Abstract
Hypothesis. Renin–angiotensin system (RAS) has been considered not only as a regulator of systemic volume and electrolyte balance but also has been recently involved in various pathological processes such as cancer. In the etiology of breast cancer, dietary factors have been analyzed and especially the influence of dietary fat has been studied, but the underlying mechanisms remain unclear. In this study, we analyzed RAS-regulating enzymes in serum of rats with N-methyl nitrosourea (NMU)-induced breast cancer fed with different diets. Study Design. Four groups of rats were injected intraperitoneally with 3 doses of 50 mg/kg body weight of NMU at different days after birth and were fed with an AIN-93 commercial diet or AIN-93 diets with 4% fat constituted respectively by extra virgin olive oil, refined sunflower oil, and refined sunflower oil enriched to 50% with oleic acid. Method. After sacrifice, blood and tumor samples were collected by spectrophotometric determinations of RAS-regulating enzymes in plasma and histopathology studies. Results. We show that the type of dietary fat does not influence latency period, incidence of animals with tumors, incidence of mortality, or tumor yield per rat. However, changes were observed in tumor volume and the histopathology. The type of dietary fat also differently modified the enzymes involved in RAS regulation. Conclusions. It might suggest that one of the mechanisms by which dietary fat affects breast cancer is the modification of the RAS system, which may be consider as a new target for integrative therapies.
Introduction
The etiology of human breast cancer is largely unknown. Different factors such as genetic susceptibility, hormonal effects, or environmental conditions have been considered as major determinants. In this way, dietary factors have been considered as one of the most important environmental determinants; in fact, it is generally thought that approximately one third of cancers are related to dietary factors. 1
Although many epidemiological and experimental studies have evaluated the influence of different dietary components on breast cancer, the roles of specific dietary factors are not completely understood, 2 but dietary fat has been directly related with breast cancer incidence. Human data regarding the association between dietary lipids and breast cancer are controversial, 3 but experimental rodent studies have reinforced the notion of the dietary fat–breast cancer link. In fact, these studies have demonstrated an increased mammary tumor incidence associated with high-fat diets. However, the dietary lipids effects depend not only on the quantity but also on the type of fat. 3 In general, n − 6 polyunsaturated fatty acids (PUFA) have strong tumor-enhancing effects and also saturated fatty acids are stimulator. On the contrary, n − 3 PUFA have inhibitory effects on mammary tumor growth and metastasis, but special attention has been focused on olive oil and oleic acid. Although there is growing experimental evidence regarding a putative protective effect on breast cancer, there are conflicting results too. 3 The mechanisms by which dietary fat is involved in breast cancer have also been studied, and thus some of these proposed mechanisms include the influence on hormonal status and on the differentiation of the mammary gland, the modification of cell membrane or gene expression and signaling pathways, as well as modifications in oxidative stress and changes in immune system function. 3
However, many evidences have suggested a role of renin–angiotensin system (RAS) in cancer. In fact, in addition to the production of plasma Ang II, a local RAS has been shown to be functional in various organs, leading to production of Ang II that might have a paracrine or autocrine function. 4 The circulating RAS is considered as a regulator of systemic volume, electrolyte balance, and blood pressure homeostasis, while local RAS systems have local tissue effects involving proliferation, growth, protein synthesis, and organ function. 5 Also, local RAS can participate in various pathological processes such as cancer.6,7 It is known that Ang II regulates cell turnover by promoting both cell proliferation and programmed cell death through differential actions via the AT1 and AT2 receptors, 8 and also it is known that AT1 and AT2 receptors are expressed in breast cancer. However, Ang II is not the only element of RAS that participates in cancer. The possibility that angiotensin III may have a key role has also been suggested in studies with N-methyl nitrosourea (NMU)-induced breast tumors. 9
In the same way, our research group has studied the influence of dietary fats on the regulatory enzymes of the RAS. 10 Accordingly with these previous data, the aim of present work was to analyze the enzymes involved in RAS regulation in the serum of rats with NMU-induced mammary tumors and to evaluate the putative role of dietary fats in this regulation in order to elucidate the mechanisms by which dietary fat may affect breast cancer.
Materials and Methods
Animals and Treatment
Seventy-three female virgin Wistar rats (149.4 ± 2.7 g body weight) were used in this work. The animals were provided from the animal house care of the University of Jaén and maintained in an environment controlled under constant temperature (25°C) with a 12 hour light/12 hour dark cycle. All animals were allowed access to water and food ad libitum. The experimental procedures for use of animals and care were in accordance with the European Community Council directive (86/609/EEC). The rats were randomly divided into 5 groups. Four groups of 16 animals were injected intraperitoneally with 3 doses of 50 mg/kg body weight of NMU at 50, 80, and 110 days after birth, as previously described. 11 Tumors induced by this method are estrogen-dependent. 12 All rats were at estrus at the first NMU injection, verified by daily vaginal smears. Three of these groups were fed with AIN-93 semipurified diets containing 4% of fat, constituted respectively by extra virgin olive oil (EVOO), refined sunflower oil (SO), and refined sunflower oil enriched to 50% oleic acid (OAESO). The fourth group received a commercial AIN-93 diet with 4% fat. Finally, the fifth group was considered as control and was injected intraperitoneally with the vehicle only and was fed with the commercial diet.
