Abstract
Women are exposed to indoor and outdoor artificial light at night (ALAN) in urban and rural environments. Excessive exposure to hazardous ALAN containing short wavelength light may suppress pineal melatonin production and lead to an increased breast cancer (BC) risk. Our objective was to address the differences in BC risks related to light exposure in urban and rural communities. We examined indoor and outdoor light habits of BC patients and controls that had lived in urban and rural areas in a 5-year period, 10 to 15 years before the time of the study. Individual data, night time sleeping habits and individual exposure to ALAN habits were collected using a questionnaire. A total of 252 women (110 BC patients and 142 controls) participated in this study. The sample was divided to subgroups according to dwelling area and disease status. Age matching was completed between all subgroups. Odds ratios (ORs) with 95% confidence intervals (CIs) were estimated for urban and rural women separately, using binary logistic regression. OR results of urban population (92 BC patients and 72 control) revealed that BC risk increases with daily use of cellphone (OR = 2.13, 95% CI = 1.01-4.49, P < .05) and residence near strong ALAN sources (OR = 1.51, 95% CI = 0.99-2.30, P < .06). Nevertheless, BC risk decreases if a woman was born in Israel (OR = 0.44, 95% CI = 0.21-0.93, P < .03), longer sleep duration (OR = 0.75, 95% CI = 0.53-1.05, P < .1), and reading with bed light illumination before retiring to sleep (OR = 0.77, 95% CI = 0.61-0.96, P < .02). Furthermore, in the rural population (18 BC patients and 66 control) BC risk increases with the number of years past since the last menstruation (OR = 1.12, 95% CI = 1.03-1.22, P < .01). However, BC risk decreases with longer sleep duration (OR = 0.53, 95% CI = 0.24-1.14, P < .1), reading with room light illumination before retiring to sleep (OR = 0.55, 95% CI = 0.29-1.06, P < .07), and sleeping with closed shutters during the night (OR = 0.66, 95% CI = 0.41-1.04, P < .08). These data support the idea that indoor and outdoor nighttime light exposures differ between urban and rural women. Therefore, we suggest that women can influence BC risk and incidence by applying protective personal lighting habits. Further studies with larger sample sizes are needed to strengthen the results.
Introduction
Different studies suggest significant but moderate excess of breast cancer (BC) in urban compared with rural areas. 1 Likewise, humans are directly exposed to natural and artificial light at night (ALAN) in urban and rural areas, together with other environmental and individual factors pertaining to different communal environments in those areas of dwelling.2-4 Hence, this study does not address the different incidence rates of BC in urban versus rural communities; rather it explores the differences in BC risks related to light exposure in these 2 communities.
Since the time of the inventor Thomas Alva Edison (1879), humans have constantly increased their indoor and outdoor ALAN exposure using incandescent bulbs containing mainly low-level yellow wavelength to light emitting diode (LED) illumination that contains short blue wavelength light. 5 Exposure to ALAN containing mainly short wavelength (SW) light (~440-520 nm) influences body physiology via the suppression of the nocturnal production and secretion of the pineal neurohormone melatonin (MLT). Therefore, this exposes women to an increased risk for developing BC.6-11 Short wavelength (SW) lighting within the blue range (440-520 nm) is the dominant daytime wave length. Melanopsine, the photo pigment of the non–image forming photoreceptors is sensitive to it and therefore, exposure to SW lighting suppresses pineal melatonin production even at low intensities.12,13 Exposure to SW light is a common treatment for seasonal affective disorder (SAD). 14 The longer wave lighting of 560 nm is the dominant lighting at night (moonlight) and only at very high intensities will partly suppress MLT production. 15 Moreover, our previous results concerning indoor and outdoor light habits of BC cases and controls in a 5-year period, 10 to 15 years prior to disease diagnosis reinforce the nexus between exposure to ALAN and increased BC risk. 3 The study shows increased BC risk for women living near strong illumination sources. However, it reveals decreased BC risk for long sleepers, women who had been using reading lamps, and women who had slept with closed shutters. The associations revealed indicate that indoor and outdoor lighting habits may influence the overall population regardless of their age and dwelling area.
