Abstract
The aim of this study was to evaluate clinical effects of weekly (UD/week) and biweekly (UD/2-weeks) low-frequency ultrasound debridement (UD) on wound healing, healing time, wound area, granulation tissue, and transcutaneous oxygen pressure (TcPO2) in patients with diabetic foot ulcers (DFUs), compared to the standard of care (SoC). A pilot randomized clinical trial was conducted involving 30 patients with DFUs who received either UD/week (n = 10), UD/2-weeks (n = 11), or SoC (n = 9) every week over a 12-week treatment period. Healing rates at 6 months were 70% in the UD/week, 54.4% in the UD/2-weeks, and 33.3% in the SoC (P = .27). The healing times were 11 (IQR 7-19) weeks for UD/week, 18 (IQR 15-36) weeks for the SoC, and 24.5 (IQR 16-30) weeks for the UD/2-weeks group (P = .036). Wound area was reduced by 87.5 ± 18.5% in the UD/week, 68.6 ± 26.2% in the UD/2-weeks, and 38.9 ± 16.6% in the SoC (P = .014). Wollina wound scores were 7 (IQR 6-7) in the UD/week, 7 (IQR 6.5-7) in the UD/2-weeks, and 4 (IQR 3-6) in the SoC (P = .002). UD/week showed not significantly higher healing rates than the UD/2-weeks and SoC, but experienced significantly shorter healing time, a greater percentage of wound area reduction and higher Wollina wound score. These findings suggest that UD/week could be a valuable therapeutic option in clinical practice, particularly for patients in whom sharp debridement is not a viable option or who require an alternative approach to promote wound healing. This study is registered in ClinicalTrial.gov
Introduction
Diabetic foot ulcers (DFUs) are among the most serious complications associated with diabetes mellitus (DM). 1 Standard of care (SoC) for patients with DFUs requires a multifaceted approach, encompassing infection control, usage of pressure off-loading devices, PAD (Peripheral Arterial Disease) management, local wound care, diabetes metabolic regulation, and treatment of comorbidities. 2 In recent years, significant advances have been made in the development of new technologies and therapies for local wound care. To structure local wound care, a group of experts developed the MOIST concept (Moisture, Oxygen, Infection, Supporting, and Tissue). 3
Debridement, a critical aspect of local wound care that involves the removal of both viable and non-viable components, is essential for effective tissue management. The primary goal of this technique is to minimize the presence of these components using the most effective methods with minimal side effects, thereby optimizing healing and preventing infections. These components include necrotic tissue, slough, biofilms, microorganisms, and foreign materials. 4 The International Working Group on the Diabetic Foot (IWGDF) identifies sharp debridement as the gold standard for local DFUs treatment. 5 However, these debridement methods require specific clinical skills due to the potential for extensive wound bed damage with bone, joint tissue, or ligament exposure. Furthermore, they are not recommended for patients with poor vascular status or wounds with poorly controlled pain.6,7 In areas that cannot be selectively debrided, such as tendons and nearby delicate structures, or when the wound size does not reduce by 50% after 4 weeks of standard treatment, advanced therapies like low-frequency contact ultrasound debridement (LFCUD) are recommended. 8
LFCUD generates sound waves that are transmitted through a fluid medium to the wound surface. It is here that the effects of micro-streaming and cavitation occur. Cavitation refers to the formation and oscillation of gas microbubbles in a fluid medium. When this phenomenon happens, microbubbles undergo various process of expansion, contraction, and implosion. This sequence aids in the removal of non-viable tissue, the disruption of biofilms, and the destruction of bacterial walls, ultimately decreasing the bacterial load on the wound surface. In a similar vein, micro-streaming refers to the movement of interstitial fluids driven by the vibrations of an ultrasound device, this phenomenon unexpectedly impacts cell membrane permeability and the activity of second messengers, promoting enhanced protein and collagen synthesis, mast cell degranulation, and increased production of growth factors. Together, these effects stimulate neo-angiogenesis and activate fibroblasts within the wound bed.9–11
Previous studies have demonstrated a positive impact of LFCUD on healing rates, significant reduction of wound area, bacterial load, and healing time of wounds.12–14 Nevertheless, the differing effects of LFCUD on DFUs when applied weekly or biweekly have not yet been examined. Therefore, building upon previous findings, this pilot randomized clinical trial aims to conduct an intention-to-treat (ITT) analysis to assess the clinical effects of weekly and fortnightly LFCUD on wound healing, healing time, wound area, wound bed granulation tissue, pain, and transcutaneous oxygen pressure (TcPO2) in patients with DFUs.
Materials and Methods
Study Design
We conducted an open-label, pilot randomized clinical trial featuring three treatment arms, involving 30 patients with DFUs who were admitted to the specialized diabetic foot unit from April 2021 to October 2023. The study protocol received approval from the local Ethics Committee and is registered in ClinicalTrial.gov.
Participants
The inclusion criteria required patients to be over 18 years old with type 1 or 2 diabetes, an HbA1c value ≤ 10% (85.8 mmol/mol) within the 3 months preceding the study, and the presence of DFUs with a duration of 1–24 months and a wound area of 1–30 cm2 at the time of inclusion. Wound stages, as per the University of Texas Wound Classification System, needed to fall within categories IA, IIA, IB, IIB, IC, IIC, ID, or IID. As per the IWGDF/Infectious Diseases Society of America (IDSA) Classification, the presence or severity of infection needed to be defined as grade 1 (uninfected), grade 2 (mild infection), or grade 3 (moderate infection).15,16 Additional eligibility criteria comprised an ankle-brachial index (ABI) > 0.9 or ABI ≤ 0.9 with Toe-Brachial Index (TBI) > 0.7, or TBI ≤ 0.7, and an ankle systolic blood pressure (ASBP) ≥ 70 mm Hg, or toe systolic blood pressure (TSBP) ≥ 50 mm Hg. 17
Patients were excluded from the study if they were diagnosed with critical limb ischemia, 17 had a life expectancy of less than 6 months, or had a clinical suspicion of diabetic foot osteomyelitis. Additionally, women who were pregnant or lactating, along with patients diagnosed with hepatitis or human immunodeficiency virus (HIV) infection, or other local/systemic conditions that could impair tissue repair were also excluded.
