Abstract
The radioprotective effect of thymol (TOH), a monoterpene phenol, on radiation-induced DNA damage was analyzed in vitro. Chinese hamster lung fibroblast cells (V79) were treated with different concentrations of TOH (0-100 µg/mL) for 1 hour before exposure to 3 Gy gamma irradiation, and then cytokinesis-blocked micronucleus and single-cell gel electrophoresis (comet assay) assays were used to evaluate the radiation-induced cytogenetic damage and genotoxic effects. Furthermore, the modulating effect of TOH on radiation-induced cell death was assessed by apoptotic and necrotic cell detection by staining with ethidium bromide/acridine orange using fluorescence microscopy. To understand the mechanism of TOH-imparted cytoprotection, mitochondrial membrane potential (MMP) was detected by flow cytometry after staining the cells with Rhodamine 123. Pretreatment of V79 cells with various concentrations of TOH (0-100 µg/mL) for 1 hour reduced the radiation-induced micronuclei as well as percent tail DNA and mean Olive tail moment with a maximum protective effect observed at TOH (25 µg/mL). Apoptosis by microscopic, MMP measurements indicated that the V79 cells exposed to gamma radiation alone showed a maximal increase in the number of early and late apoptotic and necrotic cell death associated with a significant loss of the MMP. Pretreatment with TOH (25 µg/mL) showed a significant (P < .01) decrease in the level of apoptotic fraction as well as necrotic cells and suppressed the radiation-induced collapse of MMP when compared with the radiation alone group. These results suggest that TOH suppresses radiation-induced genotoxicity, apoptosis, and necrosis primarily by the free radical scavenging and modulation of oxidative stress.
Introduction
Exposure to ionizing radiation is not only from natural (terrestrial and cosmic) sources but also from man-made sources. The main source of human exposure from man-made devices is in medicine, where radiation is used for both diagnosis and treatment of diseases. The ionizing event involves the expulsion of an orbital electron from a molecule, producing a positively charged or “ionized” molecule. Although this process forms positive ions, the essential factor from the biological perspective is that these molecules contain an unpaired electron in one of its orbital, forming free radicals. These free radicals exist only for a fraction of second but are extremely reactive and may lead to permanent damage of the affected biomolecule. Most of the energy deposited within a cell results in the production of aqueous free radicals, because approximately 70% to 80% of the cell is water. Thus, water radiolysis fabricates molecules such as hydrogen peroxide (H2O2), molecular hydrogen (H2), and a number of highly reactive radicals, such as hydrogen (H•), hydroxyl (OH•), hydroperoxyl (HO•), and superoxide (O2•). Because of their high yields and their reactivity, the OH radical and H2O2 are of great importance in a number of human ailments. 1
For the past few years, the DNA molecule has been considered to be the essential target of ionizing radiation in the cell, resulting in the induction of base damage, cross-links, DNA strand breaks such as single-strand and double-strand breaks.1,2 The DNA damage is mainly induced by abstraction of the H atom from the C-4 position of the deoxyribose on each strand or by the attack of bases via hydroxyl radicals produced by radiolysis of water. 3
One of the mechanisms of protection, involving free radical scavenging, is based on the understanding that free radicals resulting from the radiolysis of water are the main cause of radiation damage to cells. 4 Antioxidants prevent free radical–induced cellular damage by scavenging them or by promoting their decomposition 5 and can act as radioprotectors. Radioprotectors are therefore defined as agents that are administered before, during, or after radiation to prevent or reduce damage to normal cells by radiation.
Although many chemicals such as cysteine, cysteamine, and WR-2721 have proved to be promising radioprotectors, the inherent toxicity of some of these synthetic agents at their radioprotective concentrations necessitated further search for safer and effective radioprotectors for their clinical applications. Many naturally occurring antioxidants demonstrate a long window of protection, including postirradiation protection against lethality and mutagenesis. 6 The fascinating fact of natural antioxidants is that they are comparatively less toxic than most of the well-known synthetic radioprotectors.
