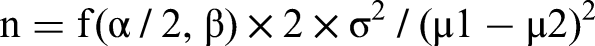Abstract
Objective
To measure the effect of neuromuscular stimulation of the common peroneal nerve on the microvascular blood flow within the wound bed of diabetic foot ulcers.
Research Design and Methods
13 patients with diabetic foot ulcers (11 neuropathic, 2 neuroischemic) were recruited. Microvascular flux and pulsatility were measured in real time in the wound bed, and at the wound edge, using Laser Speckle Contrast Imaging (Moor FLPI-2; Moor Instruments Ltd, Axminster, United Kingdom). Changes from baseline were measured when the leg muscle pump was activated by 1 Hz neuromuscular stimulation of the common peroneal nerve, using a wearable device (geko, Firstkind Ltd Daresbury, United Kingdom).
Results
In the 11 neuropathic ulcers, significant increases were seen in all microvascular parameters. Wound bed flux increased by 36% (95%CI 11%-68%, P = .002), and peri-wound flux increased by 92% (95%CI 46%-160%, P = .001). Pulsatility in the wound bed increased by 183% (95%CI 61%-517%, P = .005), while pulsatility in the peri-wound increases by 359% (95%CI 264%-455%, P = .001). Additionally, an increase in flux and pulsatility was observed in the neuroischemic ulcers.
Conclusions
Neuromuscular stimulation of the common peroneal nerve presents a mode of activating the leg muscle pump without additional pressure insult to the foot and has potential to support the microcirculation during wound-healing, effectively emulating exercise without any of the attendant risks of exercise to the neuropathic or neuroischemic foot.
Keywords
Introduction
Diabetes Mellitus (DM) has been growing in prevalence over the last several decades,1,2 and no slowing to this growth is foreseen. 3 Accompanying this worldwide epidemic is a commensurate growth in the complications and sequelae to DM, including macrovascular and microvascular disorders, retinopathy, nephropathy, neuropathy, and diabetic foot ulcers (DFU). 4
It is estimated that already more than 25 million people worldwide develop new DFUs annually. 5 Development of a DFU is associated with a 5% mortality in 12 months, 42% within 5 years, and a 2.5-fold increase in the risk of death compared to diabetic patients without DFU. 6 Patients with DFU also have increased morbidity, reduced health-related quality of life, worse psychosocial adjustment, 7 and a higher burden of healthcare interactions. 8
Although treatment for DFU represents one- third of the total cost of diabetic care, 9 the arsenal of current treatments has historically suffered from a sketchy evidence base, 10 and guidance for routine care has largely relied on expert consensus, 11 with only local oxygen therapy having substantial outcomes evidence.12,13 Recently, the demand for better quality evidence has begun to be satisfied, and new technologies are beginning to emerge. 14
One such technology is intermittent 1 Hz neuromuscular electrostimulation (NMES) of the common peroneal nerve so as to elicit a small twitch of the muscles of the leg, activating the venous pump and augmenting blood flow in the leg. 15 This is a multi-centre, open-label study measuring the effect of neuromuscular stimulation of the common peroneal nerve on the microvascular blood flow within the wound bed of diabetic foot ulcers.
Methods
This study is reported in line with The Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) guidelines 16 and conducted in accordance with the principles of the Declaration of Helsinki (2013) and in compliance with ISO14155:2011. Ethical approval (IRAS 229471) was obtained prior to the study investigation. The trial was registered on ClinicalTrials.gov (ID: NCT03186560). Patients attending outpatient clinics at the Aneurin Bevan University Health Board, Cardiff and Vale University Health Board and University Hospitals of Leicester NHS Trust were screened over a 4 year period (timeline lengthened due to the curtailment of research activities during the COVID-19 pandemic). DFUs were previously diagnosed, categorised and being treated as either neuroischemic or neuropathic ulcers.
Inclusion criteria were: age ≥18 years; intact healthy skin at the site of device application; able to understand the Patient Information Sheet; willing and able to give informed consent; willing and able to follow the requirements of the protocol; and diagnosis of a DFU by a wound healing expert.
Exclusion criteria were: significant clinical wound infection (not colonization) either acute or chronic; history of significant haematological disorders or deep vein thrombosis within the preceding 6 months; pregnancy; presence of a pacemaker or implantable defibrillator; current use of any other NMES; use of investigational drug or device within the past 4 weeks that may interfere with this study; recent surgery that may affect the study in the opinion of the chief investigator (such as abdominopelvic or lower limb); and recent trauma to the lower limbs that would prevent stimulation of the leg with NMES; obesity (body mass index BMI > 35).
The geko® Device
The geko® device (Firstkind Ltd, Daresbury, UK) is a CE and UK CA marked, self-adhesive, disposable, internally powered NMES that is applied externally to the leg indicated “…to increase blood circulation, for the prevention and treatment of oedema, to promote wound healing and for the treatment of venous insufficiency and ischemia.”
The negative electrode of the device is placed over the fibular head in the region of the popliteal fossa, such that it lies directly above the common peroneal nerve. The device setting is increased until a visible rhythmic dorsiflexion once per second (upwards and outward twitch of the foot) is observed. If no dorsiflexion is observed at the highest setting of the device, the patient is classified as a non-responder, and deemed unsuitable for treatment with the device. Reasons may include severe edema, or the presence of emollients disrupting electrical contact to the skin. In this mechanistic study, the device was applied to patients for a few minutes to make measurements (following a short equilibration period). In contrast, standard treatment for promoting wound healing (not measured in this study) comprises 12 h of geko® device use daily, until the ulcer heals.