Clinical Assessment
At baseline, the clinical characteristics of patients enrolled in the study were consistently assessed by the same clinician. From the patient's medical records, we recorded nephropathy, cardiovascular, and retinopathy statuses, as well as hypertension, hypercholesterolemia, and smoking history, along with any previous ulcerations or amputations. We collected data related to the type and average duration of diabetes, along with glucose levels and glycated hemoglobin values from the most recent blood test.
A neurological assessment was conducted using a Horwell biothesiometer (METEDA S.r.l., San Benedetto del Tronto, Italy) and a Semmes-Weinstein 5.07/10 g monofilament (Novalab Iberica, Spain). Patients who failed to feel one of the two tests were diagnosed with diabetic neuropathy. 18 Vascular screening was based on distal pulse palpation, ABI, TBI, and TcPO2. Brachial systolic blood pressure and ASBP were evaluated using an 8-MHz manual Doppler (Huntleigh Healthcare Ltd, Cardiff, UK). TSBP was measured by digital plethysmography (Systoe, Atys Medical, Madrid, Spain) and TcPO2 using the TCM4 measuring device in the dorsal midfoot (Radiometer, Copenhagen, Denmark). 17 PAD was diagnosed if the patient exhibited an absence of distal pulses and an ABI < 0.9. For patients with an ABI within the normal range (0.9-1.3) or >1.3 (arterial calcification), and absence of distal pulses PAD was considered if TBI < 0.7 and TcPO2 < 30 mm Hg. 19
In this study, DFUs were graded using the University of Texas Wound System 15 and SINBAD classifications. 20 DFU conditions were assessed based on location, duration of wound evolution, type of tissue presents on the wound bed, volume of wound exudate, peri-wound skin conditions, and wound area, respectively. Wound areas were measured using digital planimetry with a Visitrak device (Smith & Nephew, Hull, UK).
Intervention
All patients who fulfilled the inclusion criteria were randomly assigned to one of three treatment groups. Patients in the control group were managed solely with SoC in the management of DFUs. On the other hand, patients in the weekly low-frequency ultrasound debridement group (UD/week) and the biweekly low-frequency ultrasound debridement group (UD/2-weeks) received ultrasound debridement (UD) additionally to SoC, applied weekly and fortnightly, respectively.
Participants were monitored during a 12-week treatment period, which entailed one visit per week, or until the wounds healed. Following treatment period, patients underwent a follow-up period, completing a total of 6 months from the study initiation, at which time the healing rate was evaluated. During each visit, we observed the DFUs after debridement, considering the wound bed tissue, wound exudate levels, condition of the peri-wound skin, and wound area. We utilized sterile saline weekly to cleanse all wounds before evaluation.
Standard of Care (SoC)
All patients received a SoC for DFUs management in line with the IWGDF guidelines. 2 This care incorporated careful, sharp debridement of tissue, was carried out by a clinician expert in the management of DFUs and performed with scalpel as necessary to remove non-viable tissue from the wound bed and to eliminate peri-wound callus and maceration. Additionally, we used moist wound dressings for wound care, off-loading devices, and controlled infections as necessary. The chosen off-loading devices varied with wound location, deploying a heel relief post-surgical shoe for rearfoot wounds, a removable knee-high device for forefoot and midfoot plantar wounds, and a post-surgical shoe for all other wounds. For infection control, patients presenting with mild or moderate infection received empirical antibiotics during the treatment period in line with IWGDF/ IDSA guideline recommendations, 16 pending the availability of deep tissue culture results. Upon obtaining the tissue culture results, we adjusted the antibiotic treatment in consideration of the cultured bacteria.
Low-Frequency Contact Ultrasound Debridement (LFCUD)
Patients in the UD/week group received LFCUD every week for a 6-week treatment period, whereas those in the UD/2-weeks group received LFCUD at weeks 0, 2, 4, and 6 of the follow-up. After the 6-week treatment period, DFUs were managed following the standard of wound care previously described.
In both the UD/week and UD/2-weeks groups, the LFCUD of DFUs was carried out using a Sonoca 185 contact device (Söring GmbH, Germany) in the same room and by the same experienced clinician, who has more than 5 years of experience handling the UD device. The LFCUD system includes a handpiece equipped with various sonotrodes (double ball, hoof, and spatula) that generate a low-frequency ultrasound at 25 kHz. The chosen sonotrode depends on the wound depth, the wound edges, and the type of tissue on the wound bed. The technique used with the sonotrodes involved sliding with rotation and milling. The sonotrode would come into contact with the wound, removing and lifting unwanted tissue mechanically with the help of the technique and instrument. The equipment settings were as follows: the saline flow rate (irrigation) was adjusted to 60%, the ultrasound intensity (amplitude) was set to 40%, and the treatment time was 20 s per cm2. 21
Outcome Measures
Primary Outcomes
The primary outcome measures included, first, wound healing, defined as complete epithelialization of the DFUs, confirmed at least 2 weeks after wound closure, but additionally healing rate of each treatment group was calculated (n° of healed patients/ n° of patients per group × 100). 22 Second was the time to healing, which is defined as the time from the day of ulcer inclusion in the study to wound healing, measured in weeks. The third measure was the percentage of wound area reduction, calculated by comparing the wound area at week zero to the wound area at week 12.