For tumor detection and growth control, rats were examined by palpation 2 days each week after the second NMU injection. The number of tumors was recorded, and the major and minor diameters of each tumor were measured with a caliper to determine the tumor volume. The mean tumor number per rat (n/t), defined as the number of tumors per rat in animals developing at least one tumor were also determined. After 122 days of the first NMU injection, animals were sacrificed under equithesin anesthesia (2 mL/kg body weight). Blood samples were obtained through the left cardiac ventricle and centrifuged 10 minutes at 3000g to obtain the serum. These samples were frozen and stored at −80°C, until use.
Tumor samples were also obtained, fixed (formaldehyde 4%), dehydrated, and embedded in paraffin for histopathology studies. To characterize the tumor malignant phenotype, the degree of tumor morphological aggressiveness and the tumor growth were determined. We applied the Nottingham modification of the Scarff–Bloom–Richardson grading method, 13 slightly adapted to rat mammary carcinomas as described by Solanas et al. 14 This global histopathology grade adds together the pattern differentiation (acinus formation), the nuclear atypia/pleomorphism, and mitosis count. The global histopathology grade in each carcinoma was determined as follows: 3 to 5 points corresponded to a grade I or well-differentiated adenocarcinomas, 6 to 7 points to a grade II or moderately differentiated adenocarcinomas, and 8 to 9 points to a grade III or poorly differentiated adenocarcinomas. Tumor mitotic activity was distributed in 3 categories, according to the number of mitoses reported in 10 high-power fields, at 400× magnification: (a) ≤4 mitosis, (b) 5 to 9 mitosis, and (c) ≥20 mitosis. Tumor volume was defined as 1/2a(b), 2 where a is the long diameter and b is the short diameter.
Renin–Angiotensin System–Regulating Aminopeptidase Activities Assay
Aspartyl Aminopeptidase (ASAP) Activity Assay
ASAP was measured fluorometrically using aspartyl-β-naphthylamide (AspNNap) as the substrate. Briefly, 10 µL of each sample was incubated in triplicate for 30 minutes at 37°C with 100 µL of the substrate solution: 100 µM AspNNap, 1.3 µM ethylenediaminetetraacetic acid (EDTA), and 2 mM MnCl2 in 50 mM of phosphate buffer, pH 7.4. All the reactions were stopped by adding 100 µL of 0.1 M acetate buffer, pH 4.2.
Aminopeptidase A (APA) Activity Assay
APA activity was measured in the same way using glutamyl-β-naphthylamide (GluNNap) as the substrate. Ten microliters of each sample was incubated in triplicate for 30 minutes at 37°C with 100 µL of the substrate solution: 100 µM GluNNap, 0.65 mM dithiothreitol (DTT), and 50 mM CaCl2 in 50 mM of phosphate buffer, pH 7.4. All the reactions were stopped by adding 100 µL of 0.1 M acetate buffer, pH 4.2.
Aminopeptidase N (APN) Activity Assay
APN was measured fluorometrically using alanyl-β-naphthylamide (AlaNNap) as substrate. Ten microliters of each sample were incubated by triplicate for 30 minutes at 37°C with 100 µL of the substrate solution containing 100 µM of AlaNNap and 0.65 mM DTT in 50 mM phosphate buffer, pH 7.4.
Aminopeptidase B (APB) Activity Assay
APB was measured fluorometrically using arginyl-β-naphthylamide (ArgNNap) as substrate. Ten microliters of each sample were incubated by triplicate for 30 minutes at 37°C with 100 µL of the substrate solution containing 100 µM of ArgNNap and 0.65 mM DTT in 50 mM phosphate buffer, pH 7.4.
Insulin-Regulated Aminopeptidase (IRAP) Activity Assay
IRAP was measured fluorometrically using leucyl-β-naphthylamide (LeuNNap) as substrate. Ten microliters of each sample were incubated by triplicate for 30 minutes at 37°C with 100 µL of the substrate solution containing 100 µM of LeuNNap and 0.65 mM DTT in 50 mM phosphate buffer, pH 7.4.