Thymol (TOH) is a monoterpene phenol derivative of cymene, found in oil of thyme and plants such as Thymus vulgaris, citrus plants, and so on. It exhibits multiple biological activities, such as antibacterial, 7 antifungal, 8 anti-inflammatory, 9 and also possesses antioxidant, 10 free radical scavenging, 11 and antilipid peroxidative 12 properties. These properties are likely to play a curtail role in the sequence of events leading toward radiation-induced cellular damage.
Recently, we reported gamma radiation–induced cell damage and its protection by TOH via scavenging of free radicals and inhibition of oxidative stress. 13 In continuation of our previous study, the present study was undertaken to assess the effect of TOH given in combination as a protective regimen against radiation-induced DNA damage, assessed by micronuclei and comet assay in Chinese hamster lung fibroblast cells.
Materials and Methods
Chemicals
TOH (CAS No. 89-83-8) was purchased from ACROS Organics (Morris Plains, NJ), and fetal calf serum (FCS), penicillin, streptomycin, L-glutamine, low-melting-point agarose (LMPA), normal-melting-point agarose (NMPA), cytochalasin-B, Rhodamine 123, ethidium bromide (EtBr), Tris–HCl, and acridine orange (AO) were purchased from Sigma (St Louis, MO). Ethylene diamine tetra-acetic acid (EDTA), sodium bicarbonate, sodium chloride, potassium hydrogen phosphate, disodium hydrogen phosphate, potassium chloride, and hydrochloric acid were purchased from Qualigens Fine Chemicals (Mumbai, India).
Cell Lines and Culture
The radioprotective activity of TOH was studied using Chinese hamster lung fibroblasts cells (V79) procured from the National Center for Cell Sciences, Pune, India. The cells were routinely grown in 25-cm2 flasks with loosened caps, containing minimal essential growth medium (MEM) supplemented with 10% FCS and other required components at 37°C in a CO2 incubator (NuAire, Plymouth, MN), in an atmosphere of humidified 5% CO2 in 95% air.
TOH Preparation and Irradiation Procedure
TOH was dissolved in 0.01% of absolute alcohol in double distilled water at a concentration of 1 mg/mL and diluted with MEM immediately before use. The cultured V79 cells were treated with or without TOH before exposing to gamma radiation using the irradiation facility of Shirdi Saibaba Cancer Hospital, Manipal University, Manipal. The radiation is generated by a 60Co gamma teletherapy facility (Theratron, Atomic Energy Agency, Ontario, Canada) at a dose rate of 1 Gy/min and with a source-to-surface distance of 73 cm. All dosimetric calculations were done by a medical physicist. The different radiation doses used to assess the radioprotective potential were selected on the basis of earlier studies.14,15
Cell Culture Experiments
A fixed number (5 × 105) of exponentially growing cells were inoculated into several individual cultures in 25-cm2 T-flasks, allowed to grow for 24 hours to attain a semiconfluent exponential stage, and used for all the experiments.
Selection of optimum dose of TOH
The exponentially growing V79 cells were trypsinized and appropriate number (5 × 104) of cells per culture flask were seeded in to 25-cm2 T-flasks and the optimum radioprotective dose of TOH was selected by treating the cells as follows:
TOH + sham-irradiation: The cultures of this group were treated with (0, 5, 10, 25, 50, and 100 µg/mL) of TOH for 1 hour before sham-irradiation (0 Gy).
TOH + irradiation: The cultures of this group were treated with 0, 5, 10, 25, 50, and 100 µg/mL of TOH for 1 hour before exposure to 3 Gy gamma radiation.
Radioprotective/antigenotoxic effect of TOH
The optimum dose of 25 µg/mL TOH selected from the previous experiment was used to assess the radioprotective/antigenotoxic effect in V79 cells, wherein cell cultures were set up and treated as follows:
Irradiation alone (IR): The cultures of these different groups were exposed to different doses (0, 1, 2, 3, and 4 Gy) of gamma radiation.
TOH + irradiation: The cultures of these groups were treated with 25 µg/mL of TOH for 1 hour before exposure to different doses (0, 1, 2, 3, and 4 Gy) of gamma radiation.
After the various treatments, media was removed, and the cells were dislodged by trypsin–EDTA treatment. The radioprotective and antigenotoxic effect of TOH was analyzed by carrying out cytogenetic analysis.
