Abstract
Cardiotoxicity as a result of cancer treatment contributes to autonomic dysfunction and decreased cardiorespiratory fitness among cancer survivors. These deleterious cardiovascular outcomes reduce the survival prognosis for cancer patients and contribute to poor quality of life among survivors. Exercise interventions have been shown as effective in mitigating treatment-related side effects. However, previously published interventions have not explored the potential for improvement in autonomic dysfunction (heart rate variability, HRV). This study examined cardiovascular adaptations in cancer survivors (n = 76) who participated in a 26-week intervention consisting of combined aerobic and resistance training (CART). The most noteworthy improvements occurred during the first 13 weeks of training and were maintained throughout the end of the 26-week period. HRrest improved from baseline (PRE) to the midpoint (MID) (P = .036) and from PRE to POST timepoints (P = .029). HRV and VO2max did not initially appear to change in response to CART. However, after stratification on time since treatment, participants who were 5 or more years from their last treatment experienced improvements (ie increase) in the HRV characteristic of HF power (P = .050) and also in VO2max (P =.043), when compared to those experiencing less than 5 years of time since their last treatment. These findings highlight a need for more attention to address the cardiorespiratory deficits experienced by those who have recently completed cancer treatment. In conclusion, the CART intervention is effective in improving cardiorespiratory fitness and autonomic dysfunction. The structure of the intervention is feasible for cancer survivors to continue with at home, using minimal resources, and without supervision. This at-home model may be even more acceptable to recent survivors that may be homebound immediately following treatment.
Keywords
Introduction
Cardiovascular disease ranks second as the leading cause of death among individuals who underwent cancer treatment. 1 Cardiotoxicity can be induced among cancer survivors by chemotherapeutic agents and by radiation.2,3 Cardiotoxicity appears as damage to the heart muscle, arrhythmias, cardiac ischemia, pericardial disease and heart failure. 4 As a result of cardiotoxicity, cancer patients and survivors may experience a disruption of vagal regulation (ie autonomic dysfunction) and a decrease in cardiorespiratory fitness.5,6 Regular exercise participation, including combined aerobic and resistance exercise, may promote adaptations in vagal tone and cardiorespiratory fitness among cancer survivors.
Disruption of vagal tone, also known as autonomic dysfunction, can be measured as a detrimental change in heart rate variability (HRV) and/or an increase in resting heart rate (HRrest). Changes in HRV and HRrest may not be perceived by an individual cancer survivor as an immediate concern, however, the long-term impact is deleterious and can contribute to increased mortality among cancer survivors as well as an increase in daily fatigue.3,6,7 Autonomic dysfunction is associated with reduced parasympathetic efficacy and increased sympathetic overdrive, which can negatively affect heart health by increasing cardiovascular strain and increasing the risk for premature death. 8 Analysis of HRV is the standard autonomic regulation assessment method and allows for the quantification of variances in the sinus rhythm.
HRV can be expressed using a frequency domain method as different spectrum powers to compare parasympathetic and sympathetic modulation. 9 Specifically, low frequency (LF) power (ms2) indicates both sympathetic and parasympathetic control and high frequency (HF) power is the “respiratory sinus arrhythmia” and indicates primarily parasympathetic control.10,11 Increases in HF power measurements have been observed to correlate with resting bradycardia (low resting heart rate), improved physical fitness, and a decreased risk for cardiovascular events.12,13 Given that increased parasympathetic control signifies improved heart health, an improvement in HRV is indicated by an increase in HF power (parasympathetic control) or a decrease in the LF/HF power ratio.9,14 Measures of HRV and HRrest have been associated with physical fitness in healthy populations, but vagal control adaptations in response to exercise in cancer survivors are not well understood. 15 The expected exercise-related improvements in vagal tone may be salient in cancer survivors given the long-term effects of cardiotoxicity on morbidity and mortality in this population.
In addition to vagal tone, disruption of cardiorespiratory fitness is often the most reported cardiovascular consequence of cancer treatment because of fatigue and the reduced ability to partake in activities requiring higher metabolic demands. 16 Further, cardiorespiratory fitness is a predictor of cancer-related mortality. 16 Cardiorespiratory fitness (VO2max) can be measured directly or indirectly, and the latter can be carried out by assessment of maximal effort or estimated from submaximal values. Using submaximal exercise is a well-established practice for assessment of older individuals and individuals with a history of chronic conditions, such as cancer.17,18 Previously published studies support the efficacy of aerobic exercise prescription in improving cardiorespiratory fitness among cancer survivors post-treatment. 19 However, less is known regarding the benefits of a combined aerobic and resistance training (CART) intervention among cancer survivors on cardiorespiratory fitness. HRV is understudied in general in cancer patients/survivors and, specifically, there is a paucity of research on the effect of CART on HRV in this population. Expanding the current understanding of cardiovascular adaptation among cancer survivors beyond HRrest and cardiorespiratory fitness to include HRV will provide oncologists, physiatrists, and clinical exercise physiologists with a more comprehensive assessment of the long-term cardiovascular prognosis for their patients. Furthermore, exploring the potential for changes in both vagal tone and cardiorespiratory fitness in response to a CART intervention will align with the intent of the current American College of Sports Medicine (ACSM) recommendations for weekly bouts of both aerobic exercise and resistance training.
