Abstract
The inhibition of tumor incidence by hydro-alcoholic extract of S.cumini seed was evaluated in mice on two stage process of skin carcinogenesis induced by single application of 7, 12-dimethyl benz(a)anthracene (100 µg/100µl of acetone), and 2 weeks later promoted by repeated application of croton oil (1% acetone/thrice in a week) till the end of the experiment (i.e. 16 weeks). Oral administration of extract at a dose of 250mg/kg b.wt./day at the peri-initiational stage (i.e. 7 days before and 7 days after DMBA application), promotional stage (i.e. from the time of croton oil application) and at both the stages (i.e. 7 days prior to DMBA application & continued till the end of experiment) to the mice, recorded a significant reduction in tumor incidence to 37.5, 50 & 25% respectively in comparison to the carcinogen treated control, where tumor incidence was found as 100%. Tumor yield and Tumor burden were also significantly reduced by SCE. Similarly, the cumulative number of papillomas after 16 weeks was 68 in the control group, which was reduced to 15, 21 & 8 in the animals treated with the SCE continuously at peri-, post- and peri- & post- initiation stage respectively. A significant impairment was noticed in the levels of reduced glutathione, superoxide dismutase, catalase & protein and enhancement in LPO in liver and skin of carcinogen treated control mice as compared with vehicle treated mice. All such parameters were returned to near normal value by administration of SCE to DMBA treated mice. These results suggest a possible chemopreventive property of S.cumini against DMBA induced skin carcinogenesis in mice.
Introduction
Free radicals are “any species capable of independent existence that contain one or more unpaired electrons.” 1 Because of their very high chemical reactivity, free radicals are able to induce cellular damage in a variety of ways. 2 The most deleterious effects of free radicals are damage to DNA, 3 which is associated with the process of carcinogenesis.
The process of carcinogenesis passes through multiple stages of biochemical and molecular alterations in target cells. Skin carcinogenesis involves the stepwise accumulation of genetic changes, ultimately leading to malignancy.4-6 The 3 main steps in multistage skin carcinogenesis are (1) initiation, which involves free radicals, and induced genetic changes 7 ; (2) a much slower stage of carcinogenesis, promotion, which is believed to involve selective and sustained hyperplasia, leading to the specific expansion of initiated cells into papillomas 6 ; and (3) tumor progression, characterized by a high level of genetic instability that leads to chromosomal alterations. 8 To avoid such deleterious effects, the biological systems have well-developed, efficient, and widely distributed defensive enzyme systems, which include glutathione S transferase, glutathione peroxidase, superoxide dismutase (SOD), and catalase (CAT), which can eliminate and/or detoxify the free radicals. The damaging effect of free radicals can also be reduced by natural or synthetic antioxidants.
Antioxidants can terminate the free-radical chain reaction by donating hydrogen or electrons to free radicals and converting them to more stable products. Thus, antioxidants may either delay or inhibit the initiation step of carcinogenesis. In the promotional stage of skin carcinogenesis, stem cells in the basal layer of the epidermis divide more frequently, leading to the formation of premalignant lesions. This step can be reversed by antipromoters or growth inhibitors or by the induction of apoptosis.
Increasing evidence substantiates the fact that a prudent diet has protective effects against various cancers versus a Western diet, which may be detrimental. The Western dietary pattern with its higher intake of red and processed meats, sweets, desserts, and fried foods has been associated with a higher risk of colon, breast, prostate, and several other cancers. On the other hand, a prudent dietary pattern with its higher intake of fruits, vegetables, whole grains, and cereals, contributing to increased levels of antioxidants and essential minerals, such as selenium, dietary fibers, and so on has been associated with a markedly lower risk of many cancers. Thus, it may be said that what really sets apart the oriental or prudent diet from the Western diet is the presence of a very vital component—dietary phytochemicals.9-11
Epidemiological studies on diet and cancer have provided leads in the search for naturally occurring anticancer agents. 12 There is general agreement that plant-based diets reduce the risk of various types of tumors, but the basis of this protective effect is not well understood. There is experimental evidence suggesting the possible involvement of free-radical-scavenging capabilities of these natural products. Therefore, Syzygium cumini, was evaluated for chemopreventive activity against DMBA (7,12-dimethylbenz(a)anthracene)-induced skin cancer in mice.