The mechanisms by which light can influence the physiology of humans, and therefore increase BC risk, have been described in many studies in recent years.6,16-18 The results of those studies and others led the World Health Organization in 2007 to announce “shift work that involves circadian disruption is probably carcinogenic to humans (2A).”19(p1065) This decision refers to occupational exposure but not to environmental exposure. Furthermore, in 2012 the American Medical Association concluded in their annual report that “More direct health effects of nighttime lighting may be attributable to disruption of the sleep-wake cycle and suppression of melatonin release. Even low intensity nighttime light has the capability of suppressing melatonin release.” 20
Urbanization is a worldwide process in developing and developed countries, and therefore its ecological and sociological consequences are challenging. Today, more than half of the world’s population lives in urban areas. Although urbanization is a force for better health, urban living is also associated with increased risk for chronic diseases, a more stressful social environment, and greater social disparities.21,22 The definition of urban and nonurban/rural areas varies widely among countries. Still, the focal characteristics to define an urban area are population size, density, heterogeneity, and distance from other centers. 23 In Israel, according to the Israeli Bureau of Statistics (IBS), urban and rural areas are defined using a “threshold” population size of 2000 residents with no reference to illumination infrastructure, social, or other mentioned characteristics. 24 However, rural settlements in Israel, compared with urban settlements, are characterized with a shared communal lifestyle to different extents. 25
Because of the different characteristics of the dwelling areas and lifestyle of BC cases and controls, we suggest testing the following hypothesis: If exposure to ALAN illumination is associated with higher BC risk, then women who had lived in urban areas and were affected by their lifestyle will therefore, ceteris paribus, exhibit different indoor and outdoor ALAN variables as BC risks, than women who had lived in rural areas, 10 to 15 years after exposure.
To test the suggested hypothesis our study examines the multilevel association between the settlement’s level of urbanicity, individual personal variables, sleep and light exposure variables together with BC risk. In order to suggest a model for the nexus between past lighting habits (during a 5-year period, 10-15 years prior to diagnosis) and BC risk, we compared lifestyle and light exposure habits between BC patients and controls in urban and rural areas separately, using a questionnaire.
The study was aimed to distinguish between different urban and rural past indoor and outdoor light exposure habits of BC patients and controls representing the general Israeli-Jewish population, excluding shift-workers. As such, we predicted that exposure to indoor and outdoor ALAN illumination differs between urban and rural dwelling areas due to different infrastructure and lifestyle.
Materials and Methods
Study Participants and Questionnaire
Study participants were split into urban/rural according to the IBS definition with a threshold of 2000 residents in their place of dwelling. 24 Within and between the groups BC cases and controls were matched by their age. All participants were recruited between the years 2010 and 2014 and were Israeli-Jewish non–shift-workers, aged 36 to 79 years. The sample consists of subsamples representing urban and rural dwelling areas. The sample size of BC cases and controls representing the rural population is in a ratio of about 1:4 and the ratio in the urban population is about 1:1. In general, urban women were less willing to respond therefore, it was difficult to recruit a bigger sample of controls from the urban population. Urban communities in Israel are represented in towns and cities while in rural areas we find small community settlements, Moshav (noncooperative agricultural settlement) and Kibbutz (a cooperative settlement) that are characterized with a shared communal life. 25 BC patients and controls maintained their dwelling area during the reference period of the study. Cases were BC patients attending the Comprehensive Cancer Center in Soroka Medical Center (SMC), Beer-Sheva and the Baruch Padeh, Poria Medical Center (PMC) in Tiberius while healthy control subjects with no documented cancer were friends of BC patients and women enrolled through personal meetings in schools. Participants were asked by the medical staff or by the researcher to fill out a questionnaire attaining information on indoor and outdoor light exposure habits and lifestyle 10 to 15 years before diagnosis (BC cases) or the time of the research (controls).
The questionnaire used in this study to obtain information regarding indoor and outdoor light exposure habits and lifestyle was part of a previous research conducted by the authors and is detailed elsewhere. 3 Nevertheless, study BC participants and controls were asked to relate to their habits during a period of 5 years, 10 to 15 years before diagnosis or the time of the research, respectively. Main variables of interest included individual variables like years past since first birth as proxy for age at first birth (in order to avoid null values for women who never gave birth, we calculated the variable as: age minus age at first birth), birth rates (births plus full pregnancies), BC-familial history, menstruation (age at first menstruation and again, in order to avoid null values we calculated years past since the termination of menstruation as proxy for age at last menstruation, ie, age minus age at last menstruation), eating routine and the extent of a balanced diet, alcohol and coffee drinking habits together with sleeping habits and light exposure variables.
Light exposure variables included subjective light intensity in the bedroom during the night, sleeping with light at the intensity for reading, reading with room light illumination before retiring to sleep, reading with bed light illumination before retiring to sleep, sleeping with the TV on for most of the night, sleeping with light penetrating the room from outside, sleeping with dim light during the night, sleeping with closed shutters during the night, residing near strong ALAN sources, and using LW- illumination or SW- illumination as bedroom and/or bed light illumination. In order to allow more accurate decision regarding the type of illumination, pictures of various illuminations such as fluorescent, halogen, and incandescent bulbs were presented in the questionnaire.