The psychosocial and physiological benefits of regular exercise among cancer survivors are well documented, including improvements in quality of life, body composition, cardiorespiratory fitness (ie VO2max), and insulin sensitivity. 19 Initially, the majority of exercise interventions among cancer survivors focused on aerobic exercise and its relationship with improved cardiorespiratory fitness and body composition. 19 However, resistance training should be an integral component when designing exercise interventions for cancer survivors to combat the muscle atrophy associated with most cancer treatments. 20 Accordingly, the utilization of CART protocols have increased, with the majority of studies involving participants performing aerobic and resistance training on separate days or include aerobic exercise followed by a resistance training circuit of traditional strength exercises (ie leg press, chest press, seated row).21 -23 These CART interventions among cancer survivors have demonstrated significant improvements in cardiorespiratory fitness,24,25 yet there are potential limitations to the sustainability of a traditional gym-based CART approach. The gym-based protocols often include a high training volume (intensity and frequency) and participant time commitment, require specialized equipment, and are individualized which means that the important element of social support found in partnered or group-exercise is often missing. Furthermore, studies that have implemented more feasible training protocols such as body-weight resistance exercises (ie crunches, burpees), may not be suitable for cancer survivors with severe physical limitations. 26
Overall, there is a need to determine the efficacy of feasible CART protocols that are appropriate for cancer survivors at various post-treatment stages and that can be implemented in group or community-based settings. The pilot study presented herein aims to address this need by examining cardiovascular adaptations, including vagal tone, heart rate variability and cardiorespiratory fitness, among cancer survivors following a 26-week CART intervention administered in a group setting. First, we hypothesize that participants will experience significant improvements in vagal tone (ie HRV and HRrest) and cardiorespiratory fitness (ie VO2max) at mid- and post-intervention timepoints. Second, we hypothesize that in light of the cardiotoxic effects of treatment, those individuals with a shorter time since treatment cessation will experience less improvement in vagal tone and cardiorespiratory fitness compared to those who have reached the 5 or more years milestone for treatment cessation.
Methods
Overview of the Intervention
The Improving Physical Activity After Cancer Treatment (IMPAACT) Study used an exercise prescription of combined aerobic and resistance training (CART) with the addition of core strengthening and flexibility. Primary outcomes were measured at 3 timepoints over a 26-week intervention period and included baseline-to-midpoint and midpoint-to-post-intervention changes in vagal tone and cardiorespiratory fitness by measuring HRV (Low Frequency Power, High Frequency Power, LF/HF ratio), HRrest, and VO2max in cancer survivors. The study protocol was approved by the Institutional Review Board at Loyola Marymount University and the State of California Committee for the Protection of Human Subjects.
Participants
Cancer survivors were recruited using the Los Angeles Cancer Surveillance Program as well as convenience sampling and physician referral. Volunteers provided written informed consent to participate in the study and were asked to complete a questionnaire that used a closed-question structure to collect information pertaining to demographic characteristics, medical history, and health behaviors. Volunteers were excluded if at least 1 of the following conditions was present: (1) recurrent cancer or new primary diagnosis, (2) currently undergoing chemotherapy, (3) had suffered myocardial infarction or stroke within 1 year of enrollment date, (4) had history of fainting while exercising, (5) had history of chest pain or shortness of breath as a result of physical activity, and (6) had lymphedema or inflammation of lower extremities or pelvis. Prior to the intervention, participants were asked to inform their physicians of their planned participation and to review the exercise and assessment protocols.
Intervention Design
The IMPAACT Study incorporated a CART program consisting of one-hour sessions, offered 3 times per week, for 26 weeks at an academic facility. Each session included 20 minutes of cardiorespiratory training, 30 minutes of circuit-style resistance training and balance exercises, and 10 minutes of static and dynamic stretching. A Certified Clinical Exercise Physiologist® and other study personnel certified by the American College of Sports Medicine (ACSM) designed the program according to the ACSM Guidelines for Exercise for Cancer Survivors. 27 Participants wore heart rate monitors during each session and monitored their exertion based on prescribed individual intensity of the training heart rate zone. In combination with medical history and current level of physical activity and aerobic fitness, each participant’s target heart rate zone (% of heart rate reserve) was derived from the Karvonen formula, using age-predicted HRmax.
Participants engaged in cardiorespiratory training by either walking/jogging outdoors or using a stationary bike or elliptical machine indoors to reach their personalized target heart rate zone. The designated route for walking/jogging was mapped out for participants on an academic campus in close proximity to the academic circuit/strength/flexibility training facility and was without incline, obstacles, or trip hazards. The route was designed as a one-third of a mile loop with the beginning of the loop at the entry point to the academic training facility. Initial heart rate zone prescription for all participants was 35% to 50% depending on clinical/medical history and functional and physical ability as determined by baseline testing. Over the course of the study, heart rate zones were reviewed once weekly and adjusted every 2 weeks with an average change of 0% to 5%.
