640 media under standard conditions of 10% fetal bovine serum, 1% pencillin-streptomycine antibiotics, and 5% CO2 at 37°C. HEK293 were cultured in minimum essential media (MEM) with sodium pyruvate (1×) and non essential amino acid (1×). Human mesenchymal stem cells cultured in DMEM with 10% fetal bovine serum and 1% pencillin-streptomycin were isolated from adipose tissue of patients subjected to abdominoplasty.
Materials
Boswellia serrata extract (super) containing 30% acetyl-11-keto-β boswellic acid (AKBA) was purchased from Sami’s Lab Benguluru (Table 1). Enzalutamide (MDV3100) from Shelleckchemicals, USA, 5 α dihydrotestostetone (DHT) from Sigma (Cat # D-073-1ML), RPMI-1640 media from Himedia (Cat # AL162S), FBS (South American origin), and penicillin-streptomycin as antibiotics from Invitrogen, Human PSA-total ELISA kit from Ray Biotech (Cat#ELH-PSAtotal), Histopaque from sigma (Cat # 10771-100ML), and AR antibody (#3202 cell signaling).
Composition of Boswellia serrata extract.

Stock Preparation of the Drugs
BS extract powder was stored at 4°C. Stock solutions [102.5 µg/ml, corresponding to 30 µg/ml of AKBA] were prepared by weighing the powder, dissolving it in DMSO, and storing it at −20°C. The working solutions were prepared in media at the time of the experiments. Similarly, enzalutamide was also prepared in DMSO in order to obtain desired concentrations in the range 0.75 µM to 96 µM.
Composition of the Boswellia serrata Extract
The extract was prepared by isopropyl alcohol extraction of the gum resin exudates. Physical parameters like solubility (insoluble in water, soluble in alcohol), moisture content (2.41%w/w) and ash content (0.46% w/w) were analyzed. The compound was found to be dextrorotatory (+55.23°). Traces of heavy metals <10 ppm (µg/g) were also found. The detailed chemical composition of the resinous part is shown in Table 1. Apart from the pentacyclic triterpenes, the extract contains mono-, di-, tri-, and tetra triterpenes. 20
Methods
Baseline Effect of DHT on LNCaP Cells
The effect of DHT alone on cell viability was characterized by seeding LNCaP cells in a 96-well plate at a density of 1 × 104 cells and exposing them to different concentrations of DHT from 0.01 nM to −1000 nM. After 48 hours the cells were subjected to starvation by removing FBS-containing media and replaced with 10% charcoal-stripped FBS (CS-FBS) containing media. After 24 hours, a hormone induction medium containing 6 different concentrations of DHT ranging from 0.01 nM to 1000 nM was given. Cell viability was assessed using MTT (3-(4,5-Dimethyl-2-thiazolyl)-2,5-diphenyl-2H-tetrazolium bromide). The optical density was measured at a wavelength of 570 nm and 660 nm using a Beckmann Coulter Elisa Plate reader (BioTek Power Wave XS).
Morphological Analysis
The DHT-exposed LNCaP cells were monitored for 6 days and captured using a Nikon Eclipse TE2000-U inverted microscope.
Prostate Specific Antigen (PSA) Determination
Immuno fluorescence staining for PSA in the presence of DHT
The presence of DHT can affect the secretion of PSA. To determine the localization of PSA, LNCaP cells were plated at 2 × l04 cells on poly-L-lysine-coated cover slips which were placed inside 24 well plates. After 48 hours, cells were subjected to starvation by removing FBS and replacing it with 10% CS-FBS for 24 hours. Hormone induction was given by adding DHT along with media supplemented with 10% CS-FBS. The incubation was continued for another 48 hours and cells were fixed in 4% paraformaldehyde for 15 minutes following permeabilization with 0.2% triton-X 100 for 5 minutes. After permeabilization, the cover slips were incubated in blocking buffer (1% FBS in PBS) for 30 minutes. Cells were stained with primary antibody diluted in blocking buffer for 1 hour at room temperature. After washing with PBS, the cover slips were incubated overnight with secondary antibody tagged with Alexafluor 488. Then, the cover slips were washed to remove the unbound antibody and counter stained with DAPI to visualize the nucleus. After drying, the cover slips were mounted with DPX mount and then imaged under a fluorescent microscope.