All the reactions were stopped by adding 100 mL of 0.1 M acetate buffer, pH 4.2. The amount of β-naphthylamine released as the result of the enzymatic activities was measured fluorometrically at 412 nm emission wavelength and 345 nm excitation wavelength. Proteins were quantified also in triplicate by the method of Bradford, using bovine serum albumin (BSA) as standard. Specific enzyme activities were expressed as picomoles of the corresponding aminoacyl-β-naphthylamide hydrolyzed per minute per milligram of protein, by using a standard curve prepared with the latter compound under corresponding assay conditions.
Statistical Analysis
All values represent the mean of the individual determination ± standard error of the mean (SEM). Statistical analysis of the qualitative data was carried out using the χ2 test. Quantitative data were analyzed by multiple analysis of variance (MANOVA) plus Newman–Keul’s post hoc test, using IBM Pass V.19 software. Values of P < .05 were considered significant.
Results
No significant differences were found between groups either in the latency period, the incidence of animals with tumors, and the incidence of mortality in animals with tumors or the tumor yield per rat. On the contrary, the tumor volume showed a significant increase (P < .01) in NMU-treated rats fed on OAESO in relation with all the other groups (Figure 1), which did not show significant differences between them.
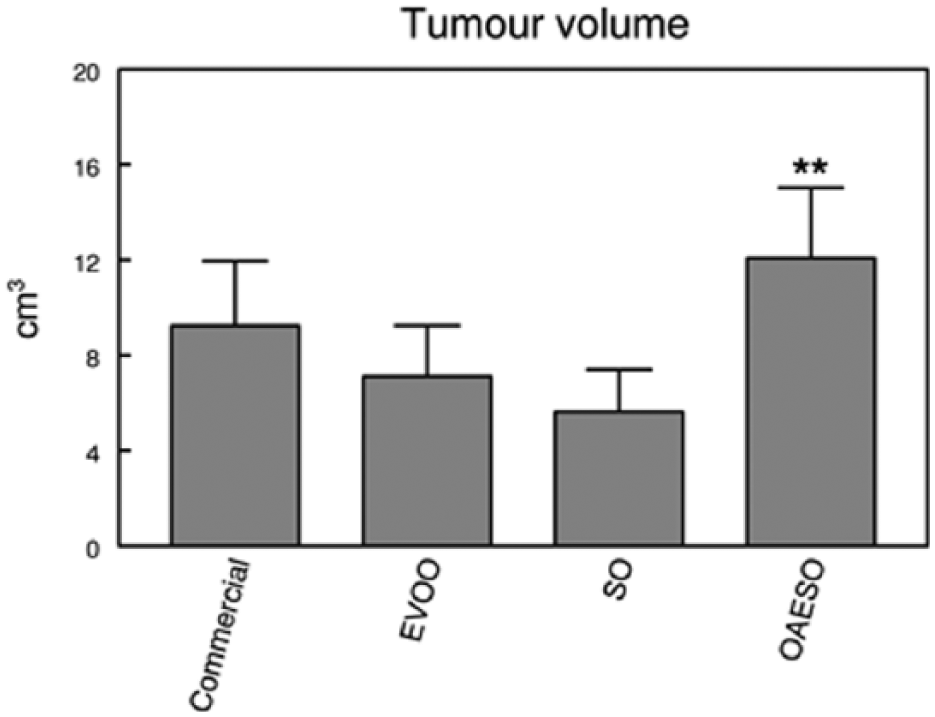
Tumor volume in animals with breast cancer induced by N-methyl-nitrosourea (NMU) fed with an AIN-93 commercial diet, or with AIN-93 diets with 4% of fat constituted, respectively, by extra virgin olive oil (EVOO), refined sunflower oil (SO), and refined sunflower oil enriched to 50% oleic acid (OAESO). Results are expressed in cubic centimeters (mean ± SEM; n = 7-13; **P < .01).
Histological analysis (Figure 2) showed a significant decrease in the percentage of tubules in tumors of animals fed with SO and OAESO when compared with animals fed with commercial diet and those fed EVOO. A significant decrease (P < .05) in the percentage of mitosis was also observed in the tumors of animals fed with EVOO and OAESO (Table 1).