The circuit-training portion followed the cardiorespiratory training and included 10 stations of alternating upper and lower body resistance-based exercise, balance activities, and vibration plates. During the first 2 weeks of training, participants completed 2 sets of the 10 exercise stations for a total of 2 minutes per station with 60-second transition/rest periods between stations. Longer time at each station allowed for participants to familiarize themselves with exercise technique and allowed the trainers to determined most appropriate resistance/modality at each station. The training progressed toward 3 sets of the 10 exercise stations with each station performed for 45 seconds with a 20-second transition/rest period between stations. Variations in exercise intensity were provided at each resistance exercise station including different band strengths or dumbbell weights. Exercise intensity was monitored via heart rate and participants were encouraged to maintain their target heart rate zones throughout the cardiovascular and resistance training portions. Further, principles of overload and progression were considered and intensity levels (repetitions, sets, weight, and color of band or tubing) were recorded on participant’s exercise cards. Progression of intensity was no more than 5% and 10% for upper body and lower body exercises per week, respectively. Sample exercises included in the exercise circuit were resistance band rows, squats, forward/backward and lateral lunges and stepping, abdominal exercise variations, push-ups, or overhead press. Participants used resistance bands and tubing or light hand and ankle weights to increase the load. Balance exercises included a variety of movements that targeted visual, vestibular and somatosensory systems, postural control and strategies, as well as center of gravity control and gait pattern variations exercises. Two stations included vibration plates on which the participants progressed from standing with knees slightly bent to partial and full squats and calf raises based on tolerance. All exercises were designed to potentially mediate deficits in body composition, aerobic fitness, muscular strength, bone density and neuropathy, conditions known to be present among cancer survivors. 28
All participants were offered customized modification at each station depending on their physical ability. The circuit session concluded with a 10-minute period of approximately 5 core exercises performed for 2 sets for 30 seconds each (ie chair leg raises, curl-ups, seated side bends) followed by 5 minutes of static stretching of major muscle groups with participants choosing to stretch while seated or while standing with support from a chair.
Fidelity to the Intervention
Attendance was recorded during each exercise session to measure program adherence. Participants self-monitored prescribed heart rate zones by wearing heart rate monitors during the entire exercise session. Throughout the intervention the prescribed zones began at 35-50% of participants’ heart rate reserve, with the individual range increasing based on participant’s ability to maintain the intensity, their perceived exertion, and regular attendance. Participants strove to exercise continuously during the duration of each station while maintaining their intensity within the prescribed heart rate zones. The ratio of trainers-to-participants at each session was minimally 1:4. Trainers were a consistent source of encouragement and could regularly remind participants to check their heart rate monitors to maintain their exertion levels and to breathe rhythmically without holding the breath. Trainers also helped participants prepare to transition to the next station during circuit training. Participants were able to walk alone or with 2 or more walking buddies during the aerobic exercise portion. Participants were paired with 1 or 2 other people at each circuit station and the static and dynamic stretches were performed as a group.
For each session, 1 trainer held the responsibility of circulating among participants and recording heart rates and exertion levels at the beginning of the aerobic session and at the beginning of each lap that participants made on the designated walking/jogging route. These data were collected again at the end of the aerobic session and prior to the start of the circuit training, with participants being reminded to continue to walk around and keep their legs moving while they had a water break. At the end of the circuit training session and prior to the core/flexibility session another round of data collection occurred. The last point of data collection took place at the end of the core/flexibility session.
Outcome Measures
Vagal tone and HRV
Prior to testing, participants were instructed to abstain from caffeine-containing foods and drinks and other stimuli known to affect heart rate as well as vigorous exercise at least 24 hours before assessment and were asked to refrain from large meals 3 hours prior to testing. No participants were prescribed hormonal treatments over the course of the study and 6 participants were prescribed beta blockers. Participants completed exercise testing while on their prescribed medications and researchers ensured participants were on the same dose of medication at all testing timepoints. Further, participants completed testing at the same time of day for pre, mid, and post testing timepoints. Upon arriving at the laboratory, participants were familiarized with testing procedures. To assess resting heart rate, (HRrest), participants rested supine in a laboratory room with decreased sensory stimulation for a 10-minute period. Breathing was paced at 12 cycles of inhalations and exhalations per minute using a metronome to minimize the effect of respiratory influence. The 10-minute recording started once the participants comfortably synced their breath with metronome and a 7-minute period was eventually evaluated; it was selected by excluding recordings with large R-R interval outliers and by adhering to R-R interval equidistance. 29 HRrest was determined as the lowest number of beats per minute (bpm) during the 10-minute period and was recorded from a Polar RS800CX (Polar Electro, Lake Success, NY) training computer. R-R intervals were measured simultaneously on the same device and were then transferred to the Kubios software package (version 2.2) for analysis of HRV. 30 HRV data was pre-processed within Kubios to automatically remove artifacts and ectopic beats using a medium (0.25 seconds) threshold value for each participant. Specific outcome measures of HRV include LF power (ms2), HF power (ms2), and LF/HF ratio (ms2).