Secondary Outcomes
The secondary outcome measures included the presence, quality, and consistency of wound bed granulation tissue, assessed using a validated wound scoring system known as the Wollina wound score, with scores ranging from a minimum of zero to a maximum of 7. 23 Another secondary outcome was the variation in TcPO2 value between week 0 and week 12, intended to evaluate the effect of the LFCUD on this microvascular test. Additionally, during each visit, patient pain was assessed and scored using a numerical pain rating scale from zero to 10, where zero meant the absence of pain, and 10 represented maximum pain. 24
Blinding and Randomization
The randomization process was executed using a simple randomization model with numbers randomly generated by the Epidat version 4.1 software package (Consellería de Sanidade, Xunta de Galicia, Spain). Patients were randomized into three treatment groups by a researcher who was unaware of the participants’ identities. Regarding blinding, none of the participants, care providers, or outcome adjudicators were blinded to the interventions after assignment.
Statistical Analysis
All statistical analyses were conducted following the ITT principle using the software package, SPSS version 25.0 (IBM SPSS Statistics for Macintosh, Version 25.0. Armonk, NY, USA: IBM Corp.). Qualitative variables were described using frequency distributions and percentages. To identify differences in categorical variables, the chi-square test for independent samples or the McNemar test for related samples was performed.
The assumption of normality for all continuous variables was confirmed using the Shapiro-Wilk test. Variables with a normal distribution (Shapiro-Wilk test with P ≥ .05) were reported as means and standard deviations (SD), whereas non-normally distributed variables (Shapiro-Wilk test with P < .05) were reported as medians and interquartile ranges (IQR). To compare normally distributed quantitative variables for independent samples, the Student's t-test and the analysis of variance (ANOVA) test were performed. In the case of non-normally distributed variables, the Wilcoxon-Mann Whitney test and the Kruskal-Wallis test for independent samples, as well as the Wilcoxon test for related samples, were implemented. When all groups were analyzed effect size was calculated by eta-squared for the ANOVA test and by epsilon-squared for the Kruskal-Wallis test, whereas when two-by-two groups and intragroup were analyzed, effect size was given by the relative risk for the categorical variables, by medians difference for the non-normally distributed variables and by the Coheńs d for the normally distributed variables.
Additionally, a survival analysis was executed to examine the time required for DFUs to heal during the follow-up period. Differences in survival between the treatment groups were assessed using the log-rank test and demonstrated by Kaplan-Meier curves. To further estimate the effect of treatment on wound healing, a Cox proportional hazards model was applied to calculate the Hazard Ratio (HR) for each treatment group, adjusting for potential confounders. Statistical significance was established at P < .05 with a 95% confidence interval.
Sample Size
The sample size was calculated to include 20 patients per treatment group, ensuring a an α level of 0.05, a β level of 0.2, and an anticipated drop-out rate of 20% using the GRANMO Sample Size Calculator version 7.12 Online (Municipal Institute of Medical Research, Barcelona, Spain). Considering evidence from the literature suggests that the sample size of a pilot study should be 10% of the total sample, a pilot randomized clinical trial will be conducted with half of the intended sample (n = 30). 25
Results
This study started in April 2021 and the follow-up period ended in October 2023, during which time forty-three patients were selected as potential participants, of whom 30 were included in the study. These individuals were randomly assigned to one of three different treatment groups: the control group (n = 9), UD/week group (n = 10), and the UD/2-weeks group (n = 11). A flow diagram of the course of the study can be found in Figure 1.
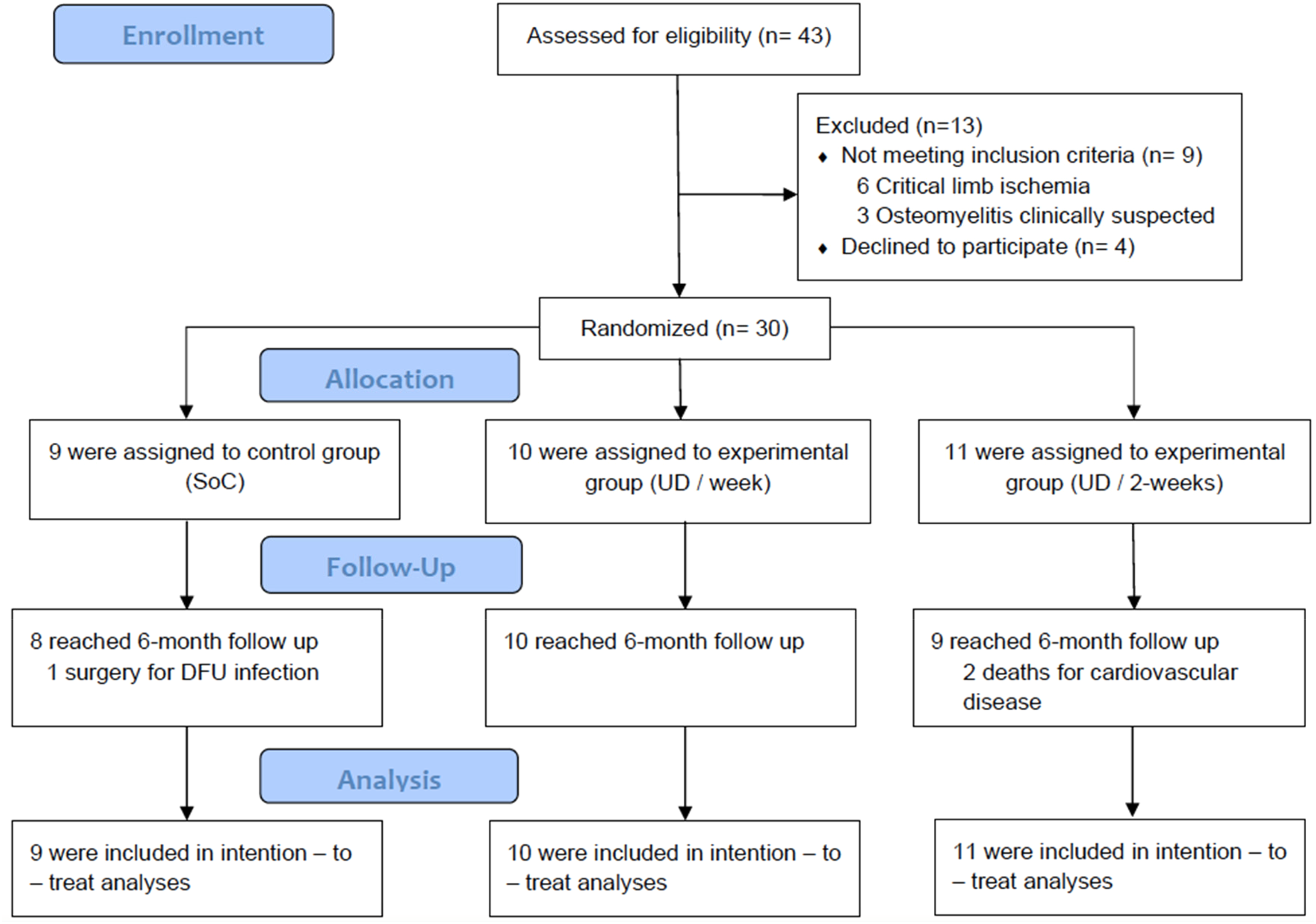
Study flow diagram.
