Abstract
Mulberry fruit water extract (MWE) has been reported to synergistically enhance the cytotoxic effect of paclitaxel by promoting mitotic catastrophe to induce apoptosis in bladder cancer cells in our previous work. The aim of this study was to evaluate and to mechanistically explore the effects of MWE on bladder cancer responses to ionizing radiation (IR) by treating TSGH 8301 bladder carcinoma cells with MWE after exposing to IR. The results of MTT assay showed a synergistic cytotoxicity of IR with the co-treatment of MWE (IR/MWE) by inducing G2/M phase arrest as demonstrated by flow cytometry analysis in TSGH 8301, HT1367 and HT1197 bladder carcinoma cells lines. The IR/MWE-treated cells expressed increased levels of the G2/M phase arrest-related proteins cdc2/cyclin B1 and displayed giant multinucleated morphology, a typical characteristic of mitotic catastrophe. Immunofluorescent confocal microscopy revealed that the combined strategy inhibited Aurora B phosphorylation through Ras/Raf/MEK/ERK signaling cascade as demonstrated by Western blotting analysis. IR/MWE also caused an inhibitory effect on Plk1 and the subsequent downstream regulator RhoA repression and Cep55 induction, which would influence cell cycle progression in the early steps of cytokinesis. A profound tumor growth suppression and inactivation of Aurora B activity in the tumor tissues by IR/MWE treatment were confirmed in the TSGH 8301 xenograft model in vivo. These data demonstrated that MWE could be an effective auxiliary to synergize with radiation on the anticancer efficacy by promoting mitotic catastrophe through inhibition of Aurora B, providing a novel and effective therapeutic option for bladder cancer management.
Introduction
The bladder, a hollow organ in the lower part of the abdomen, functions as a storing unit of urine until it is passed out of the body. Bladder cancer begins frequently in the urothelium that lines the inside of the bladder. The most common type of bladder cancer is transitional cell carcinoma, also called urothelial carcinoma. Based on the occurrence of muscle invasion, bladder cancer can be clinically classified as muscle-invasive (MIBC) or non–muscle-invasive (NMIBC) bladder carcinoma. 1 Up to the present, the standard treatment for MIBC is radical cystectomy whose perioperative complication rate and mortality rate are significant improved because of medical techniques improvement. 2 For the MIBC patients who are not suitable for or refuse radical cystectomy, an alternative treatment choice is transurethral resection of bladder tumor followed by radiation therapy and systemic chemotherapy with a benefit of preserving bladder function. The previous study showed that MIBC patients received radiation therapy or radical cystectomy had similar survival rate. 3
Clinical therapeutic application of ionizing radiation (IR) is not only limited by daily variations in bladder size and position due to differences in bladder filling and other factors that can negatively affect the ability to accurately deliver radiation to the bladder but also by the IR-prompting damages, including direct DNA double-strand break lesions and indirect induction of cellular free radicals. 4 Previous studies showed that one of the factors that influence the extent of DNA damage following radiation exposure is cellular oxygen that is believed to prolong the lifetime of the short-lived free radicals produced by the interaction of X-rays and cellular H2O.4,5 When the DNA of tumor cells is damaged by IR, the cell cycle will be delayed at one of the checkpoint by inhibiting cyclin-dependent kinases (CDKs) until DNA repair is complete. 6 The heterodimer of cdc2/Cyclin B1, also known as the M phase-promoting factor, assists the conversion of centrosomes to mitotic microtubule organizing center and the reorganization of microtubule cytoskeleton in late prophase.7,8 During DNA repair, cdc2/Cyclin B1 remains activated in the nucleus until DNA is fixed. However, continually active cdc2 prompts cancer cells to stay at M phase, when Cyclin B1 could not be degraded via the ubiquitin-proteasome pathway. 9
Mitotic catastrophe (MC) is a mechanism of sequential events that are the consequence of premature or inappropriate entry of cells into mitosis due to chemical or physical stresses including ionizing radiation.9,10 Many of mitotic kinases have been reported to regulate the mechanism of MC. The Aurora serine/threonine protein kinases family, one of those that regulates mitotic process, has members of Aurora A, B, and C. Aurora A locates at the spindle poles and functions in mitotic entry, centrosome maturation and maintenance of spindle bipolarity. 11 Accurate chromosome segregation during mitosis requires the assistance of Aurora B in the formation of chromosomal passenger complex with other regulatory proteins, 12 a process that needs auto-phosphorylation of Aurora B on Thr-232 through interaction with the inner centromere protein. 13 Overexpression of Aurora B was found in primary human colorectal cancers, non–small cell lung carcinoma and hepatocellular carcinoma cells lines.14-16 In preclinical cancer therapy researches, Aurora B inhibitor induced loss of normal chromosomal alignment and chromosomal segregation to result in MC,17,18 and inhibited small-cell lung cancer cells growth in vitro and in vivo. 19
The strategy of adding nutritional compounds from botanical as auxiliary agents to reduce side effects and toxicity of traditional cancer therapies is a noticeable trend in cancer research in recent years. 20 Mulberry is an important medicinal plant belonging to the Moraceae family; and several phenolic compounds and many flavonoids have been identified from mulberry fruit. 21 Previous researches has shown that mulberry fruits extract had potential antitumor effects such as suppressing acute colitis and colorectal tumorigenesis via preventing lipopolysaccharide (LPS)-induced NF-κB/p-ERK/MAPK signals, promoting human glioma cell death in vitro through reactie oxygen species (ROS)-dependent mitochondrial pathway and inhibiting glioma tumor growth.22,23 In addition, our previous study discovered that mulberry water extract (MWE) had synergistic effects to strengthen the toxicity of paclitaxel by promoting MC to induce apoptosis in bladder cancer cells. 24
In this study, we found that the assistance of MWE enhanced the cytotoxicity of IR via stimulating MC through a mechanism in which the inhibition of Aurora B activity interfered cytokinesis procedure in bladder cancer cells. These results suggested that MWE could be used as an auxiliary to improve IR therapy via inducing MC.
