Abstract
Background. Homoeopathic medicines treat diseases, including cancer, using ultradiluted preparations. Earlier studies indicated that homoeopathic medicines are cytotoxic to tumor cells and reduced animal tumors. However, the mechanism of homoeopathic medicines at the cellular level is not known. Methods. The following drugs were used in the study: Ruta 200C, Carcinosinum 200C, Hydrastis 200C, Thuja 200C, and Thuja 1M. These drugs were tested for their ability to induce apoptosis as seen by morphology, DNA laddering, expression of genes related to apoptosis, and TUNEL assay. Similarly, the effect of homoeopathic medicines on apoptosis was measured by microarray analysis. Activity of Ruta 200C was compared with that of the mother tincture. Results. Ruta 200C produced morphological changes in the Dalton’s lymphoma ascites tumor cells and induced DNA laddering. Carcinosinum 200C increased apoptotic gene p53 and Ruta 200C decreased antiapoptotic gene Bcl2. Administration of potentiated homoeopathic drugs to tumor-bearing mice induced TUNEL-positive cells in the tumor, showing increased apoptosis of tumor cells. Microarray analysis of cells treated with homoeopathic drugs indicated that many enzymes related to apoptosis were increased by homoeopathic drugs. Conclusion. These data indicate that apoptosis is one of the mechanisms of tumor reduction of homeopathic drugs. A comparison of potentiated drugs with their mother tincture indicated that the potentiated drugs have biological activity similar to that of their mother tincture in spite of ultradilution.
Introduction
Complementary and alternative medicine (CAM) has gained importance worldwide because of its effectiveness in controlling various ailments. In the United States, approximately 38% of adults (about 4 in 10) and approximately 12% of children (about 1 in 9) are using some form of CAM. Homeopathy, introduced by Samuel Hahnemann, is a widely used CAM in many European and Asian countries; it uses dynamized medicines to induce the body’s self-healing mechanisms to bring about symptom or disease resolution. 1 It is one of the most frequently used treatments in CAM2,3 therapy and is used with or without conventional treatment in patients with cancer, 4 especially in European countries.
The efficacy of homeopathic drugs has seldom been tested in in vitro models or in animal models, and their mechanism of action has not been experimentally proven. There are only a few clinical studies on the efficacy of homeopathic medicines in cancer treatment. In 2003, Pathak et al 5 reported the use of Ruta as an effective drug for patients with brain tumors. The mechanism of action of Ruta has been reported to be the induction of telomere erosion, mitotic catastrophe, and apoptosis in cancer cells. The efficacy of Ruta against intracranial cysticercosis has also been reported. 6 In our previous study, it was found that Ruta 200C and Hydrastis 200C could significantly increase the lifespan of transplantable tumor-bearing animals by 49.7% and 69.4%, respectively. Moreover there was approximately a 95% reduction in solid tumor volume in Ruta 200C– and Hydrastis 200C–treated animals. Hydrastis 1M was found to significantly inhibit the growth of developed solid tumors produced by Dalton’s lymphoma ascites (DLA) cells and increase the lifespan of tumor-bearing animals. Moreover, significant antimetastatic activity was found in B16F-10 melanoma-induced animals treated with Thuja1M, Hydrastis1M, and Lycopodium1M. 7 In another study, concomitant administration of homeopathic drugs like Ruta 200C, Hydrastis 200C, Lycopodium 200C, and Thuja 200C retarded the tumor growth and significantly reduced the elevated marker enzymes level in nitrosodiethylamine-induced hepatic tumors. 8 It was also found that Ruta 200C and phosphorus 1M could reduce the incidence of 3-methylcholanthrene-induced sarcomas and increase the life span of mice harboring sarcomas. 8 Amelioration of both p-dimethylaminoazobenzene (p-DAB) and azodye-induced hepatocarcinogenesis in mice by the homeopathic drug Chelidonium has also been reported.9,10 Studies also showed evidence of protective effect against chronic arsenic poisoning by Arsenicum album-200C in mice. 11