The demographic characteristics, diabetic and foot complications, along with the vascular status of patients at baseline and the characteristics of DFUs of participants included in each treatment group are summarized in Table 1. Upon comparing the data, we noted no significant differences between the treatment groups concerning diabetic complications, location, and properties of the ulcers.
Baseline Characteristics of the Patients and the DFUs.
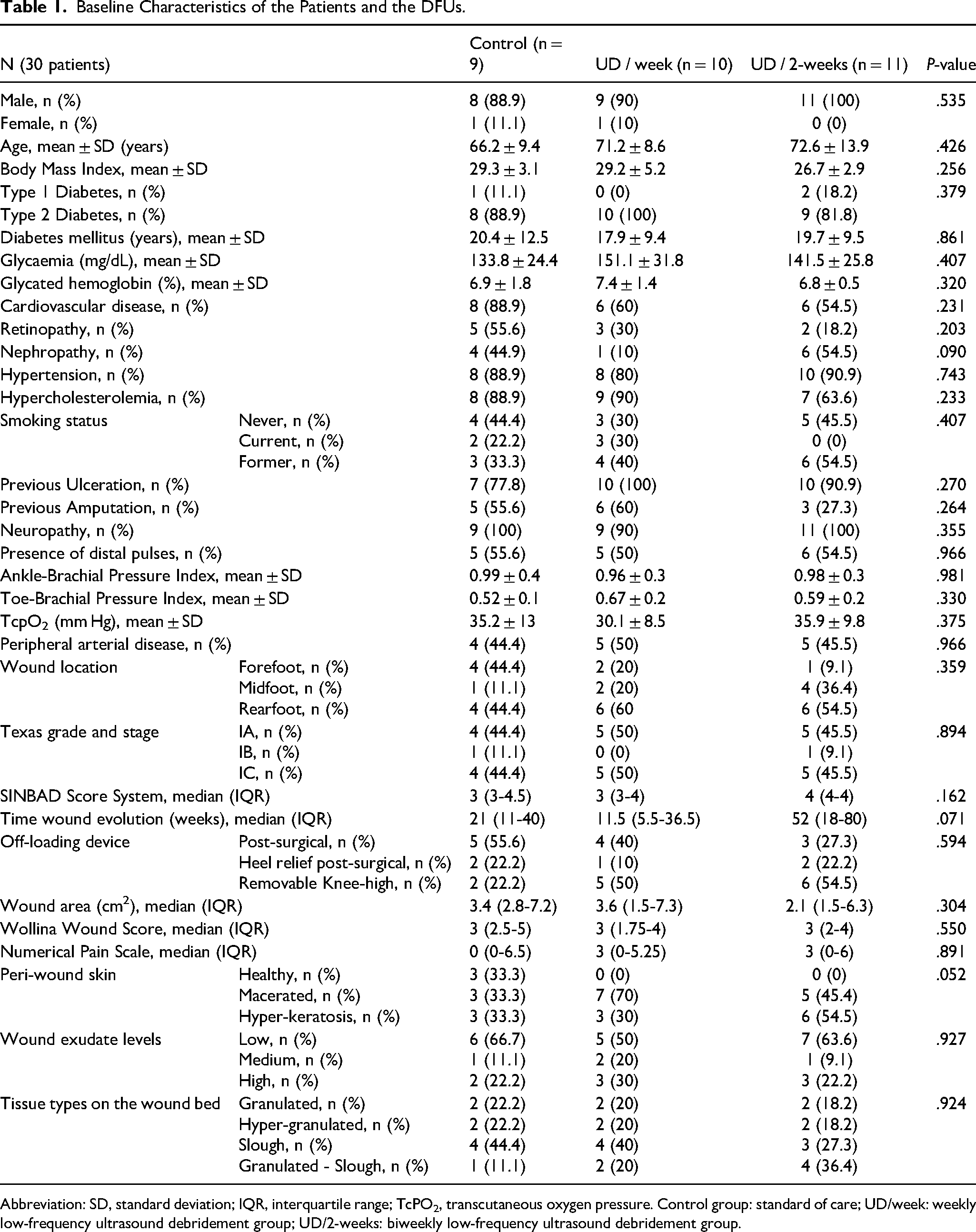
Abbreviation: SD, standard deviation; IQR, interquartile range; TcPO2, transcutaneous oxygen pressure. Control group: standard of care; UD/week: weekly low-frequency ultrasound debridement group; UD/2-weeks: biweekly low-frequency ultrasound debridement group.