Laser Speckle Contrast Imaging
Microcirculatory flow in the wound bed and peri-wound area was measured using LSCI (moor FLPI-2™; Moor Instruments Ltd, Axminster, England), calibrated according to the manufacturer's recommendations. A 785 nm laser illuminates tissue to a maximum of 1 mm depth, giving a colour coded tissue perfusion image. Perfusion measurement is based on a speckle pattern resulting from the interaction between photons and moving red cells. This non-invasive tool is therefore used to measure changes in microcirculatory flux where blood vessels are superficial. 17
The flux measurement obtained from the LSCI is a unitless quantity proportional to the speed of particles within the blood vessels. Pulsatility (an indication of the strength of the pulse in the signal) is calculated as the root mean square deviation (also unitless) from the mean flux value. The LSCI imager was positioned 30 cm from the target area to record images in the wound bed, the peri-wound area, and the reference marker. The device recorded a baseline of 5 min with the geko® switched off (inactive) followed by 30 min with the geko® device active.
Study Overview
The overall study process and measurements are outlined in Figure 1. Each subject was placed in a recumbent position at room temperature with the leg outstretched resting on an evacuated bean-bag cushion to immobilize the leg for measurements. Any excess slough or liquid was removed prior to making any measurements. The investigator selected 2 areas on each patient for data capture: the wound bed and wound edge (peri-wound). A reference marker, consisting of an opaque foil strip, was affixed to the skin adjacent to the wound, to allow for correction of movement artifact, according to a method previously described. 18
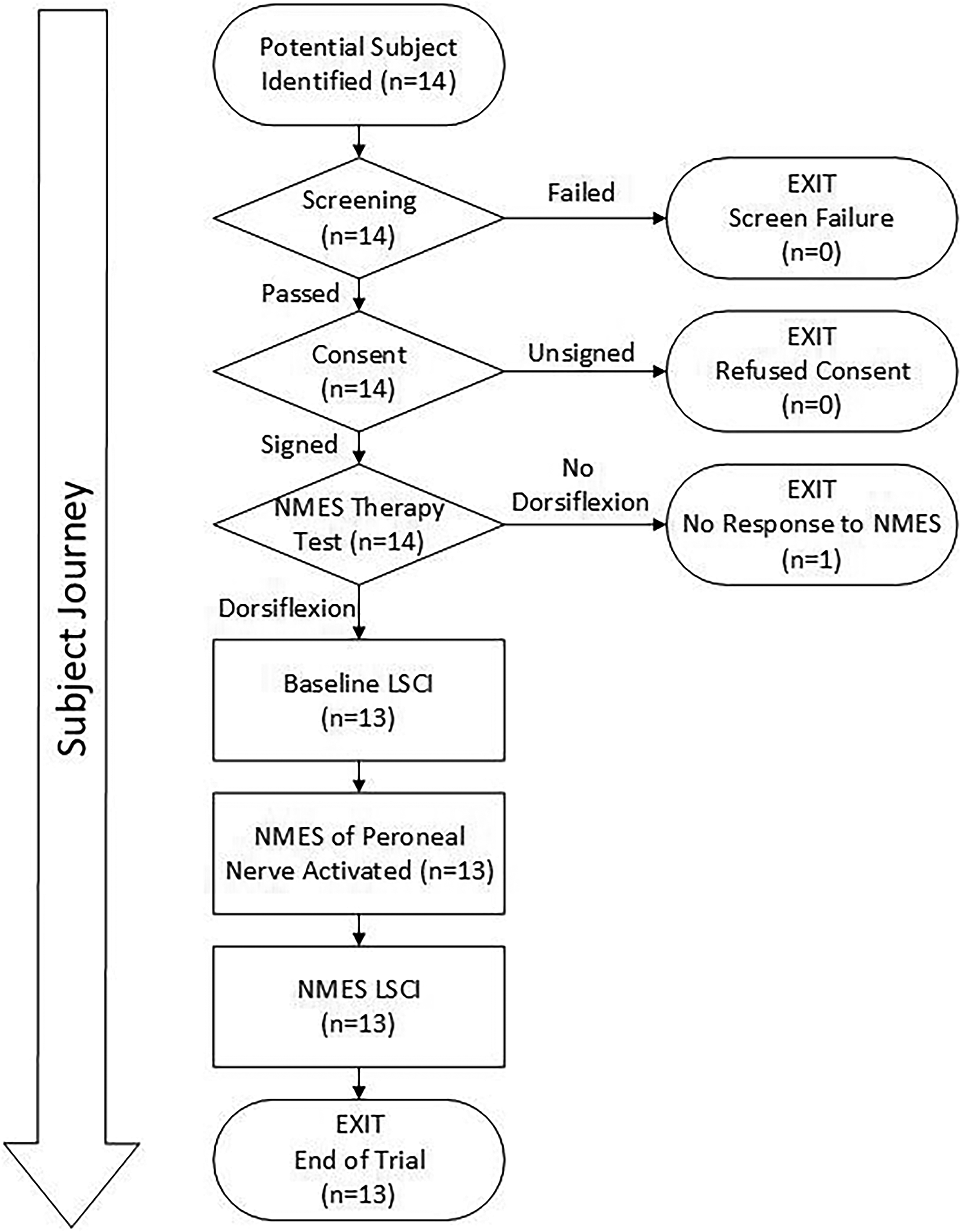
Flow chart illustration of the study design and subject accountability.
Statistical Analysis
Sample size was based on pilot data which suggested that geko® increases Speckle flux by 152% ± 61%. Thus, assuming P = .05 and power = 90%, based on the below formula:
n = 4 subjects would be required to demonstrate an effect. This was increased to 10 subjects to improve confidence. Individual patient data for flux and pulsatility are demonstrated in line graphs, with their standard error of difference. Differences between pre- and during stimulation recordings were calculated using a paired Student's t-test. P < .05 was considered significant.
The datasets generated and analysed in the current study are available from the corresponding author upon reasonable request.
Results
Demographics
Eleven neuropathic (6 male, 5 female), and 2 neuroischemic subjects (1 male, 1 female) were recruited. Mean and (standard deviation) demographics were as follows. Neuropathic group: age 58 (16) years; height 1.75 (0.13) m; weight 86 (15) kg; Body Mass Index 27.6 (3.4), wound areas 1.96 (0.76) cm2. Neuroischaemic group: age 56 years, height 1.83 m, weight 102 kg, BMI 30.2, wound area 5.53 cm2, ABPI was not measurable due to calcification. A high degree of heterogeneity between subjects was observed in both groups (eg, BMI, weight, ulcer size and so on). However, since each participant is self-controlled, this heterogeneity is accommodated in the analysis.
