Abstract
Background: Although epidemiological research demonstrates that there is an association between lifestyle factors and risk of breast cancer recurrence, progression of disease, and mortality, no comprehensive lifestyle change clinical trials have been conducted to determine if changing multiple risk factors leads to changes in biobehavioral processes and clinical outcomes in women with breast cancer. This article describes the design, feasibility, adherence to the intervention and data collection, and patient experience of a comprehensive lifestyle change clinical trial (CompLife). Methods: CompLife is a randomized, controlled trial of a multiple-behavior intervention focusing on diet, exercise, and mind-body practice along with behavioral counseling to support change. The initial exposure to the intervention takes place during the 4 to 6 weeks of radiotherapy (XRT) for women with stage III breast cancer and then across the subsequent 12 months. The intervention group will have 42 hours of in-person lifestyle counseling during XRT (7-10 hours a week) followed by up to 30 hours of counseling via video connection for the subsequent 12 months (weekly sessions for 6 months and then monthly for 6 months). The primary outcome is disease-free survival. Multiple secondary outcomes are being evaluated, including: (1) biological pathways; (2) overall survival; (3) patient-reported outcomes; (4) dietary patterns/fitness levels, anthropometrics, and body composition; and (5) economic outcomes. Qualitative data of the patient experience in the trial is collected from exit interviews, concluding remarks, direct email correspondences, and web postings from patients. Results: Fifty-five patients have been recruited and randomized to the trial to date. Accrual of eligible patients is high (72%) and dropout rates extremely low (5%). Attendance to the in-person sessions is high (95% attending greater than 80% of sessions) as well as to the 30 hours of video counseling (88% attending more than 70% of sessions). Adherence to components of the behavior change intervention is high and compliance with the intensive amount of data collection is exceptional. Qualitative data collected from the participants reveals testimonials supporting the importance of the comprehensive nature of intervention, especially the mind-body/mindfulness component and social support, and meaningful lifestyle transformations. Conclusion: Conducting a comprehensive, multicomponent, lifestyle change clinical trial for women with breast was feasible and collection of biobehavioral outcomes successful. Adherence to behavior change was high and patient experience was overwhelmingly positive.
Introduction
Breast cancer accounts for more than 40 000 deaths and costs billions of dollars each year in the United States. 1 It is the most common cancer in women, accounting for 15% of all cancer-related deaths in women and ranking as a leading cause of cancer-related mortality among women 20 to 59 years of age. 1 In 2016, an estimated 246 660 new cases of invasive breast cancer and 61 000 cases of ductal carcinoma in situ are expected in women in the United States. 1
Epidemiological research demonstrates that after a diagnosis of breast cancer there are associations between lifestyle factors and the risks of breast cancer recurrence, disease progression, and mortality. 2 Excess weight, unhealthy diet, low levels of physical activity, and excessive sedentary behavior all are associated with worse clinical outcomes than are healthier lifestyle choices. 2 Functional transcriptomic analysis showed that obesity induces multiple biological functional changes linked to cancer hallmarks in estrogen receptor–positive breast cancer in humans and mice. 3 In addition, preclinical in vivo and clinical research shows that psychosocial factors such as stress, social support network, and mindfulness influence treatment outcomes. One possible mechanism for this influence is that these factors play an important role in the modification of cancer-related behaviors and biological processes that affect tumor biology.
Research demonstrates the importance of the tumor microenvironment for facilitating and controlling cancer growth. Microenvironmental and systemic processes—including gene mutations, angiogenesis, apoptosis, hormone function, cytokine regulation, cell-signaling pathways, and immune function, to name a few—determine whether cancerous cells will grow. These cancer-related processes are influenced by lifestyle factors such as body mass index (BMI), diet, physical activity, stress, and social support. Initial research shows that intervening on these factors, particularly by cognitive-behavioral approaches and other methods grounded in behavioral theory, can modify cancer-related biological processes and influence long-term outcomes.4-9 However, few clinical trials have examined multicomponent, comprehensive lifestyle change to determine the effects on clinical outcomes and evaluating the underlying biopsychosocial mechanisms.
Epidemiological studies in many cancer types, including breast cancer, have reported associations between adherence to lifestyle guidelines in the areas of weight, diet, and physical activity and both cancer incidence and outcomes.10-13 Importantly, these studies have found that the more guidelines that are followed, the greater the reduction in cancer risk. Few studies have assessed the effect of physical activity and the independent effect of inactivity on survival and disease recurrence after a breast cancer diagnosis; however, such studies have had convincing results, finding that more activity and less sedentary behavior improve clinical outcomes.14-20 Studies examining the effects of diet suggest that eating a primarily plant-based diet that is low in total and saturated fat with moderate alcohol consumption lowers the probability of disease recurrence.21-23 There is also evidence demonstrating the health-damaging effects of chronic stress. Research has shown that chronic stress affects almost every biological system in the body 24 and can promote cancer growth and progression.25-27 The underlying mechanisms for such effects are complex and involve chronic activation of the sympathetic nervous system and the hypothalamic-pituitary-adrenal axis.28,29 Sustained elevations from these pathways can result in diverse effects such as inflammation, angiogenesis, stimulation of cancer invasion, and even reduced efficacy of chemotherapy drugs.30-32 The underlying signaling pathways 33 offer opportunities for designing new biobehavioral approaches for disrupting the effects of stress biology on cancer biology.