In the analysis of outcome measures for the three treatment groups, we noted the following results after a 6-month follow-up: the healing rates of DFUs were higher in the UD/week group (seven patients [70%]) compared to the UD/2-weeks group (six patients [54.4%]) and the control group (three patients [33.3%]). However, no significant differences were observed between the groups (P = .27). Regarding the healing time, the survival analysis of the time to healing of DFUs showed significantly different survival curves when assessing the probability of wound healing. This was confirmed by the log-rank test, as displayed in the Kaplan-Meier curves of the different study groups (Figure 2). The time to healing was less in the UD/week group, with a median of 11 (IQR 7-19) weeks, compared to 24.5 (IQR 16-30) weeks in the UD/2-weeks group, and 18 (IQR 15-36) weeks in the control group (P = .036).
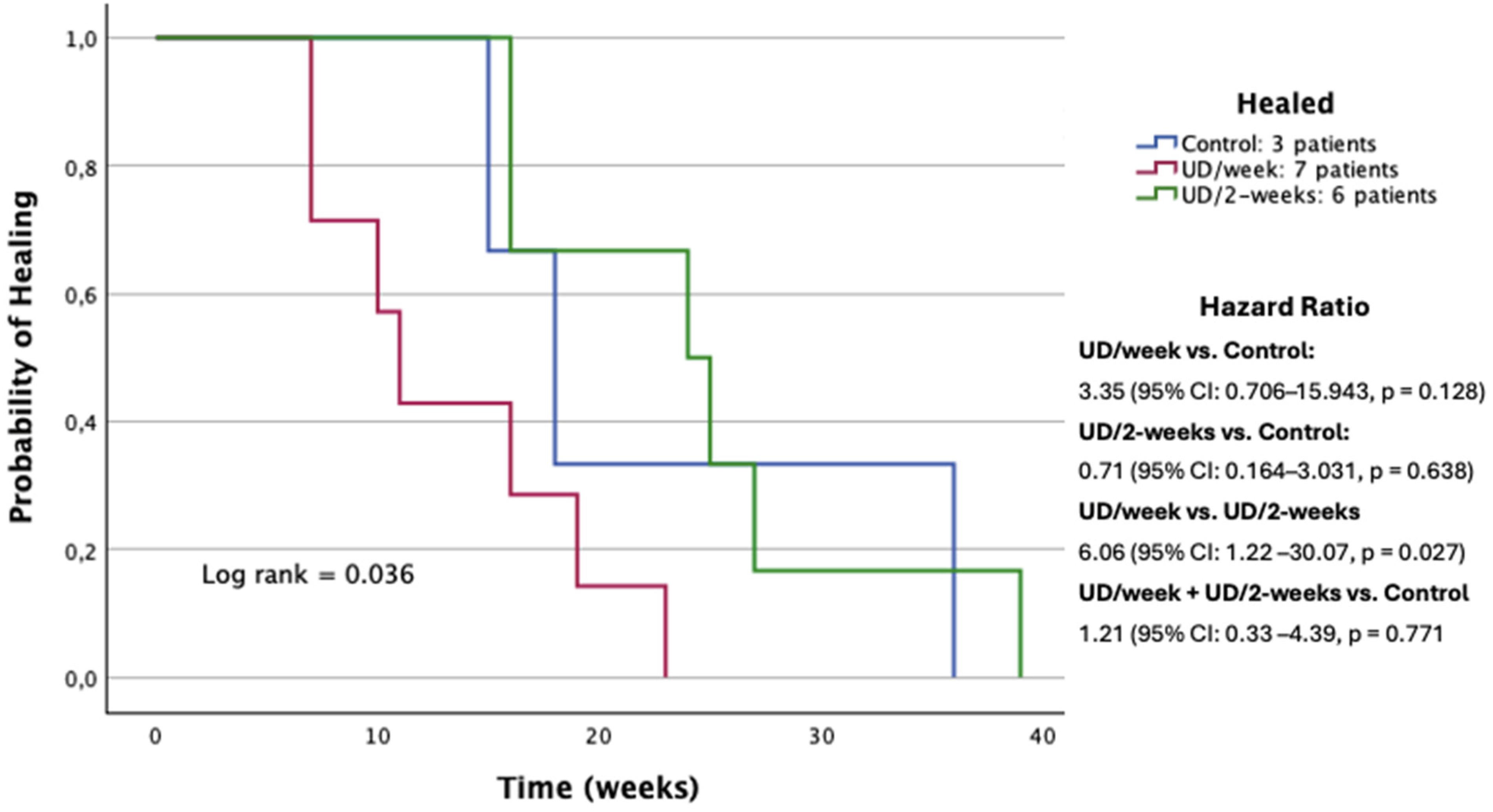
Kaplan-Meier survival curves. Time to the healing of DFUs by study group.
Furthermore, the Cox proportional hazards model indicated that patients in the UD/week group had a higher probability of wound healing compared to the SoC group, with a HR of 3.35 (95% CI: 0.706-15.943, P = .128), although this difference did not reach statistical significance. In contrast, the UD/2-weeks group showed a lower probability of healing than the SoC group, with an HR of 0.71 (95% CI: 0.164-3.031, P = .638), also without statistical significance.
In addition, after the 12-week treatment period, the UD/week group exhibited a larger percentage of wound area reduction (87.5 ± 18.5%) as compared to the UD/2-week group (68.6 ± 26.2%) and the control group (38.9 ± 16.6%). Significant differences were observed between these groups (P = .014), and effect size calculated by eta-squared, was 0.279 (95% CI: 0.13-0.480), indicating a large effect. Wollina wound scores also improved; the median score in the UD/week group was 7 (IQR 6-7), in the UD/2-week group it was 7 (IQR 6.5-7), and in the control group, it was 4 (IQR 3-6). Notably, significant differences were detected among the different treatment groups (P = .002), and effect size calculated by epsilon-squared, was 0.359 (95% CI: 0.275-0.469), suggesting a large treatment effect. As for the TcPO2 values at week 12, no significant differences were noted, with the UD/week, UD/2-week, and control groups having values of 34 ± 10, 41.7 ± 9.9, and 34 ± 12.3 mm Hg respectively (P = .237). The numerical pain rating scale showed a decrease in the study groups. It was 0 (0 for all patients) in the UD/week group, 0 (0 for all patients) in the UD/2-week group, and 1 (IQR 0-4.75) in the control group. Despite this decrease, no statistically significant differences were found among these groups (P = .20).