In an animal model for prostate cancer, use of the homeopathic drugs Conium maculatum, Sabal serrulata, Thuja occidentalis, and Carcinosin produced a 23% reduction in tumor incidence and a 38% reduction in tumor volume compared with untreated controls. Tumors in these treated animals showed a 19% increase in apoptotic cell death and reduced PCNA-positive cells. 12 A study on the in vitro and in vivo effect of a homeopathic preparation of S serrulata was also reported. The prostate tumor xenograft size was found to be significantly reduced in S serrulata–treated mice. In vitro treatment with S serrulata resulted in a 33% decrease of PC-3 cell proliferation at 72 hours and a 23% reduction of DU-145 cell proliferation at 24 hours. 13
Some reports on the in vitro activity of homeopathic drugs in cultured cells are given below. The mother tinctures as well as some potentiated medicines showed significant cytotoxicity to Dalton’s lymphoma ascite and Ehrlich ascite carcinoma cells during short-term incubation and with L929 cells during long-term incubation. The potentiated medicines were also found to inhibit CHO cell colony formation and thymidine uptake in L929 cells. The potentiated medicines produced the characteristic morphological changes and the laddering pattern in agarose gel electrophoresis of DNA, which indicated the induction of apoptosis in DLA cells. 14 Thangapazham et al 15 studied the induction of apoptosis in prostate and breast cancer cells using several potentiated drugs but could not see this effect. Another drug, Traumeel S, was shown to inhibit the production of interleukin-1, TNF (tumor necrosis factor)-α, and interleukin-8 by human T cells and monocytes in culture. 16 Phase 6 and Flu Terminator, 2 homeopathic formulations, which contain a combination of homeopathic drugs, were reported to stimulate the production of proinflammatory and anti-inflammatory cytokines by human leukocytes. 17 These results indicate that homoeopathic medicines have the capacity to inhibit cell proliferation and reduce animal tumors. Although not scientifically documented, homoeopathic physicians claim the usefulness of homeopathic drugs in cancer treatment.
In the present study, we have tried to evaluate the action of selected potentiated preparations of Ruta, Hydrastis, Carcinosinum, and Thuja in in vitro and in vivo systems using molecular biology techniques. The selection of these drugs was done as per the suggestions of Dr P. Banerjee, a famous homoeopathic physician from Kolkata because homoeopaths frequently use these medications for treating cancer patients. The activity of the potentiated preparation of Ruta was also compared with that of its mother tincture.
Materials and Methods
Source of Chemicals and Reagents
Dulbecco’s Modified Eagle’s Medium (DMEM) and Minimum Eagle’s Medium (MEM) were purchased from Hi-Media, Mumbai, India. Fetal calf serum (FCS) was purchased from Biological Industries, Israel. Oligonucleotide primer sequences of p53, Caspase-3, Bcl-2 gene, and GAPDH for reverse transcription–polymerase chain reaction (RT-PCR) were purchased from Maxim Biotech Inc (San Francisco, CA). Cell-to-cDNA II kit was purchased from Ambion Inc, Texas. The kit for cell cycle analysis—CycleTEST PLUS ApopTag peroxidase in situ apoptosis detection kit (TUNEL assay)—was purchased from Chemicon, USA. The DNA reagent kit was purchased from Becton Dickinson Immunocytometry Systems, USA. The slides used for microarray analysis were of Agilent’s Mouse Array AMADID: 016352. The labeling kit was Agilent’s low-input RNA linear amplification kit one color. The labeling method was T7 promoter–based linear amplification to generate labeled complementary RNA (one-color microarray-based gene expression analysis). The total RNA and cRNA purification kit used was Qiagen’s RNeasy minikit. The hybridization kit used was Agilent’s in situ hybridzation kit 5184-3568. The RNA quality was checked using Bioanalyzer. All the other reagents and chemicals used were of analytical reagent grade.
Preparation of Mother Tincture of Ruta graveolens
Aerial parts (leaves and tender stem) of R graveolens were purchased from a local market in Thrissur, Kerala. The voucher specimen after authentication was deposited at the herbarium of Amala Cancer Research Centre, Amala Nagar, Kerala, India, with number Ru02. A 75% hydroalcoholic extract was prepared and concentrated in a vacuum rotary evaporator at 50°C and reconstituted in distilled water. The yield was found to be 8.3%. HPLC analysis indicated that rutin was the major constituent in this preparation (data not given).