Cardiorespiratory fitness
Cardiorespiratory fitness (VO2max) was estimated from a submaximal YMCA protocol on a cycle ergometer. 31 The YMCA test has demonstrated strong criterion validity against a maximal treadmill test in adults (r = 0.77). 31 A validated submaximal exercise test was chosen for this study instead of a maximal exercise test because of participants’ history of cancer and cancer treatment and self-reporting of reduced exercise capacity and physical functioning during recruitment and screening. 32 The YMCA test is an incremental exercise test during which the heart rate response is logged in response to progressive resistance load at a cadence of 50 revolutions per minute (rpm). The test concludes successfully when the participant reaches 2 steady-state periods marked by heart rate readings of no more than 6 beats apart per minute within an intended range of 110 bpm and 85% of the age-predicted maximal heart rate. Participants maintained a cadence of 50 rpm during a cool-down period.
Statistical Analysis
Descriptive statistics are given as mean ± standard deviation for participant characteristics or mean with 95% confidence intervals for outcome measures, and number with percentage for qualitative variables. Normality was verified for all variables using histograms and box-plots. Time since treatment was collected as a continuous variable (years) and coded as a categorical variable (<5 years and ≥5 or more years).33,34 Given the emphasis on the cardiotoxicity associated with chemotherapy and radiation, the treatment variable was collapsed into a two-category variable of chemotherapy and/or radiation exposure versus no chemotherapy/radiation exposure. Mann-Whitney U test procedure was used to examine whether there was any difference in the change of each outcome at different timepoints by race and treatment type. Separate repeated measures analysis of variance (ANOVA) tests were used to determine changes in outcome measures across the 3 timepoints (PRE, MID, and POST). Sample size for each analysis may vary as a result of incomplete assessment data. Post hoc tests were done on least-squares means using a Tukey adjustment for multiple comparisons. Log transformation was used when the assumptions were not met. For outcome measures that improved significantly across time points, a Pearson Chi-square test was performed to determine the proportion of participants that improved measures of cardiovascular function from PRE to MID and from PRE to POST timepoints for groups attending ≤50% of sessions and those attending >50% of sessions throughout the intervention. All statistical tests were performed at an α = 0.05 significance level. Statistical analyses were performed using SPSS (Version 26; IBM Corporation 1989, 2019) and SAS (Version 9.3; SAS Institute Inc.).
Results
Demographics
Participant demographics for the pooled cohort (n = 76) at baseline are shown in Table 1. Demographics for the 2 individual cohorts have been published previously. 34 Participants had a mean age of 62.9 ± 10.1 years and reported their primary cancer site as follows: breast (39.5%), endometrial (15.8%), gynecological (10.5%), colorectal (9.2%), Hodgkin’s lymphoma and uterine (5.3% each), melanoma and thyroid (3.9% each), prostate (2.9%), and other (12.9%). At baseline, our sample was characterized as overweight with an average BMI of 28.5 ± 6.4 kg/m2.
Participant Characteristics at Baseline for the IMPAACT Study (n = 76).
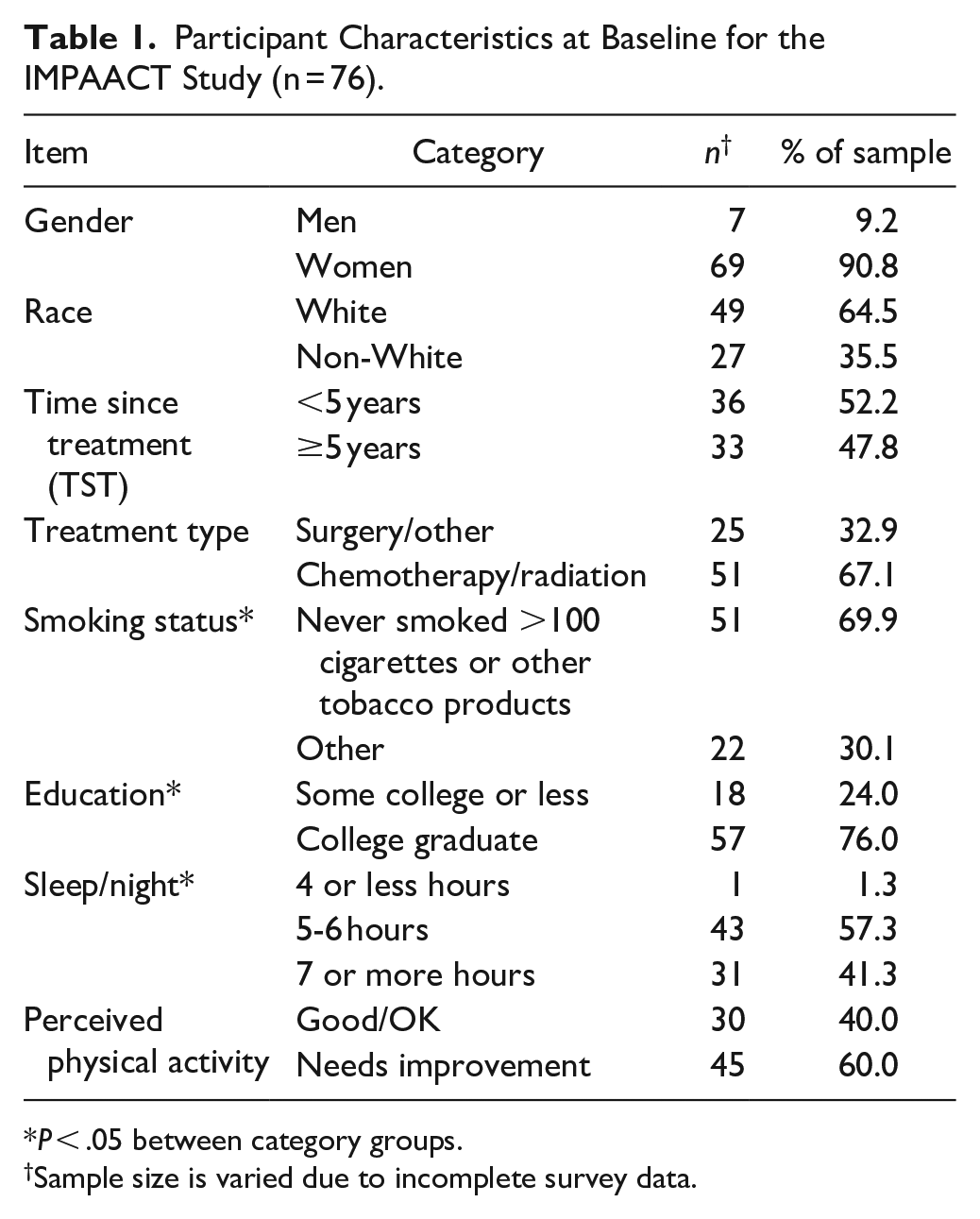
P < .05 between category groups.