Table 2 presents the outcome measures derived from a comparison of the treatment groups two-by-two after a 12-week treatment period. Statistically significant differences were observed in terms of both healing time and wound area reduction percentages; these differences were seen when comparing the UD/week to the UD/2-weeks, with P-values of .022 and .04 respectively, indicating favor towards the once-weekly group. Additionally, higher values were observed both for the Wollina wound score (P = .004 for the UD/week group and P = .004 for the UD/2-weeks group) and the percentage of wound area reduction (P = .005 for the UD/week group and P = .058 for UD/2-weeks group) in both the once-weekly and bi-weekly treatment groups compared to the control group.
Analysis of Outcome Measures in Two-by-Two Groups.
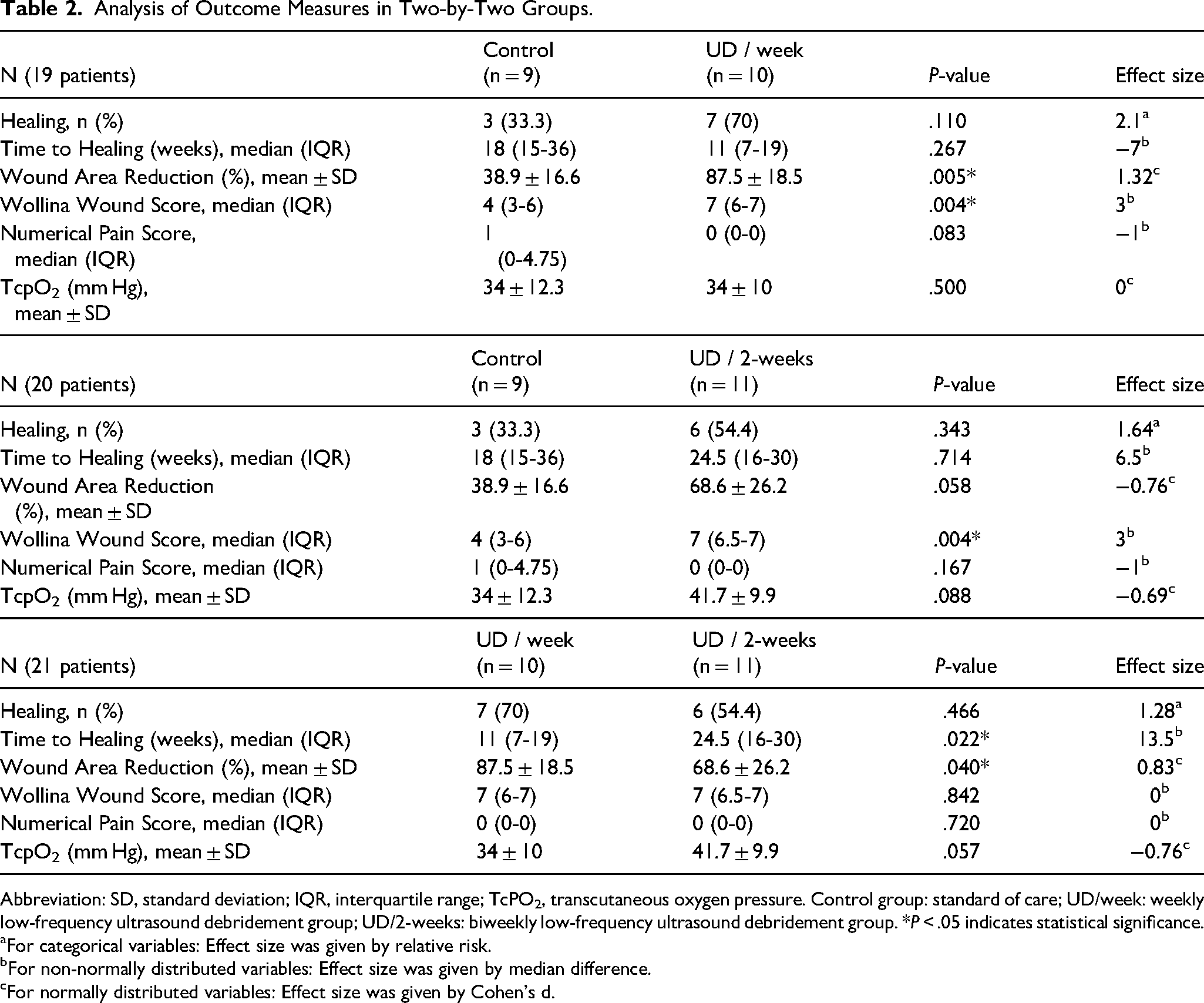
Abbreviation: SD, standard deviation; IQR, interquartile range; TcPO2, transcutaneous oxygen pressure. Control group: standard of care; UD/week: weekly low-frequency ultrasound debridement group; UD/2-weeks: biweekly low-frequency ultrasound debridement group. *P < .05 indicates statistical significance.
For categorical variables: Effect size was given by relative risk.
For non-normally distributed variables: Effect size was given by median difference.
For normally distributed variables: Effect size was given by Cohen's d.
Table 3 presents the outcome variables for each treatment group at the onset (week 0) and the conclusion of the treatment phase (week 12). The results reveal statistically significant disparities in the UD/week and UAW/2-week groups, but not in the control group.
Analysis of outcome measures intragroup.