Figure 2 shows a typical example of the measurement process. In Figure 2a, the foil reference marker can be seen proximal to the ulcer. LSCI Measurements were obtained at baseline (Figure 2b) and during treatment (Figure 2c). Activation of the NMES device results in a visible increase in flux throughout the foot, and especially in the bed and periphery of the wound. The opaque foil reference continues to show no flux, verifying that movement artefact is not contributing to the measured augmentation of flux.
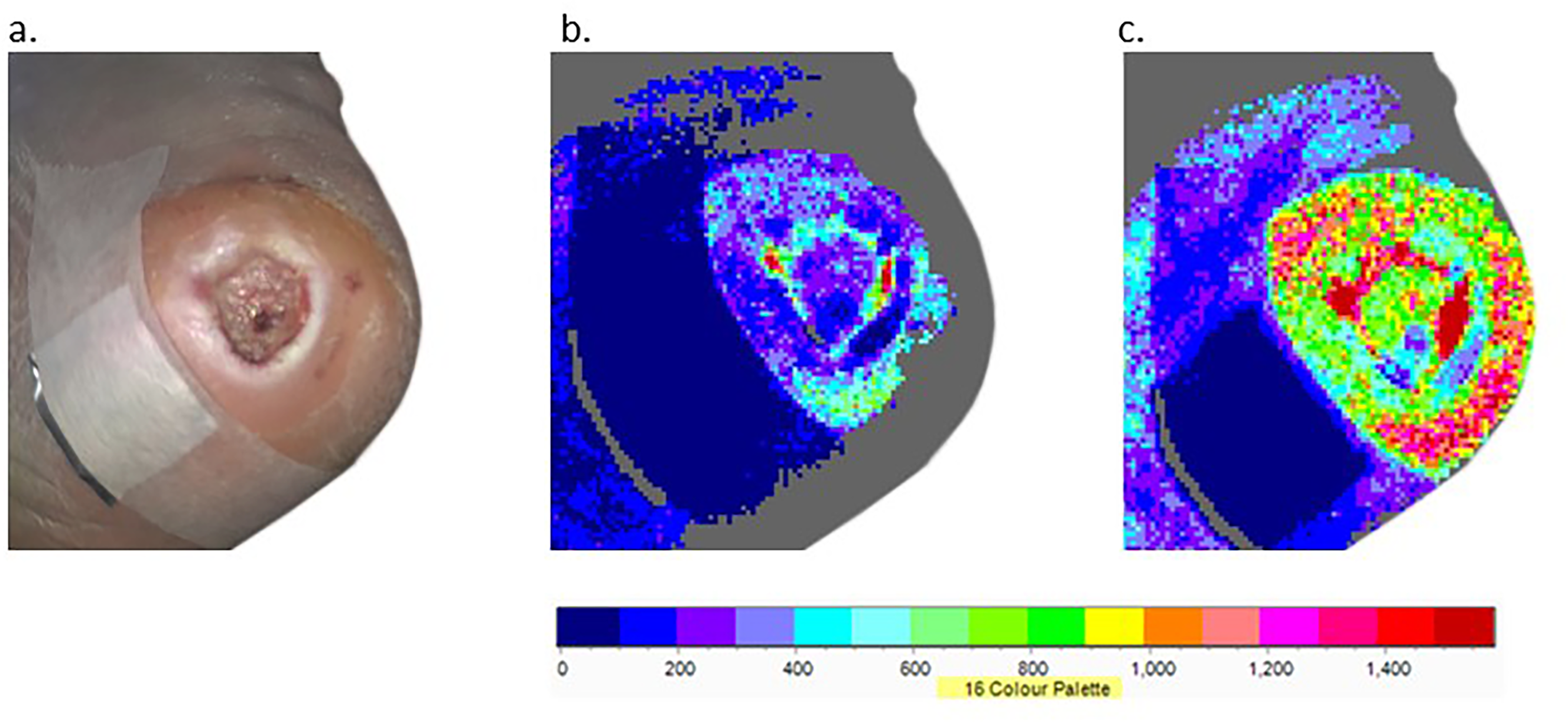
Example DFU with baseline reference marker (a), LSCI at baseline (b) and LSCI with NMES active (c).
Figure 3 shows the aggregated flux data for all 11 neuropathic ulcers.