The psychological and behavioral effects of stress can also influence health behaviors, often leading to poor diet, lack of exercise, substance abuse, poor sleep, and decreased quality of life (QOL).34-39 The mechanisms underlying the role of lifestyle factors in cancer initiation and progression are diverse and multifaceted and have not been fully elucidated. However, research has linked individual lifestyle components (ie, diet, exercise, stress) to important cancer regulatory mechanisms such as immune function, inflammatory processes, gene expression, and hormone regulation.22,40-45
Few studies have tried to intervene on lifestyle factors in women with breast cancer. The Women’s Intervention Nutrition Study (WINS) assessed the effect of a reduced fat dietary intervention on breast cancer recurrence. 46 At 5-year follow-up, lower dietary fat intake in the intervention arm was associated with a reduced risk of recurrence. The Women’s Healthy Eating and Living (WHEL) Study47-49 assessed the effects of increased vegetable, fruit, and fiber intake and decreased fat intake on disease recurrence. There were no differences in recurrence between groups when diet alone was assessed; however, there was a 50% reduction in recurrence risk in women who ate at least 5 servings of vegetables and fruits per day and engaged in at least 30 minutes of physical activity 6 days a week, 49 suggesting synergy between diet and physical activity (diet is necessary, but not sufficient alone to modify outcomes). This finding has been reinforced by studies showing that adhering to multiple guidelines for weight, diet, and exercise lowers the risk of cancer-related death, including breast cancer.10-13
Several approaches are available for managing distress in cancer patients. Cognitive-behavioral therapy (CBT), cognitive behavioral stress management, and supportive expressive therapy all have been found to help improve psychosocial and QOL outcomes.50-59 A recent study of cognitive behavioral stress management found that this technique led to decreases in inflammatory gene expression 60 and longer survival. 61 Mind-body practices also have a positive effect on many systems in the body, improving QOL, reversing the harmful effects of stress, and creating fundamental changes in the way the brain functions.62-70 These practices can affect neurotransmitters, and neuromodulation is essential in the management of the stress response. 71 Evidence suggests that combining the best of what we know from conventional programs with simple mind-body practices (ie, yoga, meditation, mindfulness) can lead to more behavior change and better outcomes than using only one specific program.72,73 This combined approach is especially important when trying to modify multiple behaviors at the same time with a goal of targeting disease outcomes.
Previous studies have predominantly modified dietary factors, physical activity, or distress, but rarely have all 3 factors been targeted in one study. Andersen et al examined a comprehensive, psychosocial, didactic intervention program for women with stage II or III breast cancer who were undergoing chemotherapy. 4 Patients in the intervention arm had significantly improved behavioral and psychological health outcomes and immune function compared with patients who received usual care.74,75 Patients in the intervention group were also found to have a significantly reduced risk of breast cancer recurrence (hazard ratio = 0.55; P = .034) and death from breast cancer (hazard ratio = 0.44; P = .016). 4
The ongoing comprehensive lifestyle change clinical trial (CompLife) seeks to determine whether providing overweight, stage III breast cancer survivors behavioral counseling and support in the areas of diet, exercise, and mind-body practice can lead to longer disease-free survival (time to recurrence is the primary outcome). Multiple secondary outcomes are also being evaluated and are described in more detail below. By examining multiple psychosocial, behavioral, and biological pathways while lifestyle factors are manipulated as part of a randomized clinical trial, we will be able to better understand the role of specific mechanisms linking lifestyle factors to disease outcomes. The current article describes the design of the trial, feasibility, adherence to the intervention and extensive data collection, and the patient experience of the first third of women recruited to the study.
Methods
Design
A randomized, controlled trial is being conducted evaluating CompLife, a comprehensive lifestyle intervention for women with stage III breast cancer. The intervention is delivered during the 4 to 6 weeks of radiotherapy and then across the subsequent 12 months. The trial is being conducted and reported according to CONSORT guidelines with the cost-effectiveness analysis conducted and reported according to CHEERS guidelines. 76 One hundred and sixty patients will be randomized to either the CompLife or usual care (UC) control group.
Participants
Eligible participants need to meet the following inclusion criteria: (1) be 18 years of age or older; (2) diagnosed with stage III breast cancer and scheduled to undergo a 4- to 6-week course of radiotherapy (XRT); (3) oriented to person, place, and time; (4) BMI of 24.5 or higher; (5) have a physician release for exercise or clearance according to the Physical Activity Readiness Questionnaire (PARQ); (6) and must meet at least 2 of the following criteria related to lifestyle: (a) consume less than 3 servings of fruit and vegetable/day; (b) engage in less than 75 minutes moderate/vigorous intensity activity per week; and (c) engage in a mind-body practice less than 4 times a month. Additionally, patients are excluded if they have any of the following: a recurrent breast cancer diagnosis; another primary cancer diagnosis within 5 years of consent (not including non-melanoma skin cancers); any major thought disorder (eg, schizophrenia, dementia); communication barriers (eg hard of hearing); or extreme mobility issues (eg, unable to get in and out of a chair unassisted).
Procedures
The study has been approved by the MD Anderson Institutional Review Board (2012-0112). Written informed consent is obtained from all participants included in the trial. Participants are informed that they are not obliged to take part in the study and are free to withdraw at any time without negative consequences on their future care.