Dynamized Homeopathic Medicines
Medicines used in the study were selected based on their usefulness in cancer treatment. All the medications were purchased from the best available sources in the world in the form of potentiated preparations. Because potentiated water was not available, it was prepared by a homoeopathic manufacturer locally under our supervision. The dynamized medicines used in this study—Ruta 200C, Carcinosinum 200C, Hydrastis 200C, and Thuja 200C—were procured from Willmar Schwabe, Germany. Thuja 1M was purchased from Boiron, France. Medicines were prepared as per the original methods of Hahnemann by diluting 1 part of the mother tincture with 99 parts of ethanol and succussing 10 times. 18 This process was repeated 200 times. The 1M homeopathic drug was prepared similarly by potentiating the mother tincture in water 1000 times. Alcoholic vehicle control (30C) prepared as per original procedure was purchased from Similia Laboratories, Aluva, India. We used this potency of vehicle control as against 200C potency of the drugs because of nonavailability of the 200C potentiated vehicle.
Animals
We obtained 8-week old (20-25 g) Swiss albino mice from the Small Animal Breeding Station, Mannuthy, Thrissur, Kerala. They were housed in well-ventilated cages and fed with normal mouse chow (Sai Durga Feeds and Food, Bangalore, India) and water ad libitum. All the experiments involving animals were performed after getting approval from the Institutional Animal Ethical Committee and as per the instructions prescribed by the Committee for the Purpose of Control and Supervision of Experiments on Animals (CPCSEA), Ministry of Environment and Forest, Government of India.
Cell Lines
DLA cells were initially procured from Adyar Cancer Institute, Chennai, India, and propagated as transplantable tumors in the peritoneal cavity of BALB/c mice.
Determination of Induction of Apoptosis by R graveolens and Its Dynamized Preparation, Ruta 200C
Morphological analysis
R graveolens (100 and/or 200 µg/mL) and Ruta 200C (20 µL/mL) were added to MEM with 10% goat serum (5 mL) containing 1 million DLA cells/mL (suspension culture) and incubated for 48 hours. We have always used an alcohol concentration of 2% or less, which does not produce any cellular toxicity. The cells were washed thrice with phosphate buffered saline (PBS), were centrifuged, and the pellet was separated. A small portion of the pellet was resuspended in PBS, and cell smear was prepared on a clean glass slide and stained with hematoxylin and eosin. Apoptosis was detected by the morphological changes (chromatin condensation, nuclear condensation, and formation of apoptotic bodies).
DNA laddering
Cells treated as above were further treated with 1 mL of lysis buffer (10 mM Tris–HCl, pH 8, 10 mM EDTA, 0.9% NaCl, 0.2% Triton X-100) for 20 minutes at 4°C and further centrifuged at 10 000 rpm for 10 minutes at 4°C. Lysates were incubated sequentially with 20 µg/mL RNase at 37°C for 60 minutes and 100 µg/mL proteinase K at 37°C for 3 to 5 hours, and DNA was extracted with phenol-chloroform-isoamyl alcohol (25:24:1) and precipitated by adding 1/10th volume of 3.5M sodium acetate (pH 5.2) and an equal volume of isoamyl alcohol. DNA was electrophoresed on 1.8% agarose gel in Tris borate EDTA buffer in a horizontal gel support apparatus, and the DNA ladder patterns were viewed under UV light and photographed.