Sample size is varied due to incomplete survey data.
Intervention Participant Adherence
Average exercise session attendance was 59.0 ± 18.1%, with the lowest attendance rate at 11.3% and highest rate at 96.3%. Reasons for lack of attendance included musculoskeletal issues, injuries/accidents unrelated to exercise intervention, vacations, return to treatment, or death unrelated to the exercise intervention. Number of sessions attended (<50% vs ≥50%) in the intervention was similar between White and non-White participants (P = .099).
Intervention-Associated Changes in Outcome Measures
Participant changes in outcome measures from baseline/0 weeks (PRE), midpoint at 13 weeks (MID), and post-intervention at 26 weeks (POST) are summarized in Table 2.
Change in Outcome Measures at Baseline, 13 weeks, and 26 weeks of an Exercise Intervention in Cancer Survivors.

Data reported as mean (95% Confidence Interval): bpm = beats per minute. Sample size may vary due to the inability of a small number of participants to complete the assessment battery.
Based on log transformed variable.
Significant difference between PRE and MID and PRE and POST (P < .05).
Vagal tone and HRV
HRrest improved from PRE to MID (P = .036) and from PRE to POST timepoints (P = .029) (Table 2). These improvements were not modified by treatment type (P = .965) or race (P = .488). When HRrest was examined across categories of participation frequency (<50% sessions attended vs ≥50% sessions attended), there was no modification of the observed change in HRrest from PRE to MID (P =.703) or from PRE to POST timepoints (P = 1.000) (Figure 1). HRrest was also examined across categories of time since treatment (<5 years TST, ≥5 years TST) and data are presented in Figure 2. A larger percentage of participants in the ≥5-year TST group experienced an improvement (ie decrease) in HRrest from PRE to MID timepoints, as compared to the <5 years TST group, however, this difference was not statistically significant (P = .778).
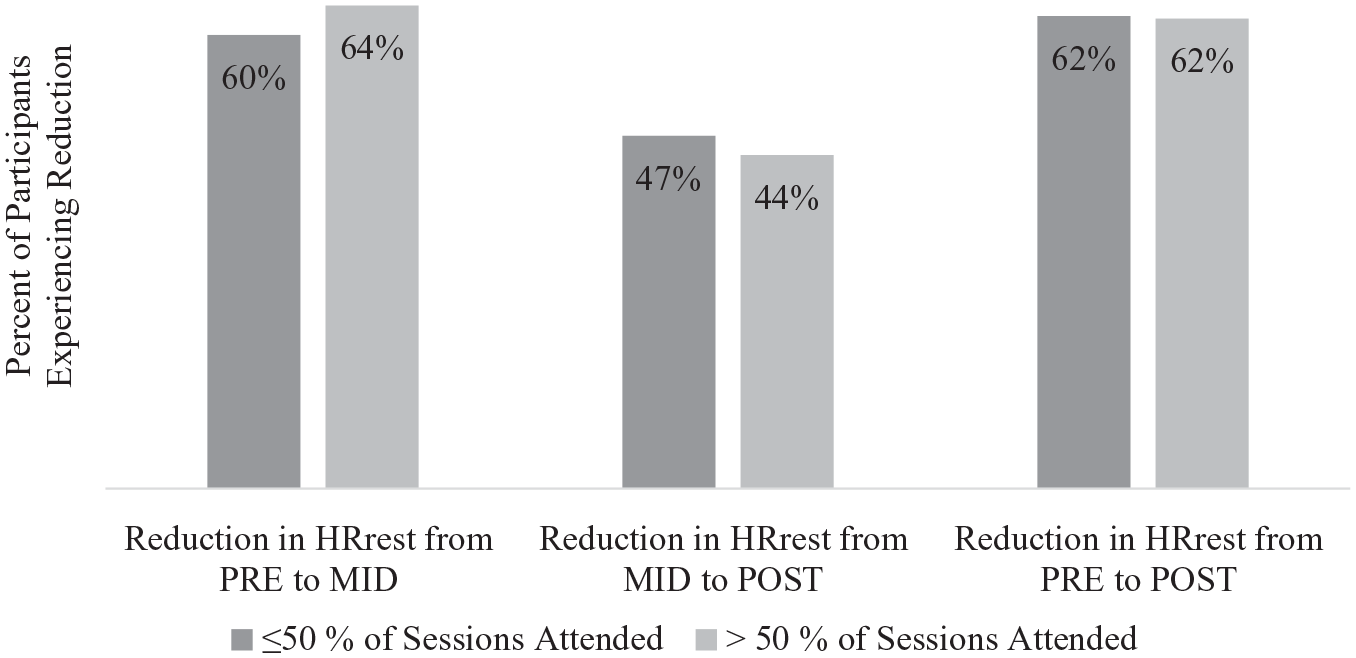
Percent of participants experiencing a reduction in resting heart rate (HRrest) from baseline (PRE) to 13 weeks (MID), from MID to 26 weeks (POST), and from PRE to POST for participants attending ≤50% of sessions compared to participants attending >50% of sessions.