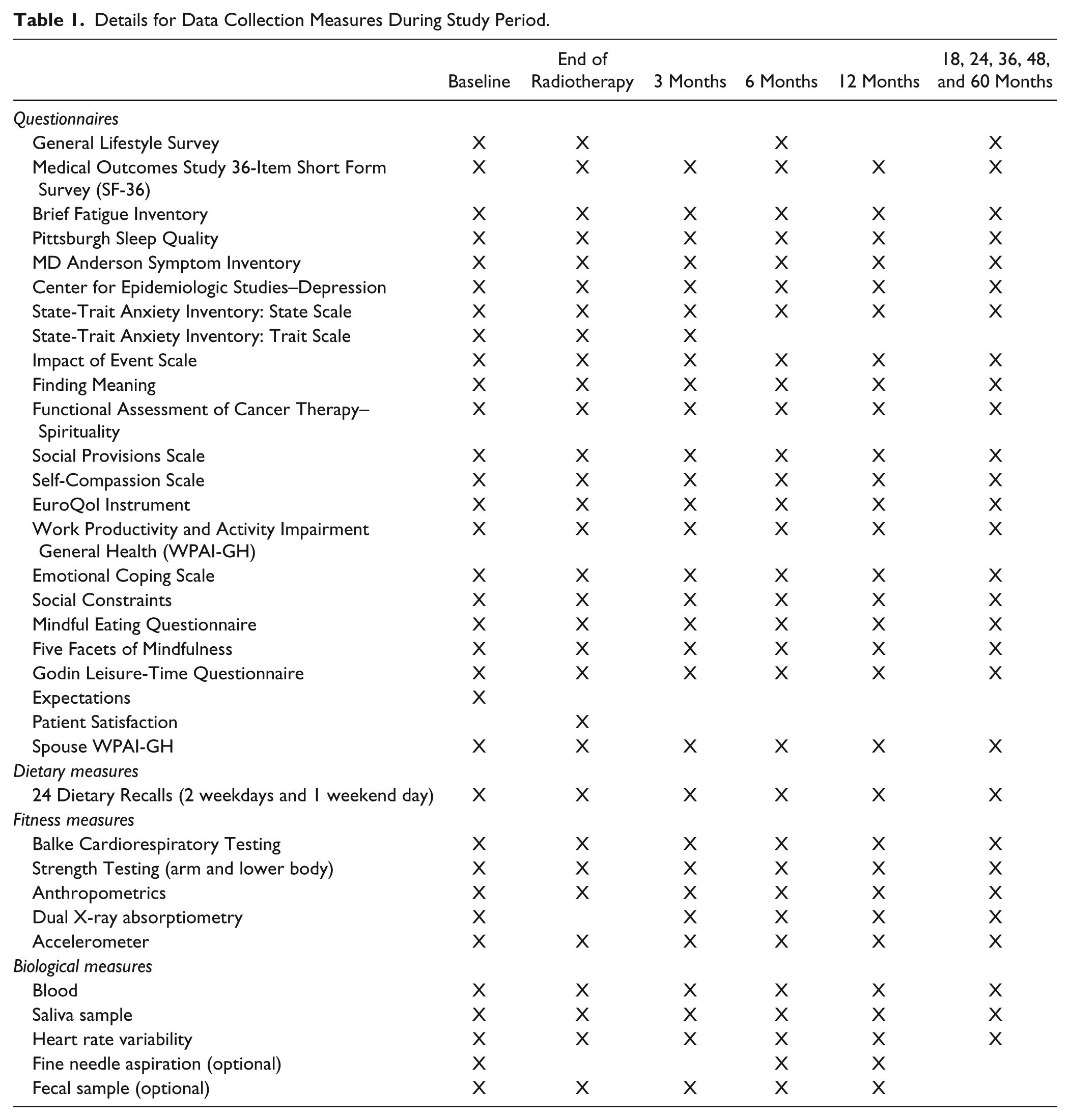
Potential study participants are identified by referring radiation oncologists and recruited and consented by study staff at the beginning of radiation therapy. After providing informed consent, patients complete the baseline assessment, which takes place prior to their first radiation treatment. The baseline assessment includes a 60- to 90-minute-long battery of questionnaires completed electronically on an iPad or online. Participants who do not have a tablet are loaned one while participating in the study. All study participants will also be issued an accelerometer to wear for 5 days. To determine the circadian nature of cortisol, 4 saliva samples a day (waking, 45 minutes later, approximately 10 hours after waking, and at bedtime) will be obtained for 3 consecutive days. All participants receive 3 randomly scheduled calls within 2 weeks of consent for three, 24-hour diet recalls. Patients undergo an electrocardiogram, fitness testing, a whole body dual-energy X-ray absorptiometry scan to measure body composition, and provide 100 mL of peripheral blood. An optional fine needle aspiration of healthy breast tissue will also be collected for those with a contralateral intact breast and provision of a fecal sample for microbiome analyses. All measures are obtained before the patient initiates radiotherapy. Patients’ parking and transportation costs are paid if they are not covered by the hospital. Patients are also given 3 small gift cards at each time point after completing each main component of the assessments (total of $100 per time point). Follow-up assessments for all participants take place during the last week of radiotherapy; 3, 6, and 12 months after baseline; and annually for 5 years from the end of radiotherapy. The intervention delivery outline is shown in Figure 1, and data collection details are shown in Tables 1 and 2.

Sample schedule of intervention sessions.
Details for Data Collection Measures During Study Period.
Details for Biological Outcomes.

A randomized pilot run-in phase of 6 patients was conducted first to determine if patients were compliant with the intervention and the intensive data collection procedures. Patients provided formal exit interviews on their satisfaction with the intervention. After the first 6 patients provided data through the end of XRT, a report was submitted to the institutional review board (IRB) and no major changes were made to the intervention. Recruitment continued after approval from IRB.
Randomization
One hundred and sixty participants (184 prior to attrition) will be randomized using a form of adaptive randomization, minimization, because simple randomization could result in covariate imbalances. 77 Minimization is similar to stratification; however, it results in better group balance and does not suffer from the limitations of stratification, especially when several participant factors are used. In minimization, group assignment is done sequentially. Before a participant is assigned to a treatment group, the number of already randomized participants with similar covariate characteristics is totaled. The totals are computed based on marginal sums so that each covariate is considered separately. The treatment assignment for a participant is then based on which treatment group assignment would produce the best overall balance with respect to the covariate characteristics. 77 In this study the patient characteristics used for group assignment are BMI (dichotomized on a running mean); diabetes (yes/no); age (dichotomized on a running mean); time since diagnosis (dichotomized on a running mean); menopausal status (pre/post); staging (A, B, or C); surgery type (segmental/mastectomy); and hormone receptor/HER2/neu status.