Statistical Analysis
In order to discover ALAN hazardous exposures which occurred during a period of 5 years, 10 to 15 years prior to disease detection, we implemented 2-level binary logistic regression models. In the first model, we conducted a stepwise logistic regression including personal, genetics, reproductive factors, and lifestyle variables. In the second model, we used hierarchal logistic regression to incorporate significant variables that emerged out of the first model (first block) together with sleep variables (second block) and ALAN variables (third block). Binary logistic regression models were performed for urban and rural participants separately, in order to obtain a prediction model for BC risk (odds ratio [OR], 95% CIs) with exposures to ALAN together with confounders. The model’s R2 is presented using Cox and Snell and Nagelkerke measures. Chi-square (χ2) test, Student’s t test and 2-way analysis of variance (ANOVA) were used to determine significant differences and interactions between cases and controls within the groups. Data were analyzed using SPSS version 19. For all analyses, statistical significance was set at P < .05 (2-tailed) where .05 < P < .1 is presented as marginal significance (MS) and P < .05 as significant (S). Marginal results should be divided by 2 (2-tailed) and although less strong, all the same support the statistical test hence indicating a trend. Ethical standards of this study were approved by the SMC Helsinki Committee, the PMC Helsinki Committee, and by the Ethical Board, University of Haifa, Israel.
Results
Descriptive Characteristics
A total of 252 women participated in this study (2010-2014) with age range 36 to 79 years. The sample consists of 110 (43.7%) BC cases and 142 (56.3%) non-BC controls. While 168 (66.7%) women lived in urban areas only 84 (33.3%) women lived in rural areas. The group of urban women consists of 92 (54.8%) BC cases and 76 (45.2%) non-BC controls while the group of rural women consists of 18 (21.4%) BC cases and 66 (78.6%) non-BC controls. All groups were age matched, therefore there is no significant difference between them (Table 1).
Age Comparison Between the Groups of the Study.

Model Analysis
Separate Evaluation of BC Risks for Urban and Rural Women
Odds ratios with 95% CIs for BC risks 10 to 15 years after exposures were calculated for urban/rural participants separately and are presented in Table 2. Two-level logistic regression models were conducted, thus the final model (model 2) comprises personal variables that emerged from the first model together with sleep and ALAN exposure variables in the second model for urban and rural women separately. The individual variables: years past since first birth, birth rates, BC familial history, age at first menstruation, eating routine and the extent of a balanced diet, alcohol and coffee drinking habits were examined separately in the first step of both populations and did not significantly affect BC risk. Supplemental Table 6: Two-way ANOVA results of reproduction variables (disease vs residential area) exhibit marginal difference between urban and rural women regarding age at first menstruation (F(1) = 3.45 P < .07, urban 12.92 ± 1.40, rural 13.35 ± 1.30) but with no significant difference between BC cases and controls (F(1) = 0.004) and with no interaction between disease and residential area (F(1) = 0.50). Also, no difference was detected between urban/rural and BC cases/controls regarding the “number of births” (F(1) = 1.95, F(1) = 0.11) and “years since first birth” (F(1) = 1.51, F(1) = 0.41). Supplemental Table 7: chi-square test results of drinking habits (disease vs residential area) show significant difference in the urban population regarding drinking alcohols weekly (χ2(1) = 4.66 P < .05 BC cases: No 78 (84.8%), Yes 14 (15.2%); controls: No 54 (71.1%), Yes 22 (28.9%), but no difference in the rural population (χ2(1) = 2.36 BC cases: No 16 (88.9%), Yes 2 (11.1%); controls: No 47 (71.2%), Yes 19 (28.8%). Furthermore, we found a marginal difference between BC cases and controls in the urban population regarding drinking coffee daily (χ2(1) = 2.92, P < .09, BC cases: No 17 (18.5%), Yes 75 (81.5%); controls: No 7 (9.2%), Yes 69 (90.8%) but no difference in the rural population (χ2(1) = 0.00, BC cases: No 2 (11.1%), Yes 16 (88.9%); controls: No 7 (10.6%), Yes 59 (89.4%). Nevertheless, as was mentioned before, the multivariate analysis did not find these variables predictive of BC risk.
Odds Ratios (ORs) of Variables Affecting Breast Cancer Risk of Urban/Rural Women (Binary logistic regression) 10 to 15 Years Before Diagnosis. a

Results are based on a sample of urban population: total population n = 168 of which cases n = 92 and controls n = 76. Likewise, a sample of rural population: total population n = 84 of which cases n = 18 and controls n = 66.
.05 < P < .1 indicates marginal significance and P < .05 indicates significance.
Urban Women
Women who used to live in urban areas 10 to 15 years before diagnosis and were born in Israel are significantly at a lower BC risk (OR = 0.44, 95% CI = 0.21-0.93, P < .03) compared with those who were born out of Israel. Moreover, urban women who used to talk daily using their cellular phone are significantly at a higher BC risk (OR = 2.13, 95% CI = 1.01-4.49, p < .05) compared with women who did not use their cellular phone daily. Nevertheless, ORs of these 2 variables were significant for urban women but not for rural women. Reading with bed light illumination before retiring to sleep lowers BC risk for urban women (OR = 0.77, 95% CI = 0.61-0.96, P < .02) but not for rural women. However, residing near strong ALAN sources 10 to 15 years before diagnosis marginally increased BC risk (OR = 1.51, 95% CI = 0.99-2.30, P < .06) among urban women but not among rural women.