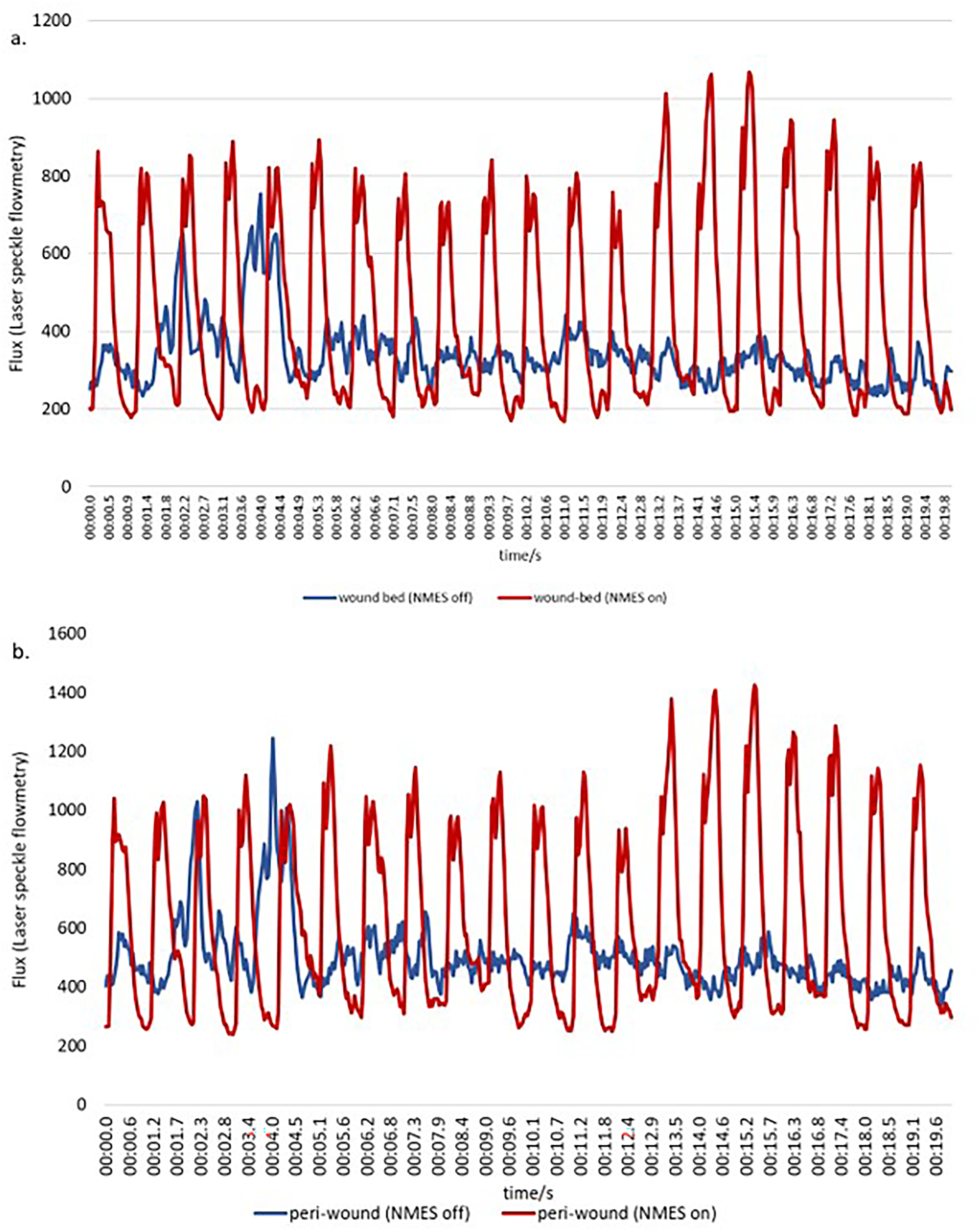
Trace of LSCI flux in wound bed of example ulcer over a 20 s period, comparing NMES on with NMES off. 3b. shows the trace of LSCI flux at periphery of example ulcer over a 20 s period, comparing NMES on with NMES off.
Wound bed flux (Figure 4) increases by 36% (95%CI 11%-68%, P = .002), and peri-wound flux (Figure 5) increases by 92% (95%CI 46%-160%, P = .001). Pulsatility in the wound bed (Figure 6) increases by 183% (95%CI 61%-517%, P = .005), while pulsatility in the peri-wound (Figure 7) increases by 359% (95%CI 264%-455%, P = .001).
Of the 13 subjects recruited, only 2 presented with neuroischemic ulcers. It was therefore not possible to perform any inferential statistics on their results, and these are presented as individual traces in Figure 8. The 2 subjects followed a very similar trajectory, with wound bed flux (Figure 8a) and wound bed pulsatility (Figure 8b), as well as peri-wound flux (Figure 8c) and peri-wound pulsatility (Figure 8d), increasing very substantially when the device was switched on. In the case of pulsatility, it increased from near-zero values.
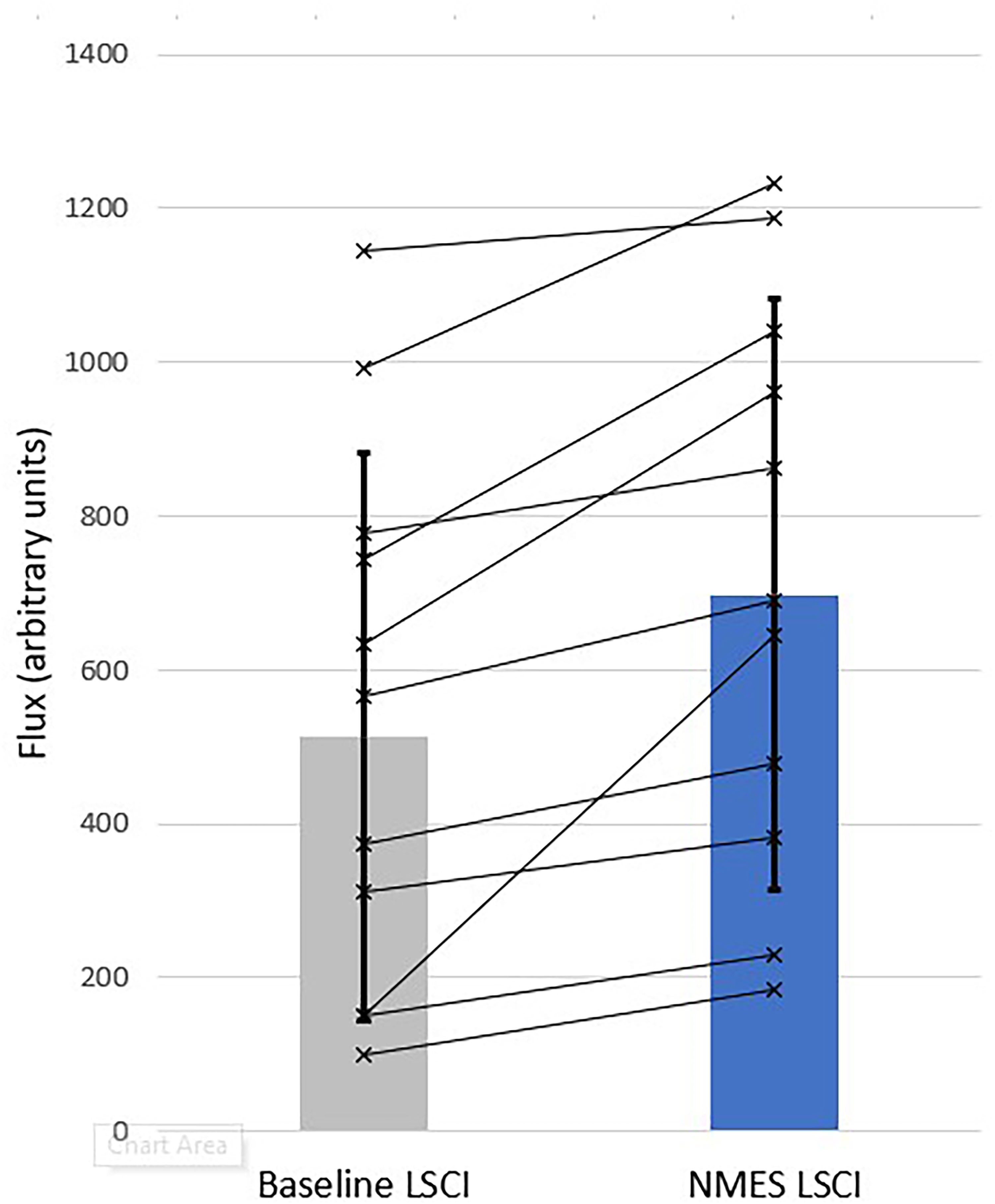
LSCI wound bed flux for all neuropathic subjects (n = 11). Bars show population means (±1SD). Lines indicate the individual subject response measures.