Apoptotic gene expression: p53, Bcl-2, and caspase 3
Gene expression study was carried out by the RT-PCR method. Cell to cDNA 11 kit, Ambion Inc, USA, was used for producing cDNA from DLA cells in culture without isolating mRNA. 19 DLA cells (1 × 104 cells/well) were seeded in the 96-well U-bottom titer plate using MEM with extract (100 and 200 µg/mL) and Ruta 200C and Carcinosinum 200C (20 µL/mL) and incubated for 4 hours at 37°C in CO2 atmosphere. After incubation, the medium was removed, and the cells were washed with ice cold PBS. Ice cold cell lysis buffer (100 µL) was added to the cells, and they were immediately transferred to a water bath, incubated for 15 minutes at 75°C and transferred to 200 µL holding nuclease–free micro centrifuge tubes. To this, 2 µL DNase-1/100 µL cell lysis buffer was added and incubated for 15 minutes at 37°C. DNase was inactivated by treating at 75°C for 5 minutes. PCR was performed with primers obtained from Maxim Biotech, Inc, USA. The primer sequences of the genes studied are given below:

All reagents provided in the kit were assembled in a nuclease-free micro centrifuge tube according to the protocol of the primer kit. This master mixture (40 µL) was mixed with 0.2 µL Taq DNA polymerase and 10 µL cDNA sample. The reaction mixture was vortexed and centrifuged, and PCR thermal cycling was performed according to the protocol of Maxim Biotech, Inc. The PCR products (8 µL) were separated by submerged agarose gel electrophoresis (1.8%), were visualized in a UV chamber, and documented using a gel documentation system.
Determination of apoptosis in vivo by TUNEL assay
In vivo apoptosis in solid tumors caused by homeopathic drugs was analyzed using the TUNEL assay. For this, solid tumors were developed by injecting 1 million DLA cells in the thigh region of Swiss albino mice. The tumors were allowed to develop for 25 to 30 days, by which time the tumor volume reached 1 cc in volume. Animals were further divided into 4 groups of 6 animals and treated with the homeopathic drugs by oral gavage as detailed below.
Group I: Tumor + vehicle (potentiated alcohol; 10 µL diluted in 0.1 mL distilled water/animal) every day
Group II: Tumor + Hydrastis 200C (10 µL in 0.1 mL distilled water/animal) every day
Group III: Tumor + Ruta 200C (10 µL in 0.1 mL distilled water/animal) every day
Group IV: Tumor + Thuja 200C (10 µL in 0.1 mL distilled water/animal) every day
Animals were killed after 20 doses of homeopathic drug were given; the tumor was dissected out, washed in ice cold saline, and fixed in 10% neutral buffered formalin; then, sections were made. The sections were then processed and stained using Apoptag kit, according to the manufacturer’s protocol, and were photographed. Apoptotic cells were counted as diamino benzaldehyde (DAB−)-positive cells (brown stained) at 5 arbitrarily selected microscopic fields at 400× magnification. The total number of cells was also counted, and the apoptotic index was calculated as the number of apoptotic cells × 100 divided by the total number of cells.
Determination of Cell Cycle Arrest by Dynamized Medicines
The effect of selected dynamized medicines on cell cycle distribution was determined by flow cytometry after staining with propidium iodide. For this, 1 million DLA cells were seeded on to a 24-well titer plate with 2 mL DMEM with 10% FCS. These cells were incubated for 10 hours at 37°C in CO2 atmosphere with and without dynamized medicines—namely, Carcinosinum 200C, Hydrastis 200C, Ruta 200C, and Thuja 200C—at a concentration of 20 µL/mL. Potentiated alcohol was also used as a vehicle control. The plate was shaken at every 1 hour interval, and at the end of incubation, the cell pellet was collected, washed in PBS, and analyzed for cell cycle changes in a flow cytometer using a Becton-Dickson-cycleTEST PLUS kit as per the manufacturer’s protocol.
Determination of the Effect of Dynamized Medicines on Gene Expression in DLA Cells Through Microarray Analysis
The gene expression profile of 15 000 genes involved in the process of carcinogenesis and apoptosis was studied. For this, 5 million DLA cells were incubated with or without drug in 24-well plates with 2 mL serum-free DMEM. The incubation was carried out in 5% CO2 incubator at 37°C for 6 hours with shaking at every 1 hour interval. The dynamized medicines used were vehicle alcohol 30C, Carcinosinum 200C, Hydrastis 200C, Ruta 200C (20 µL/mL), and Thuja 1M (100 µL/mL). Untreated cells and cells treated with 20 µL/mL potentiated alcohol were also analyzed. After incubation, the cells with medium were transferred to plastic vials and centrifuged to collect cell button. This was then frozen in liquid nitrogen and used for the analysis. Thuja 1M was compared with untreated DLA cells because it is an aqueous preparation, whereas the alcoholic preparation of Carcinosinum 200C, Hydrastis 200C, and Ruta 200C were compared with potentiated alcohol–treated cells.