S. cumini (L) Skeels is a large, commonly seen evergreen tree in the Indian subcontinent but is also found in countries of Southeast Asia and Eastern Africa. It belongs to the Myrtaceae family. In the medicine systems of Indian folklore, several medicinal properties have been attributed to it. 13 The fruits and seeds are used to treat diabetes, pharyngitis, spleenopathy, urethrorrhea, and ringworm infection. The leaves have been extensively used to treat diabetes, constipation, leucorrhoea, stomachalgia, fever, gastropathy, strangury, and dermopathy and to inhibit blood discharges in the feces.13,14 In view of the importance of this herbal plant, the present study has been undertaken to evaluate the chemopreventive potential of S cumini.
Materials and Methods
Animal care and handling was approved by our institution and was done according to the guidelines set by the World Health Organization, Geneva, Switzerland, and the Indian National Science Academy, New Delhi, India. The inhibition of tumor incidence by S cumini seed extract was evaluated on 2-stage skin carcinogenesis induced by a single application of DMBA (initiator) and, 2 weeks later, promoted by repeated application of croton oil (promoter) thrice per week, following the protocol for 16 weeks. 15
Animals
The study was conducted on random-breed male Swiss albino mice (7-8 weeks old), weighing 24 ± 2 g. These animals were housed in polypropylene cages in the animal house under controlled conditions of temperature (25°C ± 2°C) and light (14 light:10 dark). The animals were fed a standard mouse feed (procured from Aashirwad Industries, Chandigarh, India) and water ad libitum. A total of 8 animals were housed in 1 polypropylene plastic cage containing sawdust (procured locally) as bedding material. As a precaution against infections, tetracycline hydrochloride water was given to these animals once each fortnight.
Chemicals
DMBA and croton oil were procured from Sigma Chemical Co, USA. DMBA was dissolved at a concentration of 100 µg/100 µL in acetone. Croton oil was mixed in acetone to give a solution of 1% dilution.
Plant Material and Extract Preparation
The fruit of S cumini was collected locally after proper identification. The identification of the plant was done by a botanist (Voucher Specimen No: RUBL-20425) from the Herbarium, Department of Botany, University of Rajasthan, Jaipur, Rajasthan, India. The pulp was removed from the fruit and the seeds were washed properly and shade dried; then the fruit was powdered in a mixture and the hydroalcoholic extract was prepared by refluxing with double distilled water (DDW) and alcohol (3:1) for 36 (12 × 3) hours at 40°C. The liquid extract was cooled and concentrated by evaporating its liquid contents. The prepared S cumini extract (SCE) was stored at a low temperature until further use. This extract was redissolved in DDW prior to oral administration in mice.
Experimental Design
A total of 40 animals were randomly divided into 5 groups to evaluate the anticarcinogenic potential of SCE against DMBA-induced skin carcinogenesis in mice. Hair on the interscapular region of the mice were clipped 3 days before the commencement of the experiment. Only mice in the resting phase of the hair cycle were considered for the study. The body weights of the animals were recorded weekly until the end of the experiment.
Group I (n = 8): vehicle treated (negative control)
Animals of this group received topical application of acetone (100 µL/mouse) on the shaven dorsal skin and DDW equivalent to SCE (100 µL/mouse/d) by oral gavage for 16 weeks.