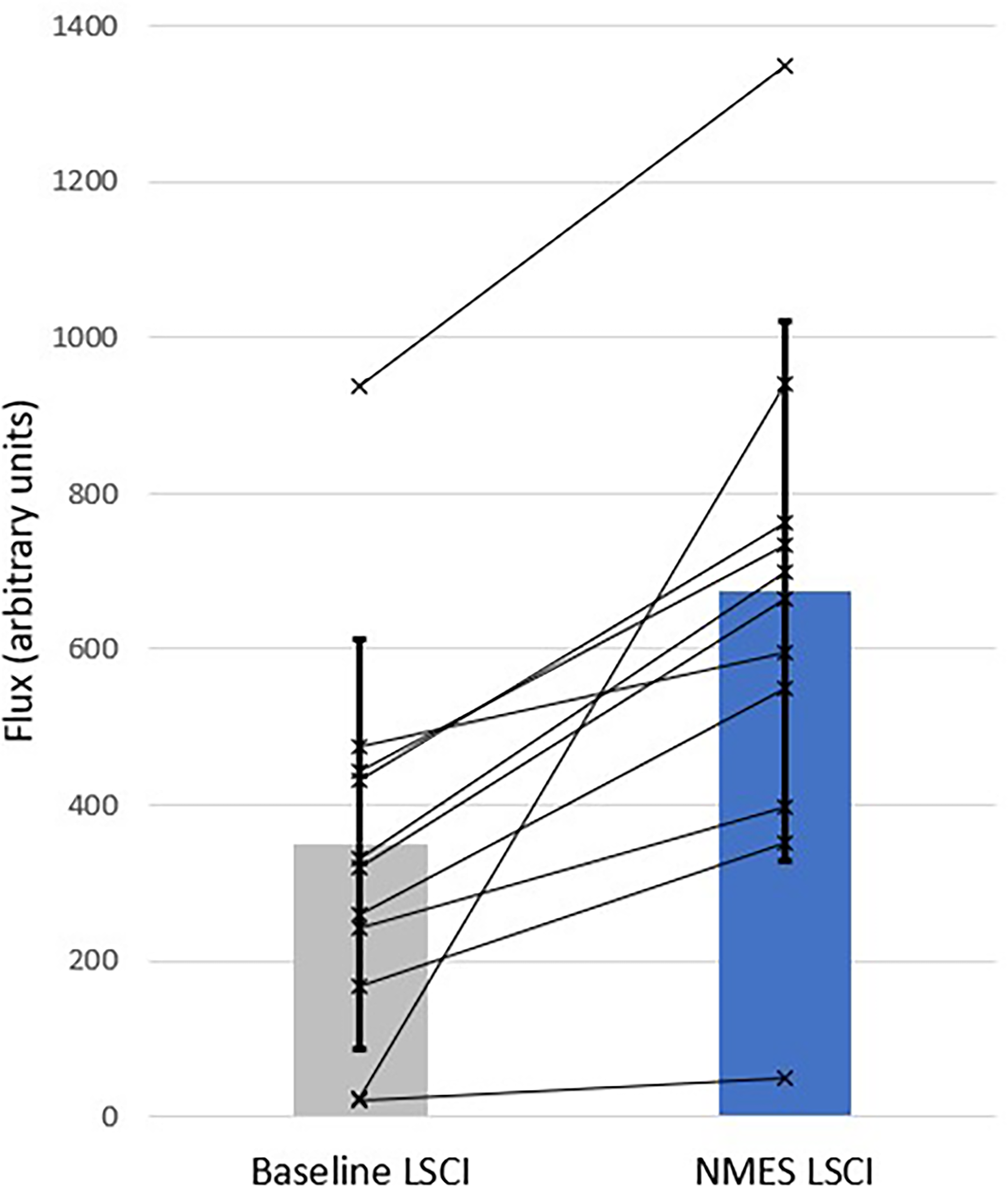
LSCI peri-wound flux for all neuropathic subjects (n = 11). Bars show population means (±1SD). Lines indicate the individual subject response measures.