Intervention
The CompLife intervention is an intensive 4- to 6-week intervention (based on whether the women are undergoing 4 or 6 weeks of radiotherapy) followed by video-conference counseling sessions over the subsequent 12 months consisting of (1) 6 weekly in-person, face-to-face counseling sessions incorporating motivational interviewing, CBT, and psycho-education, followed by 30 mobile-enabled video counseling sessions (weekly for 6 months and then monthly for the subsequent 6 months); (2) 12 exercise sessions (2×/week for 6 weeks); (3) 12 nutrition sessions, half of which are experiential cooking classes (2×/week for 6 weeks); and (4) 12 mind-body sessions (2×/week for 6 weeks). Patients undergoing 4 weeks of radiotherapy have the same amount of contact with each practitioner, simply condensed into 4 weeks. All counseling sessions are conducted one-on-one. For the other sessions, patients can invite family or caregivers to participate and patients are grouped when possible with other patients. Due to radiation scheduling issues the maximum number of patients in a class has been three. The majority of sessions are conducted one-on-one. Patients also have access to a web-based program that contains didactic information to support the 3 areas for behavior change and tools for logging physical activity, diet, and mind-body practice. It also provides a platform for posting and commenting on a shared discussion board.
At all follow-up appointments during the first year after XRT, patients have 2-hour booster sessions with the team. For 1 year after XRT, patients are also provided a family gym membership and connected to a community fitness coach. Community coaches have ongoing contact with the study personal trainer in order to ensure effectiveness and safety.
All participants receive counseling workbooks, along with yoga mats, resistance training tubes, copies of diet and exercise recommendations, 2 cookbooks, a Vitamix blender, instructional video and audio recordings, and a copy of Anticancer: A New Way of Life by Dr David Servan-Schreiber. All interventionists meet on a weekly basis to discuss progress and any particular issues experienced by study participants.
The behavior change constructs of the intervention come from multiple theories and models used to explain the cognitive, behavioral, and psychosocial determinants of health-related behavior. These include the transtheoretical model (TTM), social cognitive theory (SCT), and theory of self-determination (SDT) that work with the intervention models for clinical delivery, namely, CBT and motivational interviewing (MI). Prior evidence has demonstrated SCT’s utility in physical activity and diet for long-lasting health improvements in cancer survivors.78-80 Specific constructs shown to predict behavior in this population include SCT’s outcome expectations, self-efficacy, environmental facilitators, and TTM’s stage of change, where higher stage of change predicts greater uptake of behavior. 78 Importantly, TTM recognizes change as an ongoing process whereby the individual moves across stages and can relapse, requiring renewal of goals in an effort to change. The autonomous individual’s choices occur alongside social and environmental influences, overlapping with the SCT constructs of access in the environment and interpersonal influences. Much like TTM, the intervention methods of SDT and MI have been noted for being explicitly person-centered and process-oriented. Both emphasize optimal behavior change involves deep personal commitment and engagement to foster a sense of autonomy. 81 SDT and MI have often been used together to create long-term change in nutrition and physical activity behavior. By using multiple theories and drawing the appropriate aspects from each, we have developed a comprehensive model with the most likelihood of creating sustainable change.
Overall Goals
Physical activity
Patients undergo 30 to 60 minutes of moderate-to-vigorous intensity aerobic activity up to 6 days/week supervised and unsupervised. During XRT they meet with the exercise trainer 2 times a week where they spend 30 minutes doing resistance training with exercise bands and walk for 30 minutes. After XRT, they have continued training with a coach in the community at a local fitness center. They continue the same program with the local exercise coach who checks in with the MD Anderson exercise specialist on a weekly basis. The exercise specialist is on call as needed, as facilitated by the counselor. The exercise coach is an American College of Sports Medicine Certified Cancer Exercise Trainer, International Sports Sciences Association Certified Personal Trainer, and National Exercise & Sports Trainers Association Certified Personal Trainer with 13 years of experience as an exercise coach.
Diet/nutrition
Patients are counseled by a dietitian to follow the American Cancer Society and American Institute for Cancer Research guidelines. They are taught how to shop and cook during XRT. They learn about specific healthy foods, cooking techniques, and so on. Foods are classified as health depleting, health supporting, or neutral. Patients are asked to strive for a ratio of 90/10 of health supporting/health depleting. They are provided healthful restaurant choices and markets accessible to each patient to facilitate healthy shopping and eating. A nutritional specialist will also be on call for any additional concerns. The dietitian has more than 5 years of experience and is a licensed dietitian and registered by the Academy of Nutrition and Dietetics with training in MI.
Mind-body practice
Patients learn a seated meditation and practice a yoga-based movement practice (Sun Salute). Patients are asked to gradually increase their practice up to 12 Sun Salutes, do a brief deep relaxation technique, and up to 20 minutes of guided meditation each day. During sessions, the interventionist will provide one-on-one support concerning mindfulness, and mindful living in general, mindful eating, mindful exercise, relaxation, and so on. It is likely that relaxation and mindfulness exert influence on various processes of behavior change. The mind-body instructor is also available for additional support as facilitated if deemed necessary by the counselor contacting patients post XRT. The mind-body instructor has over 10 years of experience teaching and specialized training in oncology as well as MI training.
Behavioral counseling/social support
Participants have weekly counseling sessions rooted in CBT and MI to help support the behavior change. This critical component helps support behavior change. It also provides the participants a support person with whom they can confide and is there to support them in areas distinct from diet, exercise, and mind-body practices. Many barriers to successful behavior change need to first be uncovered and modified to facilitate long-term change. The counselor bolsters individual determination and self-efficacy so that the challenges of unspecified behavior change can be repeatedly tackled, including the challenge of relapse. If the participants required long-term psychological support they were referred for further assessment and counseling in addition to continuing with the counselor on the study. The behavioral counselor has a Master’s in Education in Counseling Psychology, extensive training in MI, over 10 years of experience, and is supervised by licensed psychologists.