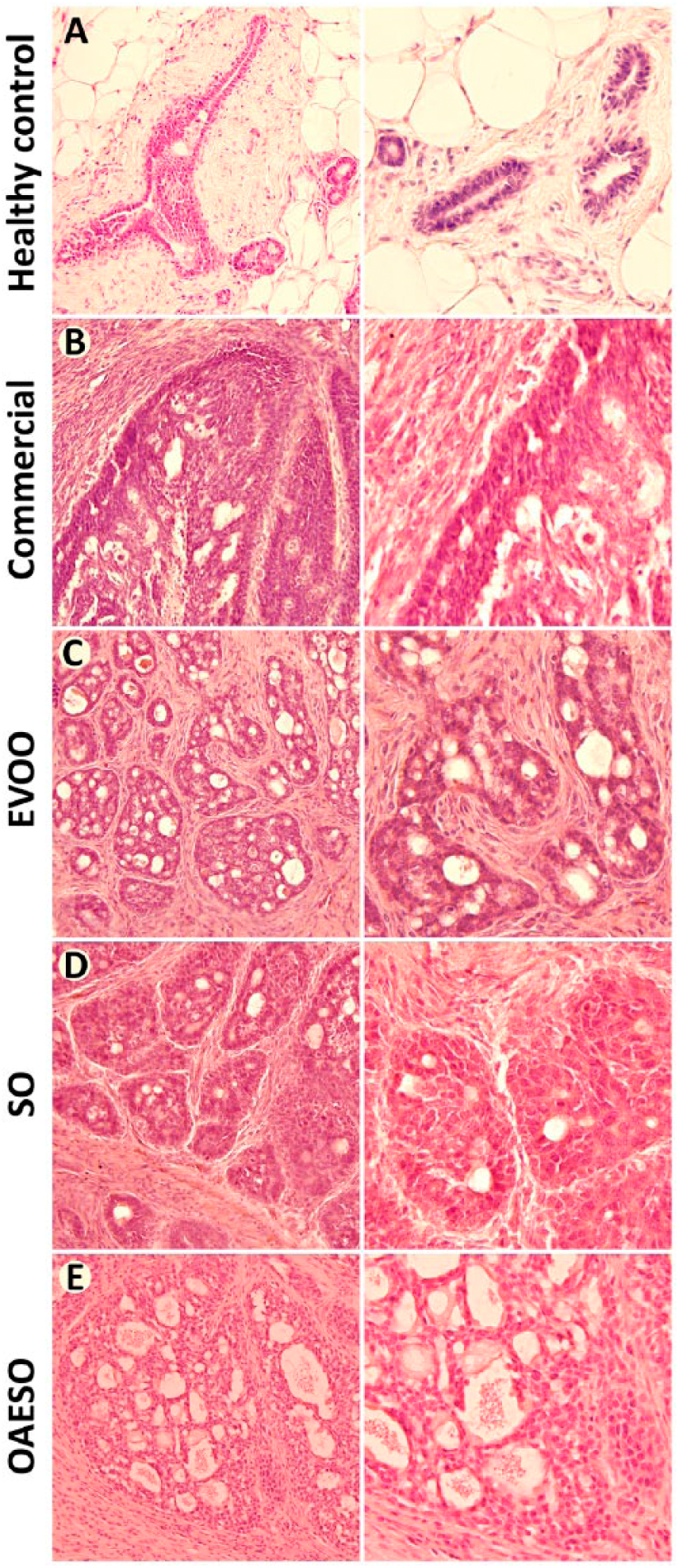
Histopathological characteristics of mammary tissue from healthy control animals (A) and from animals with breast cancer induced by N-methyl-nitrosourea (NMU) fed with an AIN-93 commercial diet (B), or with AIN-93 diets with 4% of fat constituted, respectively, by extra virgin olive oil (EVOO) (C), refined sunflower oil (SO) (D), and refined sunflower oil enriched to 50% oleic acid (OAESO) (E). Left lane, 200× magnification; right lane, 400× magnification.
Histopathological Results of the Morphological Analysis of Rat NMU-Induced Mammary Tumors From the Different Dietary Groups, Displayed as the Percentage of Tumors From Each Dietary Group Exhibiting the Different Categories of the Histopathological Feature Analyzed.