Immunoassay for PSA after combinatorial treatment
For quantitative analysis of the amount of PSA secreted by LNCaP cells treated with enzalutamide and BS in combination under different hormonal conditions, an immunoassay procedure was performed using a commercial human PSA-total ELISA kit. Briefly, the conditioned media were harvested from each well on the last day of the culture and centrifuged to remove residual cells. This assay employs an antibody specific for human PSA-total coated on a 96 well plate. Standards and samples were pipetted into the wells and the PSA-total present in a sample is bound to the wells by the immobilized antibody. The wells were washed and biotinylated anti-human PSA-total antibody was added. After washing, HRP-conjugated streptavidin was added to the wells. Then, a TMB substrate solution, provided in the kit was added and color developed in proportion to the amount of PSA. The color changed from blue to yellow, and the intensity was measured at 450 nm.
Treating LNCaP Cells with Various Combinations of Enzalutamide and BS at Different Levels of DHT
The effects of enzalutamide and BS in 3 different hormonal conditions—(i) zero testosterone, (ii) 1nM DHT, (iii) 1000 nM DHT—on cell viability were determined using an MTT assay. For this, cells were seeded on a 96-well plate at a density of 1 × 104 cells. After 48 hours, the cells were subjected to starvation for 24 hours. Then the cells were replenished with respective hormonal conditions: −0 DHT, 1 nM DHT, and 1000 nM DHT in CS-FBS media. The solution containing the each drug combination was added to the well plates. The control was without the addition of any drugs. Cell viability was assessed for 48 hours by adding MTT reagent.
Combination Index Assay
The nature of the drug interactions can be determined using the dose-effect relationship of drugs and their combinations by measuring the combination index (CI) quantitatively using the cell viability data. Interactions between enzalutamide and BS extracts were analyzed using Compusyn software based on the Chou Talalay method. 21 According to this model, interactions could be grouped into synergistic (CI < 1), additive (CI = 1), and antagonistic (CI > 1).
Drug and Drug Combination Toxicity Studies on Peripheral Blood Mononuclear Cells (PBMCs)
To verify the toxicity of the drugs in normal cells, PBMCs were isolated from blood obtained from the blood bank. Histopaque was added to the blood at a ratio of 3:4 and centrifuged at 400g for 30 minutes. The lymphocyte layer was removed to a fresh tube and washed 2 to 3 times with RPMI medium at 180g to 100g. Finally, the pellet was resuspended in 1 ml complete RPMI and cells were counted using the trypan blue exclusion method. The 2 × 105 cells were seeded into each well of 96 well plate and treated with Boswellia serrata extract and enzalutamide (both individually and in combination). Cell viability was analyzed using MTT assay after 48 hours.
Toxicity Studies in Human Mesenchymal Stem Cells (MSC) and Human Embryonic Kidney Cell Line (HEK293)
To substantiate the toxicity of the drugs in normal cells, human MSC’s, and HEK293 cells (seeding density 5 × 103 and 1 × 104 cells respectively) were treated with BS extract and enzalutamide individually and in combination for 48 hours. The cell viability was analyzed using MTT assay.
Molecular Studies of Receptor Expression Levels
Expression of P glycoprotein using flow cytometry
One aspect that may be critical is the efflux of drugs from the cells controlled by P-glycoprotein expression levels that may be dependent on testosterone levels. To assess Pgp expression, LNCaP cells (1 × 106) were grown in 4 different conditions: normal culturing conditions with 10% FBS in RPMI-1640 media (base line), with 10% CS-FBS in RPMI-1640 (serum starvation), with 1 nM DHT and 1000 nM DHT. These cells were treated with Boswellia serrata, enzalutamide and both in combination for 48 hours. The cells were harvested using trypsin-EDTA and then resuspended in PBS. The positive control cells were untreated with drugs but cultured under the above mentioned conditions. The negative control cells were treated with verapamil (25 µM), a P-glycoprotein inhibitor, for 10 minutes to 15 minutes in respective culture conditions. The cells were then incubated with anti-P-glycoprotein antibody at a ratio 1:100 for 30 minutes on ice. Finally, flow cytometry analysis was performed using the BD FACS Canto II.
Western blotting for androgen receptor expression
Androgen receptor expression was determined using western blotting. For this purpose, the freeze-thaw method was carried out for cell lysate preparation. Cell lysates were isolated using cell extraction buffer (life technologies) centrifuged and stored at 20°C. Protein estimation was done using Bradford reagent (B6916-Sigma) and 50 µg of each sample was diluted in Laemlli buffer, boiled at 95°C for 5 minutes and loaded on 8% polyacrylamide-bisacrylamidegel. After transfer, the blot was incubated in androgen receptor antibody (1:1000 dilutions) overnight at 4°C. It was then washed and incubated with secondary antibody mouse anti-rabbit IgG (1:5000 dilutions) for 2 hours. After washing, the blots were incubated in ECL substrate (Millipore Immobilon Forte) and exposed to a gel dock (BioRad).