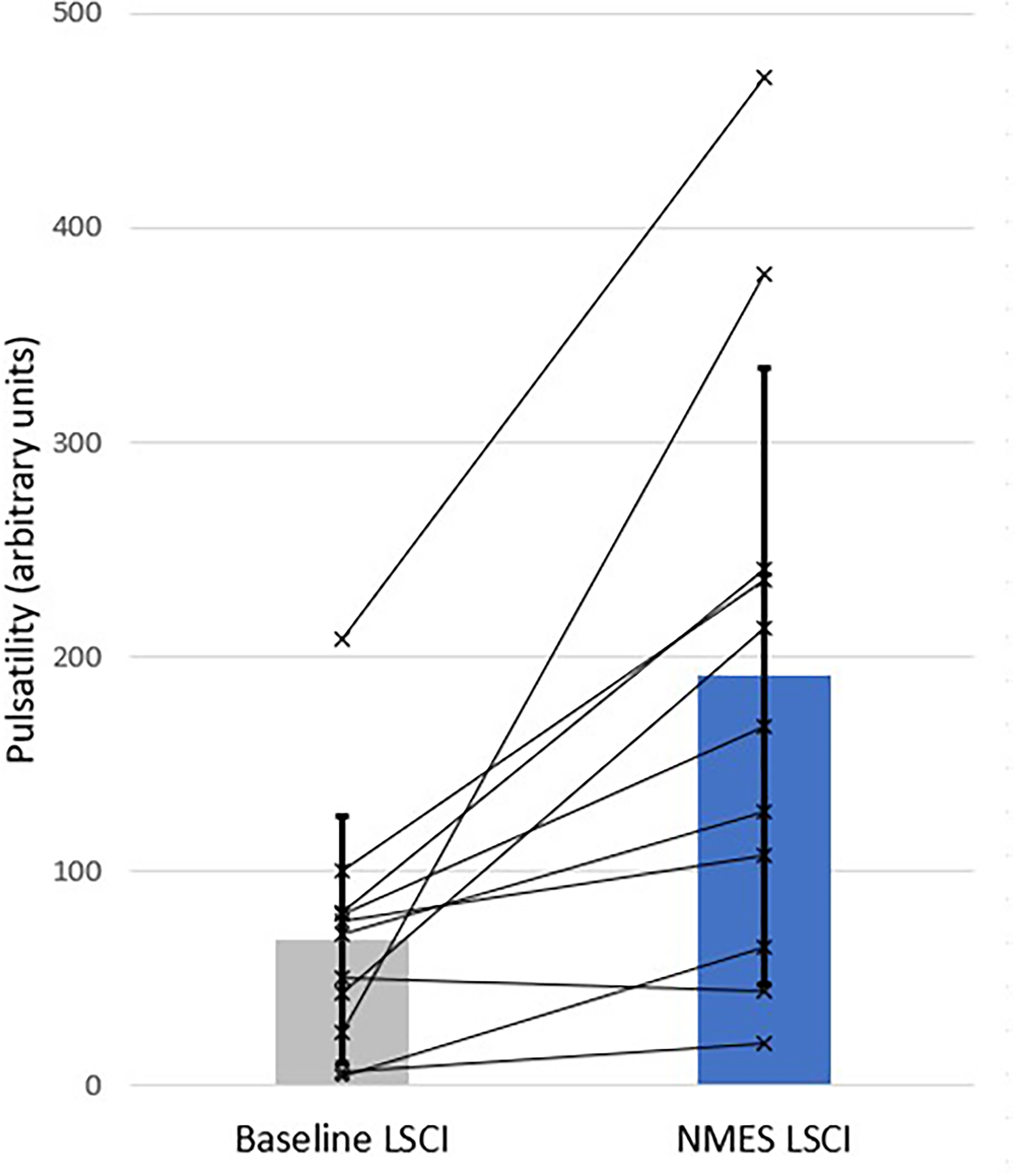
LSCI wound bed pulsatility for all neuropathic subjects (n = 11). Bars show population means (±1SD). Lines indicate the individual subject response measures.
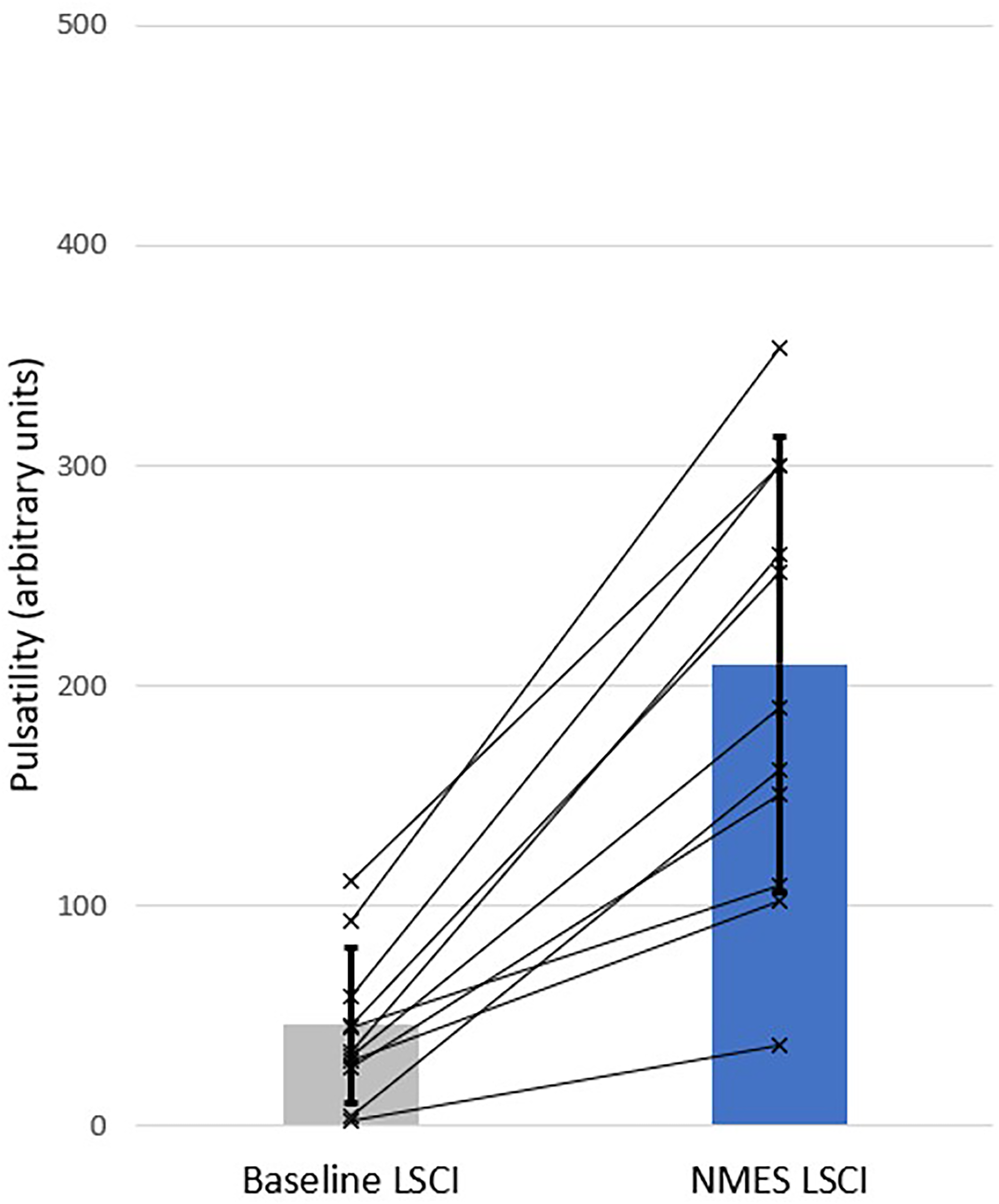
LSCI peri-wound pulsatility for all neuropathic subjects (n = 11). Bars show population