Tailored workbooks with goal-setting, cognitive restructuring, methods for addressing barriers, and so on, will be used throughout the one-on-one counseling. The research team provides patients information on how to find health promoting grocery stores, restaurants, farmer’s markets, and fitness centers in their community, increasing environmental access or at least awareness of access points, facilitating social norms, and engendering a climate of relatedness for the individual committed to long-lasting change.
Usual Care
Patients randomized to usual care are provided patient-education materials for cancer prevention and control available through the MD Anderson patient education center including information on diet, exercise, and stress management. They have regular contact with the research staff to coordinate the data collection, but no counseling is provided. Oncologists are not curtailed from making their usual recommendations to patients about lifestyle, if that is part of their practice. Patients can also access all resources at MD Anderson to support healthy lifestyle, including programs offered by the Integrative Medicine Center. Determining the programs the UC patients attend and detailed assessment of changes in their lifestyle behaviors will help determine potential drop-in rate. Due to the outcomes being assessed (recurrence of disease and overall survival) the UC group is not a wait-list group.
Compliance
We have developed online behavior-tracking tools that allow us to track compliance, and we use gaming-based theories to compensate patients for the time they spend tracking their behaviors. Points equate to dollars in a schema where participants are given a blank re-loadable gift card on which we deposit funds on an ongoing basis. This also eases patient burden as a means for quick compensation as they complete assessments. For participants without computer access, an iPad is loaned to them for use throughout the study.
To ensure safety and quality control, the mind-body and resistance training sessions are videotaped so that both visual and audio data will be available for review. Counseling and diet sessions are audio-recorded only. Ten percent of each interventionist’s sessions are randomly chosen for review.
Outcomes
Primary
Evaluation for recurrence will follow current American Society of Clinical Oncology guidelines and the MD Anderson Cancer Center algorithm.
Secondary
Multiple secondary outcomes are being collected: (1) biological (blood, saliva, and fecal samples): immune function; circulating tumor cells and epithelial-mesenchymal transition-inducing transcription factors; endocrine function; insulin and glucose metabolism; cancer-related pathways; antioxidant capactity; growth factors; oxidative stress and inflammatory mediators; nutrient levels; and gut microbiome biodiversity; (2) overall survival; (3) patient-reported outcomes: fatigue, sleep disturbances, other aspects of QOL, mental health, social support, and measures of positive growth; (4) dietary patterns, fitness levels, physical activity, anthropometrics, heart rate variability, and body composition; (5) economic outcomes including medical costs, work and/or home productivity, and cost-effectiveness; and (6) proliferation markers from healthy breast tissue acquired from fine needle aspirations. We are also freezing plasma, serum, and extracting DNA and RNA and viable cells for future miRNA studies, gene array, and so forth. Tables 1 and 2 summarize the measures and timing of data collection.
Statistical Analysis
Our primary analyses will follow the intent-to-treat (ITT) principle. We will analyze the primary endpoint of the study, disease-free survival, using a multivariate comparison of disease-free survival for the 2 groups in a Cox proportional hazards analysis. 82 We will consider the following as potential covariates in the analysis: tumor and nodal status, menopausal status at diagnosis, tumor factors, treatment regimen.
Continuous variables include changes in biological pathways (immune and endocrine function, insulin and metabolism, etc), dietary patterns, fitness levels, heart rate variability, and patient reported outcomes such as fatigue, sleep disturbances, and other aspects of QOL. These variables are repeated measurements on multiple outcomes, possibly including hierarchical structure over subscales and related outcome measures. Our desired inference includes hypotheses about treatment effects at different times and on different outcomes. Because we expect scores on the criterion measures to be correlated within individuals over time, we will use generalized linear mixed model regression. Separate sets of analyses will be conducted for each criterion variable. In modeling these data, intervention condition is a between-subject factor, time is a within-subject factor, and baseline measures and other medical and demographic factors will be included as covariates.
For the Comprehensive Lifestyle group, we will determine compliance with practice. We will also assess changes in diet, physical activity, and stress management practices in both groups. Exploratory analysis will examine change in lifestyle as a mediator of clinical, biological, and patient-related outcomes.
Missing Data and Dropouts
Our proposed primary analysis on disease-free survival naturally accounts for the censored data. Our secondary analyses will be based on all observed data. These analyses will be valid under the missing-at-random assumption. 83 However, we will conduct sensitivity analysis assuming different missing data mechanisms (including missing not at random via appropriate models such as selection models). 83 We will also explore alternative approaches to handling missing data, including using multiple imputation and pattern-mixture models, 84 to potentially reduce bias and improve efficiency in the statistical inference for the secondary outcomes. Similar results based on the sensitivity analysis will strengthen our study findings related to the secondary outcomes.
Sample Size
We expect that 50% of patients in the control group will have a recurrence within 4 years of completing radiotherapy—a median time to recurrence of 48 months. By including 80 patients in each of the 2 groups, a total of 160, with a total number of events required of 60, we will be able to detect a difference between rates of recurrence-free survival at 4 years of 50% and 70% between the 2 groups, with a 1-sided significance level of .05 and 80% power. At 5 years these rates are estimated to be 42% and 64%, and at 10 years 18% and 41%.
The hazard rates of the 2 groups are estimated to be 0.0144 and 0.0074, which translates to a hazard ratio of 0.515, which is similar to the hazard ratio for recurrence-free survival in a previously reported study. 4 The reported study included patients with stage II (90%) and III breast cancer, and the intervention was a psychological program with lifestyle recommendations that was less intensive than what is being done in the current study. By including only patients with stage III disease and utilizing a more intensive intervention program, we expect to have larger effect size than that in the Anderson study. 4
By including 80 patients in each group, we will also be able to detect and declare as significant differences between groups at any specific time point for continuous variables computed from the QOL data, blood work, and so on, that are at least 0.46 standard deviations or larger (effect size of 0.46), assuming a 2-sided significance level of .05 and 80% power. Power will in general be higher to detect the same average differences over time using the repeated measures data. In order to ensure we have 160 evaluable patients for the secondary outcomes, we will recruit 184 patients to account for 15% attrition. Evaluable is defined as having baseline and at least one follow-up assessment after the end of XRT. For those in the intervention group, this will also include completing the 6-week intensive portion of the program.
