Abstract
Dichloroacetate (DCA) is a metabolic modulator that inhibits pyruvate dehydrogenase activity and promotes the influx of pyruvate into the tricarboxylic acid cycle for complete oxidation of glucose. DCA stimulates oxidative phosphorylation (OXPHOS) more than glycolysis by altering the morphology of the mitochondria and supports mitochondrial apoptosis. As a consequence, DCA induces apoptosis in cancer cells and inhibits the proliferation of cancer cells. Recently, the role of miRNAs has been reported in regulating gene expression at the transcriptional level and also in reprogramming energy metabolism. In this article, we indicate that DCA treatment leads to the upregulation of let-7a expression, but DCA-induced cancer cell death is independent of let-7a. We observed that the combined effect of DCA and let-7a induces apoptosis, reduces reactive oxygen species generation and autophagy, and stimulates mitochondrial biogenesis. This was later accompanied by stimulation of OXPHOS in combined treatment and was thus involved in metabolic reprogramming of MDA-MB-231 cells.
Introduction
Breast cancer is a leading cause of death worldwide among women. Because of the heterogeneity and activation of different metabolic pathways, breast tumors sustain progression and evasion from various chemotherapies. Cancer cells, compared with normal cells, have different energy requirements as they grow more aggressively than normal cells and are more dependent on glycolysis than oxidative phosphorylation (OXPHOS) and generate more metabolic substrates to substitute other metabolic pathways, pentose phosphate pathway, and gluconeogenesis to meet their energy demand. 1 Warburg proposed that enhanced glycolysis is due to dysfunctional mitochondria or mutations in the electron transport chain, and as a result, cancer cells switch from the regular source of energy from OXPHOS to glycolysis, thus supporting tumor proliferation and eluding programmed cell death. 2 Recently, breast cancer has been considered a metabolic disorder that can be targeted by metabolic reprogramming and thus promoting programmed cell death. Furthermore, metabolic reprogramming is connected with the organization or morphology of the mitochondria. Focusing on metabolic modulators can be considered as an effective strategy to inhibit tumor proliferation and to initiate apoptosis. 3
Dichloroacetate (DCA) is a mitochondrial targeted generic drug that can penetrate tissues after oral administration and is also a metabolic modulator to treat lactic acidosis. 4 DCA is a regulator of pyruvate dehydrogenase (PDH), promoting influx of pyruvate formed after glycolysis into the mitochondria where the tricarboxylic acid cycle occurs. 5 DCA inhibits the phosphorylation of PDH by PDH kinase because the activated form of PDH kinase prevents the entry of pyruvate formed after glycolysis into mitochondria. There are several reports suggesting the antitumor behavior of DCA in different cancer cell lines, such as inhibiting gluconeogenesis, lipogenesis, and cholesterol biogenesis by targeting multiple pathways engaged in generating oncometabolites. 6 It has also been reported that the efficacy of DCA improved when used in combination with other drugs.7,8
MicroRNAs (miRNAs) are evolutionarily conserved (21-24 nts) small noncoding RNAs that are known to modulate the expression of target messenger RNA by binding to its 3′ UTR. 9 They can either inhibit translation or degrade the target mRNA. miRNAs are not only crucial in development but also regulate metabolic reprogramming, differentiation, calcium homeostasis, and other physiological processes. 10 Recently, dysregulation of miRNAs is associated with onset of various diseases, including cancer. Let-7a is a known tumor suppressor miRNA found to be downregulated in primary and metastatic tumors, and its loss or downregulation is associated with cancer aggressiveness. 11 Let-7a modulates the expression of cancer cells by targeting MYC, HMGA2, RAS, E2F6, and other genes in different cancers. 12 Currently, it has been shown that drugs, when delivered or used in combination with this gene, can be considered as a tool in malignancies. 13 Therefore, in the current study, DCA was used in combination with let-7a to produce more antitumor behavior by targeting metabolic reprogramming and apoptosis. 14
Material and Methods
Cell Culture and Treatments
The MDA-MB-231 cell line was procured from the National Centre for Cell Science, Pune, India, and was cultured in Dulbecco’s Modified Eagle’s Medium (DMEM) supplemented with 10% fetal bovine serum and 1% penicillin-streptomycin in a 37°C, 5% CO2 humidified incubator. The cells were treated as per the requirement of the experiment. To generate ρ0 cells, 50 µg/µL of uridine was added in culture media and provided long-term exposure of ethidium bromide (EtBr; approximately 20 days exposure at a concentration of 25 µg/mL). 15
For transfection of miRNA mimics and antisense, appropriate numbers of cells were seeded in 30-mm dishes and transfected in serum-free media with 10 nM of let-7a mimic and antagomirs, using lipofectamine 2000 reagent (Invitrogen, Carlsbad, CA).