means (±1SD). Lines indicate the individual subject response measures.
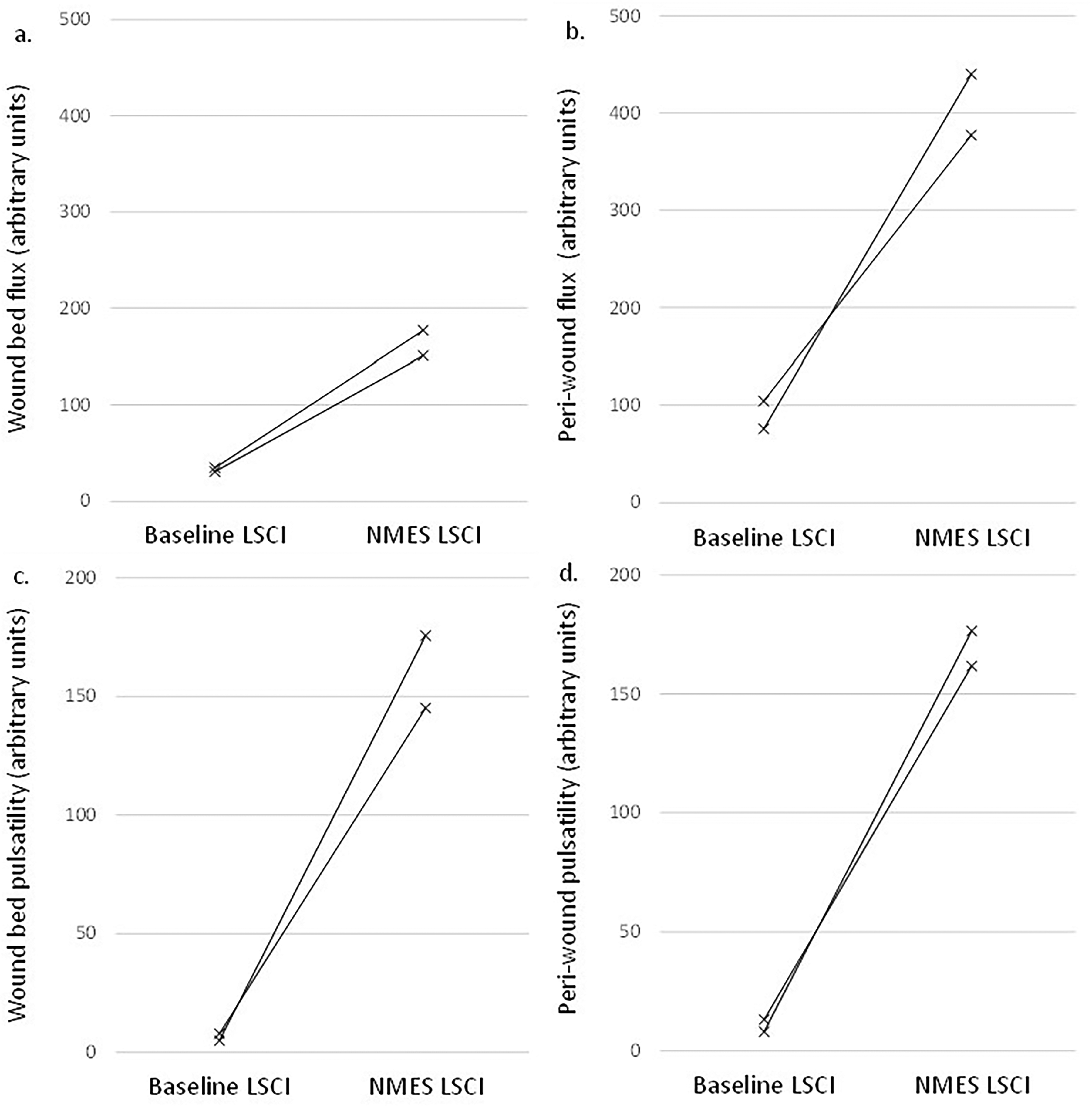
LSCI wound bed flux (a), peri-wound bed flux (b), wound bed pulsatility (c) and peri-wound bed pulsatility (d), for all neuroischemic subjects (n = 2). Lines indicate the individual subject response measures.