Group II (n = 8): carcinogen treated (positive control)
Mice of this group were treated topically with a single dose of DMBA (100 µg/100 µL of acetone) over the shaven area of the skin. After 2 weeks, croton oil (1% w/v in acetone) was applied 3 times per week until the end of the experiment. This group received DDW equivalent to SCE (100 µL/mouse) by oral gavage for 16 weeks.
Group III (n = 8): SCE treated (experimental 1)
These experimental animals received the same treatment as in group II, along with SCE at a dose of 250 mg/kg body weight/animal/d, orally for 7 days before and 7 days after DMBA application.
Group IV (n = 8): SCE treated (experimental 2)
Animals of this group received the same treatment as in group II and were administered SCE (250 mg/kg body weight/animal/d) by oral gavage, starting from the time of croton oil treatment till the end of the experiment (ie, 16 weeks).
Group V (n = 8): SCE treated (experimental 3)
Animals of this group were administered SCE (250 mg/kg/body weight/animal/d) by oral gavage, starting from 7 days before DMBA application and continuing until the end of the experiment (ie, 16 weeks).and served as the periinitiation and postinitiation group.
Morphological Parameters
The following morphological parameters were studied:
tumor incidence—the number of mice carrying at least 1 tumor expressed as a percentage incidence;
tumor yield—the average number of papillomas per mouse;
tumor burden—the average number of tumors per tumor-bearing mouse;
diameter—the diameter of each tumor;
weight—the weight of the tumors of each animal at the termination of each experiment;
body weight—the weight of the mice as measured weekly;
average latent period—the lag between the application of the promoting agent and the appearance of 50% of tumors, calculated by multiplying the number of tumors appearing each week by the time in weeks after the application of the promoting agent and dividing the sum by the total number of tumors, that is, ΣFX/n,
where F is the number of tumors appearing each week, X the numbers of weeks, and n the total number of tumors; and
inhibition of tumor multiplicity—(total number of papillomas in carcinogen control − total number of papillomas in SCE-treated experimental animals) × 100/(total number of papillomas in carcinogen control).
Biochemical Study
Biochemical alterations were measured in animals of all the groups at the time of the termination of the experiment (ie, the 16th week). At the end of the 16th week, the animals were killed by cervical dislocation. The dorsal skin affected by tumors and liver was quickly excised and washed thoroughly with chilled saline (pH 7.4). It was then weighed and blotted dry. A 10% tissue homogenate was prepared from part of the sample (skin and liver) in 0.15 M Tris–KCl (pH 7.4), and the homogenate was then centrifuged at 2000 rpm for 10 minutes. The supernatant thus obtained was taken for estimation of lipid peroxidation (LPO) and reduced glutathione (GSH). The biochemical parameters estimated in the liver and skin of mice are described below.
Lipid peroxidation
The level of LPO was estimated spectrophotometrically by the thiobarbituric acid reactive substances method, as described by Ohkhawa et al. 16 Briefly, thiobarbituric acid (0.6%), sodium dodecyl sulfate (0.1%), and trichloroacetic acid (TCA; 20%) were added to 200 µL of the tissue homogenate (10%) prepared as described above. This mixture was heated for 60 minutes, cooled, and extracted with N butanol-pyridine (15:1); the optical density was recorded at 532 nm, and the contents were expressed as nmol/mg of tissue.
Reduced glutathione
The level of GSH was estimated as total nonprotein sulfahydryl group by the method of Moron et al. 17 The homogenate was immediately precipitated with 100 µL of 25% TCA, and the precipitate was removed after centrifugation. Free endogenous GSH was assayed in a total volume of 3 mL by the addition of 200 µL of 0.6 mM 5,5′ dithio-bis (2-nitrobenzoic acid) dissolved in 0.2 M phosphate buffer (pH 8.0) to 100 µL of the supernatant, and the absorbance was recorded at 412 nm using a UV-VIS Systronics spectrophotometer. Reduced GSH was used as a standard. The levels of GSH were expressed as micromoles per gram of tissue.