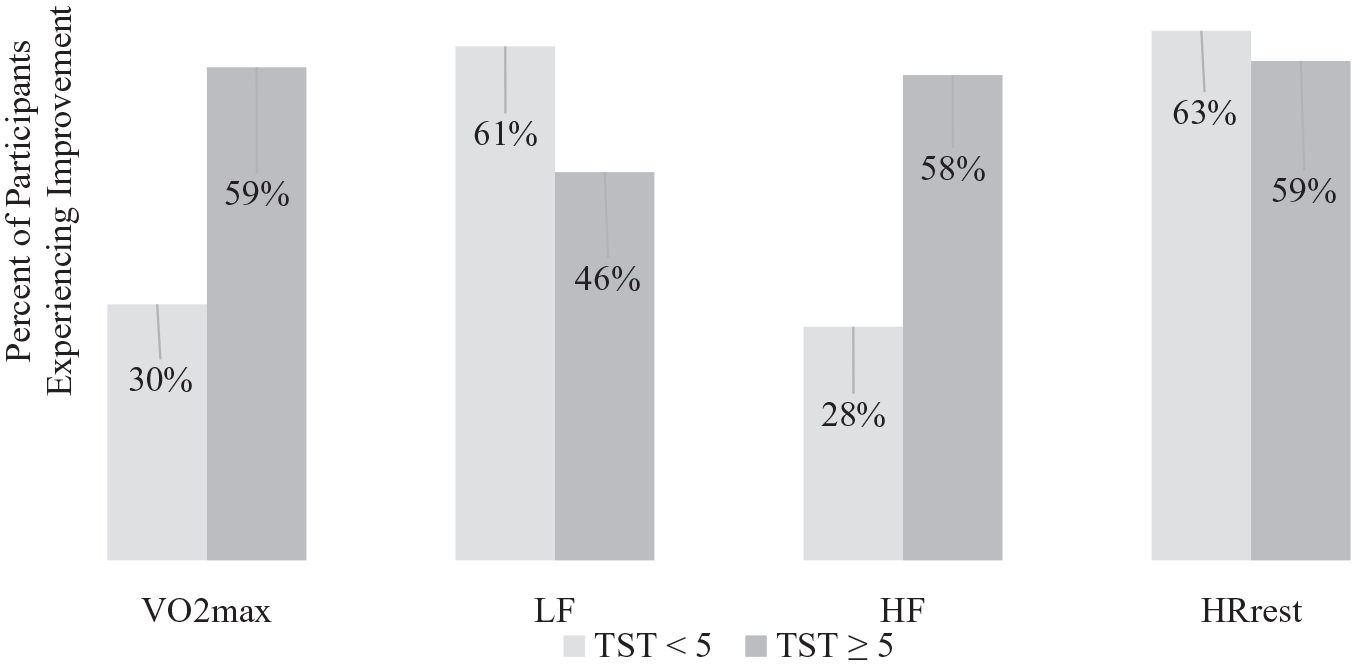
Percent of participants who demonstrated improvement in outcome measures from baseline (PRE) to 13 weeks (MID) for participants within 5 years of treatment (TST < 5 years) compared to participants who received their last treatment 5 or more years prior to study onset (TST ≥ 5 years).
With regard to HRV outcomes (Table 2), there was no observed change in LF power, HF power, or the LF/HF power ratio from PRE to MID or from PRE to POST (P > .050). Treatment type did not modify the observations for HF power (P = .710) or LF power (P = .678). When race was considered, a suggested change was noted for HF power (P > .075) between PRE to MID, but not for LF power (P > .394). When time since treatment was considered, a change was observed for HF power (P = .050), reflecting that a greater number of participants in the ≥5 year TST group experienced improvements (ie increase) in HF power between PRE and MID timepoints compared to cancer survivors in the <5 year TST group (Figure 2). There was no observed difference in the number of participants demonstrating improvements (ie decrease) in LF power between TST groups (P = .329) (Figure 2).