Micronucleus Assay
The micronucleus frequency test is a widely accepted method for in vitro and in vivo genotoxicity investigations and human biomonitoring studies.16,17 Micronuclei are DNA-containing bodies that are formed during the mitosis as a result of radiation-induced chromosomal breaks or from whole chromosomes incorporated in the daughter cells as discrete entities along with the main nucleus. The micronuclei assay was carried out according to the method of Fenech and Morley, 16 modified and adapted to our laboratory conditions. 14 Briefly, the cells from both experiments were allowed to attach for 6 hours of posttreatment and incubated further with 3 µg/mL of cytochalasin-B to inhibit cytokinesis. The cells were left undisturbed and allowed to grow for another 16 hours. Thereafter, the medium containing cytochalasin-B was removed and the cells were washed with phosphate-buffered saline (PBS). Finally, cells were dislodged with trypsin–EDTA treatment, centrifuged, and subjected to mild hypotonic treatment (0.75% ammonium oxalate) for 3 minutes at 37°C. Later, the cell suspension was centrifuged again and the resultant cell pellet was fixed in Carnoy’s fixative (3:1 methanol, acetic acid). The cells were centrifuged again, resuspended in a small volume of fixative, and spread on to precleaned coded slides. Duplicate cultures were used for each drug concentration in each group, and the results were confirmed by repetition of these experiments.
The slides containing cells were stained with 0.002% AO in Sorensen’s buffer (pH 6.8) and washed twice in the buffer. The buffer-mounted slides were observed under a fluorescence microscope equipped with a 450 to 490 nm BP filter set, with excitation at 453 nm (Carl Zeiss Photomicroscope III, Jena, Germany) for the presence of micronuclei in the binucleate cells (BNC). A minimum of 1000 BNC only with well-preserved cytoplasm was scored from each culture, and the frequency of micronucleated binucleate cells (MNBNC) was determined. The micronuclei identification was done according to the criteria of Fenech and coworkers. 18 Cytokinesis block proliferation index (CBPI) was calculated, which indicates the average number of cell cycles per cell during the period of exposure to cytochalasin-B, and used to calculate cell proliferation. To determine the cytotoxic effect, CBPI was calculated using the following formula:
Cytostasis
Assessment of cytostasis indicates the reduction in cell proliferation after various treatments when compared with the control/untreated cultures. The percentage of cytostasis was calculated using the following formula:
where CBPIT is the cytokinesis block proliferation index of the treated group and CBPIc is the cytokinesis block proliferation index of the untreated control.
Comet Assay (Single-Cell Gel Electrophoresis)
The alteration in radiation-induced DNA damage and its repair by TOH was evaluated by single-cell gel electrophoresis (comet assay). A separate experiment was conducted to assess the effect of TOH on IR-induced DNA damage and repair, where groupings and other conditions remained similar to that described for radioprotective effect, except that V79 cells were allowed to grow further for various postincubations for allowing DNA repair (30, 60, and 120 minutes); cells from the above-mentioned groups were dislodged by trypsin–EDTA (0.1%) treatment and processed for comet assay. This assay was performed under alkaline conditions at pH 13, according to the original procedure of Singh and coworkers 19 with minor modifications as described in our earlier publication. 20 Briefly, slides frosted at one end were covered with 100 µL of 0.75% LMPA prepared in PBS at 37°C and the agarose was allowed to solidify under a cover slip on ice after which the cover slips were removed. The pelleted cells were embedded in 0.75% LMPA following immersion in ice-cold lysis mixture (2.5 mol NaCl, 0.1 mol Na2EDTA, 0.01 mol Tris–HCl, pH 10, 1% Triton X-100) for 1 hour. The slides were then transferred to electrophoresis solution (0.3 mol NaOH, 1 mmol Na2EDTA, pH > 13) for 20 minutes at 4°C. A current of 25 V (0.3 A) was then applied for 30 minutes. The slides were neutralized with 0.4 mol Tris–HCl (pH 7.5) and finally stained with EtBr (5 µg/mL). All the steps were conducted under a reduced light level to prevent additional DNA damage.