Catalase
CAT activity was assayed by the method of Aebi. 18 The change in absorbance was followed spectrophotometrically at 240 nm after the addition of H2O2 (30 mM) to 100 µL of the supernatant (10% of skin homogenate prepared in 50 mM phosphate buffer and centrifuged for 10 minutes) in 50 mM phosphate buffer (pH 7). The activity of the enzyme is expressed as U/mg of tissue, where U is micromoles of H2O2 disappearance per minute.
Proteins
Total proteins were estimated by the method of Lowry et al 19 using bovine serum albumin as a standard, and the level was expressed in milligrams per gram.
Superoxide dismutase
SOD was determined by the method of Marklund and Marklund 20 by quantification of pyrogallol auto-oxidation inhibition, and the results were expressed in units per milligram protein. One unit of enzyme activity was defined as the amount of enzyme necessary for inhibiting the reaction by 50%. Auto oxidation of pyrogallol in Tris–HCL buffer (50 mM, pH 7.5) was measured by increase in absorbance at 420 nm.
Vitamin C
For tissue ascorbic acid (vitamin C) determination, the fresh organs were weighed, homogenized in acetate buffer (20 mg/mL), extracted with cold 4% TCA, centrifuged, and filtered. Ascorbic acid was determined by the method of Roe and Kuether. 21
Statistical Analysis
The results are expressed as the mean ± standard error of the mean. The data from biochemical determinants were analyzed using the Student’s t test.
Results
The present study describes the anticancer efficacy of S cumini against DMBA-induced skin cancer in mice. All DMBA and croton oil treated mice had a visible rough granular surface on the shaved skin with varying degrees of erythema and sometimes with white plaque-like lesions. The number of tumor-bearing mice was recorded from the seventh week onward because tumors started to appear from this time in carcinogen-treated controls (group II). The cumulative number of papillomas after 16 weeks was 68 in the positive control group, which was reduced to 15, 21, and 8 in the animals treated with SCE in the periinitiation, postinitiation, and periinitiation and postinitiation groups, respectively (Figure 1).

Modulatory influence of the investigative dose(250 mg/kg body weight/d) of Syzygium cumini on cumulative number of papillomas of skin in mice
At the end of the experimental period (ie, after 16 weeks), in the DMBA and croton oil–treated mice (group II), the incidence of skin tumors was 100%; whereas in the DMBA and croton oil + SCE–treated groups (groups III, IV, and V), reduced number of such tumors was evident. The incidence of papilloma-bearing mice in groups III, IV, and V was 37.5%, 50%, and 25%, respectively (Figure 2), at the end of the experiment.

Modulatory influence of the investigative dose(250 mg/kg body weight/d) of Syzygium cumini on tumor incidence in skin of mice
The mean number of papillomas/tumors in tumor-bearing mice (tumor burden) was also significantly reduced by treatment with S cumini in comparison with the carcinogen control group. The maximum inhibition of tumor burden was recorded in group V, which received SCE throughout the experimental period. The tumor burden in the carcinogen-treated control group was 8.5, and the same was reduced to 5.0, 5.25, and 4.00 in groups III, IV, and V, respectively, after 16 weeks of treatment (Figure 3).