For 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyl tetrazolium bromide (MTT) assay, 7000 cells/well were seeded in a 96-well plate for 24 hours, followed by various treatments as detailed in individual experiments for indicated time points. The treated media was replaced with MTT solution (0.5 mg/mL in 1× phosphate-buffered saline [PBS]) followed by incubation in the dark at 37°C for 3 hours. Formazan crystals were then dissolved in dimethyl sulfoxide (DMSO), and absorbance was taken at 570 nm. 16
Real-Time Quantitative PCR
Total RNA was extracted using TRIZOL reagent from MDA-MB-231 cells. Concentration and purity of the samples were determined by the NanoDrop spectrophotometer. RNA was reverse transcribed by using 200 ng of total RNA, and the real-time polymerase chain reaction (PCR) was performed using Eva Green (CFX Connect, Bio-Rad, Hercules, CA). A reaction mixture was prepared by adding 100 nM each of forward and reverse primers, 2 µL of 5-fold diluted cDNA, 5 µL of 5× SYBR Green, and then the volume of 10 µL was maintained by nuclease-free water. The reaction was initiated at 95°C for 5 minutes with 10-second denaturation; annealing and extension were carried out at 58°C and 72°C for 1 minute and 30 seconds, respectively. 17 The data were normalized to β-Actin.
Fluorescent Staining
To support the combined effect of DCA and let-7a on mitochondrial mass, MDA-MB-231 cells were stained with MitoTracker Green (localized to mitochondrial membrane, hence used for determining mitochondrial mass) and analyzed using flow cytometer and confocal microscopy. 18
Furthermore, the reactive oxygen species (ROS) level was determined after incubating the MDA-MB-231 cells with 5 µM H2DCFDA (Invitrogen, Molecular Probe) for 30 minutes in the dark. After incubation, cells were washed with 1× PBS and harvested for flow cytometer analysis. 19 For the analysis of each sample, 50 000 events were collected, and data were analyzed.
Mitochondrial membrane potential was determined using JC-1 fluorescent dye (Molecular Probe, Invitrogen) and stained with 1 µM JC-1 for 30 minutes at room temperature. Fifty thousand cells or events were analyzed using a flow cytometer with the laser 488 and emission filter 525/50 nm for the green signal and laser 561/582 nm for red signal. 20
Mitochondrial-Specific Fluorescent Staining
MDA-MB-231 cells were stained with MitoTracker Green (localized to the mitochondrial membrane, hence used for determining mitochondrial mass) and analyzed using flow cytometer and confocal microscopy.
Annexin V/Propidium Iodide
Cells were propagated as described above and treated with 25 mM DCA and transfected with a mimic of let-7a for 24 hours. After harvesting, cells were washed with 1× PBS. Further staining was carried out as per the manufacturer’s instruction. Finally, different populations were distinguished by flow cytometer, and data were analyzed.
Determination of Total ATP Content
Treated and transfected MDA-MB-231 cells were trypsinized and centrifuged. Total ATP content was determined by resuspending cells in NP40 lysis buffer containing glycerol. The samples were boiled for 5 minutes at 95°C, and ATP concentration was determined by luciferase and luciferin bioluminescent kit (Invitrogen).
Determination of Autophagy
MDA-MB-231 cells were seeded, and after 24 hours of seeding, cells were treated and transfected with above-mentioned concentrations. Cells were harvested after 24 hours and processed for determining autophagy as per the manufacturer instructions (Invitrogen).