Rural Women
OR was also calculated for “years past since the termination of menstruation” (age minus age of last menstruation) and it significantly elevates BC risk only for rural women (OR = 1.12, 95% CI = 1.03-1.22, P < .01). However, reading with room light illumination before retiring to sleep, marginally lowers BC risk for rural women (OR = 0.55, 95% CI = 0.29-1.06, P < .07) but not for urban women. Likewise, sleeping with closed shutters during the night marginally lowers BC risk (OR = 0.66, 95% CI = 0.41-1.04, P < .08) for rural women but not for urban women.
There is no significant difference between urban and rural women regarding sleep duration and BC risk (F(1) = 0.41) (Table 5). Nevertheless, longer sleep duration in a period of 5 years 10 to 15 years prior to disease detection, will marginally lower BC risk for urban (OR = 0.75, 95% CI = 0.53-1.05, P < .1) and rural women (OR = 0.53, 95% CI = 0.24-1.14, P < .1); however, to different intensities (Table 2-4).
Hierarchical Binary Logistic Regression Analysis of Variables Affecting Breast Cancer Risk in Urban Women, 10 to 15 Years Before Diagnosis.a
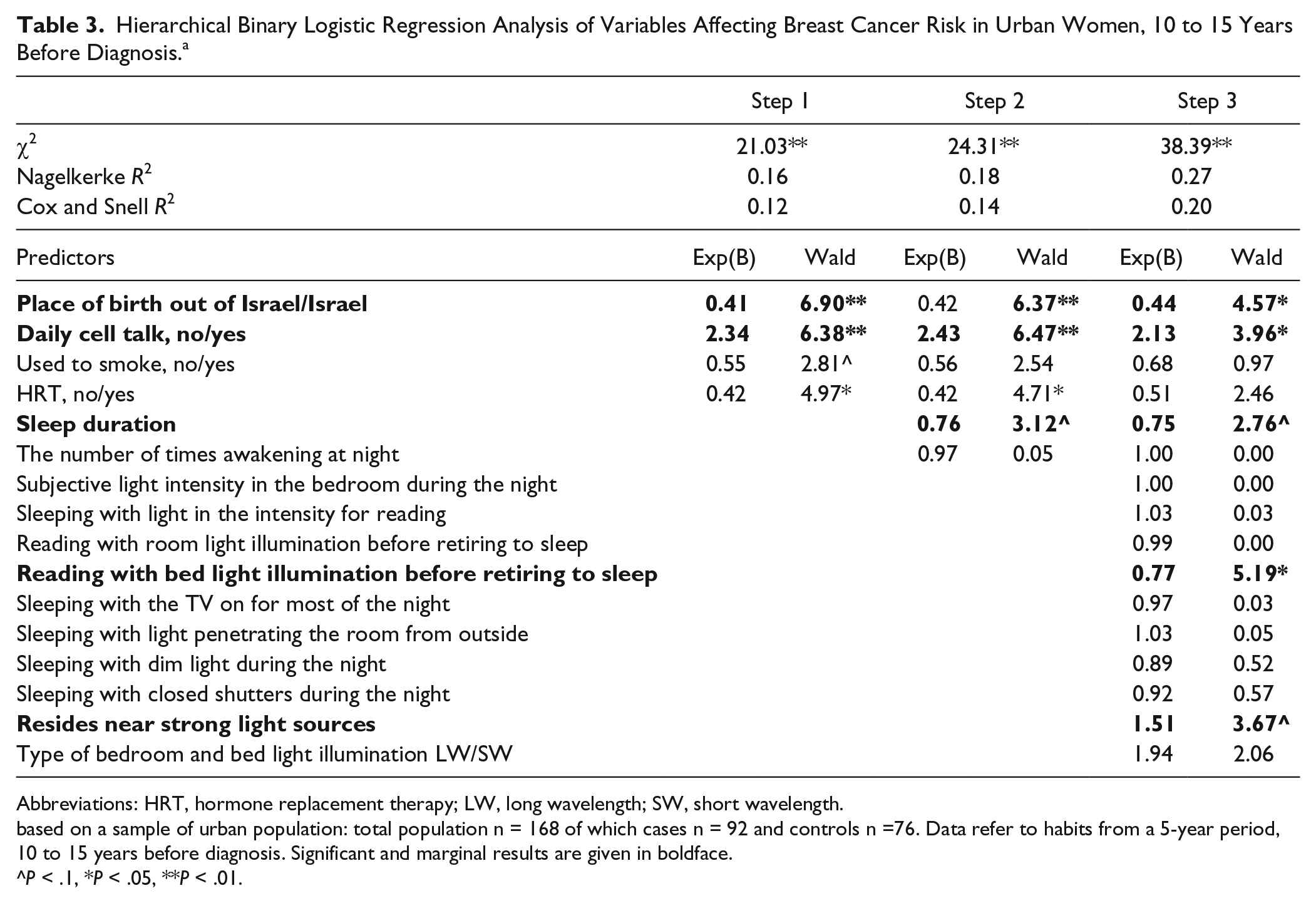
Abbreviations: HRT, hormone replacement therapy; LW, long wavelength; SW, short wavelength.
based on a sample of urban population: total population n = 168 of which cases n = 92 and controls n =76. Data refer to habits from a 5-year period, 10 to 15 years before diagnosis. Significant and marginal results are given in boldface.