Materials and Methods
Antibodies and Reagents
Primary antibodies used in Western blotting and immunocytochemical analysis were as follows: antibodies against Cdc2, phospho-Cdc2 (Tyr15), Cyclin B1, phospho- Cyclin B1 (Ser133), Aurora A, phospho-Aurora A (Thr288), Ras, B-Raf, MEK1/2, p-MEK1/2, and Lamin A/C were purchased from Cell Signaling (Danvers, MA); antibodies against Cep55, polo-like kinase 1 (Plk1) and phospho-Plk1 (Thr210) were purchased from Santa Cruz (Santa Cruz, CA); β-actin was purchased from Gene Tex Inc (San Antonio, TX). Secondary antibodies Alexa Fluor 488 and FITC were from BD Biosciences (Franklin Lakes, NJ). RPMI1640 medium, Eagle’s minimum essential medium, phosphate buffered saline (PBS), trypsin-EDTA, penicillin-streptomycin mixed antibiotics,
Extraction of Aqueous Fractions From Mulberry Fruit
Fresh mulberry fruit (54 kg) were frozen dried to obtain 5.4 kg dried mulberry fruit powder. The dried powder (100 g) was dissolved in 1000 mL of deionized water with stirring for 2 hours. The suspension was frozen dried into powder after a centrifugation at 3500 rpm for 20 minutes at 4°C. A yield of about 50% of original dried powder weight was reached. The lyophilized powder (MWE) was resuspended in distilled water and filtered (0.45 µm pore size) for subsequent uses in cell cultures and in animal study.
Cell Culture
TSGH 8301, HT1367, and HT1197 cells lines were obtained from Bioresource Collection and Research Center, Food Industry Research and Development Institute (Taipei, Taiwan). TSGH 8301 cells line was maintained in RPMI 1640 medium supplemented with 10% (w/v) fetal bovine serum; and HT1367 and HT1197 cells lines were maintained in Eagle’s minimum essential medium with fetal bovine serum to a final concentration of 10% (w/v). Cells were cultured at 37°C in a humidified atmosphere of 5% CO2/95% air incubator.
Cytotoxicity Assay
Cancer cell viability was determined by MTT reduction assay. Cells seeded in 24-well plates at a density of 3 × 104 per well were exposed to IR followed by MWE 12 hours later and then incubated for additional 24, 48, and 72 hours. After incubation, the cells were washed with PBS and then incubated with 1 mL medium containing 100 µL MTT (5 µg/mL) for 4 hours. The amount of viable cells per dish was quantified by the ability of living cells to reduce the yellow dye to a purple formazan product, which was solubilized in isopropanol and measured spectrophotometrically at 563 nm with a spectrophotometer (Hitachi U-2900/2910).
Cell Cycle Distribution Analysis
TSGH 8301, HT1367, and HT1197 cells treated with MWE and/or IR for the indicated doses and time periods were trypsinized from the culture plate and fixed in −20°C ice-cold 70% ethanol. Thereafter, the DNA content was determined by staining with a solution containing 0.1% Triton-X 100, 20 µg/mL RNase, and 20 µg/mL propidium iodide (PI) at room temperature in the dark for 30 minutes. The cell cycle distribution was analyzed using CellQuest software on a BD FACS Calibur machine (Mountain View, CA).
Western Blot Analysis
From the treated cells, the cytoplasmic and nuclear proteins were extracted by NE-PER nuclear and cytoplasmic extraction reagents (Thermo Fisher Scientific) and quantified using a Bio-Rad protein assay (Bio-Rad, Hercules, CA) with bovine serum albumin (BSA) as a standard. Each lysate (20 µg of protein) was resolved on denaturing polyacrylamide gels and transferred electrophoretically to nitrocellulose membranes. After blocking with 5% nonfat dried milk in TBS-Tween 20 (TBST), the membranes were incubated with primary antibodies overnight at 4°C. Immunoreactive proteins were detected with horseradish peroxidase (HRP)-conjugated secondary antibodies for 1 hour at room temperature. After washed with TBST, the reacted bands were developed using the enhanced chemiluminescence kit (GE Healthcare Biosciences) and identified using the ImageQuant LAS 4000 mini.
Hematoxylin and Eosin Stain
After the treatment, the cultured cells were fixed with 95% (v/v) alcohol for 1 minute at room temperature followed by washing 3 times with purified water. The cells on the coverslips were stained for nuclei with hematoxylin for 3 to 5 minutes, and then washed with ammonia solution for 30 seconds and with purified water 3 times. The cells were dehydrated by passing through 70% (v/v) and 95% (v/v) ethanol for 30 seconds each. The cells were then stained with eosin for 1 minute followed by immersing in 95% (v/v) and 100% (v/v) ethanol for 30 seconds each. The coverslips were immersed in xylene for 1 minute and then mounted onto slides.