Western Blot Analysis
MDA-MB-231 cells were treated and transfected with the above-mentioned protocol, and after 24 hours of treatment and transfection, cells were harvested and washed with 1× PBS. Protein lysates were prepared using RIPA lysis buffer and quantified by using Bradford reagent. The 40 µg of protein sample were subjected to SDS-PAGE on 10% and 12% gel, which were then transferred onto nitrocellulose membrane. The membranes were probed overnight with primary antibodies p53 (1:1000, Invitrogen), Bax (1:1000, Invitrogen), AMP-activated protein kinase (APMKα; 1:1000; Sigma, St. Louis, MO), peroxisome proliferator–activated receptor γ coactivator 1-α (PGC-1α; 1:1000; Sigma), MFN2 (Mitofusin 2; 1:1000; Sigma), and β-Actin (1:3000; Sigma). After overnight incubation, membranes were subsequently washed with PBS with tween 20 and incubated with horseradish peroxidase–conjugated secondary antibodies (1:10000). β-Actin was taken as housekeeping gene. 21
Oxygen Concentration Measurement
Measurement of mitochondrial respiration is essential to gain insight into mitochondria-mediated metabolism and can be accomplished by measuring the oxygen consumption rate using high-resolution oxygraph. Respiration of permeabilized MDA-MB-231 cells was investigated by substrate-uncoupler-inhibitor titrations (SUIT) protocol with slight modifications. Approximately 1.5 × 106 cells were added in the oxygraph chamber containing serum-free DMEM media. Sixteen microliters (8 µM) of digitonin was injected into an oxygraph chamber containing cell suspension for permeabilization, and respiration was recorded for 10 minutes. Malate and succinate were added in the presence of the complex-I (CI) inhibitor rotenone to determine CI- or CII-linked respiration. ADP (adenosine diphosphate) was added at 2.5 mM to obtain OXPHOS capacity. After adding substrates and inhibitors, respiration was recorded for 10 minutes each until the signal becomes stable. 22
Results
DCA Induced Mitochondria-Independent Cell Death in Cancer Cells Under Normoxia
Dichloroacetate is a small molecule targeting mitochondria that regulate the metabolism of cancer cells. 4 To explore the anticancer activity of DCA, we first investigated the effectiveness of DCA in inhibiting cell proliferation under normoxia and hypoxia and whether DCA activity was dependent on mitochondria or not. To assess the stated hypothesis, ρ0 (mitochondrial DNA [mtDNA]-deficient cells) of MDA-MB-231 cells were generated, and both wild-type (WT) and ρ0 MDA-MB-231 cells