Results
Participants
The first 6 patients recruited to the trial were compliant with data collection and the intervention components. Only one major modification was made to the exclusion criteria to exclude patients with uncontrolled diabetes; the first patient had uncontrolled diabetes and had a recurrence within 6 months of consent. After IRB approval, expanded recruitment began March 2014; a total of 57 participants have been recruited to the trial (June 2016; CompLife N = 26 and UC N = 29). Of the 76 initially eligible approached, 57 consented for a consent rate of 75%. Reasons for not consenting included lack of time, lack of interest, and other reasons. Of the 57 patients who consented, 2 were found ineligible before randomization. To date, 5 patients have dropped out all within the first 6 weeks after recruitment (see Figure 2). Table 3 shows demographic and medical information for the 55 eligible participants randomized. In terms of meeting eligibility criteria of not engaging in healthy lifestyle (diet, exercise, and mind-body practice), 67% did not engage in 2 of the lifestyle factors and 33% did not engage in all 3 lifestyle factors. It is noteworthy that 50% of the women are of a minority racial group, 38% have less than a college degree, and 20% have a household income of less than $50 000/year. There was a large distribution in age (range 25-81 years), BMI (range 24.5-41.3), and number of children (range 0-5).

Consort diagram through the end of radiation treatment.
Study Participant Baseline Demographic and Medical Factors.

Abbreviations: BMI, body mass index; TNBC, triple negative breast cancer; HR, hormone receptor; HER, human epidermal growth factor receptor; BRCA1 and BRCA2, breast cancer susceptibility gene 1 and breast cancer susceptibility gene 2; lifestyle factors, not engaging in lifestyle behaviors at the time of study consent.
Compliance With Intervention
Of the 26 patients randomized to CompLife, one was not compliant and dropped out before undergoing any sessions and 2 are currently going through the program. Compliance with the 6-week intervention (a total of 42 sessions over 6 weeks) has been high (21/23 missed ≤3 sessions; these 2 patients missed sessions due to hospitalizations). Attendance at the video counseling sessions has been high, with 10/23 (44%) attending 100% of the available sessions and 44% attending 70% to 99% of the available sessions.
Participants also tracked daily food intake using the tracking tool that automatically calculates the ratio of heath supporting to health depleting food choices. By the end of the 6 weeks, 21/23 participants exceeded the 90:10 goal, and the other 2 patients met the goal and showed improvement in dietary pattern. In terms of exercise, about 96% of the participants exercised 30 minutes per day or more, with one participant exceeding an hour per day (based on self-report). Of the 23 participants who have completed the 6-week intervention and tracked daily, none practiced meditation before commencing the program. By the end of XRT, 17/23 patients (74%) exceeded or practiced the daily prescription of 20 minutes of meditation a day (average = 35 minutes/day; range = 20-80 minutes). The remaining 6 patients practiced 10 to 15 minutes daily. In terms of the sun salutations, 57% reached 10 to 12 daily rounds and 30% were doing at least 8 rounds a day by the end of the 6 weeks, with the remaining three doing less than 8 rounds a day.
There is a concern about drop-ins because of the nature of lifestyle research. To date, of the patients randomized to the UC group, 5 participants can be classified as dropping-in to one or more components as evident by self-reported activity, but none to all 3 areas. Observed drop-in behavior attests to the feasibility of the intervention components as desirable practices for cancer survivors. There have been no reported adverse events due to the study.
Compliance With Data Collection
Compliance with data collection has been exceptionally high for both groups. Complete data on each of the measures in the categories of food recall interviews, fitness measures, and blood and saliva collection are available for at least 90% of the participants. This is higher than our previous studies and exceeds our expectations for the ongoing trial. Of the participants eligible for the optional fine needle aspiration procedure 30% have consented. Similarly, 67% of participants agreed to provide the optional fecal sample.
Patient Experience
We conducted formal exit interviews with the first 3 patients randomized to the intervention group to determine their satisfaction with the intervention. Patient experiences are also collected throughout the study through interaction with the 4 primary instructors, through evaluation of the recorded final session at the completion of XRT with the behavioral counselor, and a 12-month exit interview. The qualitative data can be found organized by theme in Table 4. The qualitative reports indicate overwhelming support for the program and life transformations. The overarching categories included fundamental shift; gratitude; mindfulness; information sharing; and learning and preparedness for change. One aspect that we had hoped would take place was the information sharing of what the patients were learning with their family and friends. Patients indicated that they were teaching meditation to their families and modifying the diet of the whole family. For example, one patient indicated, “This whole program has changed me drastically. I will pass it on to my friends and family.”
Qualitative Reports.
Below are responses to open-ended interview questions, concluding patient remarks from final in-person sessions, or end of intervention sessions organized thematically. Themes were not set a priori; rather, they emerged as interview transcripts were reviewed. Emerging themes included fundamental shift in one’s self-understanding; gratitude; mindfulness; information sharing; and learning and preparedness for change.
Quotes From 60-Minute Exit Interviews
After the intensive 6-week portion of the program, the first 3 intervention participants (CL001, CL003, and CL006) were interviewed. Example Question: “Overall, how did you feel about the program?”

It is clear from many of the patient testimonials that the support they receive from the 4 counselors is a key factor supporting their success in behavior change. Another patient wrote,
I know the changes to my diet, the tracking, the talks with Lisa [the counselor] strengthened me beyond measure and I feel that my continued adherence to this program is no longer just following a plan. I’ve changed my lifestyle for the better and you all had a hand in it!