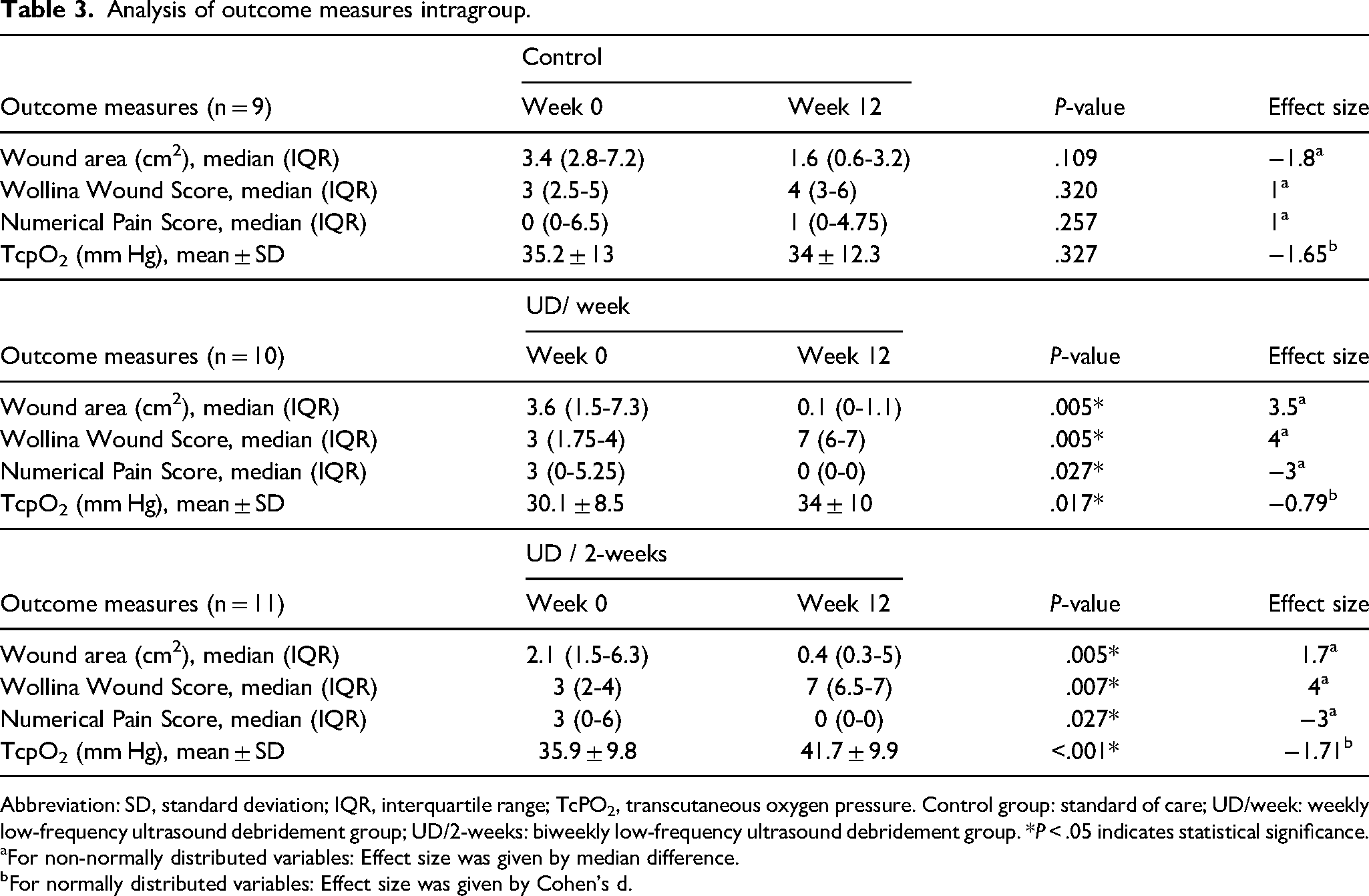
Abbreviation: SD, standard deviation; IQR, interquartile range; TcPO2, transcutaneous oxygen pressure. Control group: standard of care; UD/week: weekly low-frequency ultrasound debridement group; UD/2-weeks: biweekly low-frequency ultrasound debridement group. *P < .05 indicates statistical significance.
For non-normally distributed variables: Effect size was given by median difference.
For normally distributed variables: Effect size was given by Cohen's d.
Discussion
In comparing the three study groups, our primary results indicate that after a 6-month follow-up, the healing rates were 70% in the UD/week group, 54.4% in the UD/2-weeks group, and 33.3% in the SoC group; however, no significant differences were found between the study groups (P = .27). Similarly, Amini et al 26 conducted a randomized clinical trial (RCT) involving 40 patients, reporting no differences in healing rates between ultrasound therapy (60%) and SoC (55%) in treating DFUs. Likewise, Lázaro-Martínez et al 12 found no significant differences in healing rates between the UD group (85.1%) and SoC (83%) in a RCT involving 51 patients with neuroischaemic DFUs. Nevertheless, recent meta-analyses from Liu et al 27 and Chen et al 28 identified statistically significant differences in DFUs healing rates when comparing UD versus standard wound care. As for time to healing, our survival analysis of the healing time for DFU revealed significantly distinct survival curves upon probability analysis of wound healing using the log-rank test. The UD/week group required the least amount of healing time, with a median time of 11 (IQR 7-19) weeks, followed by the control group with a median time of 18 (IQR 7-19) weeks, and the UD/2-weeks group, with a median time of 24.5 (IQR 16-30) weeks (P = .036). In fact, Liu et al 27 reported a significant difference in healing time for DFUs while comparing ultrasound's effect with SoC. Regarding the outcome measures evaluated after a 12-week treatment period in our study, the percentage of wound area reduction was significantly higher in the ultrasound groups (UD/week [87.5 ± 18.5%] and UD/2-weeks [68.6 ± 26.2%]) compared to the SoC group ([38.9 ± 16.6%], P = .014), echoing the findings of previously published meta-analyses.14,27
Wollina wound scores also improved across all treatment groups, with significant differences observed between them (P = .002). This contrasts with the findings from Lázaro-Martínez et al's RCT, 12 which did not reveal any statistically significant differences between UD and sharp debridement. As far as we know, our RCT is the first to assess the impact of LFCUD on TcPO2 values and pain measured on a numerical rating scale. TcPO2 values at week 12 increased in the ultrasound groups (UD/week [34 ± 10 mm Hg] and UD/2-weeks [41.7 ± 9.9 mm Hg]) but not in the control group (34 ± 12.3 mm Hg). However, there were no reported differences between groups (P = .237). In terms of the numerical pain rating scale, it decreased in the UD/week group (0 [0-0]) and UD/2-weeks group (0 [0-0]), but not in the control group (1[0-4.75]), though no statistically significant differences were found between them (P = .20).