Immunofluorescence and Confocal Microscopy
TSGH 8301 cells seeded at 3 × 104 cells on coverslips overnight were fixed in 4% paraformaldehyde on ice for 15 minutes, washed 3 times with PBS, and then permeabilized with 0.1% Triton X-100 in PBS for 15 minutes at room temperature. The cells were further blocked in 10% goat serum for 1 hour before the required primary antibody was applied. After washing with PBS, the cells were incubated with Alexa 488- or FITC-conjugated secondary antibody (BD Biosciences). Cell nuclei were counterstained with DAPI 32670 (Sigma). After extensive washing, coverslips were mounted onto glass microscope slides, and cells were viewed with a fluorescence confocal microscope (Zeiss, Jena, Germany).
Xenograft Model and Treatment
Four-week-old BALB/c male nude mice were purchased from National Laboratory Animal Center (Taipei, Taiwan) and maintained for 1 week in a specific pathogen-free room with an irradiated 5058-PicoLab Mouse Diet (LabDiet, Inc, St Louis, MO) at 22°C and 55% humidity with a 12-hour diurnal system. TSGH 8301 cells (1 × 107 cells/mouse) were mixed with an equal volume of BD Matrigel (Becton, Dickinson and Company, Franklin Lakes, NJ) and injected into the right inguinal region of a nude mouse. When the tumor size reached approximately 140 mm3, the mice were randomly divided into 4 groups receiving the following treatments: IR combined with MWE (12 mice), MWE alone (8 mice), IR alone (12 mice), and sterile deionized water (the control group, 8 mice). A single dosage of IR (10 Gy/mouse) was applied to the tumor site at the beginning of the treatment; and MWE (4 mg/kg) in sterile deionized water was given with a stomach sonde needle daily. Tumor volumes were monitored using a caliper every week during the entire experiment. Tumor volumes were calculated using the following formula: tumor volume = major axis × (minor axis)2 × 0.52. All animal care and experimental procedures were carried out in strict accordance with the guidelines for the care and use of laboratory animals of Chung Shan Medical University, and approved by the Institutional Animal Care and Use Committee. The article does not contain clinical studies or patient data.
Statistical Analysis
Statistical analyses were performed by Sigmastat software (Jandel Scientific, San Rafael, CA). Results are expressed as mean ± SD, and data were analyzed by Student’s t test or 1-way analysis of variance with post hoc Dunnett’s test for significant difference with P < .05 considered significant.
Results
Effects of MWE Combined With IR on the Cytotoxicity via Inducing G2/M Phase Arrest in Bladder Carcinoma Cell Lines
MWE used in this study was rich in phenolic compounds and flavonoids as analyzed by HPLC-DAD-ESI-MS and reported in our previous study. 24
Three bladder carcinoma cell lines at different malignant stages, TSGH 8301, HT1136, and HT1197, were tested for the cytotoxic effect of IR, MWE, or combination of both for different time periods. The results of MTT assay showed that the supplement of MWE (0-1500 µg/mL) along with a single IR exposure (10 Gy) enhanced, dose- and time-dependently, the toxicity of IR alone, while MWE (500 µg/mL) alone was not toxic to TSGH 8301 cells (Figure 1A). The 2 other cell lines demonstrated results with a similar trend (Figure 1B). To understand the possible death mechanism regarding the synergistic cytotoxic effect of MWE and IR, flow cytometry analysis of the treated cells was performed. Although the cell cycle distribution of MWE alone group was comparable to that of control group (no treatment), the addition of MWE along with IR treatment increased the population of cells arrested at the G2/M phase from ~40% (IR alone) to ~60% (MWE 750-1500 µg/mL + IR) at 24 hour (Figure 1C). The cell cycle arrest at the G2/M phase was even more profound (~80% of cell population) at 48 hour. This observation indicated that MWE enhanced the IR-induced G2/M arrest at early time point. Similar outcomes were obtained when the same strategy was applied to HT1367 and HT1197 cells showing a more apparent dose-dependent manner of MWE (Figure 1D). TSGH 8301 cell line was used thereafter in this study because it was more susceptible to the treatment strategy than the other 2 cell lines, which would let us to clarify the mechanism.

Effects of mulberry water extract (MWE) combined with ionizing radiation (IR) on the cytotoxicity via inducing G2/M phase arrest in bladder carcinoma cells lines. (A) and (B) TSGH 8301, HT1367, and HT1197 bladder carcinoma cells were treated with IR (10 Gy), MWE (500 µg/mL), or IR/MWE with the indicated concentrations of MWE for 24, 48, and 72 hours before being subjected to the MTT assay for cell viability. The data are reported as means ± SD from 2 different experiments performed at least in triplicate and are expressed as a percentage of control (not treated). One-way analysis of varaince with post hoc Dunnett’s test was used to calculate the p value for each dose treatment compared with IR alone, (+P < .05; ++P < .01) and between time points (*P < .05; **P < .01). (C) TSGH 8301 cells and (D) HT1367 and HT1197 cells were treated with IR (10 Gy), MWE (500 µg/mL), or IR/MWE with different concentrations of MWE for the indicated time point and then subjected to cell cycle distribution analysis by flow cytometry. (E) Nuclear protein samples obtained from TSGH 8301 cells of different treatments at 24 hours were analyzed by Western blotting for phosphorylation and protein levels of Cdc2 and Cyclin B1. The numbers under each blot are the intensity of each band relative to that of the control (not treated) or IR alone. The blots were reprobed with an anti-Lamin A/C antibody to confirm equal loading of the samples. Arrow head indicated the band used for quantitation. The results of C to E are representative of 3 independent experiments with similar results.