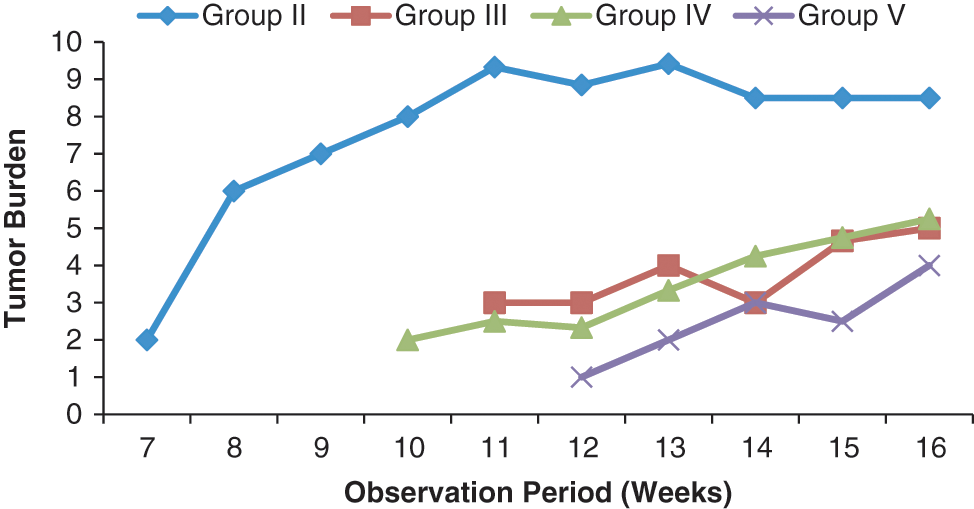
Modulatory influence of the investigative dose(250 mg/kg body weight/d) of Syzygium cumini on tumor burden in skin of mice
In the control group, the average number of papillomas per animal was 8.5, whereas in the SCE-treated groups (groups III, IV, and V), the corresponding numbers were 1.87, 2.62, and 1.00, respectively, after the 16th week of carcinogen (DMBA) administration (Figure 4). The average latency period in positive control mice was recorded as 7.88, whereas the same was found to be increased to 12.00, 11.14, and 12.625 in the animals of groups III, IV, and V, respectively (Figure 5). The maximum inhibition of multiplicity of papillomas occurred in the SCE-treated group V (Figure 6). At the end of the experiment (ie, at 16 weeks), the tumor size in SCE-treated animals was much smaller than that in carcinogen-treated animals (Table 1).

Modulatory influence of the investigative dose(250 mg/kg body weight/d) of Syzygium cumini on tumor yield in skin of mice

Modulatory influence of the investigative dose(250 mg/kg body weight/d) of Syzygium cumini on average latent period of tumor appearance in skin of mice

Modulatory influence of the investigative dose(250 mg/kg body weight/d) of Syzygium cumini on inhibition of tumor multiplicity in skin of mice
Modulatory Influence of the Investigative Dose of Syzygium cumini on Weight Gain Profiles, Tumor Size, and Tumor Weight

Treatment schedule of each group is specified in the Materials and Methods section.
The levels of LPO in the liver and skin of control and experimental animals are depicted in Figure 7. These were found to be significantly (P < .001) increased in group II cancer-bearing mice when compared with vehicle-treated animals (group I). SCE treatment resulted in a significant decrease in LPO levels of mice belonging to group III (P < .01), IV (P < .01; P < .05) and group V (P < .01; P < .001) when compared with the carcinogen-treated controls (group II).

Modulatory influence of the investigative dose (250 mg/kg body weight/d) of Syzygium cumini on tissue lipid peroxidation (LPO) in mice: statistical comparison—normal vs control; control vs experimental
Activity of GSH was found to be significantly reduced in the liver and skin of group II (P < .001) carcinogen-treated animals when compared with group I (negative control animals). These changes were reverted to near normal values on treatment with SCE in group III (P < .001; P < .01), group IV (P < .001; P < .01), and group V (P < .001; Figure 8).

Modulatory influence of the investigative dose(250 mg/kg body weight/d) of Syzygium cumini on tissue reduced glutathione (GSH) in mice: statistical comparison—normal vs control; control vs experimental; significance levels: *P ≤ .05,**P ≤ .01, ***P ≤ .001
Administration of DMBA (group II) significantly reduced (P < .001) the extent of enzymatic antioxidant enzymes, CAT, and SOD in group II in comparison to the vehicle-treated group (group I). In contrast, treatment with SCE elevated the levels of such enzymes in groups III (P < .001; P < .01), IV, and V (P < .001; P < .05) in comparison to positive controls (group II; Figures 9 and 10).