P < .1, *P < .05, **P < .01.
Hierarchical Binary Logistic Regression Analysis of Variables Affecting Breast Cancer Risk in Rural Women, 10 to 15 Years Before Diagnosis. a

Abbreviations: LW, long wavelength; SW, short wavelength.
Based on a sample of rural population: total population n = 84 of which cases n = 18 and controls n = 66. Data refer to habits from a 5-year period, 10 to 15 years before diagnosis. Significant and marginal results are given in boldface.
P < .1, *P < .05, **P < .01.
The model summary in 3 steps (hierarchical logistic regression) is presented in Table 3 for urban women and in Table 4 for rural women.
R2 of Urban Women: BC Risk by Independent Individual, Sleep, and ALAN Predictors
Accumulative explained variance of all the variables in the model of urban women (Table 3, step 3) is 27% (χ2(16) = 38.39, P < .01 Nagelkerke R2 = 0.27, Cox and Snell R2 = 0.20). Step 1, personal characteristics, includes place of birth, daily cellphone talk, smoking and using hormone replacement therapy during menopause explains 16% of the variance (χ2(4) = 21.03, P < .01, Nagelkerke R2 = 0.16, Cox and Snell R2 = 0.12). Sleep habits, including sleep duration and the number of times awakening at night (step 2), explains only 2% of the variance. Total variance for step 2 is 18% (χ2(6) = 24.31, P < .01, Nagelkerke R2 = 0.18, Cox and Snell R2 = 0.14). Adding variables representing exposure to ALAN (step 3) contributes 9% more to the variance with total of 27%. Significant and marginal OR results were obtained in step 3 for the variables: place of birth, daily cell talk, sleep duration, reading with bed light illumination before retiring to sleep, and residing near strong ALAN sources.
R2 of Rural Women: BC Risk by Independent Individual, Sleep, and ALAN Predictors
Accumulative explained variance of all the variables in the model of rural women (Table 4, step 3) is 36% (χ2(13) = 22.17, P < .05, Nagelkerke R2 = 0.36, Cox and Snell R2 = 0.23). Step 1, personal characteristics, which includes “years past since the termination of menstruation” explains 17% of the variance (χ2(1) = 9.62, P < .01, Nagelkerke R2 = 0.17, Cox and Snell R2 = 0.11). Sleep habits, including sleep duration and the number of times awoken at night (step 2) explains 5% of the variance. Total variance for step 2 is 22% (χ2(3) = 12.76, P < .01, Nagelkerke R2 = 0.22, Cox and Snell R2 = 0.14). Adding variables representing exposure to ALAN (step 3) contributes 14% more to the variance with total of 36%. Significant and marginal OR results were obtained in step 3 for the following variables: years past since the termination of menstruation, sleep duration, reading with room light illumination before retiring to sleep and sleeping with closed shutters during the night.
Interaction Between Dwelling Area, Disease, and the Tested Variable
A 2-way ANOVA was conducted and significant results are presented in Table 5.
Two-Way Analysis of Variance With Interactions: Disease Versus Dwelling Area. a

Significant and marginal results are given in boldface.
P < .1, *P < .05, **P < .01.
Sleep Duration
There is no significant interaction between dwelling area, disease, and sleep duration (F(1) = 0.41) (Table 5). Nevertheless, the difference between BC cases and controls is significant (F(1) = 7.22, P < .01) as was reported in our previous study. 3 Implementing sleep duration separately into each population model results that longer sleep duration, in a period of 5 years 10 to 15 years prior disease detection, will marginally lower BC risk for urban (OR = 0.75, 95% CI = 0.53-1.05, P < .1) and rural women (OR = 0.53, 95% CI = 0.24-1.14, P < .1); however, to different intensities.
The Number of Years Past Since the Termination of Menstruation
Significant interactions were revealed between the number of years past since the termination of menstruation (after controlling for age), disease, and dwelling (F(1) = 6.48, P < .01). In the rural population for BC cases, the mean time that passed since the last menstruation was 13.78 ± 8.86 years and for the control group it was 7.51 ± 6.65 years (t(82) = 3.29, P < .01). There was no such difference in the urban population (t(166) = 0.07). In the total population, BC cases display higher mean time of 9.95 ± 8.22 years that passed since the last menstruation than the control group 8.37 ± 7.77 years (F(1) = 6.83, P < .01). There was no significant difference between urban and rural women with regard to this variable (F(1) = 1.49).