When comparing the treatment groups two-by-two, it was observed that there were no significant differences between the ultrasound groups (UD/week and UD/2-weeks, P = .466) and the control group concerning healing time (P = .11 and P = .343). However, significant differences emerged when the ultrasound groups (UD/week and UD/2-weeks) were compared with each other, favoring the UD/week group (11 [7-19] weeks vs 24.5 [16-30], P = .022). Regarding the percentage of wound area reduction, the UD/week group showed a significantly higher percentage (87.5 ± 18.5%) compared to both the control group (38.9 ± 16.6%, P = .005) and the UD/2-weeks group (68.6 ± 26.2%, P = .040). However, no significant difference was noted when comparing the UD/2-weeks group versus the control group (68.6 ± 26.2% vs 38.9 ± 16.6%, P = .058).
As for the Wollina wound score, both the UD/week group and the UD/2-weeks group, with median scores of 7 (IQR 6-7) and 7 (IQR 6.5-7) respectively, showed enhanced granulation tissue formation compared to the control group with a median of 4 (IQR 3-6), yielding statistically significant differences (P = .004). This outcome can be attributed to the effect of LFCUD on cell proliferation, which induces a significant augmentation of collagen and fibroblasts at the wound site, compared to the SoC. 12 However, no remarkable differences were observed in the Wollina wound scores when comparing the ultrasound groups.
Upon comparing both the TcPO2 values and the numerical pain rating scale between the three groups (UD/week, UD/2-weeks, and the control group), the results did not show any statistical differentiation (Table 2).
As the first RCT to examine the clinical effect of weekly and biweekly LFCUD in patients with DFUs, it is not feasible to establish comparisons with other similar studies conducted in the past.
Finally, when analyzing intragroup outcomes at baseline and the end of this RCT, it was observed that in the control group, there was a reduction in the wound area and an increase in the Wollina wound score. Similar results were obtained in terms of the numerical pain rating scale and TcPO2 values, with no statistical differences observed between week 0 and week 12 of the study. However, in both the UD/week group and UD/2-weeks group, there was a significant decrease in the wound area and the numerical pain rating scale, as well as a significant increase in the Wollina wound score and TcPO2 values (Table 3). The higher TcPO2 values can be explained by the effect of LFCUD on the increased production of growth factors, and enhanced proliferation of endothelial cells, and cytokines. This results in an increased density of microvessels and thus neo-angiogenesis, as previous studies have shown.11,12,29
The primary limitations of this RCT were its small sample size and the lack of blinding among participants and care providers, which might introduce bias in the outcome of the varying treatments. Additionally, a multivariate analysis was not performed to adjust for potential confounding factors, which may have influenced the observed differences between groups. As previously discussed, this is the first RCT to evaluate the comparative clinical impacts of weekly versus fortnightly LFCUD administration and SoC in patients suffering from DFUs over a 6-month tracking period.
Furthermore, this research suggests that weekly LFCUD could serve as an effective alternative to sharp debridement (SoC) when it is contraindicated or unavailable for patient use. To solidify our conclusions, future studies should involve larger sample sizes and blind treatments for investigators, and, where possible, for patients. Moreover, this study did not include a cost-effectiveness analysis, which would be valuable in determining the clinical efficacy of ultrasound debridement in routine clinical practice. Future research should incorporate economic evaluations to reinforce the clinical and economic effectiveness of this technology.
Conclusion
After a 6-month follow-up, patients in the UD/week group demonstrated statistically non-significant tendency to higher healing rates compared than those in the UD/2-weeks and SoC groups. However, the time to healing of DFUs was notably shorter in the UD/week group than in the UD/2-weeks and SoC groups. Following the 12-week treatment period, the wound area reduction percentage was greater in the UD/week group than in the UD/2-weeks and control groups, with significant differences between the study groups. Furthermore, concerning the Wollina wound score, a significantly higher score was observed when comparing the UD/week group versus the SoC group. However, similar scores were observed when comparing the UD/week group to the UD/2-weeks group, with no significant differences noted between them. Both the numerical pain rating scale and TcPO2 showed no significant changes when comparing the UD/week group to the UD/2-weeks group and the SoC group. Nevertheless, significant differences were detected when comparing the numerical pain rating scale score and TcPO2 values within the UD/week group and the UD/2-weeks group at baseline and at the end of the study.
From a clinical perspective, these results suggest that UD/week may serve as a beneficial adjunct therapy in the management of DFUs. The significant reduction in healing time and wound area emphasizes its potential as an effective alternative for patients who are not candidates for sharp debridement or require a less invasive approach. The implementation of UD/week in clinical practice could improve wound healing outcomes and potentially reduce complications associated with delayed healing. Future studies with larger sample sizes and cost-effectiveness analyses are needed to validate these findings and support their integration into standard wound care protocols.
Footnotes
Acknowledgments
None declared.
CRediT Authorship Contribution Statement
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Ethics Statement
The study was conducted according to the guidelines of the Declaration of Helsinki and approved by the Ethics Committee of Hospital Clínico San Carlos, Madrid, Spain (protocol code [C.P.-C.I. 21/596-E] and date approval [09/23/2021]) and is registered in ClinicalTrial.gov (Registration no.: NCT04716790).