Isolation and Micro-Ex-Plant Culture of Prostate Cells from Patient Samples
The prostate samples were collected and processed with the prior consent of the patients and the study was approved by the Institutional Ethical Committee [IEC-AIMS-2018-NANO-043]. To analyze the efficacy of the drugs in combination with human samples, a total of 3 prostate cancer samples (Gleason score 8-10) were obtained. Small sections of prostate cancer tissue were dissected with the help of pathologists and collected into a sterile tube containing HBSS and pencillin-streptomycin antibiotics and stored on ice 1 hour to 2 hour prior to processing. After several sterile washes, the specimen was minced with a sharp blade and mild enzymatic digestion was done using collagenase (250 U/ml) for 1 hour to 2 hour. The digestion mixture of the cells, cell clumps, and micro-explants was washed by centrifugation (50g-100g for 20 seconds) for 2 or more times and the pellet seeded in growth medium (RPMI 1640 with 5 μg/ml insulin, 30 nM sodium selenite, 10 nM hydrocortisone) plus 10% FBS onto collagen-coated dishes.
Cytotoxicity Assessment of Drugs in Patient Samples
The cells obtained from the solid tumors were single cells or small cell clusters with ≥ 80% viability. Cytotoxicity experiments were done at the third passage of the cells. For this, cells were seeded on a 96-well plate at a density of 5 × 103 cells in triplicate. After 48 hours of drug treatment, the viability was measured using the MTT reagent.
Results and Discussion
DHT Stimulated the Growth of LNCaP Cells in a Dose-Dependent Manner
Effect of DHT on LNCaP cells was studied and it was seen that, as the concentration of DHT increased from 0.01 nM to 10 nM there was an increase in cell viability when going from Day 2 to Day 6 and an increase in proliferation 1.4 to 1.6 fold. However, further increase of DHT up to 1000 nM resulted in a gradual decrease in cell viability. There was no overall decrease in viability below that of the control cells (Figure 1a). Morphological differences were also observed when cells were grown in different concentrations of DHT (Figure 1b). Slight morphological variations were observed—the cells were somewhat elongated below 10 nM concentration of DHT, whereas above that the cells became more spherical in shape, the latter suggesting less viability.

Effect of DHT on androgen dependent LNCaP cells. (a) Serum starved cells were the control cells. The rate of proliferation was calculated in percentage with respect to the control group of the respective days. Data shown as mean ± SD for triplicate wells for n = 3 experiments. (b) Morphological changes were imaged for LNCaP cells up to 6 days under different hormonal concentrations.
Enzalutamide and Boswellia serrata Showed Synergistic Interactions in the Presence of DHT
Figure 2a to c showed that the individual action of BS extract, AKBA, and enzalutamide was suppressed by DHT. This is consistent with the action of DHT in enhancing AR expression. 22 IC50 value was approximately 32.8 µg/ml for BS (Figure 2a), but it was not achieved for the equivalent w/v of AKBA (Figure 2b). In both 1 nm and 1000 nm DHT, the action of the drug was less efficacious than for the serum-starved condition, which is strong evidence for the suppression of drug action when DHT is present. Interestingly, when BS and enzalutamide were used in combination (Figure 2d), the drug efficacy was higher at 1 nM DHT than when DHT was absent or when DHT levels were high (1000 nM). Note that normal DHT levels in blood are on the order of 1 nM. 23 At this 1 nM DHT level, IC50 was achieved when BS was ≥4.1 µg/ml and enzalutamide was ≥6 µM in the combination of the 2 drugs. However, when AKBA and enzalutamide were used in combination, the efficacy was not appreciable compared to that when the extract was used in the combination.

Percentage of cell viability analyzed by cytotoxicity assay. (a to c) The individual cytotoxic effects of BS extract, AKBA, and enzalutamide respectively. (d and e) Cells treated with BS extract and AKBA in combination with enzalutamide. Data shown as mean ± SD of triplicate wells and are representative of 3 independent experiments.