were treated with different concentrations of DCA followed by an assessment of cell viability using the MTT assay. The data obtained after performing the MTT assay showed IC50 values of 25 mM in WT MDA-MB-231 cells and 5 mM in ρ0 cells (Figure 1A and B). The ρ0 cells showed more sensitivity to DCA at lower concentrations as compared with WT cells, indicating that mitochondria are central to the survival of cancer cells on DCA treatment. Interestingly, under hypoxic conditions, the results were precisely opposite where WT cells were more sensitive to DCA compared with ρ0 cells, though IC50 values were more than 35 µM in WT cells. It is well documented that the tumor hypoxia promotes an aggressive phenotype and chemotherapeutic failure; thus, it can be concluded that DCA loses its efficiency under hypoxic conditions (Figure 1C and D). In ρ0 cells, DCA does not inhibit the cancer cell proliferation indicating the importance of mitochondria in its effect.
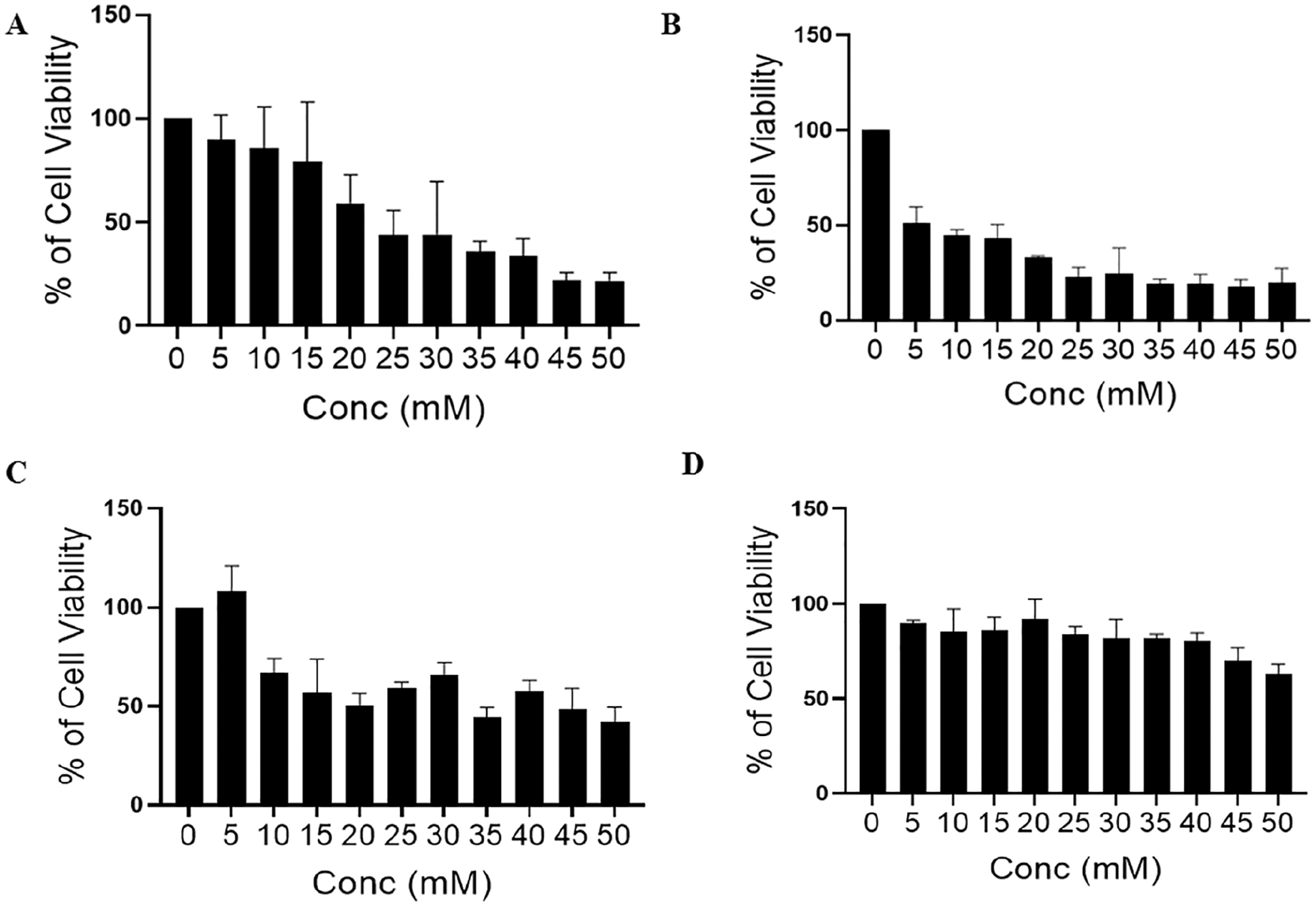
Effect of dichloroacetate (DCA) on cell viability was evaluated by MTT assay. (A and B) Showing the effect of DCA on wild-type (WT) and ρ0 MDA-MB-231 cell viability, respectively, treated with different concentrations of DCA (0, 5, 10, 15, 20, 25, 30, 35, 40, and 45 mM), and after 24 hours of treatment, MTT assay was performed in normal condition. (C and D) Cell viability assessed by MTT assay in hypoxia in WT and ρ0 MDA-MB-231 cells, respectively.
Camptothecin, etoposide, doxorubicin, and 5-fluorouracil (5-FU) are known to inhibit cell proliferation and induce apoptosis in cancer cells. 23 We thus further combined the treatment of DCA with the above-mentioned drugs, and cell viability was assessed. The viability of MDA-MB-231 cells was determined after pretreating and posttreating cells with different concentrations of DCA and 5-FU at different time points. It was observed that DCA inhibits cell proliferation by 50% at 25 mM, whereas the same percent of inhibition was observed at 5 µg and 10 µg of 5-FU. Furthermore, cells were pretreated with DCA and 5-FU for 6 hours followed by 5-FU and DCA for 24 hours, respectively, to determine whether they increase the sensitivity of each other or not. The results obtained showed that both drugs are neither synergistic nor additive but act independently of each other (Figure 2A). Similar results were obtained when DCA was used in different combinations with other drugs such as doxorubicin and camptothecin as observed under normoxia and hypoxia (Figure 2B and C).