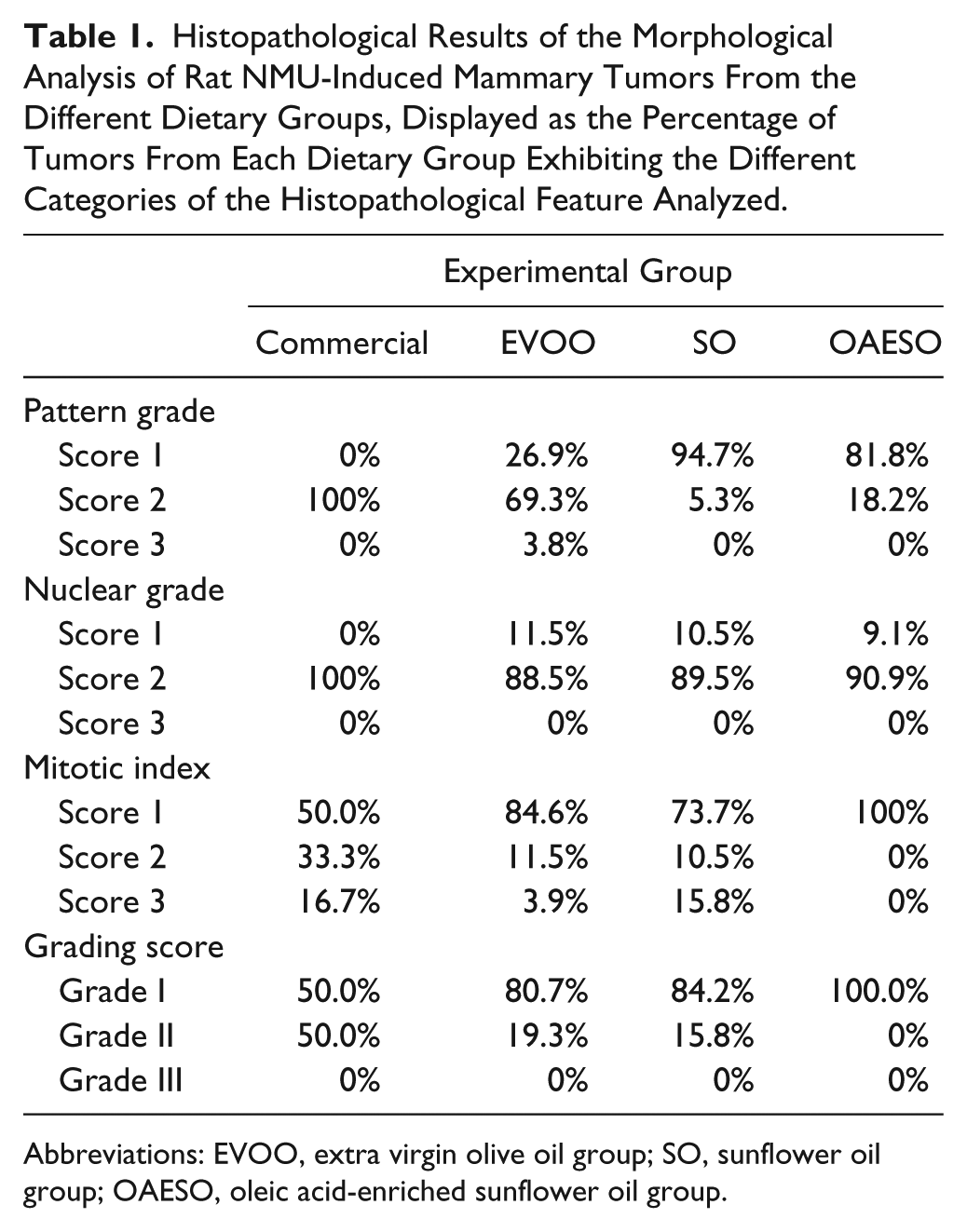
Abbreviations: EVOO, extra virgin olive oil group; SO, sunflower oil group; OAESO, oleic acid-enriched sunflower oil group.
The study of serum RAS regulatory enzymes has showed a significant increase (P < .05) in specific ASAP activity in EVOO and SO groups when compared with control healthy animals (Figure 3A), whereas no differences were observed between the other groups. In the same way, APA specific activity increased significantly in all animals with breast cancer when compared with the control healthy animals. However, the highest signification was found in EVOO and SO groups (P < .01), and in a lesser degree, in commercial and OAESO groups (P < .05; Figure 3B).
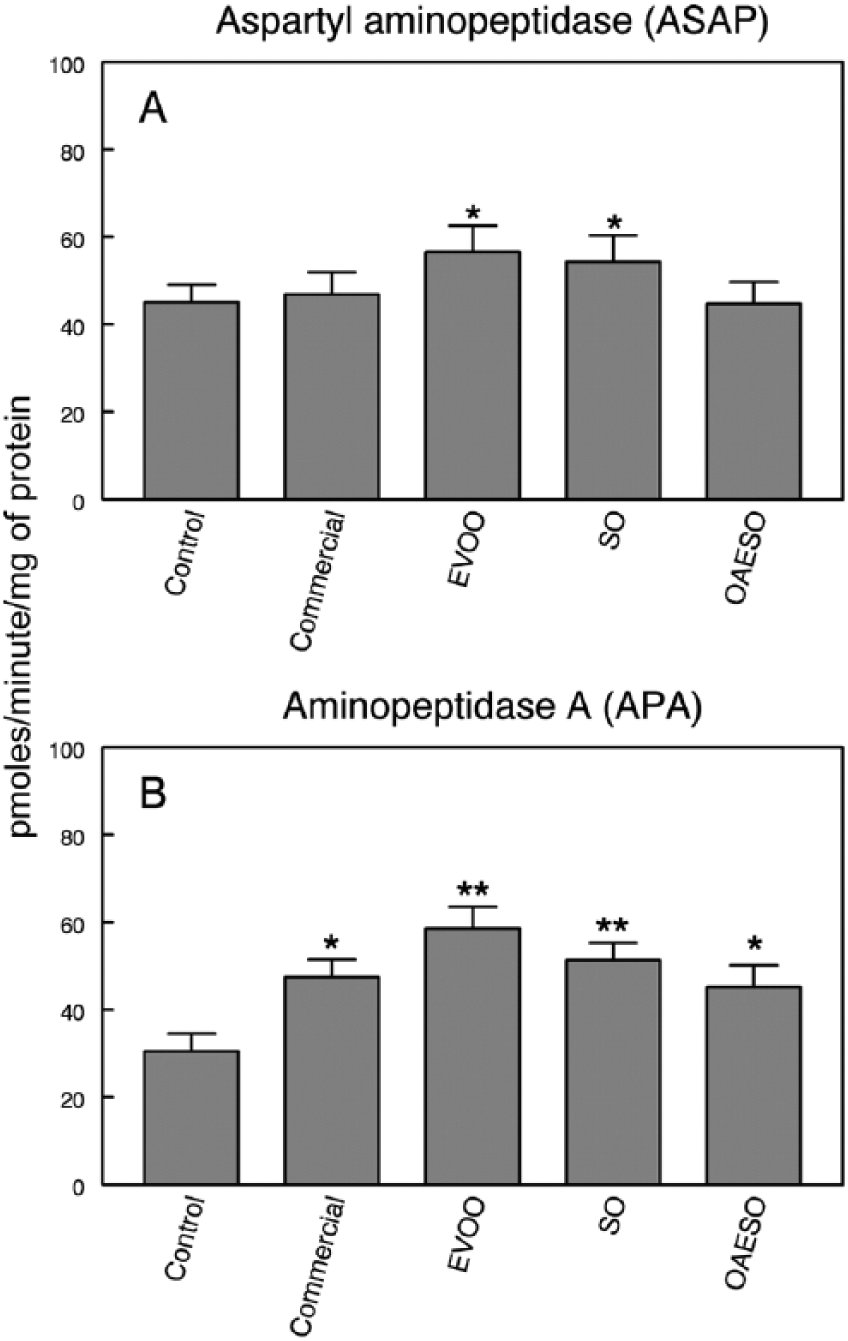
Aspartyl aminopeptidase (ASAP) (A) and aminopeptidase A (APA) (B) activities in serum of nontumor healthy control animals and animals with breast cancer induced by N-methyl-nitrosourea (NMU) fed with an AIN-93 commercial diet, or with AIN-93 diets with 4% of fat constituted, respectively, by extra virgin olive oil (EVOO), refined sunflower oil (SO), and refined sunflower oil enriched to 50% oleic acid (OAESO). Results are expressed in picomoles of aspartyl- or glutamyl-β-naphthylamide hydrolyzed per minute and per milligram of protein (mean ± SEM; n = 7-13; *P < .05; **P < .01).
Specific APN activity showed a significant increase (P < .01) exclusively in the SO group, and a significant decrease (P < .05) in the OAESO group when compared with control healthy animals. No changes were detected in animals fed a commercial or EVOO diet (Figure 4A).
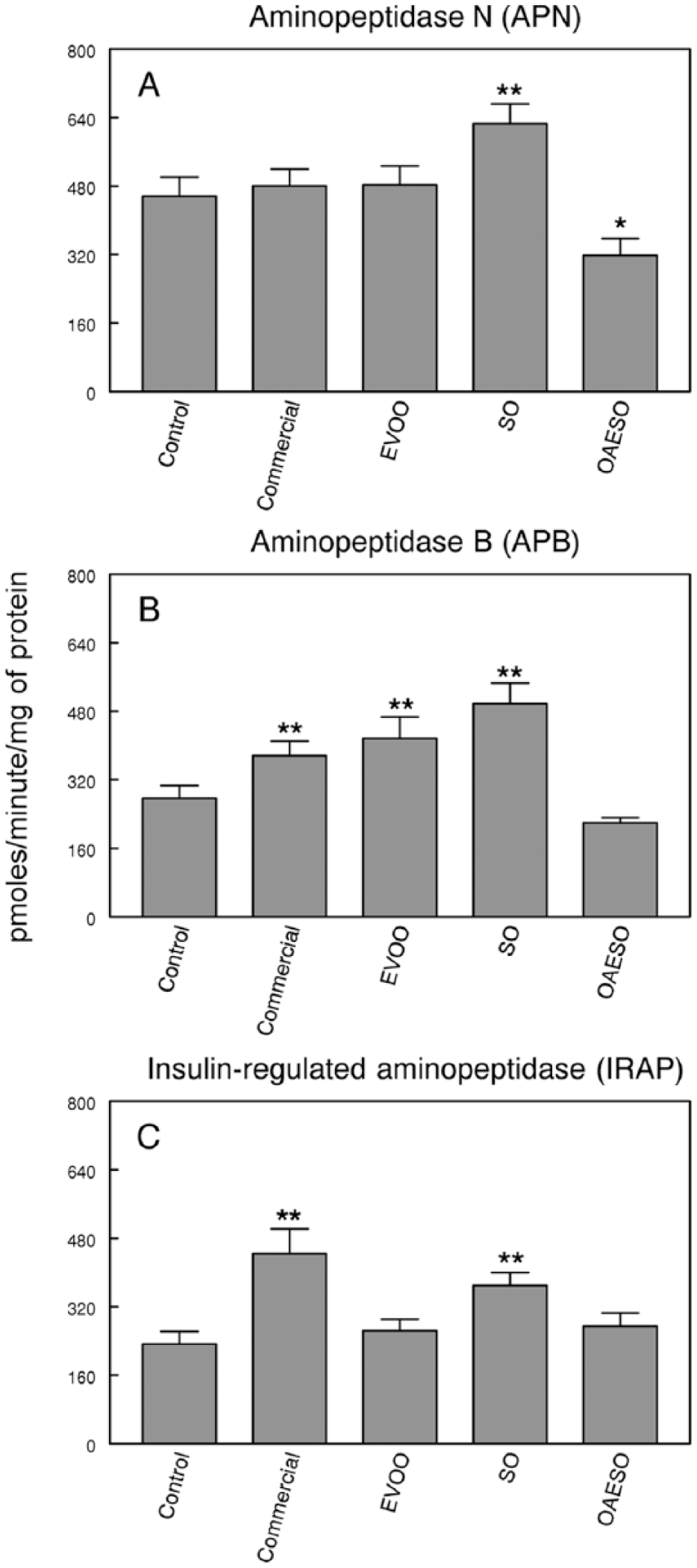
Aminopeptidase N (APN) (A), aminopeptidase B (APB) (B), and insulin-regulated aminopeptidase (IRAP) (C) activities in serum of nontumor healthy control animals and animals with breast cancer induced by N-methyl-nitrosourea (NMU) fed with an AIN-93 commercial diet, or with AIN-93 diets with 4% of fat constituted, respectively, by extra virgin olive oil (EVOO), refined sunflower oil (SO), and refined sunflower oil enriched to 50% oleic acid (OAESO). Results are expressed in picomoles of arginyl-β-naphthylamide hydrolyzed per minute and per milligram of protein (mean ± SEM; n = 7-13; **P < .01).