Ethidium bromide–stained DNA on each slide was visualized at 40× magnification using fluorescence microscopy as “comets” with the fluorescent measurements in the head portion of the comet and in the tail by capturing the images using an epifluorescence microscope (Olympus BX51, Olympus Microscopes, Tokyo, Japan) equipped with 515 to 535 nm excitation filter, a 590 nm barrier filter, and a CCD camera (CoolSNAP-Procf Digital Color Camera Kit Ver 4.1, Media Cybergenetics, Silver Spring, MD). For the determination of percentage DNA damage, a total of 100 images per group was captured and analyzed using Komet software (Version 5.5, Kinetic Imaging, Bromborough, UK).
Morphological Study of Apoptotic Cells
Apoptosis was determined morphologically after staining the cells with EtBr/AO using fluorescence microscopy as described earlier. 21 Briefly, exponentially growing V79 cells (106) were seeded in 25-cm2 T-flasks and allowed to attach and grow overnight. The cells were grouped as follows:
Control group: The culture of this group was left untreated (without TOH/radiation).
TOH alone group: The culture of this group was treated with optimal concentration (25 µg/mL) of TOH for 1 hour before sham-irradiation.
Irradiation alone group: The culture of this group was exposed to 10 Gy gamma radiation.
TOH + IR group: The cells of this group were treated with optimal concentration (25 µg/mL) of TOH for 1 hour before exposure to 10 Gy gamma radiation.
After various treatments, cells were further allowed to grow for 24 and 48 hours. Both adherent and floating cells were collected and washed with cold PBS and stained with a mixture of AO (50 µg/mL) and EtBr (10 µg/mL). The cellular morphology was evaluated using fluorescent microscope (Olympus BX51, Olympus Microscopes) at 40× magnification. Altogether 200 cells were analyzed and differentiated as living (green intact nucleus), early apoptotic (bright green condensed or fragmented chromatin), late apoptotic (disrupted cytoplasmic membrane with orange-stained condensation or fragmentation chromatin), and necrotic cells (uniformly orange-stained cell nuclei).
Assessment of Mitochondrial Membrane Potential (MMP)
MMP was estimated by measuring the uptake of the fluorescent probe Rhodamine 123 as previously described by Scaduto and Grotyohann 22 using flow cytometric analysis. Briefly, the exponentially growing V79 cells (5 × 105) were divided into 3 groups—Group I (control), untreated; Group II (IR alone), cells were exposed to 4 Gy gamma radiation; Group III (TOH + IR), cells were treated with 25 µg/mL of TOH for 1 hour before exposure to 4 Gy gamma radiation and the cells were allowed to grow for a further 1 hour. At the end of the treatment period, fresh medium (MEM without FCS) was added. Rhodamine 123 (5 µg/mL) was added to the media and incubated for 30 minutes in the dark at 37°C. After 2 washes in PBS, the fluorescence intensities were analyzed by FACSCalibur flow cytometer (Becton Dickinson, San Jose, CA) and the data were analyzed using WinMDI Software Version 2.9.
Statistical analysis
Statistical analysis was performed using one-way analysis of variance (ANOVA) followed by Bonferroni’s post hoc test to determine the statistical significance between various groups. The values are mean ± SEM.
Results
Micronucleus Assay
TOH showed protection against radiation-induced DNA damage, as evidenced by the significant (P < .01) reduction in MNBNC after various doses of TOH treatment in V79 cells. TOH by itself was not toxic to DNA. Pretreatment of V79 cells with various concentrations of TOH (0-100 µg/mL) for 1 hour reduced the radiation-induced micronuclei. Although the reduction in the yield of radiation-induced micronuclei was observed in all the doses, TOH at a dose of 25 µg/mL resulted in the maximum inhibition in the yield of MNBNC when compared with other TOH-treated groups. However, increase in the dose of TOH did not further enhance the radioprotective potential. Therefore, 25 µg/mL of TOH was selected as an optimal dose for further studies (Table 1). CBPI was calculated to assess the effect of TOH on the cytotoxic effect of irradiated V79 cells. The values of CBPI in IR alone showed delay in cell proliferation. TOH pretreatment modulated the proliferative activity in the combination groups with an increase in CBPI, indicating the surmounting of cytotoxic effects caused by ionizing radiation (Table 1).