Statistical Analysis
The results are expressed as mean ± standard deviation. Statistical evaluation of the data was done by 1-way ANOVA followed by Dunnet’s test (post hoc) using the In Stat 3 software package.
Results
Effect of R graveolens and Ruta 200C on the Induction of Apoptosis
Morphology and DNA laddering
Results indicated that R graveolens as well as Ruta 200C induced apoptosis, as seen by the morphological features of the treated DLA cells. The classical features of apoptosis like blebbing, chromatin condensation, and formation of apoptotic bodies were seen in treated cells (Figure 1). These features were not seen in vehicle-treated cells. DNA isolated from cells treated with R graveolens and Ruta 200C showed typical DNA laddering after electrophoresis indicating the induction of apoptosis by these medications (Figure 2).

Morphology of Dalton’s lymphoma ascite (DLA) cells after treatment with Ruta graveolens and Ruta 200C (in vitro) (400×): A. untreated DLA cells; B. vehicle control (1% alcohol); C. cell treated with R graveolens showing blebbing; D, E. cell treated with R graveolens showing apoptotic bodies; F. cell treated with Ruta 200C showing chromatin condensation; G, H. cell treated with Ruta 200C showing apoptotic bodies
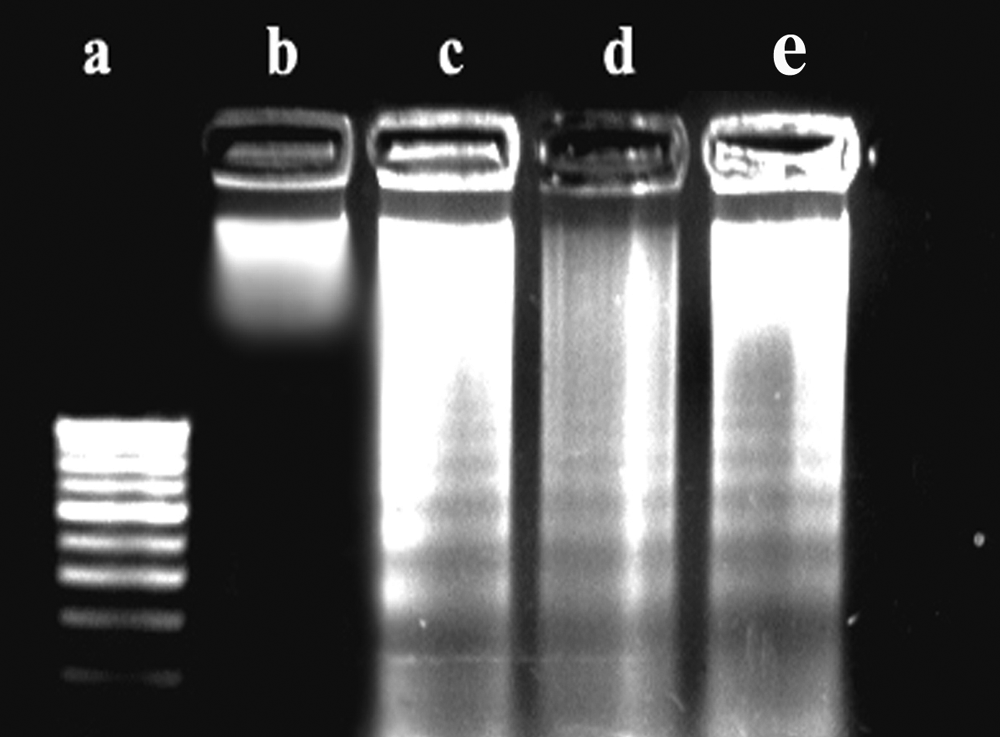
DNA laddering after treatment with Ruta graveolens and Ruta 200C (in vitro): lane a, molecular weight marker (100 bp); lane b, untreated control; lane c, treated with R graveolens (100 µg/mL); lane d, treated with R graveolens (200 µg/mL); lane e, Treated with Ruta 200C (20 µL/mL)
Expression of apoptotic genes
The expressions of proapoptotic genes p53 and Caspase 3 and the antiapoptotic gene Bcl-2 were checked with R graveolens, Carcinosinum 200C, and Ruta 200C. There was no expression of p53 and Caspase 3 when incubated with R graveolens and Ruta 200C. However, the expression of the Bcl-2 gene (antiapoptotic) was found to be inhibited by R graveolens and Ruta 200C. Carcinosinum 200C induced p53 gene expression in the DLA cells. The internal control GAPDH was found to be expressed in all the samples (Figure 3).