Cardiorespiratory fitness
Changes in VO2max were not apparent from PRE to MID or from PRE to POST timepoints (P >.05) (Table 2). VO2max across timepoints was not modified by race (P = .657) or treatment type (P = .732). In a Chi Square analysis of time since treatment, more participants in the ≥5-year TST group experienced improvements in VO2max (P = .043) compared to participants in the <5-year TST group (Figure 2).
Discussion
This pilot study examined cardiovascular adaptations (ie HRV and cardiorespiratory fitness) following a 26-week combined aerobic and resistance training (CART) intervention, implemented in a group setting. Improvements in outcomes were most prominent between the PRE to MID timepoints, suggesting that the initiation of participation in CART was most beneficial during the first 13 weeks of the intervention. Changes in outcomes did not reach statistical significance from MID to POST, which indicates that participants may have experienced a plateau effect in their training adaptations. However, improvement in HRrest was progressed during MID to POST timepoints and the improvements in several outcomes from the PRE to MID training period were sustained during the MID to POST training period.
Improvements in Vagal Tone
HRrest improved from PRE to MID timepoints, demonstrating improved cardiovascular function following our CART intervention. Interestingly, improvements in HRrest did not vary by treatment type, race, or time since treatment, suggesting that our CART program may be sensitive to elicit improvements in cancer survivors independent of their time since treatment status. Specifically, the decrease in HRrest observed may be attributed to an improvement in parasympathetic modulation and/or improvements in cardiorespiratory fitness in our sample of cancer survivors. 11 However, it is difficult to attribute potential mechanisms to explain or our findings given that there were no changes initially observed for cardiorespiratory fitness or HF power (parasympathetic control) or LF power (parasympathetic/sympathetic control) across timepoints and no variation with treatment type, time since treatment (cardiorespiratory fitness only), or race (LF power only). Nonetheless, the variation suggested with race for HF power should be explored further to separate potential biological characteristics of cardiovascular adaptation and race from external characteristics (eg psychosocial stressors) that may influence parasympathetic control and time since treatment.
In the current study, participants in the ≥5-year TST group experienced the greatest improvement in HF power, likely because they are further from the cardiotoxic effects of treatment. Similar to our findings, Caro-Morán et al 15 examined vagal tone in breast cancer survivors who were less than 1 year from treatment cessation and demonstrated significantly lower HF power among cancer survivors compared to age-matched controls. These differences in HRV have been frequently attributed to cancer therapies that include the anthracycline- and trastuzumab- containing regimens, or to new adjuvant cytotoxic (taxane-containing) agents and hormonal regimens (aromatase inhibitors) in which several of the participants in the present study completed. 35 However, Cramer et al 36 found that patients with colorectal cancer had compromised vagal tone regardless of whether they underwent chemotherapy or not, suggesting that a different mechanism might negatively affect autonomic function. For example, vagal tone disparities may resolve over time with a washing out of treatment effects which may explain why participants longer from treatment experienced greater improvements in parasympathetic control. Findings from this current pilot study are consistent with the aforementioned studies in that: (a) the effect of treatment appears to lag well into the survivorship period with an eventual washout of cardiotoxicity after 5 or more years, (b) treatment type does not appear to modify vagal tone and perhaps there is another determinant of the cancer experience that is influencing HRV. Another potential mechanism that may explain the lack of effect of exercise on vagal tone is the intra-individual variability in daily HRV measures. For example, participants completing cancer treatment more recently may experience greater variability and therefore using an isolated daily HRV measurement as in the present study may have confounded the results. Therefore, future research investigating the effects of exercise training on HRV among cancer survivors may complete more robust HRV analyses including weekly coefficients of variation as performed in athlete monitoring investigations 37 to account for the high variability in recent cancer survivors.
Improvements in Cardiorespiratory Fitness
Our study demonstrated no changes for VO2max across timepoints and no variation by race or treatment type. However, similar to certain vagal tone outcomes, we observed a better outcome for those survivors that are further from the cardiotoxic effects of treatment. In contrast to our findings, Dieli-Conwright and colleagues demonstrated an increase in estimated VO2max in breast cancer survivors following a 6-month aerobic and resistance training intervention with a prescribed exercise intensity of 65-85% heart rate maximum. 22 The inability to detect a change in cardiorespiratory fitness in the current study may be attributed to our sample size and/or to our use of a submaximal protocol for assessing VO2max. For example, some participants were unable to match their efforts during the MID or POST YMCA submaximal bicycle tests due to physical limitations at the time of assessment. Further, prescribed intensity for aerobic exercise was individualized based on the current health status and physical limitations of the participant, and the intensity may not have been sufficient to elicit significant improvements in VO2max comparable to previous studies that utilized a higher exercise intensity. 22
Limitations and Strengths
This pilot study’s sample size is small, however, it remains comparable in size to other studies examining exercise interventions in cancer survivors.38,39 The study is designed as a prospective analysis of a single intervention group and is limited by the absence of a control group. The study is strengthened by a relatively long intervention period of 26 weeks, which allows for longitudinal assessment of individual progress at multiple, meaningful timepoints. Our sample was heterogeneous in terms of types of cancer site, however, researchers have previously reported that cancer type did not affect exercise-mediated cardiovascular outcomes. 40 Further, this study examined a more feasible CART program implemented in a group-based setting compared to traditional gym-based CART programs requiring specialized equipment and a high training volume. 22
The midpoint assessments took place at the conclusion of the first 13-week training period and were followed by a month-long break in supervised training due to the academic training setting and respect for observance of religious/ethnic holidays. Participants were provided with at-home instructions for the CART intervention in order to maximize intervention fidelity. The intervention at the academic facility was designed to be adaptable to a home setting and without reliance on specialized equipment or spaces. As such, the at-home adaptations were identical to those in the academic facility and participants were already trained in the intervention protocol by participating for the first 13 weeks. Participants were given resistance bands and hand weights to use at home, along with a printed intervention protocol guidebook with pages to record daily progress and HR monitor readings, and their own Polar watches/chest straps. The month away from the supervised program may have contributed to a lack of improvement in outcomes during the MID to POST timepoints, but it did not have a deleterious effect on the gains from the PRE to MID timepoints. Participant improvements noted at MID were maintained throughout the second 13 weeks of the training program (MID through POST).