Effect of TOH on Micronuclei Induction in the V79 Cells Exposed to 3 Gy Gamma Radiation a

Abbreviations: CBPI, cytokinesis blocked proliferation index; TOH, thymol; IR, radiation.
All results are shown as mean ± SEM from the data of 3 independent experiments.
P < .01, when compared with IR-alone group.
P < .001, when compared with IR-alone group.
P < .05, when compared with IR-alone group.
The optimal dose of TOH (25 µg/mL), which had the greatest reduction in micronuclei, was further used in combination with various doses of gamma radiation (0, 1, 2, 3, and 4 Gy) exposed 1 hour after TOH treatment to assess the antigenotoxic potential even at the higher doses of radiation. A linear dose-dependent MNBNC% increase in radiation-alone group was observed, whereas 25 µg/mL TOH significantly resulted in 26.1%, 24.8%, 30.5%, and 21.1% reduction of MNBNC when compared with the respective radiation-alone groups (Table 2).
Alteration in the Frequency of Micronuclei in Binucleate Cells by Pretreatment of 25 µg/mL TOH on the V79 Cells Before Exposure to Different Doses of Gamma Radiation a

Abbreviations: TOH, thymol; IR, radiation; MN, micronuclei.
All results are shown as mean ± SEM from the data of 3 independent experiments.
P < .05, when compared with IR-alone group.
P < .01, when compared with IR-alone group.
P < .001, when compared with IR-alone group.
Comet Assay
V79 cells treated with 3 Gy of radiation caused a significant (P < .01) DNA damage as evident by the increase in tail DNA and olive tail moment (OTM) when compared with the control group. The optimum protective dose of the TOH was selected by treating V79 cells with 0, 5, 10, 25, 50, and 100 µg/mL TOH for 1 hour before exposure to 3 Gy of gamma radiation at 60 minutes of postirradiation. Treatment of V79 cells with different doses of TOH reduced the DNA strand breaks significantly, with the highest reduction being observed at 25 µg/mL TOH (Figure 1).

Determination of olive tail moment (OTM) by the comet assay in V79 cells treated with various concentration of thymol (TOH; 0-100 µg/mL) for 1 hour before exposure to 3 Gy of gamma radiation
To examine the extent of DNA repair at different postincubation time periods, the cells from both groups were harvested at 30, 60, and 120 minutes. IR-alone groups showed a maximum DNA repair in the radiation-induced DNA strand breaks at 120 minutes of posttreatment time with a reduction in the tail DNA and OTM. Treatment with optimal dose of TOH (25 µg/mL) for 1 hour before exposure with different doses of IR caused a significant reduction (P < .05) in the percentage of tail DNA and mean OTM values at all the postirradiation time points when compared with their respective IR-alone groups (Table 3).
Effect of Optimum Concentration of TOH (25 µg/mL for 1 Hour) on V79 Cells Before Exposure to Different Doses of Gamma Radiation Measured by Comet Assay at 30, 60, and 120 Minutes After Irradiation a

Abbreviations: OTM, olive tail moment; TOH, thymol; IR, radiation.
All results are shown as mean ± SEM from the data of 3 independent experiments.
P < .05, when compared with respective IR alone group.
P < .01, when compared with respective IR alone group.
P < .001, when compared with respective IR alone group.
Morphological Analysis of Apoptosis by EtBr/AO Staining
To assess the type of cell death induced by IR, V79 cells were analyzed by microscopic EtBr/AO dual-staining method. At 24 and 48 hours after the TOH alone the percentage of apoptotic and necrotic cells did not exceed 3% of the total number of cells (Figure 2). As expected, cells exposed to IR (10 Gy) alone showed a maximal increase in the number of early and late apoptotic (about 28% and 31%, respectively) and necrotic cells (7% and 9%, respectively) both at 24 and 48 hours of postincubation time periods. To investigate the anti-apoptotic effect of TOH, the nuclear morphology of the irradiated (10 Gy) cells on pretreatment with TOH (25 µg/mL) was analyzed. There was a significant (P < .01) diminution in the level of apoptotic fraction and necrotic cells, as compared with the IR-alone group, both at 24 and 48 hours of postirradiation (Figure 2A and B).