When combined with the neuropathic patients, the neuroischaemic patients further amplify the increases observed. In the combined group (n = 13) the wound bed flux increases by 40% (95%CI 12%-68%, P < .001), and peri-wound flux increases by 104% (95%CI 61%-146%, P < .001). Pulsatility in the wound bed increases by 220% (95%CI 112%-329%, P < .001), while pulsatility in the peri-wound increases by 405% (95%CI 298%-513%, P < .001).
Discussion
Activation of the leg muscle pumps by NMES of the common peroneal nerve has been previously shown to augment blood flow and velocity in the venous, arterial, and microvascular systems,19–21 and reduces claudication pain. 22 It increases the rate of healing in venous leg ulcers (VLU), 23 as well as augmenting microvascular flow and pulsatility in the wound bed of VLU 24 and arterial leg ulcers (ALU). 25 This study demonstrates that the microcirculatory benefits delivered by NMES that has been observed in other patient groups is also observed in DFU patients, with underlying neuropathic and neuroischemic pathologies. Additionally, the NMES device is safe to use in these populations.
Given the immense heterogeneity of patients with diabetic foot ulcers in general, it is unsurprising to see some degree of intra-group variation, and this (perhaps inevitable) level of heterogeneity would be problematic in a classic inter-cohort RCT design. However, in this self-controlled design, each subject's baseline LSCI is compared with his/her own NMES LSCI, so accommodating these differences.
The NMES device results in a very significant increase in both microcirculatory flux and pulsatility, as measured by LSCI. This increase occurs in both the wound bed and the wound periphery. The increase in pulsatility is of particular interest, since pulsatile flow provides many components considered essential for wound healing. 26 Pulsatile blood flow results in intermittent shear forces at the endothelial surface and results in chemical signalling and transduction, production of growth factors, nitric oxide production, and the reduction of oxidative stress. 27 The stated object of many wound treatment regimens including negative pressure therapy, alternating positive pressure therapy, ultrasound therapy, and other energy-based modalities is to generate pulsatile flow. 28 However, this study is the first to show statistically significant improvement in pulsatility in the DFU wound bed and peri-wound.
Off-loading the foot was established 140 years ago as one of the principles of DFU treatment. 29 Its relevance today is underpinned by our understanding of the etiology of DFU: damage to tissue caused by small vessel occlusion resulting from a combination of angiopathy and peripheral neuropathy.30,31 Peripheral neuropathy removes the essential feedback to the subject that mechanical damage is being done when the extremity is loaded.
Off-loading the foot, using a cast or other such device, has the benefit of reducing some restrictions to perfusion to the foot, and, if correctly fitted and monitored, potentially helping to prevent further mechanical damage. 32
Exercise has also been identified as beneficial to DFU healing, as it activates the calf muscle pumps, so increasing perfusion in the foot, 33 and has been found to reduce many risk factors for DFU. 34
However, a trade-off exists between off-loading and exercise. Some approaches to offloading, such as the total contact cast, restrict mobility and impede exercise, and particularly the action of the leg muscle pumps. Conversely, many forms of exercise present an increased risk of repetitive mechanical damage to the insensate foot.35,36
Conclusion
This study demonstrates that NMES of the common peroneal nerve increases microvascular flow in the wound bed of DFU. This presents a mode of activating the leg muscle pump without additional pressure insult to the foot and has potential to support the microcirculation during wound-healing, effectively emulating exercise without any of the attendant risks of exercise to the neuropathic or neuroischemic foot.
Highlights
Why did we undertake this study? We wanted to explore the mechanism by which a new intervention might increase healing rate in diabetic foot ulcers. What is the specific question(s) we wanted to answer? We wanted to see if the intervention increased microvascular flow in the wound bed, and/or at the wound edge. What did we find? We found that neuromuscular stimulation of the common peroneal nerve increased microvascular flux and pulsatility in the wound bed and the wound edge for neuropathic and neuroischemic diabetic foot ulcers What are the implications of our findings? Neuromuscular stimulation provides a mechanism of action by increasing micro-circulatory flow which may underpin a new treatment for diabetic foot ulcers.
Footnotes
Author Contributions
Keith Harding wrote the protocol, David Bosanquet was the Chief Investigator for the trial, primary research on subjects was delivered by Melissa Blow and Faye Ashton. All authors reviewed and approved the final version of the manuscript.
Conflict-of-Interest Statement
Keith Harding has received payments for consulting work from Firstkind Ltd.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The authors acknowledge the assistance of Firstkind Ltd, Hawk House, Peregrine Business Park, Gomm Road, High Wycombe, HP13 7DL for sponsoring the study (Grant Ref: FSK-SPECKLE-001) and provided the NMES devices for the trial.
Guarantor
Keith Harding is the guarantor of this work and, as such, had full access to all the data in the study and takes responsibility for the integrity of the data and the accuracy of the data analysis.
Twitter Summary
Microcirculatory flux and pulsatility was measured in the wound bed of 13 diabetic foot ulcers. Neuromuscular stimulation of the common peroneal nerve increased microcirculatory flux and pulsatility.
Appendix: list of participating investigators.
Chief Investigator: Professor Keith Harding
Principle Investigator: David Charles Bosanquet – Consultant Vascular Surgeon
Co-investigator: Melissa Blow: Principal Podiatrist
Site: Aunerin Bevan University Health Board, Cwmbran Clinic, Tudor Rd, Cwmbran, NP44 3YA.
Site: Welsh Wound Innovation Centre
Principle Investigator: Sarah Bradbury – Clinical Director
Previous Principle Investigator: Nicola Mary Ivins – Director Of Clinical Research, Wound Healing.
Address: Rhodfa Marics, Ynysmaedy, Pontyclun, CF82 8UX, Wales, United Kingdom.
Site: Glenfield Hospital:
Principle Investigator: Matthew James Bown – Professor of Vascular Surgery
Co-investigator: Faye Ashton – Research Nurse
Address: Groby Road, LE3 9QP, Leicester, United Kingdom.