On the contrary, a significant increase (P < .01) in specific APB activity was observed in commercial, EVOO, and SO dietary groups of animals with breast cancer when compared with control healthy animals. No change in APB activity was found in animals fed OAESO diet (Figure 4B).
Finally, the study of IRAP activity showed a significant increase (P < .01) in rats with breast cancer fed with commercial diet and SO when compared with control healthy animals. No changes were found in EVOO or OAESO groups (Figure 4C).
Discussion
In this work, we analyzed the influence of the type of dietary fat on the NMU-induced rat mammary tumors. In our experimental design, all NMU-treated rats were fed ad libitum with an AIN-93 standard diet containing solely 4% fat (extra virgin olive oil, refined sunflower oil, or refined sunflower oil enriched with 50% oleic acid) or an AIN-93 commercial diet containing 4% undefined fat. The control group of healthy animals, which was only treated with the vehicle, was also fed a commercial diet. It has been shown that the timing of fat intake appears to be crucial in the development of breast cancer, 2 so it is important to note that dietary manipulation under our conditions begin early in the animal’s lives.
We have showed that the type of dietary fat does not seem to influence the parameters of the carcinogenesis, because no significant differences were found between groups either in the latency period, the incidence of animals with tumors, and the incidence of mortality in animals with tumors or the tumor yield per rat. However, important changes have been observed in tumor volume in the animals of the OAESO group. Similarly, the histological study also showed that the animals that