Reading With Room Light Illumination Before Retiring to Sleep
Urban and rural women present significantly different habits regarding reading with room light illumination before retiring to sleep (F(1) = 4.85, P < .05). While urban women (BC cases and controls) used to read sometimes (2.37 ± 1.47) with room light illumination before retiring to sleep, rural women more rarely used (2.09 ± 1.27) room light illumination for reading. The interaction reveals that BC cases of the rural population rarely (1.56 ± 1.15) read with room light illumination before retiring to sleep while controls sometimes (2.24 ± 1.27) read with room light illumination before retiring to sleep (t(82) = 2.06, P < .05). Still, there is no such difference between BC cases and controls in the urban population (t(166) = 0.18).
Discussion
Urban populations are growing worldwide together with the increase in BC incidence rates.26,27 The results of several studies explore BC risks in urban and rural populations around the world.28-32 The analysis of 168 urban and 84 rural Israeli women regarding habits in a 5-year period 10 to 15 years before diagnosis reveals different individual and ALAN variables which affect BC risk in those populations.
The following discussion will first refer to sleep duration, a variable that differs between BC cases and controls (as was demonstrated in our previous study 3 ). Next, we will discuss individual variables that affect the 2 tested populations separately. The variables “place of birth” and “daily use of cellphone” affect the urban population and the variable “years since the termination of menstruation” affects the examined rural population. Subsequent to this, we will discuss ALAN variables and their association to BC risk in both tested populations.
Sleep Duration
Both populations’ BC risk is affected by sleep duration, nevertheless with different intensities. Several studies have explored sleep duration in relation to BC risk but their findings are inconsistent.33-38 However, our result indicates that longer sleep in a 5-years period 10 to 15 years before diagnosis is associated with decreased BC risk for the controls in the total population (cases 7.49 ± 1.07, controls 8.12 ± 1.01; F(1) = 7.22, P < .01). There was no significant difference between urban women and rural women regarding sleep duration (Table 5). In this study, sleep duration is the only variable that affects BC risk among the 2 tested populations and with the same trend. Therefore, it may imply that sleep duration affects BC risk regardless of the dwelling area and its characteristics. Nevertheless, in the rural population we find greater protection with longer sleep duration. Rural populations constitute a unique “rural culture” including more commonly employed in agriculture and small business. 39 Rural areas in Israel are composed of “Kibbutz” and “Moshav” agricultural settlements with a typical lifestyle that is better aligned with the natural light cycles as well as a shared communal lifestyle. 40 Results from the literature regarding variances in BC risks comparing urban/rural areas describe different BC risks but, to the best of our knowledge, not differing ORs of sleep duration.28,29 Therefore, we suggest that a conserved lifestyle with less disturbed circadian rhythms may interplay with sleep duration to greatly reduce BC risk.
Cellphone Use
Our results regarding daily use of a cellphone in a 5-year period 10 to 15 years before diagnosis reveal that BC risk increased with daily cell talk for urban women but not for rural women. In 2011 the World Health Organization classified radiofrequency electromagnetic fields—the type emitted by mobile phones—as “possibly carcinogenic to humans” (Group 2B). 41 Nevertheless, in the same year, a panel of the International Commission on Non-Ionizing Radiation Protection had concluded that regarding brain tumor in adults, the evidence weighed against causation. 42 There is some evidence from laboratory research supporting the influence of electromagnetic radiation (EMR) from mobile phones on cell processes in breast cancer cells. However, there is not enough evidence on increased BC risk related to EMR exposure.43,44 Notwithstanding the previous, our results support an increased BC risk for urban women who used their cellphone daily in a period of 5 years 10 to 15 years prior to diagnosis. A mechanism that may be linked to prolonged EMR exposure is that EMR may suppress the production of melatonin.44-46 The fact that urban women are highly exposed to ALAN together with EMR presumably undergo a significant decrease in melatonin levels, which may affect their odds to nullify EMR hazardous exposures. On the other hand, we must consider professional or economic differences between women who used their cellphones daily versus those who did not. These data were not collected, therefore becoming a limitation in this study. Nevertheless, data obtained from the IBS regarding the prevalence of cellular phones in Israeli households by the type of locality in the years 2000-2004 (similar to the time of the study) show that in the year 2000 in the urban population, 62.5% of the households had at least 1 cellular phone and 25.9% had 2 cellphones or more. Also, in the rural population 79.4% of the households had at least 1 cellular phone and 41.9% had 2 cellphones or more. In 2004, in the urban population, 83.4% had at least 1 cellular phone and 52.2% had 2 cellphones or more while in the rural population 89.5% had at least 1 cellular phone and 66.4% had 2 cellphones or more. 47 These data show that the Israeli population holds at least 1 cellular phone in most households and that it is in common use in any dwelling area. However, contrasting old technologies, today’s technologies provide us with cellular phones that emit not only EMR but also hazardous SW illumination. Therefore, we may possibly expect it to further affect BC risk in the years to come.