Modulatory influence of the investigative dose(250 mg/kg body weight/d) of Syzygium cumini on tissue catalase (CAT) in mice: statistical comparison—normal vs control; control vs experimental; significance levels: *P ≤ .05, **P ≤ .01, ***P ≤ .001
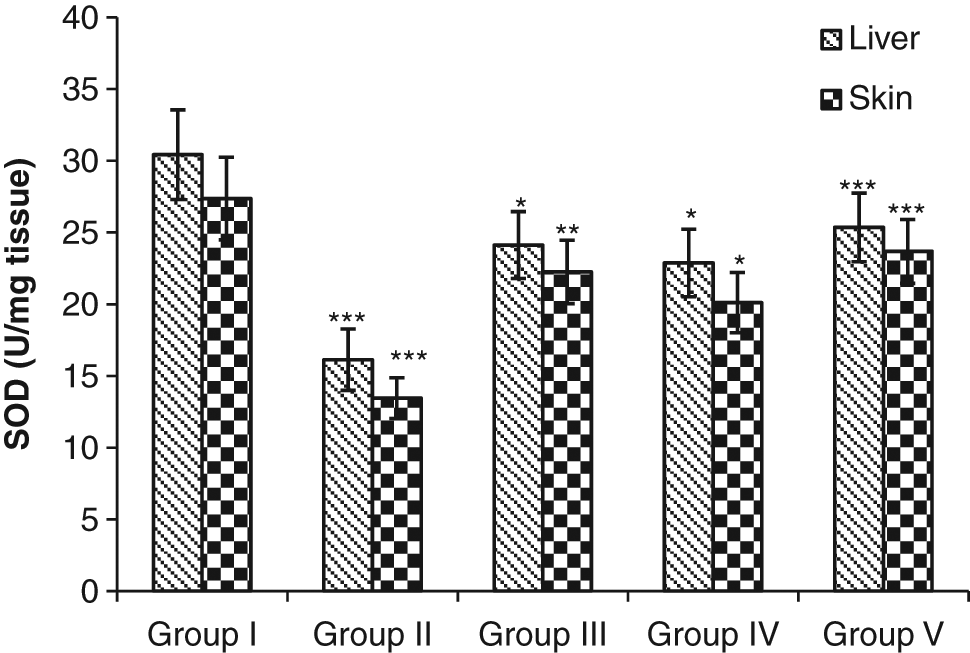
Modulatory influence of the investigative dose(250 mg/kg body weight/d) of Syzygium cumini on tissue superoxide dismutase (SOD) in mice: statistical comparison—normal vs control; control vs experimental; significance levels:*P ≤ .05, **P ≤ .01, ***P ≤ .001
S cumini treatment during periinitiation (group III), postinitiation (group IV), and periinitiation and postinitiation stages (group V) significantly (P < .001) elevated the level of total proteins in the liver and skin when compared with positive controls (group II; Figure 11).

Modulatory influence of the investigative dose(250 mg/kg body weight/d) of Syzygium cumini on total proteins in mice: statistical comparison—normal vs control; control vs experimental; significance levels: ***P ≤ .001
Vitamin C content in liver and skin was drastically lowered in the control group. On the other hand, administration of S cumini extract significantly enhanced the vitamin C content in the liver (P < .05, P < .01, and P < .001) as well as in the skin (P < .05, P < .01, and P < .001; Figure 12).