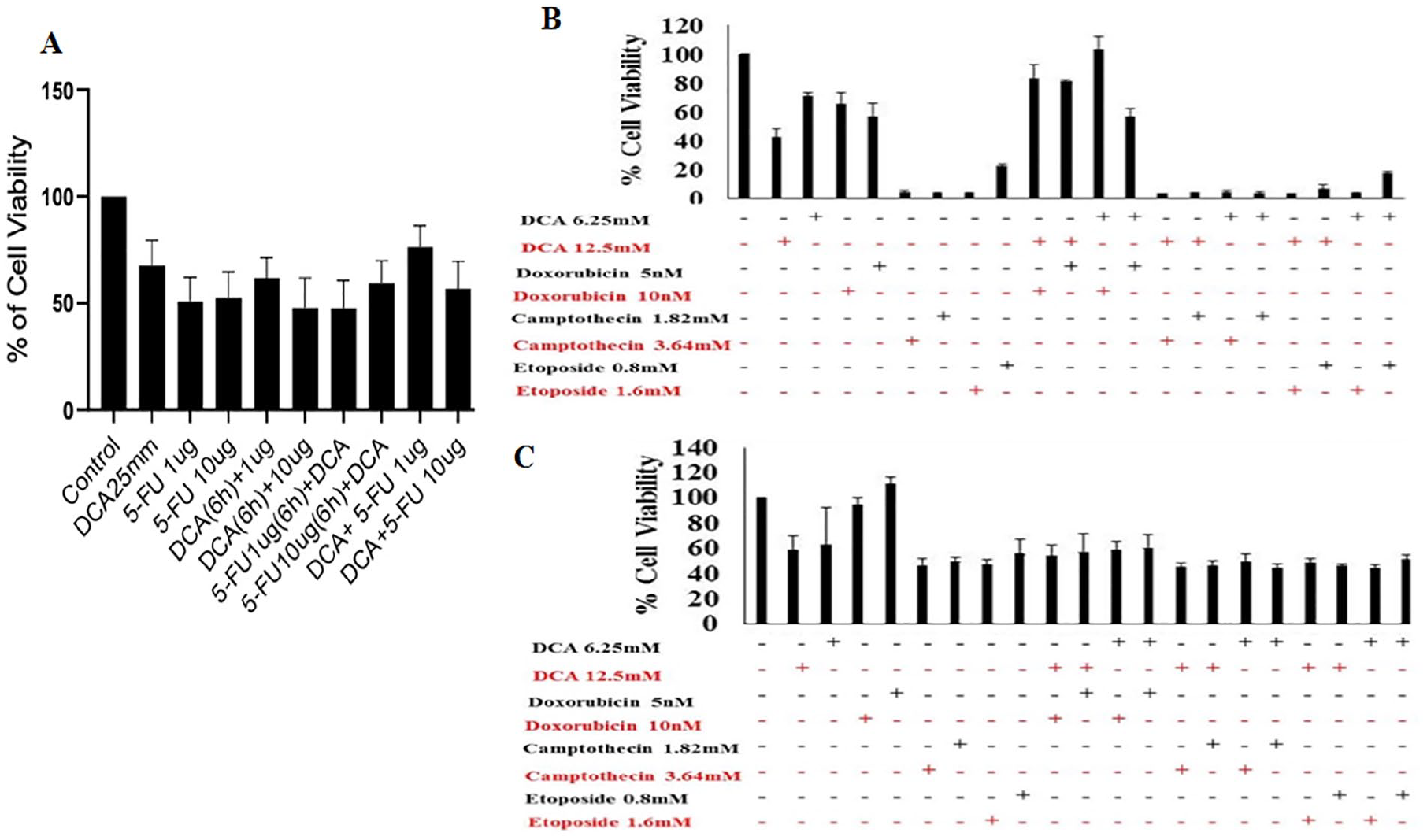
(A) Cell viability was assessed in MDA-MB-231 cells after pretreating and posttreating cells with dichloroacetate (DCA) and 5-fluorouracil (5-FU) at different time points. Cells were treated with DCA (25 mM) and different concentrations of 5-FU. (B and C) Showing percent of cell viability using MTT assay after sensitizing cells with different combination of drugs under normoxic and hypoxic conditions, respectively.
DCA Induces Expression of Tumor Suppressor miRNAs
To understand the regulation of various miRNAs on DCA treatment, we performed quantitative PCR analysis of some important miRNAs. The result showed that tumor suppressor miRNAs let-7a, let-7b, and miR-494 are upregulated on DCA treatment (Figure 3A). Let-7a is known for its tumor suppressor activity by exerting an antiproliferative effect that might be due to the activation of the apoptotic pathway; however, the exact mechanism is not known. 24 Previous studies have suggested the antiproliferative activity of let-7a in breast cancer as well as other cancerous cell lines. 25 Furthermore, miR-494 is considered as a tumor suppressor miRNA in breast cancer, directly targeting PAK1. Our data also showed that DCA upregulates the expression of miR-494, which inhibits the cell proliferation. On the other hand, it suppresses the expression of oncogenic miR-3908, which is known to inhibit the migration of breast cancer cells.26,27 Moreover, the expression profile obtained after quantitative real-time PCR showed the downregulation of oncogenic miR-1973, which was earlier reported to reduce apoptosis. 28

Representative plots showing effect of dichloroacetate (DCA), let-7a, and combination of DCA and let-7a on different processes using MDA-MB-231 cells. (A) Showing differential expression of miRNAs on DCA treatment. (B) Effect of DCA and let-7a on mitochondrial membrane potential using JC-1. (C) Impact of DCA on mitochondrial DNA–encoded genes was assessed by qPCR. (D) MDA-MB-231 cells were treated and transfected with DCA and let-7a mimic/inhibitor, respectively, and MTT assay was performed after 24 hours and 48 hours. (E) Percentage of apoptotic cells after staining with annexin/PI by flow cytometer. Graphical representation of percentage of apoptotic and necrotic cells. (F) Western blot analysis of selective apoptotic markers.