Gene expression of p53 and bcl2 after treatment with Ruta graveolens, Carcinosinum 200C, and Ruta 200C (in vitro). (i) Expression of p53 gene: lane a, positive control for p53 (205 bp); lane b, control Dalton’s lymphoma ascite (DLA) cells without p53 expression (205 bp); lane c, treated with R graveolens (100 µg/mL) showing no expression; lane d, treated with R graveolens (200 µg/mL) showing no expression; lane e, treated with Ruta 200C (20 µL/mL) showing no expression; lane f, treated with Carcinosinum 200C (20 µL/mL) showing expression. (ii) Expression of Bcl2 gene: lane a, positive control for Bcl2 (235 bp); lane b, control DLA cells showing Bcl2 expression (235 bp); lane c, treated with R graveolens (100 µg/mL) showing no expression of Bcl2; lane d, treated with R graveolens (200 µg/mL) showing no expression of Bcl2; lane e, treated with Ruta 200C (20 µL/mL) showing no expression of Bcl2
TUNEL assay
TUNEL staining has been used extensively to identify cells with nuclear DNA fragmentation. Cells were scored as apoptotic when they were TUNEL positive (brown staining), and TUNEL-negative cells (green colored) were visualized by counterstaining with methyl green (Figure 4). Quantification of the number of apoptotic cells showed significant increase in the TUNEL-positive cells in the treated group of animals. In control animals, apoptotic cells were found to be 4.3% ± 0.39%. After treatment with Hydrastis 200C, the apoptotic cells in solid tumor were found to have increased to 67.2% ± 7.12 %( P < .001). In Ruta 200C– and Thuja 200C–treated animals, the number of apoptotic cells were found to be 24% ± 3.86% and 50.04% ± 0.96% (P < .001), respectively. These results show the ability of these homeopathic drugs to induce apoptosis in solid tumors.

Immunohistochemistry for TUNEL-positive cells of tumor section after treatment with homeopathic drugs (in vivo) (400×): A. vehicle (potentiated alcohol)-treated cells (10 µL); B. treated with Hydrastis 200C (10 µL); C. treated with Ruta 200C (10 µL); D. treated with Thuja 200C (10 µL)
Cell Cycle Analysis
The cell cycle arrest was analyzed in a flow cytometer. It was found that none of the dynamized preparations produced any profound change in cell cycle within 10 hours of the incubation period studied.
Gene Expression Profile of DLA Cells Through Microarray Analysis
The expression profile of selected dynamized preparations was analyzed using a microarray. The expression of 15 000 genes involved in apoptosis and cancer was analyzed. The profile of Thuja 1M–treated cells was compared with that of untreated cells because it is an aqueous preparation. The effects of Carcinosinum 200C, Hydrastis 200C, and Ruta 200C were compared with that of vehicle-treated DLA cells. The fold change used for upregulation was >1, and for downregulation, it was <−1.
When the DLA cells were incubated with Carcinosinum 200C, it was found that 30 genes were upregulated and 50 genes were downregulated. In the case of Hydrastis 200C, 28 genes were upregulated, and 58 were downregulated. Ruta 200C caused the upregulation of 44 genes and downregulation of 51 genes. It was found that 280 genes were upregulated and 326 were downregulated by Thuja 1M treatment. The genes involved in signaling pathways were found to be altered by Thuja 1M treatment (Figure 5). Genes that are involved in apoptosis and carcinogenesis, which were up/downregulated by treatment with the homeopathic preparations are listed in Tables 1 and 2. The overall view of differentially regulated genes with Carcinosinum 200C, Hydrastis 200C, Ruta 200C, and Thuja 1M are shown in Figures 6 and 7.