Results of apoptotic and necrotic cells assessed by ethidium bromide/acridine orange staining of V79 cells treated with optimum concentration of thymol (TOH; 25 µg/mL) for 1 hour before exposure to 10 Gy gamma radiation: (A) 24 hours; (B) 48 hours.
Assessment of Mitochondrial Membrane Potential
MMP was estimated by measuring the uptake of the fluorescent probe Rhodamine 123. The changes in the mitochondrial membrane integrity are one of the early events of apoptotic cascade of events. To examine IR-induced loss of the MMP, V79 cells were exposed to 4 Gy of gamma radiation, which showed a significant loss of the MMP as measured by the uptake of fluorescent cationic dye Rhodamine 123 at 60 minutes postincubation time, compared with the control. Pretreatment with TOH (25 µg/mL) for 1 hour significantly delayed the IR-induced collapse of MMP; however, MMP was not as that of control levels (Figure 3).
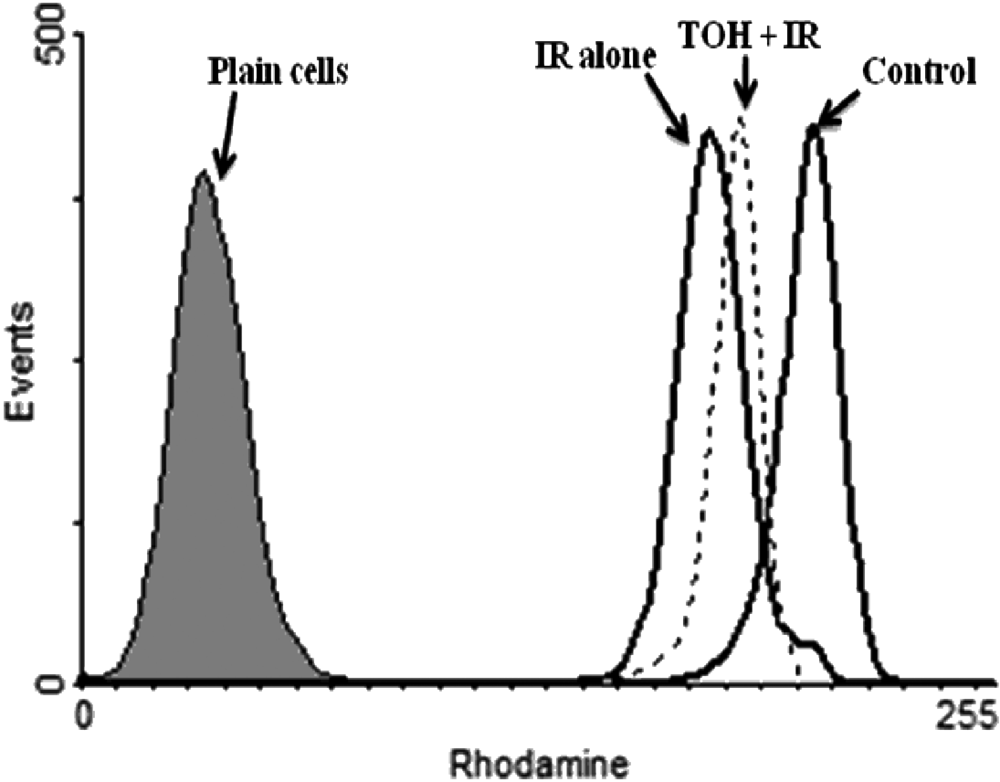
Detection of mitochondrial membrane potential with Rhodamine 123. V79 cells were treated with thymol (TOH; 25 µg/mL for 1 hour) and exposed to 4 Gy of gamma radiation and postincubation for 60 minutes
Discussion
The present study was aimed at understanding the role of natural antioxidant TOH in streamlining radiation-induced DNA damage under in vitro conditions. Although being one of the DNA-damaging sources, radiation has been known to be used successfully in cancer treatment. The ability to kill cancer cells with minimal damage to normal cells determines the success of radiation therapy. 23 It is used as palliative treatment (where cure is not possible and the aim is local disease control or symptomatic relief) or as therapeutic treatment (where the therapy has survival benefit and it can be curative). Radiation being a well-known carcinogenic xenobiotic agent can also be the cause of cancer and also induce secondary malignancies seen in a small minority of patients receiving a curative course of radiation treatment. Because of the damaging effects of ionizing radiation, researchers have long been interested in identifying new nontoxic, effective, and expedient compounds to protect humans against radiation-induced normal tissue injuries.