Another patient said: “It’s been 24 years since I’ve taken care of myself and now they have given back all of the tools I needed to get back to normal. The support and learning from all of you means so much.”
The participants are also getting support from people in their own communities to sustain the changes. For example, one patient said her family was holding her accountable to the dietary aspects of the study and her children said, “Mom, should you really be eating that?” Another participant with an 11-year-old son said she wanted to be healthy for him so that instead of sitting and watching him play, she would have the energy to join him. Her son told her that he wanted to join a gym and said, “Mom, we need to change the way we eat and I want to get fit.” He also told her he wanted to practice yoga with her. The parallel process of both of them making changes and strongly supporting one another is an important aspect for making sustainable lifestyle changes. One patient said, “If this is going to work, I have to find a way to include the entire family.” This was especially true with the exercise and diet components. Not only did she want to include her family, she also wanted to model the changes for her children so that they could learn healthy habits from a young age.
Another theme that was consistent across participants was the importance of mindfulness and the stress management component. In joining the study none of the patients had a meditation practice or a regular mind-body practice, yet they were experiencing a lot of stress. One patient noted, A lot of times in life you’re buried in everything you have going on and you’re not taking care of yourself. That’s not the case anymore. Because of this program, I’m just happy. I’m at peace. I’m a new person.
Patients noted how important this component was in supporting their other behaviors. For example, one patient noted, “I am doing so much better because I am being more mindful with eating, exercise and life. You take the moment and you inhale and exhale. I really am so much more mindful.” A strong statement of the more profound changes that are taking place comes from the following patient quote:
I don’t know if anyone has ever felt like this. To have so many questions, but the answers are trapped in you. That’s how I’ve felt for so long. Well, I’ve come to realize that through meditation and deep relaxation that my answers are flowing. I can honestly say that I’ve never been in such deep thought about my life.
Reinforcing the multicomponent aspect of the study and that all 3 components play an important role, when one participant was asked to discuss which area of lifestyle change (mind-body, exercise, and/or nutrition) had the most influence on her health and well-being, she noted, “It’s really a toss-up because all three areas of wellness had an impact.”
Discussion
The ultimate goal of the Comprehensive Lifestyle study is to determine whether making changes in the areas of diet, exercise, and mind-body practices in women with stage III breast cancer who are overweight and not following the American Institute for Cancer Research guidelines for cancer prevention and control can reduce the probability of recurrence of disease. The trial will also provide evidence for the biobehavioral mechanisms by which behavior modifies disease pathways. Although there was concern when designing the trial that the intensity of the behavior modification program would lead to low recruitment, poor adherence to behavior change, and missing data due to the extensive measures being collected, to date the trial has been extremely successful in all of these areas. The trial has demonstrated high recruitment rates, ongoing compliance with the intervention is good, there is high adherence to data collection in both groups, initial indication of behavior change, and high patient engagement as indicated through the qualitative data. It is also important to note that the trial has been successful in recruiting patients from minority groups.
Most important, the patients reported engaging in behavior change in the areas of diet, exercise, and mind-body practices. This is something that will be tracked over time, but the initial indication is high adherence even at 6 and 12 months after the end of radiotherapy. If patients can maintain this level of adherence through 12 months, research indicates the likelihood of continued success is high. 85 From a data collection perspective, the trial has also been successful with over 90% of the participants providing complete data.
A strength of the trial is that it is examining multiple health outcomes and diverse disease pathways. Furthermore, delivering the program during radiotherapy provides a unique time when patients are already coming to the medical center for their treatment and allows for in-person sessions in order to optimize learning, skills demonstration, mastery, autonomy, and compliance. Other ongoing studies in the area of lifestyle change are either focusing on just trying to modify BMI through either diet, exercise, or both or are studies examining diet and exercise alone or combined.86-90 As stress and psychosocial issues are such a salient factor for these women, missing this component will compromise the benefits of these programs. Evidence shows that stress can have a direct effect on cancer biology 27 and an indirect effect on outcomes through influencing the health supporting behaviors we are trying to modify.35-39 As the patients are noting, the stress management component from the mind-body practices and the social support and behavioral counseling they are getting are critically important to them in order to fully engage the behavior change in the areas of diet and exercise.
This is also the first trial to our knowledge of behavior change of this magnitude that is using an incentivization structure for gamification of behavior change and tracking. Claiming points as a reward in theory leads to feelings of competence, increasing self-efficacy and motivation. In addition, unlike other trials that are using wellness coaches to teach diet and exercise, experts in each area lead all the counseling sessions in the Comprehensive Lifestyle trial. This ensures that participants have the necessary support to engage optimally in the 3 behaviors of mind-body practices, exercise, and health promoting diet. Also, an important strength is that the remote counseling over the course of 12 months allows for continued psycho-education and support in the home environment. This level of support is important as a key feature of this study is teaching participants how to carry on these behaviors in their homes and in their communities once the “training” component is over.