The inhibitory effect of combined treatment with enzalutamide and BS in LNCaP cells was analyzed using Compusyn software, wherein a combination index less than unity indicates strong synergism, whereas index values greater than 1 indicate anti-synergistic effects (the index of unity is indicative of additive effect).The results in Figure 3 show positive synergism only when DHT is present but negative synergism when no DHT is present; that is, the combination is worse than the action of the individual drugs in the absence of DHT. Hence, DHT promotes the combination action of the drugs.

Combination index (CI) of LNCaP cells for 48 hours of combinatorial treatment of BS extract and enzalutamide in 3 different conditions.
This can be explained in another way. LNCaP cells which are androgen dependent cells require an optimum concentration of androgen for its growth. The 1 nM DHT showed acceptable and better cell growth than lower concentration of DHT. The cell will obviously undergo morphological changes deprived of hormones which may lead to cell death irrespective of drug treatments. Therefore, the difference in cell viability due to drug treatments will not be demonstrable in serum starvation conditions and will be more marked in the presence of DHT.
Localization of PSA
Immunofluorescence of PSA in LNCaP cells was done to monitor the presence and localization of PSA when treated with different concentrations of DHT. Figure 4 is a compilation of the respective pictogram of LNCaP cells showing PSA staining. When the cells were cultured in the absence of DHT, PSA production was very low and no proper localization was observed. When the concentration of DHT was increased to 1 nM, the relative number of cells that stained for PSA increased and most of the cells showed localization of PSA in the nucleus. Similarly, when cells were treated with 10 nM DHT, the nuclear localization of PSA was more visible with higher signal strength. However, at higher concentrations of DHT (1000 nM), it was difficult to hold the cells on to the cover slip as they were washed off during the staining protocol.
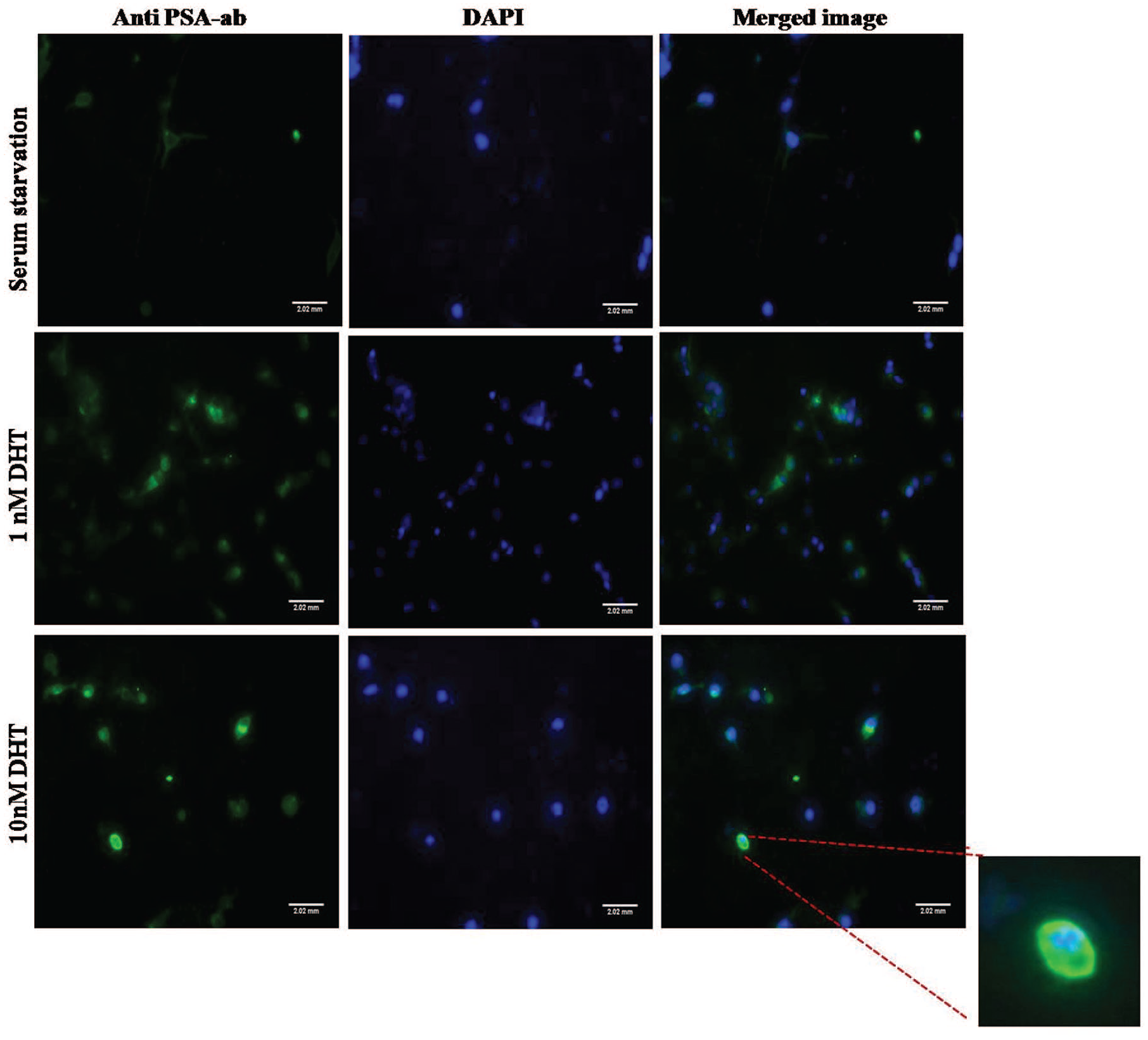
LNCaP cells stained with anti-PSA antibody. (a) Cells in serum starved condition secreted low amount of PSA and localization of PSA was not detected. (b) and (c) Treatment with DHT stimulated PSA production and nuclear localization was observed. The images were captured in 20× magnification.