There has been an increased interest in the role of free radicals on either metabolic or radiation-induced carcinogenesis.24,25 Some antioxidants have been shown to protect cells against DNA damage induced by ROS, 6 and they can also have their potential to reduce oncogenesis.26,27 If such antioxidants are from dietary sources, it would be easily acceptable with their biologically achievable concentrations and may be more safely manipulated in patients undergoing radiotherapy. Our earlier study indicated that TOH is one such antioxidant that can scavenge free radicals effectively, 13 and in the present study, we attempted to evaluate the potential of TOH for the modulation of radiation-induced genotoxicity and apoptosis in V79 cells.
Ionizing radiation can cause DNA damage leading to chromosomal aberrations4,28 or cell death. 29 Despite having a well-equipped enzymatic repair system, some of the DNA lesions, especially double-strand breaks, are converted into cytogenetic alterations that may be visualized. 30 The extent of DNA damage and repair could be detected efficiently by the alkaline comet assay, 31 which detects single-strand and double-strand breaks at the level of DNA molecule such as sites of incomplete repair, alkali labile sites, and DNA–DNA and DNA–protein cross-links. The comet assay has been effectively used in this study, and the degree of DNA migration was correlated to the extent of DNA damage occurring in each single cell. Furthermore, to infer precisely the biological relevance of the results obtained, comet assay was coupled with other measure of DNA damage, that is, micronuclei, as proposed by Fenech and Morley. 16 The difference between the effects measured in the comet assay and cytogenetic tests (micronuclei) is basically because of the variations in type of DNA alterations that the test system detects, that is, cytogenetic tests detect fixed mutations that persist for at least 1 mitotic cycle, whereas the comet assay merits the detection of plethora of DNA lesions. Therefore, these assays have been used as established methods and have become a very reliable indicator of radiation-induced genetic damage. In the present study, we substantiated whether the risk of genomic instability of cells induced by IR was reduced with TOH using micronucleus and comet assay. The damage to DNA was evidenced by increased OTM with increasing doses of IR and decrease in the OTM of the comets treated with IR in the presence of TOH (25 µg/mL) in comparison with the cells treated with IR only. Our observations are consistent with the finding that TOH rendered a protective effect against mitomycin C and 2-amino-3-methylimidazo [4,5-f]-quinoline induced DNA breakage in human lymphocytes. 32
Radiation-induced double-strand breaks led to micronuclei formation and are considered to be one of the principal indicators of chromosomal damage because these are the chromatin fragments/whole chromosomes that are not incorporated into the main nucleus during mitosis. 33 The micronucleus assay, an important endpoint for the assessment of cytogenetic damage, was also used in previous studies with polyphenolic compounds to assess their antigenotoxic potential.20,34,35
The present study showed that TOH when applied in vitro did not cause significant formation of micronuclei, indicating its nontoxic effect at the doses we used; however, there was a significant reduction in the micronuclei frequency in the cells treated with TOH (25 µg/mL) before irradiation when compared with irradiated groups. These observations clearly indicate the protective effect of TOH against gamma radiation–induced DNA damage in V79 cells. Even though the protective effect showed a dose-dependent increase, there was an optimum concentration of 25 µg/mL, beyond which increase in this effect was nonsignificant. Although the actual mechanism for the optimum protection of TOH at 25 µg/mL is unclear, it could possibly be suggested that lower concentrations of TOH may not be enough to quench all the free radicals and conversely at higher concentration TOH might have reacted with some other ligands in the system and thus might not be completely available for quenching the free radicals generated by ionizing radiation. Furthermore, this is in agreement with our earlier in vitro results, wherein TOH showed maximum cytoprotection and anti-apoptotic activity against gamma radiation at an optimal concentration of 25 µg/mL and further increase in dose did not increase the radioprotective potential. 13 A similar observation has been reported by several authors for other antioxidant protectors in vivo as well as in vitro like Orientin and Vicenin, 2-mercapto-propionylglycine, and DMSO.36-38