Recently, let-7a was found to be downregulated in different cancer types such as colon cancer and lung cancer. Let-7a is a tumor suppressor molecule as it can inhibit the expression of RAS and c-MYC at the translational level. 29 Since let-7a is one of the well-known tumor suppressor miRNAs and was upregulated on DCA treatment, we sought to determine the combined effect of let-7a and DCA on mitochondrial membrane potential. It is known that most cancer cells have hyperpolarized membrane due to which potassium channels remain closed, thus inhibiting the release of apoptotic molecules. 30 Our results showed the reduced mitochondrial membrane potential as indicated by the reduced green population in combined treatment compared with DCA alone (Figure 3B).
Interestingly, MTT and restoration of mitochondrial membrane potential gave some compelling indications about the involvement of mitochondria in the anticancer effects of DCA. Furthermore, we sought to determine the impact of DCA on mitochondrial encoded genes. Our results showed that both mtDNA-encoded genes, ND3 and ND4, were upregulated on exposure to DCA (Figure 3C). Both code for subunits 3 and 4 of mitochondrial encoded NADH dehydrogenase, respectively, and are part of CI.31,32 CI is the largest enzyme complex that binds to the NADH substrate and initiates the first step in the electron transport process, which leads to energy production in the form of ATP. Their upregulation indicates increased CI activity and results in higher ATP production. 33
We then studied the impact of combined treatment on cell viability or induction of apoptosis. To accomplish this, let-7a mimic was transfected with or without DCA treatment followed by MTT assay. The results showed that while DCA, as well as let-7a overexpression, can inhibit proliferation independently in time-dependent manner, their combination did not affect to that extent (Figure 3D). These results were further validated using flow cytometer–based annexin V/propidium iodide (PI) where contradictory results were obtained: no significant difference was observed between DCA and let-7a alone, but when used in combination, the percent of late apoptotic cells increases (Figure 3E). The results of these experiments indicate that DCA upregulates let-7a expression, but its apoptotic effect is independent of let-7a. Western blot analysis revealed upregulation of p53 and Bax levels, which is more prominent in DCA-treated cells, indicating that p53 is involved in DCA-induced apoptosis (Figure 3F).
To further understand this complex interplay, we carried out experiments with combined treatment of DCA and let-7a overexpression. First, we found that ROS, as measured by H2DCFDA staining, showed more reduction in combined treatment as compared with DCA and let-7a alone (Figure 4A). The data from ROS and JC-1 experiments show that combinatorial treatment leads to consistent effects on cellular parameters. Autophagy is another crucial parameter as it is often observed that cancer cells survive via autophagy during energy-deficient conditions. Our results indicate that DCA-treated cells have a slightly lower autophagy index compared with control cells. However, in combined treatment of DCA and let-7a overexpression, there is significant inhibition of autophagy (Figure 4B). These results are especially significant indicating that inhibition of autophagy might sensitize the cells to the presence of DCA, thus increasing the percent of apoptotic cells.

Combined effect of dichloroacetate (DCA) and let-7a alters different parameters. (A) Levels of reactive oxygen species (ROS) were detected after staining MDA-MB-231 cells with H2DCFDA using flow cytometer. Representative peaks infer the intensity of the dye. (B) Showing the inhibition of autophagy in MDA-MB-231 cells. Mitochondrial membrane depolarization and reduced ROS generation indicate increase in mitochondrial mass. (C) Bars showing the combined impact of DCA and let-7a on ATP level. (D) Representative graph showing the bars of fluorescence intensity of MitoTracker Green obtained by flow cytometer. (E) Confocal images showing the increased staining of MitoTracker Green in cells treated with DCA and transfected with mimic of let-7a. (F) Western blot images showing markers for mitochondrial biogenesis.
Since it was increasingly evident that DCA induces mitochondria-mediated apoptosis as revealed by the expression of Bax and p53, we explored for changes in various mitochondrial parameters on DCA and let-7a alone and in the combination of both treatments. The most exciting observation was the restoration of mitochondrial membrane potential in combined treatment, indicating potential induction of mitobiogenesis. Furthermore, ATP output analysis showed a partial increase in ATP levels in cells treated with DCA alone or with let-7a overexpression (Figure 4C). Our flow cytometer as well as confocal microscopy–based analysis revealed the increased green intensity indicating the number of mitochondria increases in combined treatment as compared with DCA and let-7a alone (Figure 4D and E). Protein expression of mitochondrial biogenesis markers was determined by western immunoblot. As shown in Figure 4F, the expression of MFN2 and AMPKα increases significantly on combined treatment as compared to individual treatments, but no change was observed in PGC-1α. These results overall reflect that DCA, as well as let-7a individually or in combination, leads to an increased number of mitochondria in cancer cells, which in turn produces more energy.