Immigration
Israel provides an ideal context to explore differences between immigrant women and local residents because it contains many Jewish immigrants from different origins but with reduced genetic diversity. 48 Our study presents a decreased BC risk for nonimmigrant Jewish women in urban areas of Israel, but no effect in rural areas. Several studies in other countries concluded that lifestyle and dwelling had an impact on BC risk for immigrant women and their daughters.49,50 Despite their cultural diversity, immigrants may experience similar health-related problems. These problems are designed by the nature of the migration experience as well as the immigrants’ marginal status in the host countries, particularly for women. These experiences may result in higher levels of stress, partial access to health services together with cultural boundaries, and lower health perceptions.51-53 The combination of migration together with the urban environment and individual lifestyle may produce an elevated health risk, which in turn may also influence BC risk,21,49 as was revealed in our present study.
Menopausal Status
A wide study including 118 964 women with BC from 117 epidemiological studies conducted by a Collaborative Group on Hormonal Factors in Breast Cancer demonstrated a greater BC risk among premenopausal than postmenopausal women of the same age. 54 The interaction we revealed in our study demonstrates an elevated BC risk regarding “years since the termination of menstruation” for rural women only, therefore differs from these results. Nevertheless, our results did not refer to premenopausal and post-menopausal women in a specific age group rather than to the menopause stage (controlled by age) of the participants in urban and rural areas. Enquiries of the data show similar age range and median in the urban versus rural population (urban—age range 36-79 years median 59 years; rural—age range 47-74 years median 58 years) but with no mean age difference (Table 1). These differences may affect our results, thus becoming a limitation. Nevertheless, different studies point a decrease in MLT levels along the years that affects BC risk.55,56 These data were not collected in this study. Still, we may possibly consider that the decrease in MLT will influence BC risk in any dwelling area. The rural dwelling area provides its inhabitants a unique environment and lifestyle; therefore, in this case we suggest to further examine the unique characteristics and trends of this group.
Exposure to ALAN
Different ALAN variables affect BC risk in urban and rural women. These associations were still present after controlling for age and other potential confounders that were measured.
Exposure to Outdoor ALAN
In the urban population, our results revealed that residence near strong ALAN sources (exposure to outdoor ALAN) is associated with increased BC risk. Our result is consistent with those of others3,57-59 claiming that women residing in regions with high ALAN levels may experience an elevated BC risk. However, our results are marginally significant (2-tailed) only for the urban population but not for the rural population. A study conducted by Kim et al 29 in South Korea that was published in 2015 addressed the nexus between ALAN, BC risk and dwelling area. The authors concluded that ALAN is the most significant BC risk factor for women from both urban and rural areas. Therefore, regarding the urban population, it supports our results. 29 However, there are major differences in the structure of the 2 studies as well as in the definition of urban and rural areas. An urban area was defined by the authors using a “threshold” population size of 50 000 residents with no reference to illumination infrastructure or social characteristics. Therefore, as to population size only, urban areas in Israel may have been categorized as rural in the above study. However, our results of increased BC risk in urban areas reflect the fact that intense ALAN sources are common in urban areas in Israel. Most of past and present lighting used outdoors, such as metal halide, mercury vapor, fluorescent lamps, and LEDs, contain SW light. SW light can inhibit MLT production and secretion even at low intensities and as a result influencing human physiology and increasing BC risk.12,59-61 Integrating the results of the 2 studies may be a way to reassure that the observed increased OR/Relative Risk (RR) does not reflect the higher BC rates in urban areas alone rather than the excessive exposure to ALAN, lifestyle, and infrastructure.62,63
In the rural population, the influence of exposure to outdoor ALAN is reflected with a decreased BC risk due to preventing any outdoor lighting by sleeping with closed shutters. These data are supported by previous significant results and trends,3,59,64 which emphasize the importance of outdoor ALAN and elevated BC risk. This distinct habit in the rural dwelling may be due to security issues where homes are on ground level and need to be secured rather than due to avoiding light, still as was found in our research, this reduces BC risk.
Exposure to Indoor ALAN
Indoor exposure to ALAN may affect BC risk as well. Our results of urban women show a significant decrease in BC risk by reading with bed light illumination before retiring to sleep. Outdoor urbanized areas are becoming rapidly exposed to excessive ALAN. We therefore suggest that there is a clear contrast between outdoor and indoor lighting, and exposure to both may create an accumulative effect with hazardous consequences. However, when using low-intensity indirect illumination emerging from bed light at night time, mainly in urban areas, may help in keeping the accumulative exposure to hazardous light under a certain threshold. This moderate accumulative signal possibly allows the body to stay aligned with natural light and dark cycles despite intense outdoor or/and indoor light. As a result, the body continues to produce and secrete MLT habitually. Deviation from habitual MLT levels can potentially influence body physiology and health, including BC.7,19,65 The results of this study refer to a time period when LW illuminations (incandescent/halogen) with a dominant wavelength greater than ~550 nm were more prevalent in use (BC patients 79.3% and controls 82.7%); therefore, as was demonstrated by Cajochen et al, 10 these moderately affect MLT production and secretion.