Mitochondrial damage is known to play an important role in the regulation of apoptosis. 39 Radiation-induced ROS may cause a variety of changes in the intra and extra mitochondrial membrane function and the resultant changes in MMP. A reduction in MMP is believed to be mediated by the opening of the mitochondrial permeability transition pore, a multiprotein complex 40 leading to the release of cytochrome c from mitochondria to cytosol and the subsequent activation of the caspase pathway to promote cellular apoptosis. 41
Many methods have been proposed to estimate MMP modifications, mainly by flow cytometry with cationic lipophilic dyes. 42 Rhodamine 123 is a cell permeable, green fluorescent dye that is readily confiscated by active mitochondria and is known for its low cytotoxicity and insignificant fluorescence quenching. 43 In the present study, we found that V79 cells when exposed to IR caused redistribution of the dye through the cytoplasm and form a new equilibrium with the external medium, resulting in a loss of dye from the cell leading to decrease in fluorescence. Our study is in agreement with several other reports revealing the fact that a significant decrease in Rhodamine 123 florescence intensity is seen on triggering of apoptotic stimuli, which is associated with a considerable fall in MMP.44,45 Whereas treatment with TOH for 1 hour before exposure to IR restored the mitochondrial membrane potential significantly when compared with IR-alone treated cells. These observations are consistent with the earlier finding that the antioxidant Eckol completely suppressed IR-induced apoptosis in V79 cells by preventing mitochondrial membrane depolarization. 15
Polyphenols are well known free radical scavengers because of the high reactivity of their hydroxyl substituents. 46 The exact mechanism by which TOH mediates anticlastogenic potential is still unknown. But this may be because of the effective antioxidant potential of TOH by scavenging free radicals responsible for DNA damage. Because the high concentrations of scavengers must be present at or near the critical target sites within the hydration shell of DNA to be effective in protecting against the radiation-induced damage resulting from OH radicals, 47 the 1 hour pre-irradiation treatment of TOH was found to be optimal in this study. These observations strongly indicate that TOH should be inside the cells before and during the time of irradiation to impart radioprotection. Earlier, we have reported that the increase in the dose of radiation depletes the antioxidant status and increases lipid peroxidation in the V79 cells. 13 Treatment with TOH prior to radiation exposure increased the antioxidant status at both enzymatic antioxidant (superoxide dismutase and catalase) and nonenzymatic antioxidant (GSH) levels and decreased the level of malondialdehyde (MDA). This effect was because of its antioxidant property and shows that TOH acts as a good scavenger against free radical generation and thereby inhibits lipid peroxidation as well. Furthermore, it is convincingly demonstrated that lipid peroxidation is a complicated radical chain reaction leading to the formation of various products, such as lipid hydroperoxides, conjugated dienes, and MDA, and may also contribute to the increased DNA damage. 48 Earlier we demonstrated the antilipid peroxidative potential of TOH against radiation-induced lipid peroxidation. 13 Thus, the increase in the antioxidant status during TOH pretreatment might have further decreased the attack of such free radicals on biomolecules including DNA as well as membrane lipids and thereby decreased the deleterious effects of radiation on V79 cells.
Taken together, our data put forth the fact that TOH, a naturally occurring phenolic compound, effectively protected cells against radiation-induced genotoxicity and apoptosis. The mechanism of radioprotection by TOH could be ascribed to its antioxidant and free radical scavenging activities.
Footnotes
Acknowledgements
The authors are thankful to Dr K Satyamoorthy, Director, Manipal Life Sciences Centre; Dr P. Laxminarayana Rao, Department of Physiology and Neurosciences; and Dr Poornima Baliga, Dean; Dr Mamatha Ballal, KMC-IC, Manipal University, Manipal, for their help and encouragement during this study. We also thank Prof Vidhyasagar, Head, Radiotherapy, and Prof J. G. R. Solomon, medical physicist, Shiridi Saibaba Cancer Hospital, Manipal, for the use of the irradiation facility and for radiation dosimetry, respectively.
The author(s) declared no potential conflicts of interest with respect to the authorship and/or publication of this article.
The author(s) received no financial support for the research and/or authorship of this article.