During DNA damage, active cdc2/Cyclin B1 complex is recruited to the nucleus and remained active to avoid cells escaping from the G2/M phase. 9 To confirm that cdc2/Cyclin B1 complex was activated in the G2/M phase arrest cells, Western blotting was performed to determine the phosphorylation of cdc2 and Cyclin B1, a known G2/M markers and events. The treatment of MWE alone did not affect the protein levels and phosphorylation state of cdc2 and Cyclin B1 as compared with the control cells. IR exposure increased the levels of cdc2 and phosphorylated cdc2 and Cyclin B1, which were further enhanced dose-dependently by the co-treatment of MWE at 24 hours (Figure 1E). These data provided evidence that the combined treatment of IR/MWE promoted cell death through inducing G2/M phase arrest in bladder carcinoma cells.
Induction of Mitotic Catastrophe by IR/MWE Treatment in TSGH 8301 Bladder Carcinoma Cells
The cytochemistry H/E stain of the IR alone or IR/MWE treated TSGH 8301 cells displayed significant number of multinucleated cells (black arrows) at 24 and 48 hours (Figure 2A). This phenomenon was significantly boosted as MWE concentration increased at both time points (Figure 2A, right panel). Giant multinucleated cells characterized by missegregated and uncondensed chromosomes are often the morphological markers of MC. The recent studies discovered that Aurora A would cooperate with Bora, a G2/M expressed protein, to induce Plk1 activation, and the activated Plk1 could delay cancer cells to escape from the G2/M phase via activating cdc2/Cyclin B1.25,26 We analyzed the protein levels of MC-related proteins, p-Aurora A/Aurora A and p-Plk1/Plk1, at 24 and 48 hours. As a moderate elevation of p-Aurora A was observed in the MWE and IR/MWE, but not IR alone, treated cells at 24 hours, there was a profound and dose-dependent induction of the phosphorylation of Aurora A at 48 hours (ratio to the control was up to 4.24-fold) under a circumstance that its protein level remained unchanged (Figure 2B and C). On the other hand, while all treatments induced slightly the activation of Plk1 (p-Plk1), only IR and IR/MWE increased total protein levels at 24 hours as compared with those of untreated cells. When the treatment time extended to 48 hours, the phosphorylation of Plk1 was diminished unexpectedly, while the total protein level remained elevated at a time point that G2/M arrest was robust (Figure 2B and C).
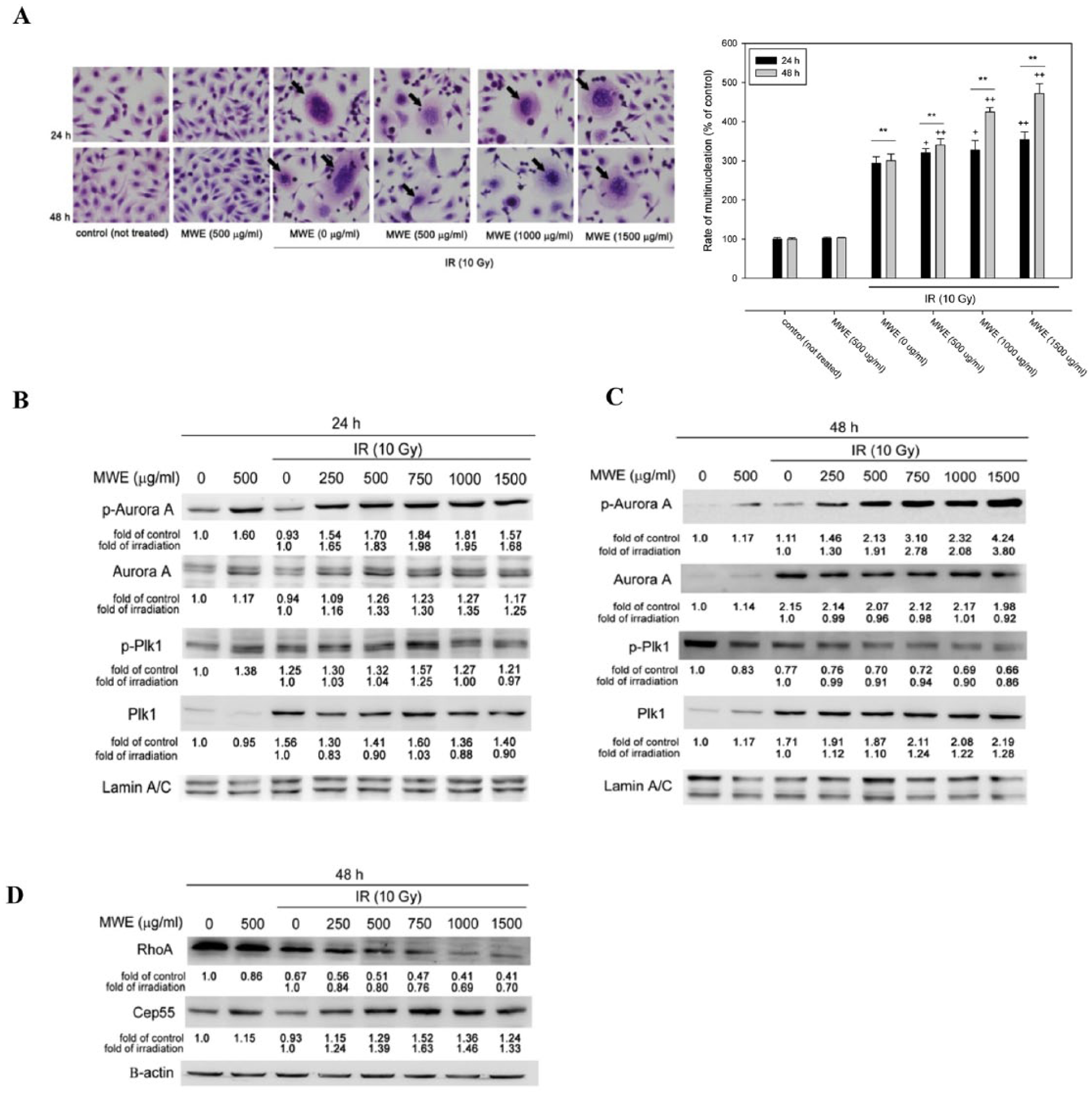
Mulberry water extract (MWE) enhance ionizing radiation (IR) ability to induced multinucleation and expression of mitotic catastrophe–related proteins in TSGH 8301 cells. (A) TSGH 8301 cells were subjected to hematoxylin and eosin (HE) staining after the indicated treatments and times. The arrows point to the cells with multinucleation (left panel). Right panel represents the quantitation of the number of multinucleated cells/100 cells that was expressed as percentage of control. The data are reported as means ± SD from 2 different experiments performed at least in triplicate. One-way analysis of varaince with post hoc Dunnett’s test was used to calculate the P value for each dose treatment compared with IR alone, (+P < .05; ++P < .01) and between time points (*P < .05; **P < .01). (B) and (C) Nuclear proteins prepared from the TSGH 8301 cells treated with IR, MWE or both for 24 and 48 hours were subjected to Western blotting analysis for the levels of phosphorylated form and total protein of Aurora A and Plk1. (D) Cytoplasmic lysates were extracted from TSGH 8301 cells to assess Rho A and Cep55 expression. The numbers under each blot are the intensity of each band relative to that of control (not treated) or IR alone. The relative protein amounts were quantified, and the results are normalized to that of β-actin (cytoplasm) or Lamin A/C (nucleus). The results shown are representatives of 3 independent experiments with similar results.
The phenomenon of high cellular activated Aurora A could coerce cancer cells to stagnate in G2/M period. However, our data showed that the phosphorylation of Plk1, a downstream factor of activated Aurora A, was not concurrently increased along with the elevated levels of p-Aurora A in the IR/MWE treated TSGH 8301 cells. Previous studies have revealed that Plk1 could induce activation of RhoA, a central regulator of cellular contractility, and negatively regulate Cep55, a microtubule-bundling protein, to assist cytokinesis in progress.27,28 In the present treatment strategy, the level of RhoA was decreased by IR exposure, and was further diminished by the co-treatment of MWE (Figure 2D). On the contrary, IR/MWE induced the expression of Cep55 at 48 hr. These observations that coincide with the downregulation of Plk1 could sabotage the progression of cytokinesis, and that together with the continuous activation of Aurora A could finally lead to severe MC.
Inhibition of Aurora B Phosphorylation by IR/MWE Treatment in TSGH 8301 Bladder Carcinoma Cells
The study of Shao et al 29 demonstrated a novel pathway that Aurora B phosphorylates Plk1, triggering its kinase activity at the kinetochores, to ascertain proper chromosome biorientation and accurate chromosome segregation in mitosis. Using Western blotting we found that the combined treatment cause a reduction in the phosphorylation of Aurora B as compared with the MWE and IR alone groups (Figure 3A) while the protein levels was slightly increased. Confocal microscopy immunofluorescent analysis was then used to detect the localization and expression of p-Aurora B. As shown in Figure 3B, p-Aurora B located mainly in the nucleus and concentrated at the kinetochores in some cells (white arrows) in the control and MWE alone groups; however, the florescent intensity was ~30% less in the latter (Figure 3C). The cells received IR exposure revealed a phenotype of large endopolyploid cells that are fairly well characterized for MC 30 and had about 10% less of p-Aurora B that was mainly distributed in the nucleus without concentrated localizing at the kinetochores (Figure 3B and C). In the combined treatment group, the expression of p-Aurora B was dissipated to ~20% of control, and giant endopolyploid cells were observed (Figure 3B and C). This evidence demonstrated that the combined strategy could integrate the effects of IR and MWE via inhibiting the activity of Aurora B to interfere with cancer cells division that led to the formation of multinucleation and giant cell of MC in TSGH 8301 bladder cancer cells.