Further, leg fatigue or arthritic related complaints were the most common reasons reported by the participants who could not successfully finish YMCA submaximal test, which resulted in unequal sample sizes at the assessment timepoints. Our assessment experience raises a question regarding the best way of determining cardiorespiratory fitness in a population of cancer survivors. It is challenging to accommodate the varied ability levels coupled with a wide age range in a highly heterogeneous group. Whereas the walking mode can serve participants with higher mobility or those unable to stay seated for longer periods of time (eg patients treated for colorectal cancer), cycling provides a safer mode for cancer survivors suffering from treatment-related balance issues. 41
Conclusion and Practical Implications
This pilot study examined the effects of a CART intervention on both cardiorespiratory fitness and vagal tone in cancer survivors. It is worth noting that the CART intervention was acceptable to older cancer survivors that could be characterized as overweight and that self-reported a need to improve their physical activity levels. Attendance among participants varied, however, frequency of participation did not significantly change the intervention efficacy. This may be due to the ease at which participants could self-monitor fidelity to the intervention using a heart rate monitor and basic Likert scale assessment of exertion, regardless of the type of exercise they were engaged in. In addition to the observed improvements in resting heart rate for cancer survivors in response to CART, this study also demonstrates potential synchronicity between improvements in cardiorespiratory fitness and vagal tone (HF power/parasympathetic tone).
This study identifies a potential health disparity in the gains in both cardiorespiratory fitness and vagal tone as a result of proximity to treatment. Based on our findings, future studies should focus on exercise prescriptions that might be tailored to cancer survivors who have recently completed treatment in order to reduce vagal tone disparities in a safe and efficacious manner. During recovery and after cessation of physical activity, sympathetic withdrawal coupled with increased vagal input helps the body return to a resting state. 42 HRV-guided training can help to individualize exercise prescriptions not only for athletes but also for clinical populations. Enhanced levels of vagal input before an exercise bout as well as sufficient recovery state, that is, fully recovered vagal tone, both lead to better training response. 43 Whereas the current exercise guidelines for cancer survivors are comprehensive, the protocol does not include the use of HRV as part of individualized exercise prescription. As the scientific community continues to explore the most effective forms of physical activity interventions, HRV lends itself to aid this process. 38
The potential use of an at-home program as a “maintenance therapy” after an initial supervised intervention increases the feasibility and scalability of CART to improve cardiovascular adaptations among cancer survivors. At-home interventions may also be implemented on virtual platforms to encourage partnered or group exercise while mitigating transportation barriers and restrictions on social distancing if a cancer survivor is experienced immune compromise as a treatment-related side effect.
Footnotes
Acknowledgements
Silvie Grote was affiliated with Loyola Marymount University as a Visiting Assistant Professor during study design and data collection and with Loma Linda University as a Doctoral Candidate during data analysis. Jeanette M. Ricci was affiliated with Loyola Marymount University as an Undergraduate Student during data collection.
We would like to sincerely thank the participants of the Loyola Marymount University IMPAACT Study. We are grateful for our research collaborators, Hawley Almstedt, Stephanie Perez Beaudion, Todd Shoepe, and Sarah Strand. We would also like to thank the following divisions and programs at Loyola Marymount University for their support of student research assistants: Frank R. Seaver College of Science and Engineering, the McNair Scholars Program, the Rains Research Assistant Program, and the Summer Undergraduate Research Program.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by the Tower Cancer Research Foundation’s Community Partner Grant (award period: 1/1/15-12/31/15) and by an award granted by the Charitable Division of the QueensCare Foundation (award period: 1/1/15-12/31/15). The content is solely the responsibility of the authors and does not necessarily represent the official views of the National Cancer Institute or the National Institutes of Health. Tarleton received funding to cover the cost of research supplies and exercise equipment from the Tower Cancer Research Foundation (Community Partner Grant) and from the QueensCare Foundation (Charitable Division). These funding sources did not provide salary support for the principal investigator and other key personnel.
Ethical Approval
The study was approved by the Loyola Marymount University Institutional Review Board (LMU IRB 2014 SP 27 and LMU IRB 2015 SP 23) and the California Health and Human Services Agency Institutional Review Board (Protocol ID 14-02-1507) in accordance with the Helsinki Declaration of 1975, as revised in 1983, and the Declaration of the World Medical Association.