DCA Leads to Metabolic Reprogramming of the Cancer Cells
Finally, to verify the association of OXPHOS with increased mitochondrial biogenesis and ATP, we measured the oxygen consumption rate in live time scale using Oroboros O2k.
Consistent results were obtained after measuring the oxygen consumption rate or respiration rate. After permeabilization of control and treated MDA-MB-231 cells with digitonin (16 µM), addition of malate induces an increase in respiration in cells treated with DCA and transfected with let-7a relative to DCA alone as indicated by reduced oxygen concentration. ADP added at a concentration 0.5 mM increases respiration in combined treatment (measuring CI-linked OXPHOS capacity). CI- and CII-linked respiration was determined in the presence of rotenone and succinate. Respiration rate in the ADP and rotenone-stimulated state was similar, but there was a 5 times increase in respiration in the presence of succinate. Overall, it can be concluded that in control cells malate does not induces respiration, but the combined treatment of DCA and let-7a stimulates respiration, indicating restoration of mitochondria functionality (Figure 5).

Showing the measurement of oxygen concentration on addition of different mitochondria-associated substrate and inhibitors in MDA-MB-231 cells.
Discussion
Dichloroacetate is a generic, less expensive drug previously used in humans and known for modulating cancer metabolism, but its mechanism remains elusive. DCA was previously reported as regulating apoptosis, altering membranes, affecting the cell cycle, and many other processes in different cancer types.34-36 Metabolic reprogramming is a characteristic feature of cancerous cells to promote survival under stress conditions. DCA is a known metabolic modulator that inhibits glycolysis and induces OXPHOS. Considering this feature, we were intrigued by the impact of DCA and let-7a on mitochondrial parameters in MDA-MB-231 cells. This is the first report that reports the combinatorial effect of DCA and let-7a in triple-negative highly metastatic MDA-MB-231 cells. Furthermore, to gain insight into the mechanism of DCA in combination with let-7a inside mitochondria, ρ0 cells were generated by long-term exposure of EtBr at low concentration supplemented with uridine. The ρ0 cells have depleted mtDNA after prolonged and low-dose treatment of EtBr. 37 Our results showed the dose-dependent reduction in cell proliferation in in vitro culture of WT and ρ0 MDA-MB-231 cells. WT MDA-MB-231 cells showed 50% inhibition at 25 mM, while ρ0 cells showed 50% inhibition at a lower dose of DCA (5 mM). Thus, it can be deduced that diseased or dysfunctional mitochondria are responsible for the proliferation of cancer cells, providing ROS, that support cell proliferation, whereas ρ0 cell drug sensitization appears at a lower dose, indicating chemosensitivity induced after deleting dysfunctional mtDNA. 38
Besides dysfunctional mitochondria, hypoxia is another major factor contributing to cancer cell proliferation. Hypoxia increases the stability of hypoxia-inducing factor 1 (HIF-1α), thus increasing the expression of hexokinase, which, in turn, increases uptake of glucose and enhances glycolysis. 39 We thus tested the efficacy of DCA under hypoxia in both WT and ρ0 cells. Our results showed that DCA is mitochondrial-dependent under normoxia, whereas it is independent under hypoxia as indicated by the increased cell proliferation ρ0 cells. DCA was also used in combination with other anticancer agents in both conditions and did not show synergistic effects in combination with other drugs.
Furthermore, we studied the impact of DCA on selective miRNAs and mitochondrial genes that are associated with cancer progression when they are dysregulated. We found that DCA upregulates the expression of tumor suppressor miRNAs and downregulates the expression of oncogenic miRNAs.
After investigating the impact of DCA on tumor suppressor miRNA let-7a, we combined the treatment of DCA with let-7a to enhance the efficacy of DCA and studied the combined effect of let-7a and DCA on the MDA-MB-231 cells. It is well known that most of the cancer cells have hyperpolarized mitochondrial membrane due to which potassium channels remain closed, and thus inhibit the release of apoptotic proteins. 30 Our results show that the combined effect of DCA and let-7a increases red intensity after staining with JC-1, indicating restoration of the mitochondrial membrane potential. We then further explored the mechanism behind the restoration of the mitochondrial membrane potential on combined treatment and found that DCA increases the expression mitochondrial genes ND3 and ND4 indicating the association of DCA with mitochondria. Our study thus confirms the antiproliferative behavior of both DCA and let-7a alone and in combination. This result shows that the combined effect of DCA and let-7a more strongly inhibits proliferation in a time-dependent manner indicating the antiproliferative behavior of let-7a and confirming the previously reported results. 40 The inhibitory behavior of combined effect of DCA and let-7a might be due to the induction of apoptosis. Our result shows that DCA and let-7a alone do not induce more apoptosis, but the combined treatment of DCA with let-7a switches more cells toward apoptosis.