There are some weaknesses with the ongoing trial design. A generalizability and replicability issue for this trial is the amount of time required by personnel and individualized attention to each participant, though the type of personnel required (dietitians, exercise coaches, mind-body instructors, and behavioral counselors) are readily available in most communities and should be essential members of any health care team. During radiotherapy patients receive a total of 7 hours of counseling a week for 6 weeks (or 10-11 hours a week for 4 weeks), followed by weekly sessions for 6 months and then monthly sessions for the subsequent 6 months. This results in a total of 72 hours of counseling time. While this may threaten the external validity of our findings, the authors believe the protocol’s manualized program can provide training materials for similar programs. In addition, 72 hours of counseling is a relatively modest investment of time and money if this leads to improved QOL and overall health and decreased recurrence of disease. The components of the intervention that take part in the community (eg, restaurant choices, gym memberships, etc) utilize existing networks that reinforce lasting change once participants return to their usual daily lives. Due to linking the delivery of the program to patients’ radiation treatment schedules, most of the sessions are delivered in a one-on-one format because of scheduling challenges. The one-on-one format is required for the psychological counseling, yet the other sessions can include family and caregivers and other patients in the program. The lack of group support that is a known active component of many psychosocial interventions is missing in the program. However, patients are counseled on how to engage with their support networks in a healthy manner. Linking the delivery of the intervention to their radiation treatment schedule makes it easier for more patients to participate, even those from out of town. Future studies that deliver CompLife to cancer survivors who have completed treatment can be conducted in group format to capitalize on the power of social support and connection. Limiting our patients to only those with stage III disease and with BMI of 24.5 or higher also decreases the generalizability of the findings. It may turn out that making lifestyle changes does not alter the disease trajectory in women with stage III disease, yet it may for women with earlier stage disease or for those at normal weight. Future larger clinical trials can expand the inclusion criteria to include a more heterogeneous patient population.
Conclusions
Few clinical trials have examined the effects of comprehensive lifestyle change on cancer-related outcome, and they have tended to focus on the effects of 1 or 2 behaviors independently. This neglects the synergy between behaviors and the health benefits of combining healthy behaviors, both physically and mentally. This trial addresses barriers such as costs, time, and transportation through the use of remote counseling. The high rate of recruitment and adherence to the trial will enable a comprehensive evaluation of lifestyle factors and their associations with improved health outcomes, and ultimately disease-free survival, for women with stage III breast cancer. Whitlock et al 80 assert that effective interventions arising from primary care settings address the most important risk behaviors; provide resources to assist patients as they adopt behavior change; may involve receiving care from various adjunct health care team members; utilize complementary communication channels such as telephone or computer-aided counseling; and require multiple contacts over time. The current trial meets each of these criteria for successful and evidence-based intervention and sets the stage for key improvement in long-term patient outcomes. 80
Footnotes
Acknowledgements
We are immensely grateful to Dr David Servan-Schreiber who worked tirelessly to help raise the necessary funding to conduct the trial. Dr Servan-Schreiber was an adjunct professor at MD Anderson during the conception and design of the trial and worked alongside us prior to his death in July 2011. We thank our valuable community sponsorships including discounted gym memberships with the YMCA of Greater Houston and vouchers for athletic shoes from Luke’s Locker Inc. We also received material support from Kleen Kanteen and the Vitamix Corporation, and the gracious permission from the Environmental Working Group to reprint educational materials on produce. Meg Hirsberg of the Anticancer Lifestyle Program also shared her environmental health resources with us while designing the intervention. We are grateful to Zoe Finch Totten, Founder and CEO of the Full Yield Inc and its Vice President Dee Bagshaw, with whom the web platform for the program was developed. We thank our colleagues from the Department of Radiation Oncology for creating a culture of cooperative, patient-centered care. Expertise on dietary assessment methodology and data collection is provided by Dr Cynthia Thomson, University of Arizona. Dr Joseph Petrosino and Dr Nadim Ajami, Baylor College of Medicine’s Alkek Center for Metagenomics and Microbiome Research, provide expertise on microbiome research and are conducting the microbiome analyses. We are thankful to a dedicated research team of research coordinators and interns who helped put together the educational materials, coordinate the study, and/or work on this manuscript: Erika Clark, Christine Spencer, Lauren Rosenbluth, Cassie Peretore, Andrew Cusinamo, and Richard Wagner. The trial’s data collection is made possible in part by the Department of Pathology and the Patient-Reported Outcomes, Survey & Population Research (PROSPR) Shared Resource (PROSPR), and the Center for Energy Balance in Cancer Prevention and Survivorship, Duncan Family Institute, MD Anderson. The authors are grateful to the incredible hard work and determination of the interventionists: Courtney West who has taught the exercise sessions since the study commencement; dietitians including Ali Miller, who provided her guidance in the study planning phase, and Jennifer Gebhard, Joseph Gonzales, and Deema Simaan; and to our current dietitian Sue Thompson. Of greatest importance, we thank the women who choose to participate in this trial. They inspire us as we see them navigate our world with strength, courage, and fierce attention to detail in the pursuit of health and happiness through lifestyle change.
Author Contributions
Intervention development: KBE, CC, AC, LoC, LiC, RH, CH, RN, SM, PP, AmS.
Conception and design: BA, GB, KBE, CC, AC, LoC, LiC, RH, CH, YL, SM, RN, PP, GP, JR, YS, AmS, AnS, PY, SY.
Provision of study material or patients: BA, KBE, CC, AC, LoC, LiC, RH, CH, GP, PP.
Collection and assembly of data: BA, TA, LoC, LiC, RH, CH.
Data analysis and interpretation: TA, LoC, LiC, RH, AmS.
Manuscript writing: All authors.
Final approval of manuscript: All authors.
Financial support: LoC.
Administrative support: RH, AmS.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was financially supported in part by private donations from The Duncan Family Institute with support from Jan and Dan Duncan, The Bosarge Family Foundation, The Thornburg Foundation, Cindy and Rob Citrone, The Lester Family Foundation, the Todd Family Charitable Foundation, Meg and Gary Hirshberg, S3 Partners LLC, Liz and Robert Sloan, Mr Ricardo Mora, Ms Maliha Khan, the Andrew & Lillian A. Posey Foundation, Aurora Investment Management LLC, CF Global Trading LLC, Ms Lynda Arimond and her family through the Not Just Another Cancer Fundraiser, Mr Ben Latham, and the Rising Tide Foundation for Clinical Cancer Research. Hundreds of others contributed anywhere from $10 to $5000 in donations from an open-access philanthropy site that continues to educate the public about the research study today. Members of the Houston community also came together to raise funds, including through a concert hosted by Casa Argentina.