Combinatorial Effect of Enzalutamide and BS in PSA Secretion
From Figure 5, it is evident that the presence of a small amount of DHT increases PSA production by almost 10-fold. The increase in PSA going from 1 nM to 1000 nM is smaller, a factor of 1.5 fold. A significant reduction (by about 50%) in PSA was observed when the concentration of BS extract was increased in the combination beyond 10.25 µg/ml.

Reduction of PSA production in the presence of DHT in LNCaP cells when treated with BS and enzalutamide. One-way ANNOVA was performed and P value of .0073 was obtained showing statistical significance (**) between the groups. The data was given in mean ± standard deviation performed in duplicates.
Toxicity Studies on Peripheral Blood Mononuclear Cells (PBMCs)
The toxicity analysis of Boswellia serrata and enzalutmide individually and in combination were performed on PBMCs. Figure 6 shows that above 48 µM of enzalutamide toxicity sets in while the MIC value of enzalutamide alone on LNCaP cells was 96 µM. It is clear comparing Figure 6 with Figure 2 that enzalutamide is toxic at therapeutic levels. On the other hand, BS was found to be toxic above 25.6 µg/ml. Thus, nontoxic drug formulations for therapy can only be found for BS-enzalutamide combinations.

Toxicity analysis in peripheral blood mononuclear cells. The standard deviation was calculated and error bar was plotted for 3 independent PBMC isolation experiments in triplicates.
Toxicity Studies on Human MSC and HEK293 Cell Line
Toxicity analyses were done on adipose tissue isolated human mesenchymal stem cells and on HEK293 cell lines. The drugs were non-toxic to MSC’s, except for the combination with highest concentration of BS extract (51.2 µg/ml) and enzalutamide (Figure 7a). Therefore, the combinations with lower concentrations which were cytotoxic to cancer cells will be non-toxic to human MSC’s. On the other hand, HEK293 cells exhibit toxicity for BS extract from 16.4 µg/ml and above, and in case of enzalutamide 12 µM and above. The combinations above 10.25 µg/ml BS and 12 µM enzalutamide was found to be toxic to this cell line (Figure 7b). Even though, HEK293 cell lines are more sensitive to these drug treatments, the cytotoxic combination (4.1 µg/ml BS extract and 6 µM enzalutamide) in LNCaP cell lines were predicted to be non-toxic to HEK293 cell lines.

Toxicity analyses. (a) Human MSC’s treated with BS and enzalutamide. (b) HEK293 cell line treated with BS and enzalutamide. The statistical significance of reduction in cell viability was calculated comparing to the control untreated cells and plotted. Data shown as mean ± SD for triplicate wells for 3 independent experiments.
P-Glycoprotein Expression in the LNCaP Cells
Serum starvation increased P-gp expression in the untreated case. It is also clear from Figure 8 that P-gp expression decreased with DHT addition or with addition of BS, enzalutamide or their combinations.

Flow cytometry analysis of P glycoprotein expression in LNCaP cells. (a) Flow cytometric analysis of the hormone treated (1 nM and 1000 nM DHT) LNCaP cells. (b) Mean values ± SD of 3 independent experiments were shown. Cells were treated with both BS extract and enzalutamide individually and in combination under different hormonal conditions and compared with the baseline expression (RPMI media with 10% FBS) of the cells.