Synergistic effect of ionizing radiation (IR) and mulberry water extract (MWE) inhibited Aurora B activity in TSGH 8301 cells. (A) Nuclear proteins prepared from the TSGH 8301 cells treated with IR, MWE, or both for 48 hours were subjected to Western blotting analysis for the levels of phosphorylated form and total protein of Aurora B. The numbers under each blot are the intensity of each band relative to that of control (not treated) or IR alone. The relative protein amounts were quantified, and the results are normalized to that of Lamin A/C (nucleus). The results shown are representatives of 3 independent experiments with similar results. (B) TSGH 8301 cells with the indicated treatments for 48 hours were fixed with formaldehyde and then stained with anti-p-Aurora B (red), anti-Tubulin (green) and DAPI (blue) for confocal microscope observation. (C) Quantitation of the fluorescent intensity of p-Aurora B in TSGH 8301 cells. Student’s t test was used to calculate the P value for each treatment compared with the control (***P < .001).
Inhibition of Ras/MEK/ERK Signaling Pathway by IR/MWE in TSGH 8301 Bladder Carcinoma Cells
The activation of Aurora B has been reported to be regulated by Ras/Raf/MEK/ERK signaling cascade in gynecological cancer cell lines. 31 Ras/Raf/MEK/ERK signaling cascade participates in regulation of normal cell proliferation, survival, and differentiation, 32 and some mutated or aberrantly overexpressed components of the signaling pathways are involved in the proliferation, survival and metastasis of human cancer cells. 33 Ras is a small GTP-binding protein that locates at the inner surface of cell membrane and binds to the Ras-binding domain of Raf (named for Rapidly Accelerated Fibrosarcoma with 3 isoform: A-Raf, B-Raf, and C-Raf) to regulate its activation.33,34 The activation of B-Raf was reported to trigger downstream MEK/ERK activation to regulate cancer cells proliferation, invasion and metastasis. 34 As shown in the results of Western blotting assay, the combined strategy inhibited the expression of Ras and B-Raf dose-dependently, and repressed the phosphorylation of MEK (p-MEK) that was consistent with the decreased level of MEK protein (Figure 4A). The phosphorylation of ERK1/2 was also decreased by the combined treatment; however, there was an increase in the protein level.
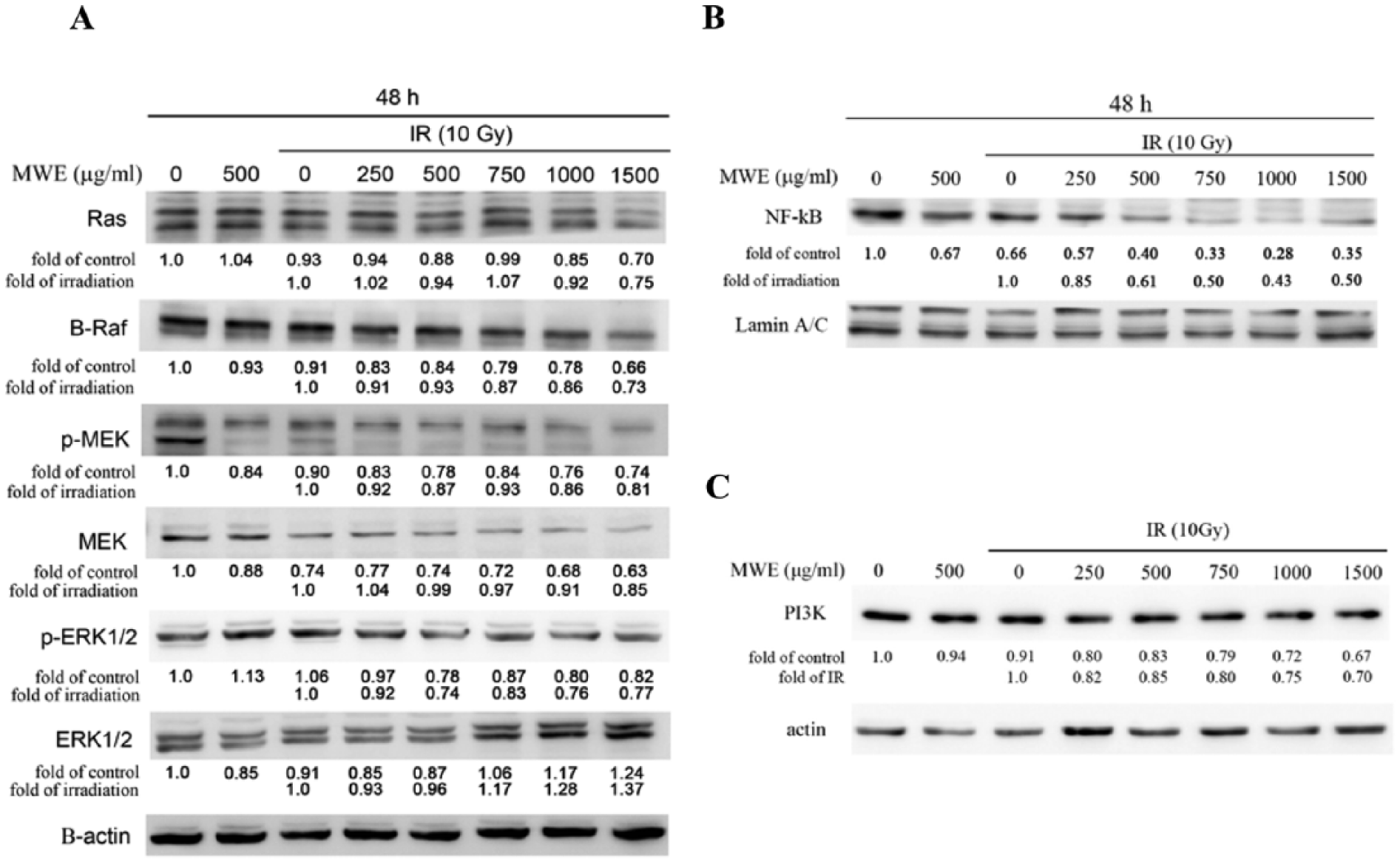
Ionizing radiation/mulberry water extract (IR/MWE) treatment inhibited Ras/MEK/ERK signaling pathway in TSGH 8301 cells. Cytoplasmic lysates (A and C) and nuclear proteins (B) prepared from the TSGH 8301 cells treated with IR, MWE, or both 48 hours were subjected to Western blotting analysis for the levels of the indicated proteins. The relative protein amounts were quantified, and the results are normalized to that of β-actin (cytoplasm) or Lamin A/C (nucleus). The results shown are representatives of 3 independent experiments with similar results.