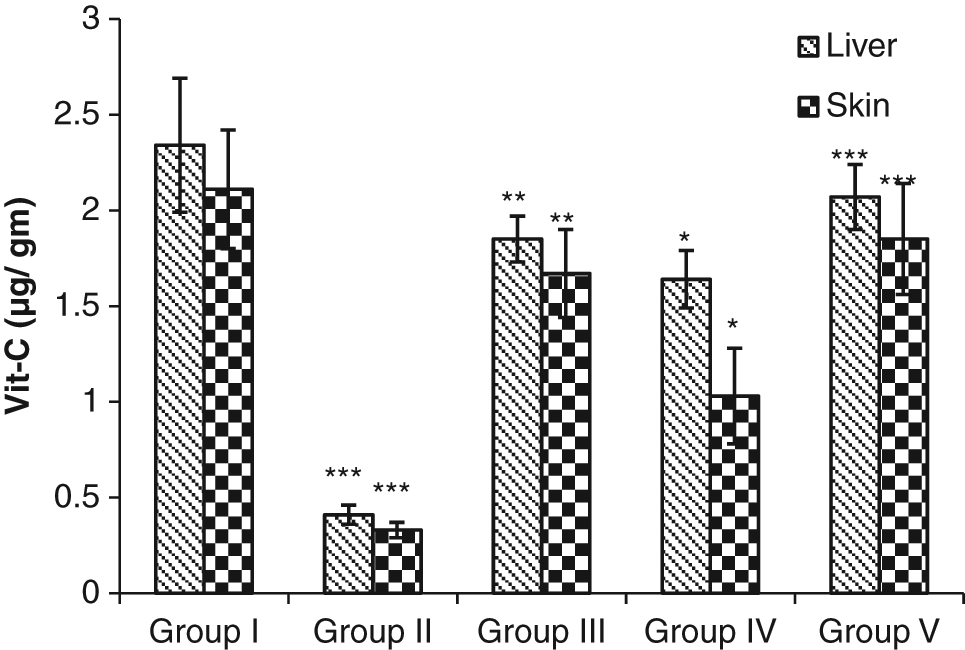
Modulatory influence of the investigative dose (250 mg/kg body weight/d) of Syzygium cumini on tissue vitamin C (Vit-C) in mice: statistical comparison—normal vs control; control vs experimental; significance levels: *P ≤ .05, **P ≤ .01, ***P ≤ .001
Discussion
The polycyclic aromatic hydrocarbon DMBA can act as a complete carcinogen or an initiator of mouse skin carcinogenesis.22,23 Oxidative stress is caused by an imbalance between the production of reactive oxygen species (ROS) and a biological system’s ability to readily detoxify the reactive intermediates or easily repair the resulting damage. All forms of life maintain a reducing environment within their cells. This reducing environment is preserved by enzymes that maintain the reduced state through a constant input of metabolic energy. Disturbances in this normal redox state can cause toxic effects through the production of peroxides and free radicals that damage all components of the cell, including proteins, lipids, and DNA. The production of ROS and the peroxidation of membrane lipids are well linked with the initiation of carcinogenesis, which further affects the normal biochemical process.24,25 Free radicals are involved both in the initiation as well as in the promotion stage of tumorigenesis, and their biochemical reactions in each stage of the metabolic process are associated with cancer development. Antioxidants are believed to help protect the body from free-radical damage. Antioxidant-based drugs/formulations for the prevention and treatment of complex diseases such as atherosclerosis, stroke, diabetes, Alzheimer’s disease, and cancer have appeared during the last 3 decades. 26 This has attracted a great deal of research interest in natural antioxidants.
MDA is an aldehyde that can be produced from free-radical attack on polyunsaturated fatty acids. Hence, it is of interest to assess MDA as a marker of oxidative stress. MDA is known to play a role in LPO and in the modulation of antioxidants during the progression of cancer. There is a natural balance between the number of free radicals generated in the body and the antioxidant defense system that scavenges them and thereby protects the body against pathogenesis. 27 It is evident from the results presented herein that the level of MDA was found to be elevated in the positive control animals, which is responsible for increased tumor incidence, tumor burden, and cumulative number of papillomas. In contrast, a reduced level of MDA was observed in S cumini–treated experimental groups, suggesting that S cumini could be a potent free-radical scavenger, which might be responsible for the decreased tumor incidence in such animals.