Inhibition of Androgen Receptor Expression
As shown in Figure 9, addition of DHT increased cellular AR protein expression. When treated with BS, AR protein decreased and was inhibited at 25.6 µg/ml. Treatment with enzalutamide alone up to 12 µM did not show a significant difference in AR protein expression. It is evident that a combination of BS and enzalutamide was more effective at reducing AR expression, which is an important part of drug efficacy.

Protein expression. (a) and (b) depicts the AR & Akt expression in 1 nm DHT and 1000 nm DHT. The intensity of the pixels was measured using Image J software and was plotted against the respective drug concentrations. (c) and (d) shows the expression AR in 1 nM DHT and 1000 nM DHT respectively. (e) and (f) shows the expression of Akt in 1nM and 1000nM DHT respectively.
Micro-Explants Culture and Drug Sensitivity in Patient Samples
The cells derived from patient prostate samples were processed a micro-explant culture technique. 24 The explants were attached (Figure 10a to d) to the coated plates within 2 weeks.

Patient sample analysis. Micro-explant culture of prostate sample. (a-b) 2 week to 5 week, (c-d) 8 week to 12 week growth. (e-f) Individual drug treatments were done. (g) Cells were treated with drugs in combination of BS extract and enzalutamide. (h) Combination index.
From Figure 10, it evident that the effect of the drug varies from patient to patient. The action of the drugs and their combination was best for patient 3 Figure 10e to g and the synergistic action of the drug combinations was also highest for patient 3 (Figure 10h).
Discussion
When the drugs BS extract or enzalutamide were added, a 50% reduction in cell viability was only achieved at toxic levels of extract or enzalutamide (compare efficacy, Figure 2a and c, with toxicity, Figure 6). However, when both BS and enzalutamide were added together, DHT promoted their synergistic action (Figures 2 and 3); that is, the combined action of the 2 drugs was higher than that of the individual drugs, giving rise to efficacy at values of the individual drug concentrations much below their toxic levels. As shown in Figure 2, IC50 values were reached at only approximately 4 µg/ml BS and 6 µM enzalutamide compared to toxic values of 51 µg/ml BS and 96 µM enzalutamide. This is a significant result that argues in favor of using the extract in combination with the standard drug enzalutamide. This synergism was not observed to this extent when the active ingredient, AKBA, was used in combination with enzalutamide (Figure 2a and b). This may be attributed to the mixture of various triterpenes in the extract, see Table 1. These triterpenes have also been reported to also have anti-cancer properties. 25
Synergism was not observed when DHT was absent (Figure 3). Nor it was less when DHT levels were high (1000 nM). This is evidence that the combined action of the drugs may be through the androgen receptor. AR expression was significantly lower at 1 nM DHT when compared to 1000 nM DHT, which may explain why 1 nM DHT was more effective in helping synergistic action.
The beneficial role of the extract can be attributed to its suppression of AR expression and PSA production (see Figures 5 and 9). PSA production is related to androgen receptor activity in stimulating PSA gene expression downstream of AR. In the androgen-dependent phase of cancer, this AR activity is ligand dependent. However, the same activity can also be caused by ligand-independent AR activation in the androgen-independent phase of the cancer 26 leading to an increase in PSA. Thus, AR suppression by extract can be the basis for reduced PSA activity also. BSA was more effective than the drug enzalutamide in suppressing AR (Figure 9).
Conclusions
In summary, BS extract in combination with enzalutamide resulted in synergistic effects on LNCaP cells in the presence of DHT, which suppressed both AR expression and PSA secretion. The combination increased efficacy at concentrations much lower than the drug toxicity levels. The combination was also effective in reducing P-gp expression, thereby reducing drug efflux from the cancer cells, potentially increasing the efficacy of the action of enzalutamide. Use of BS extract may be considered for use in combination with enzalutamide in the androgen-dependent phase of prostate cancer. Importantly, the extract was more effective than the active ingredient, AKBA, in synergizing with the standard drug enzalutamide.
Footnotes
Acknowledgements
We are thankful to Dr.Ajit Nambiar, Head of the Pathology Department Amrita Institute of Medical Science, Cochin for identifying and helping in dissecting tumor sections from patient samples.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was generously supported by the Department of Science and Technology WOS (A) scheme, India [Grant No.: SR/WOSA/LS-1154/2014].