Expression Profile of Genes With Fold Change Involved in Apoptosis and Cancer After Treatment With Homeopathic Medicines In Vitro: Upregulation

Expression Profile of Genes With Fold Change Involved in Apoptosis and Cancer After Treatment With Homeopathic Medicines In Vitro: Downregulation


Number of differentially regulated genes by treatment with dynamized medicines (in vitro)

Overall view of differentially regulated genes by treatment with dynamized medicines (in vitro): A. vehicle (potentiated alcohol)-treated cells (20 µL/mL); B. cells treated with Carcinosinum 200C (20 µL/mL); C. cells treated with Hydrastis 200C (20 µL/mL); D. cells treated with Ruta 200C (20 µL/mL)

Overall view of differentially regulated genes by treatment with Thuja 1M (in vitro): A. untreated cells; B. cells treated with Thuja 1M (100 µL/mL)
Microarray analysis showed that different genes involved indirectly in the process of apoptosis were upregulated and those acting as second messengers in the process of tumorigenesis were downregulated.
Discussion
In the present study, we tried to elucidate a possible mechanism of action of R graveolens and some other selected homeopathic medicines used in cancer treatment. Cytotoxic activity of a drug is often one of the first assessments done to determine if it could be used against cancer. Dynamized preparations showed significant cytotoxic action against cancer cell lines, and at times, the activity was higher for 200C potency than for the mother tincture. 14 The cytotoxicity produced in the cells may be either by necrosis or by apoptotic induction. It has been reported earlier that the R graveolens possessed cytotoxic action to certain tumor cells 20 and the present study showed that the observed cytotoxicity of homeopathic drugs was through apoptosis, as it produced typical morphological changes of apoptotic cell death in DLA tumor cells. This was also supported by in vivo studies in which animals treated with homoeopathic medicines significantly increased the TUNEL positive cells in the tumors, indicating that apoptosis is one of the major mechanisms through which these drugs act. Moreover, we have shown earlier that treatment with Hydrastis 1M significantly reduced already developed solid tumors in mice that were induced by DLA cells. 7 We had earlier reported 14 that out of 10 homoeopathic medicines (200C) tested, only Ruta 200C, Thuja 200C, and Carcinosinum 200C produced morphological features of apoptosis, indicating that the apoptosis is a result of the medicines rather than the action of the vehicle used. It was found that the DNA isolated from the treated cells showed the laddering pattern, which is one of the hallmarks of apoptosis, in which the internucleosomal degradation of the cellular DNA by an endonuclease resulted in the appearance of DNA fragments, which are multiples of 180 to 200 bp. The molecular pathways that lead to apoptosis are regulated by many genes, such as caspases, p53, and Bcl-2, which have direct involvement in apoptosis. It was found that the Ruta extract could inhibit Bcl-2, the antiapoptotic gene, and hence, one of the mechanisms through which R graveolens exerts its action could be through inhibition of Bcl-2. It was also found that R graveolens can induce reactive oxygen species. Hence, a possible mechanism of apoptotic induction may be through the reactive oxygen species–mediated p53 independent pathway in which the apoptosis inducible factor is involved. 21 It has been reported that rutin, the active ingredient of R graveolens, induces apoptosis in cancer cells. 22 Dedoussis et al 23 also demonstrated apoptosis induction in K562 cells by rutin, and the apoptotic activity shown has been attributed to granzyme induction or death receptor/ligand interactions. Recently, Frenkel et al 24 showed that 4 ultradiluted homoeopathic preparations induced cell cycle agents and apoptosis in 2 host cancer cell lines, accompanied by altered expression of cell cycle regulatory proteins.