In the rural population, our results revealed that BC risk decreases if a woman was reading with room light illumination before retiring to sleep and sleeping with closed shutters during the night. The gap in lighting infrastructure between urban and rural areas in Israel is still large. Many rural settlements had old illumination infrastructures. Therefore, these rural settlements experienced darkness at night (unpublished data). For that reason, we suggest that indoor lighting used by the participants 10 to 15 year before the time of the study may have influenced MLT production and secretion and therefore BC risk.7,19,65 Also, our findings show that most of rural participants used LW illumination as room light (BC patients 83.3% and controls 84.8%). When rural BC cases and controls were compared, we see that when using room light illumination, the controls have an apparent decrease in BC risk. Since it is impossible to measure timing, intensities, and duration of room light in retrospect as was set by Gooley et al, 65 we can only refer to the outcome. Using room light illumination by the controls as part of their probable conservative lifestyle may have kept the accumulative light exposure under the dangerous threshold and thus reduced their BC risk. As for the BC cases, we lack information about their personal light exposure habits prior to entering their sleeping habitat. Therefore, there is a limitation in the scope of this research as we cannot point to any incriminating timing, intensities, or duration of light habits that may have increased their BC risk.
When looking at the urban and rural populations, we see that in urban areas, the variable that stands out is the using of bed light illumination; while in rural areas, it is the using of room light illumination that is the prominent variable. Nevertheless, in both cases, supported by the knowledge that LW light moderately suppresses MLT production and secretion compared with SW light,10,15,66 and the fact that most of the participants used LW light as room light or bed light we suggest that light exposure threshold was not crossed by the individual’s use of indoor lighting.
Study Limitations
The following are some limitations of the study:
The small number of the subsamples was a major limitation in the study; therefore, it is possible that we might have missed significant association in each dwelling area due to low statistical power. However, marginal and significant results that emerged from the study support the negative influence of ALAN on women’s health regarding BC.
Dwelling area was set according to the IBS definition with a threshold of 2000 residents. Therefore, it is possible that the results could have been altered with a different cut point.
Individual data regarding timing, wavelength, intensity, and duration of ALAN in a 5-year period 10 to 15 years ago were not collected. As a result, these data of the individuals are missing and should be collected in a future study.
Individual data regarding light exposure habits out of the sleeping habitat were not collected.
The data were collected as part of another study, 3 and limitations of the data collection method are described there.
As in all studies, residual confounding cannot be completely excluded.
Conclusion
The variables that were tested in this study regarding a 5-year period 10 to 15 years before diagnosis of BC explain 27% of the variance in the urban population but 36% of the variance in the rural population. These results may be explained by the multivariable environment present in urban areas compared with those of rural areas. Furthermore, variables representing exposure to ALAN (step 3 of the model) contributes 9% to the total variance in the population of urban women and 14% to the total variance in the population of rural women. Although there is a difference in the explained variance of ALAN variables between the 2 populations, it may support the hypothesis that exposure to ALAN illumination is associated with higher BC risk in the total population. 29 Furthermore, we suggest referring to the rural population as an internal control with less affecting variables (documented or undocumented) comparing with the urban tested group.
Assessing the results regarding indoor and outdoor ALAN variables in the model, we may indicate differences in the setting of illumination together with the type of illumination. Exposure of women to indoor illumination in urban and rural environments has a significant protective effect while exposure to outdoor illumination increases BC risk significantly. These differences may point to a threshold of light exposure. Using indoor light helps maintain light levels below the threshold and therefore has a protective effect whereas exposure to outdoor illumination together with indoor illumination is probably above the threshold and as a result increases BC risk. We must bear in mind that this study refers to a time when LW incandescent and halogen illuminations were widely used rather than SW fluorescent and LED as today.
In this study, ALAN variables were prominent over other potential confounders in urban and rural environments. Even with the limitations associated with this, the fact that the association between exposure to ALAN during the scotophase in a 5-year period, 10 to 15 years ago, and that an elevated BC risk remains, even after controlling for other silent variables, suggests that our nighttime lighting habits in the past may be a significant BC risk factor in any dwelling environment.
Footnotes
Acknowledgements
We thank Dr Michael Koretz of the Elisheva Kaplan Eshkol Comprehensive Breast Health Center of Soroka University Medical Center, Beer-Sheva for allowing us to approach breast cancer patients and controls visiting the clinic and thanks to Dr David Gefen and Dr Rita Toker of the Oncology Day-Care Clinic of the Soroka University Medical Center, Beer-Sheva, for helping in handing out the questionnaires to breast cancer patients.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