consumed oleic acid in their diet (EVOO and OAESO) had a lower number of mitosis while animals fed with SO had a reduction in the number of tubules observed in tumor tissue. The tumor grade showed that animals fed a standard diet developed tumors of grade 1 and 2 in the same proportion. However, animals fed different fats showed a higher number of grade 1 tumors than grade 2 tumors, especially in animals fed OAESO where only type I tumors was observed. Therefore, there are many reasons to question whether there is a causal relationship between normal (not high) fat intake and breast cancer.1,15 Some epidemiological studies have analyzed the relationship between the normal amount of dietary fat and breast cancer, but specific conclusions have not been made. In the present study, we used a normolipidic fat level (4%) so the changes observed in the different parameters were a consequence of the type, not of the amount of fat, because all the diets used were isocaloric and had contributed to the energy intake in the same way. Many studies have suggested that monounsaturated fat in the form of olive oil may be protective against breast cancer, or at least may prevent a variety of chemically induced tumors,1,16 although other authors have suggested that the different percentages of oleic and linoleic acids have a key role in the protective effects of olive oil. 17 These data may explain the results obtained in present work. In this sense, the tumor size, which may be related to the malignancy of the tumor, was similar in animals fed commercial, EVOO, and SO diets, though the animals fed OAESO presented the highest volume; however, in these animals, the degree of malignancy of these tumors is the lowest. Therefore, it can be concluded that these tumors are larger but with better prognosis. These results may suggest that the percentage ratio between the different fatty acids in the diet is more important than the particular concentration of one fatty acid in particular.