The expression of Bax and p53 confirmed the mechanism of apoptosis. However, our results also confirm the role of DCA in induction of apoptosis and are the first report revealing the role of DCA in sensitizing let-7a. 41 After establishing the antiproliferative and apoptotic behavior of DCA and let-7a, we further explored the free radical level and mitochondrial membrane potential. Our results showed significant reduction in ROS level in MDA-MB-231 cells on combined treatment. The reduced ROS inhibits proliferation as the elevated ROS activate various growth-promoting pathways and enhance genome instability.42,43 Autophagy is an important mechanism to remove the damage to cellular proteins and maintain cellular metabolism. Recently, it has been demonstrated in LoVo cells that DCA induces autophagy due to increased ROS level. 44 Several reports are suggesting that the inhibition of autophagy increases sensitivity to chemotherapeutic drugs.45,46 Our study showed that DCA and overexpressed let-7a inhibit autophagy in MDA-MB-231 cells due to induction of apoptosis, and more inhibition was found when DCA was used in combination with let-7a.
Thus, increased apoptosis, reduced ROS, and reduced mitochondrial membrane potential indicates the alteration in the mitochondrial mass or mitochondrial biogenesis. The above-obtained results give further insight into the effect of combined treatment on energy production in the form of ATP and the mitochondrial network. A slight increase in the ATP level was observed in combined treatment, whereas the increased intensity of MitoTracker Green indicates an increase in mitochondrial mass. Hence, it can be concluded that DCA and let-7a increases mitochondrial mass and distributes mitochondria evenly and uniformly as compared with untreated cells.
Recent studies suggest that mitochondrial fusion leads to enhanced ATP production, whereas downregulation of fusion was associated with mtDNA depletion, ROS production, and dysfunctional OXPHOS. 47 The translocation of endophilin A1 inside mitochondria enhances mitochondrial biogenesis–associated proteins PGC-1α and MFN2. 42 Our result shows increased expression of PGC-1α (a master regulator of mitochondrial biogenesis) and MFN2 after treatment with DCA and let-7a alone as compared with control but expression increases after combined treatment. We also analyzed the expression of AMPKα. Under stressed conditions, cells have low AMP/ATP ratio indicating lower energy level AMPKα activation maintains energy homeostasis and also induces the expression of p53. Hence, increased ATP level, induction of apoptosis, increased intensity of MitoTracker Green, and induced expression of MFN2 and PGC-1α indicates DCA in combination with let-7a induces mitochondrial biogenesis.
It has been previously reported that mitochondrial morphology is associated with the production of ATP through OXPHOS, initiation of apoptosis, regulation of calcium homeostasis, and other processes. 47 It can be postulated that altered mitochondrial morphology might be linked with the metabolic shift induced by let-7a and DCA. Our result revealed that increased ATP level was due to increased OXPHOS as indicated by reduced oxygen concentration on treatment with DCA and let-7a alone, but more shift was observed when treatment was combined with transfection of let-7a. Combined treatment reduces the dependency of cancer cells on the glycolytic phenotype; hence, the shift of cancer metabolism from glycolysis to OXPHOS indicates induction of mitochondrial mediated apoptosis.
In conclusion, our result showed that the combined effect of DCA and mimic of let-7a reduces proliferation of MDA-MB-231 cells in a dose- and time-dependent manner (Figure 6). DCA also regulates the expression of small noncoding regulatory molecules. Our data also show induction of apoptosis, confirmed by the expression of Bax and p53, and reduced membrane potential suggesting combined treatment of DCA and let-7a induces mitochondrial apoptosis by restoring the mitochondrial morphology. Our results also align with the present literature showing partial reduction in autophagy that can be responsible for increasing the chemosensitivity of DCA. Additionally, the expression of Bax was in line with expression of PGC-1α and MFN2 indicating mitochondrial biogenesis induced by combined treatment of DCA and let-7a. Mitofusin can be connected with the activation of OXPHOS that leads to ATP generation, and hence regulates the energy-dependent mitochondrial-mediated apoptosis.

Model showing how combined treatment of let-7a and dichloroacetate (DCA) induces apoptosis by altering mitochondrial morphology and metabolism.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research work was supported by a grant from DST-SERB (SR/SO/AS-31).