Another mechanism of apoptosis induction, which is independent of p53 activation, is through bax, which can induce apoptosis 25 and which is translocated from the cytosol to mitochondria, facilitating membrane permeabilization. 26 Bax translocation and permeabilization of membranes are blocked by antiapoptotic proteins like Bcl-2, which are localized mainly in mitochondrial membranes, where they block membrane permeabilization. So an increased ratio of bax/bcl-2 leads to the formation of pores in the mitochondria, resulting in an efflux of small molecules inside the mitochondria such as cytochrome c, apoptosis inducible factor, and other proapoptotic factors. 27 Mitochondria act as a crossover point between caspase-dependent and caspase-independent apoptotic pathways. Apoptosis inducible factor, which is located in the mitochondrial intermembrane space and is released to the cytosol and to the nucleus in response to death stimuli, is a key trigger of caspase-independent apoptosis.28,29 Release of apoptosis inducible factor results in the generation of apoptotic phenotypes like chromatin condensation and phosphatidylserine exposure. It was found that there was no expression of the p53 gene after treatment with dynamized Ruta preparation, but it could inhibit Bcl-2, indicating the triggering of apoptosis through a caspase/p53-independent mechanism.30,31
The gene expression profile using microarray showed that there was upregulation of genes involved in the process of apoptosis and downregulation of oncogenes. Carcinosinum 200C upregulated A230072I16Rik, a Bcl-2 inhibitor of transcription. It is a mitochondrial protein that functions as a peptidyl-tRNA hydrolase, which when released into the cytoplasm elicits apoptosis. 32 Hydrastis 200C could upregulate Hipk2, a gene involved in the induction of apoptosis. 33 Ruta 200C upregulated the proapoptotic gene Bmf 34 and death domain kinase, RIP (receptor-interacting protein), an important gene induced during DNA damage involved in p53-independent cell death. 35 Another gene significantly upregulated by Ruta 200C is a proapoptotic gene Bif-1 (Sh3glb1), an important component of the mitochondrial pathway for apoptosis, which is a novel Bax/Bak activator, whose loss may contribute to tumorigenesis. 36 Thuja 1M has been shown to upregulate many genes involved in apoptosis, as discussed in the Results section. It downregulated the genes involved in cell motility, adhesion, and proliferation, which include Mapk2k2, Pdgfa, Rdx, Iqgap1, Wasf2, and so on. Many growth factor receptors are also downregulated. A diagram illustrating the possible mechanism of action of homeopathic drugs is shown in Figure 8.

Hypothetical diagram showing possible mechanism of induction of apoptosis by homeopathic drugs
The role of immunity in the therapeutic efficacy of homeopathic medicines has been reviewed by Bellavite et al.37,38 Gene expression profiling of macrophages with a combinatorial homeopathic drug, canova, has been reported to modulate more than 100 genes involved in transcription/translation, cell structure, immune response, and so on. 39 Likewise, these data indicate that one of the mechanisms of dynamized preparations may be through exerting cytotoxic action by modulating the gene expression of many genes involved in the process of apoptosis. Khuda-Bukhsh40,41 has proposed earlier that the action of homeopathic drugs may be through regulation of gene expression, presumably through hormone–hormone-protein complexes, the sensor gene–integrator gene–receptor gene–producer gene pathway, or through the regulator/mutator gene–operator gene–structural gene pathway. This is in line with observations from our studies. These data indicate that apoptosis is one of the mechanisms of tumor reduction of homeopathic drugs. A comparison of potentiated drugs with their mother tincture indicated that the potentiated drugs have biological activity similar to that of their mother tincture in spite of ultradilution.
However, the interaction of dynamized medicines with their cellular counterparts cannot be explained by our current knowledge of molecular symmetry and drug interaction with cellular components.
Study Limitations
In a study such as this, we should have used a similarly potentiated vehicle, purchased from the same manufacturer, as the control. However, because of its unavailability, we could not do the same.
Footnotes
Acknowledgements
The authors would like to acknowledge Samueli Foundation, USA, for the grant allowing us to carry out this work. They are also grateful to Dr R. K. Maheshwari for his kind help.
The author(s) declared no potential conflicts of interest with respect to the authorship and/or publication of this article.
Authors received financial support from Samueli Foundation, USA to carryout the work.