In addition to lipids, proteins are also affected by ROS. 28 Oxidation of protein is the result of tertiary structural alteration that causes protein aggregation. Decreased levels of total proteins in the liver and skin of animals in the carcinogen-treated control group (group II) clearly reflect the overproduction of free radicals or/and the inability of the antioxidant defense system to perform adequately during tumor growth. S cumini treatment significantly enhanced these enzyme levels, which might be responsible for the decrease in the activity of tumor promoters used in the present study.
SOD plays an important role in the antioxidant enzyme defense system. SODs convert superoxide radicals into hydrogen peroxide. 29 Because the activity of SOD was induced in mice by the S cumini extract treatment, the generation of ROS is inhibited, and the dismutation of super oxide radicals may well be accelerated by the catalyzing role of SOD. CAT, the activity of which was also augmented by S cumini treatment, helps in removing the hydrogen peroxide produced by the action of SOD. Indeed, SOD activity, along with that of CAT, explains the significant decrease in LPO seen in the present investigation, which is an indicator of oxidative stress that persists in the cell.
The current study demonstrates that S cumini extract can activate the antioxidant defense system following exposure to the carcinogen DMBA, which was found to result in decreased activity of the above-mentioned enzymes. The fruits of S cumini Skeels are edible and are reported to contain gallic acid, tannins, anthocyanins, and other components. 30 Most of the plant parts of Eugenia jambolana are used in the traditional system of medicine in India; it has promising therapeutic value because of its various phytoconstituents, such as glycosides (jamboline), ellagic acid, gallic acid, tannins, 31 fatty oil, steroids, flavonoids, triterpenes, 32 phenolics, monoterpenoids, 33 minerals, proteins, calcium, and vitamins. 34
Increase in the level of GSH by the chemopreventive action of flavonoids in mouse skin has also been reported. 35 It is thus the combined effect of modulating antioxidant enzyme(s), which may in turn lead to a shift in the intracellular oxidation/reduction balance and to a changed cell and organ sensitivity to induced tumorigenesis.
Ascorbic acid has multiple antioxidant properties, including an ability to regenerate α-tocopherol by reducing α-tocopheryl radicals at the surface of membranes. All these constituents may be responsible for the observed anticarcinogenic effects of S cumini extract in the present study.
Phenols are very important plant constituents in terms of their radical scavenging ability resulting from the presence of hydroxyl groups. 36 The phenolic content may contribute directly to the antioxidative action. 37 It has been suggested that polyphenolic compounds have inhibitory effects on mutagenesis and carcinogenesis in humans. 38
Many of the biological actions of flavonoids have been attributed to their powerful antioxidant properties. They can act in several ways, including direct quenching of ROS, chelation of metal ions, and regeneration of membrane-bound antioxidants.
Conclusions
In conclusion, the present investigation has demonstrated clearly that the S cumini seed can be used as a potential cancer chemopreventive agent by virtue of its efficacy in stimulating the antioxidant defense system. It protects against oxidative stress via the elevation of antioxidative defense enzymes while significantly reducing the specific activity of the level of LPO. All these effects considered together might result in the significant reduction of DMBA-induced papilloma genesis in mice at the periinitiation, postinitiation, and periinitiation and postinitiation periods. Since S cumini has shown no toxic effect at the tested doses, 39 it could well be applied in cancer chemoprevention to reduce the risk of cancer. However, investigation to isolate and identify the active principles and to elucidate the exact mechanism of action is warranted.
Footnotes
Acknowledgements
Grateful acknowledgement is due to the Indian Council of Medical and Research (Ref No 59/25/2005/BMS/TRM), New Delhi, India, for supporting this work.
The author(s) declared no potential conflicts of interest with respect to the authorship and/or publication of this article.
The author(s) received no financial support for the research and/or authorship of this article.