Regarding the RAS, it does not only play essential roles in the maintenance of vascular homeostasis 5 but also promotes directly or indirectly angiogenesis and growth of neoplastic cells.18,19 The 2 main bioactive peptides of the RAS, Ang II and its direct metabolite Ang III, exhibit the same affinity for type 1 and type 2 angiotensin receptors,20,21 and it is known that AT1 and AT2 receptors are expressed in breast cancer. 8 In this work, the influence of dietary fat on aminopeptidase activities involved in RAS metabolism in NMU-induced breast cancer animals has been also analyzed. We observed that APA is increased in all the studied groups; therefore, this effect could be due to tumor rather than the diet. The increase in APA activity suggests an increase in Ang III and a decrease in Ang II circulating levels. This decrease may be responsible for the overexpression of AT1 receptors described in breast cancer. 18 However, increased levels of Ang III, which exhibits the same affinity for the AT1 receptors, would facilitate the development of the disease.
APB and APN are involved in the metabolism of Ang III. The behavior of APB is similar to APA, because it increased in all the groups analyzed except in OAESO group; therefore, these changes can also be related with the tumorigenesis rather than the diet. However, APN activity showed different behaviors. The commercial and EVOO diets did not change the activity, but a significant increase and a significant decrease were observed in SO and OAESO groups, respectively. Taken together, we can conclude that the metabolism of Ang III may be modified by dietary fat because APN activity changes differently with the diet. Metabolism of Ang III should be faster in animals fed SO, since the APB and APN activities are increased.
An increase in circulating levels of APB and/or APN activities suggests that the metabolism of Ang III to Ang IV is rapid, indicating an increase in circulating levels of Ang IV. The action of Ang IV is mediated by the AT4 receptors. 22 Albiston et al 23 have identified this receptor as the insulin-regulated aminopeptidase (IRAP) and have also shown that the AT4 receptor ligands dose-dependently inhibit the catalytic activity of IRAP. Although our results did not agree with an inhibitory effect of Ang IV on IRAP activity, we can conclude that both IRAP and Ang IV forming activities are involved in this animal model of breast pathogenesis through unknown mechanisms. In previous studies, we described that IRAP activity was greatly increased in serum of rats with mammary tumors induced by NMU. Current results support these previous results because we observed a significant increase in this activity in NMU-induced rats fed a commercial diet. Interesting data were obtained when the influence of dietary fat is analyzed. No changes were observed in EVOO and AGEO groups, but a significant increase was observed in the SO group. These results may indicate the possible role of oleic acid in the regulation of this activity, because animals fed a diet containing oleic acid showed similar levels of IRAP activity to control animals. On the other hand, there are evidences that support that IRAP activity is particularly interesting in breast cancer due to its role in the hydrolysis of oxytocin (OTX). OTX has been reported to be common in cells of healthy breast tissue but is rarely or never detected in breast cancer. 24 In addition, OTX inhibits the proliferation of human breast cancer cell lines 25 and, thus, may play a role in prevention this disease. 26 In vivo, OTX significantly reduced the growth of mouse mammary carcinoma. 27 The increased levels of IRAP activity in commercial and SO groups may indicate a lower availability of OTX and this may be responsible for the increase in the OTX receptor number described by other authors in breast cancer. On the other hand, IRAP is the only aminopeptidase known to cleave vasopressin (AVP). 24 It has been described that all breast cancer expresses the AVP gene and AVP receptors. Through these receptors, AVP exercises multifaceted effects on tumor growth and metabolism. The increase in serum IRAP activity could also be an indicator of a misregulation of AVP function.
In any case, our results show that dietary fat modifies differentially the enzymes involved in the regulation of RAS. It might suggest that one of the mechanisms by which dietary fat affects breast cancer is precisely the modification of this system, which may be considered as a new target for integrative therapies.
Footnotes
Authors’ Note
MDRS is a fellow of the Consejo Andaluz de Colegios Oficiales de Farmacéuticos.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Supported by Junta de Andalucía through PAIDI BIO-296.