In the past study, it was reported that B-Raf could promote nuclear factor–κb (NF-κb) pathway via C-Raf activation to support cell proliferating and survival ability. 35 The Western blotting results showed that the level of NF-κb in the nucleus of the IR/MWE-treated cells was correspondingly decreased with the reduction in the B-Raf expression (Figure 4B). Phosphatidylinositol 3-kinase (PI3K)/AKT is another main effector pathway of Ras, participating in cellular proliferation, transformation, adhesion, apoptosis, survival, and motility. 36 The data demonstrated that IR/MWE downregulated PI3K expression, an observation consistent with the changes in Ras level (Figure 4A and C). These outcomes indicated that the combined strategy of IR/MWE could repress concurrently the pathways of Raf/MEK/ERK and PI3K/AKT, a consequence of Ras suppression.
Inhibition of Tumor Growth and Aurora B Activity by IR/MWE in a Human Bladder Carcinoma TSGH 8301 Xenograft Model
To clarify the in vivo efficacy of present strategy, TSGH 8301 xenografts in mice were treated with IR (10 Gy), MWE (p.o. 4 mg/kg), or IR/MWE (10 Gy/p.o. 4 mg/kg) when the tumors reached an average volume greater than 140 mm3. The mice were randomly grouped and treated with IR at the first day (IR alone and IR/MWE groups) and MWE every day for 3 weeks. The results in Figure 5A showed that the untreated tumors displayed a stable growth and were more than the double of the initial size at the third week. The application of IR alone or MWE alone retarded the development of the tumors at the first week and suppressed the growth rate thereafter as compared with that of control, attaining an average volume that was 150% of the initial size at the third week. IR/MWE reduced effectively 34% of the initial tumor size (P < .01) at the first week and inhibited substantially tumor growth in the later weeks. At the end of the experiment, the tumor volume of the IR/MWE group was almost identical to the initial size.

Synergistic strategy retarded tumor growth in a human bladder carcinoma TSGH 8301 xenograft model. (A) TSGH 8301 cells (1 × 107 cells/mouse) were injected into the right inguinal region of nude mice to form tumor xenografts. When the tumor size reached about 140 mm3, the mice were randomly divided into 4 groups and received the following treatments: ionizing radiation (IR) combined with mulberry water extract (MWE), MWE alone, IR alone, and sterile deionized water (control group). Tumor size was monitored every week, and the results are expressed as means ± SD. One-way analysis of variance (ANOVA) with post hoc Dunnett’s test was used to calculate the P value for each treatment compared with IR alone at each time point (**P < .01). (B) Immunohistochemical examination of p-Aurora B in the tumor sections obtained from the indicated treatment and (C) quantitation of fluorescent intensity of p-Aurora B in TSGH 8301 cells. One-way ANOVA with post hoc Dunnett’s test was used to calculate the P value for each treatment compared with the control (*P < .5; ***P < .001). (D) Western blotting analysis of the levels of p-Aurora B in xenograft tumors. The upper and lower panels are the results obtained from different animal samples. (E) Statistical quantitation of the ratio of p-Aurora B/Aurora B in tumor tissues of all animals. One-way ANOVA with post hoc Dunnett’s test was used to calculate the P value for each treatment compared with the control (***P < .001).
Since the aforementioned in vitro studies revealed that the combined treatment exerted the cytotoxic effect toward bladder carcinoma TSGH 8301 via reducing Aurora B activity, it was interesting to know whether the same mechanism occurred in vivo. Immunohistochemical fluorescent analysis was therefore used to detect the localization and expression of p-Aurora B in the tumor specimens. As shown in Figure 5B, p-Aurora B was co-localized with DAPI in the nucleus in the control and MWE alone groups; however, the florescent intensity was ~10% less in the latter (Figure 5C). The tumor slide of IR group showed irregular organization of the nuclei as DAPI stain demonstrated, and significantly eliminated level (~35% of control) (P < .001) of p-Aurora B (Figure 5B and C). In the combined treated tumor sample, the fluorescence intensity of p-Aurora B was almost undetectable and down to a level of 7% of control (P < .001). This evidence supported that with the assistance of MWE, IR treatment could achieve better inhibitory effect on Aurora B activity. To confirm the findings, we detected p-Aurora B and Aurora B contents in the tumors by Western blotting assay (Figure 5D), and expressed the activity by normalizing the level of p-Aurora B to that of Aurora B protein (Figure 5E). We discovered that Aurora B was significantly decreased in some of the IR/MWE treated tumors (Figure 5D, lower panel). The changes in the ratio of p-Aurora B to Aurora B of different groups were similar to the results of fluorescence intensity of p-Aurora B (Figure 5C) showing that IR and IR/MWE decreased significantly the activity of Aurora B in the tumor tissues to ~60% and ~40% of control, respectively.
Discussion
A treatment of transurethral resection of bladder tumor followed by radiation therapy and systemic chemotherapy is a typical procedure for the MIBC patients. Novel compounds as adjuvants to improve the efficacy and to reduce the resistance of both chemo- and radiotherapy are clearly required. The present results demonstrated a synergistic effect of MWE under IR influence via inhibiting Aurora B activity and inducing MC. Taken together with our previous research showing that MWE assisted paclitaxel, a currently used chemotherapeutic agent, to treat bladder cancer via inhibition of PTEN activity and also leading to MC, 24 MWE possesses great potential in serving as an additive to amend the effectivity of traditional treatments of bladder cancer for both radio- and chemotherapy.
DNA damage induction is the main mechanism of action of radiation therapy, and tumor cells response to it by mobilizing cell cycle–regulated proteins to repair DNA damage. It has been reported that radiation treatment induced G2/M phase arrest in meningioma cells that were then slowly recovered and formed aggressive intracranial tumors with rapid spread and morbidity unless an interference with Chk2 activation or cyclin B1/Cdc2 interaction was delivered. 37 Although the maximum energy of radiation could be delivered directly to kill tumor cells theoretically, the normal cells around tumor tissue would not tolerate radiation energy that achieved tumor-killing effect.38,39 Therefore, the employment of radiosensitizing agents can increase tumor cell killing effect for a given physical dose of radiation. In this study, we demonstrated that MWE could be used as a radiosensitizing agent to assist radiation to kill TSGH 8301 cells via inducing MC.
Aurora kinases are essential for cell proliferation and could serve as biomarkers of a poor prognosis because they are expressed at high levels in solid tumors, especially Aurora A. 40 The nuclear accumulation of active Aurora A has been recently described to regulate the Haspin-H3T3-ph-Aurora-B feedback loop for the timely formation of the chromosomal passenger complex before spindle assembly. 41 Although our data showed that the treatment of IR along with MWE caused an astonishing and MWE dose-dependent induction of Aurora A phosphorylation 48 hours after the management, we considered it was a consequence of an inhibition on Aurora-B activity that blocked mitosis progression and led to a compensated increase in Aurora A activation. The work of Tao et al 42 reported that aurora B kinase inhibitor AZD1152 was able to mediate radiosensitization in vivo by enhancing MC in HCT116 colon cancer cells. We further linked the suppression of Aurora-B to the inhibitory effect on its downstream regulator Plk1 that consequently influence the downstream signaling by repressing RhoA level and increasing Cep55 expression. The activation of RhoA and repression of Cep55 are essential for accurate chromosome segregation and proper cytokinesis during mitosis.27-29 The blockage of such actions could be the promising cause of the promoting effect of IR/MWE on MC.
The most commonly encountered problem with cancer radiotherapy is radioresistance, and PI3K/AKT signaling pathway was proved to be involved in such process. 43 It was discovered that IR stimulated epidermal growth factor receptor (EGFR) that triggered PI3K/AKT signaling pathway and drove cancer cells to generate radioresistant ability. 44 Many PI3K inhibitors have been developed that are now approved by the FDA (such as idelalisib and copanlisib) or in clinical trials. 45 Ras/Raf/MEK/ERK was another signaling pathway induced by EGFR that contributed to the radioresistance of cancer cells. 46 Clinical trials testing EGFR, BRAF, and MEK inhibitors as single agents or in combination are ongoing. However, either pathway is complex, with many feedback loops and interactions with other pathways, which make it hard to predict the outcome of the inhibition of either one of them. It has been suggested that combined therapy with inhibitors targeting both PI3K/AKT and Ras/Raf/MEK/ERK was the most effective way to treat cancer in animal models, 36 and could be a promising strategy in human cancer management. The present study showed that MWE was able to assist the efficacy of IR by inhibiting Ras/Raf/MEK/ERK activity and decreasing PI3K level (Figure 4), suggesting that this combined treatment could target these 2 pathways concurrently that would be beneficial in future clinical application.
In the past decade, complementary and alternative medicine, as known as CAM, gradually becomes an important trend in the cancer therapy. For instance, PHY-906, extracting and condensing from 4-herb Chinese Medicine Formula, and pomegranate extract are under clinical trials to serve, respectively, as neoadjuvants to assist clinical colorectal cancer and prostate cancer therapy.47,48 The use of MWE together with chemotherapeutic agent and IR that could initiate multieffects leading eventually to MC, a new direction in cancer research other than apoptosis and autophagy, provide a promising new strategy with good potency for bladder cancer.
In conclusion, we provided evidences showing that the assistance of MWE under the influence of IR caused severe MC via repressing Ras/Raf/MEK/ERK signaling pathway and Aurora B activity in bladder cancer cells. These data support further preclinical work on the application of MWE as adjuvant for radiosensitization.
Footnotes
Acknowledgements
The authors would like to thank Chung Shan Medical University and Chi Mei Medical Center for providing the necessary facilities and financial support.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by Chung Shan Medical University, Taichung City, Taiwan (Grant No. CSMU-CMMC-106-02); and Chi Mei Medical Center, Tainan City, Taiwan (Grant No. CMCSMU10602).
