Abstract
This prospective case series evaluated the clinical outcomes of skin micro-fragment therapy in managing hard-to-heal diabetic foot ulcers (DFU). A total of ten patients with chronic DFU, who were treated in a specialized diabetic foot unit, were included in this study. The primary outcome was the wound healing rate at 12 weeks. Secondary outcomes comprised the time to complete epithelialization, wound area reduction (WAR) at 4 and 12 weeks, and the incidence of adverse events. After treatment with hy-tissue micrograft (HT-MG), 6 (60%) of patients achieved complete wound closure within 12 weeks. The mean healing time was 7.8 ± 2.4 weeks. The mean WAR at 4 and 12 weeks was 67.2% ± 23.5% and 87.5% ± 24% respectively. The procedure was well tolerated with no complications observed in the donor site such as pain or infection. No adverse effects related to the infiltration procedure at wound site were recorded with a promising wound healing rate during the follow-up period. These results suggest that HT-MG could be a safe and effective treatment option for chronic DFU, promoting noteworthy wound healing and reducing healing times. Further studies are required to confirm these findings and assess long-term outcomes.
Introduction
Diabetic foot ulcers (DFU) are a significant and chronic complication associated with diabetes mellitus (DM), leading to severe health outcomes and substantial economic burdens.1,2 These DFUs often fail to heal, developing into ‘hard-to-heal’ wounds 3 that can be broadly described as those failing to respond to standard therapy in an orderly and timely manner. 4 Approximately 33% of DFUs remain open and fail to heal within a one-year follow-up period, developing into non-healing wounds. 5
Delayed healing heightens the possibility of adverse sequelae, 6 which in turn escalates the risk of soft tissue infection, osteomyelitis, and lower extremity amputations. 7 Numerous factors impact the healing of DFU. These factors can be categorized into those directly tied with wound etiology, including metabolic disorders, peripheral arterial disease, and bacterial bioburden8,9; clinical risk aspects that are not specifically related to etiology, such as age, anemia, or smoking, and ultimately, patient-related risk factors like adherence, economic status, or systemic drugs.4,10
Nonetheless, it is important to consider complex wounds that typically stall in the inflammatory phase without progression. 11 This highlights the importance of conducting a comprehensive holistic assessment. Accordingly, these situations require advanced therapies including traditional standard of care measures (SoC), 12 and new adjunct therapies. 3 A foot ulcer that fails to close by at least 40%–50% over 4 weeks despite adequate SoC is unlikely to completely heal without a more specific intervention.4,13
Highly evidenced advanced therapies include hyperbaric oxygen therapy (HBOT), topical oxygen therapy (TOT), autologous leukocyte, platelet-rich fibrin patches, placental-derived products, and sucrose octasulfate dressings. 14 Although new trends in recalcitrant DFU care are gaining increasing interest in the scientific community, they consume disproportionate amounts of medical resources and health professionals’ time, 15 with considerable variation in healing rates. 4
Recently, skin micro-fragment therapy has emerged as a promising alternative. This technique involves the mechanical fragmentation of a small quantity of skin to produce autologous skin micrografts. When applied to the ulcer, these grafts promote wound healing.16,17 Micro-fragmented skin tissue is a new mechanism able to conserve biological properties and capable of stimulating the regenerative process in chronic wounds that usually remain in the inflammatory phase. It acts as a slow-released natural scaffold that preserves the extracellular matrix components which provides support to the gradual growth of the fibroblast metabolic activity, and continuously delivers growth factors and pro- and anti-inflammatory cytokines which supports the regenerative response. 16
Previous evidence supports the idea that autologous punch grafting promotes healing in chronic DFU, particularly in cases where conservative treatment has been unsuccessful. 18 However, there are also several limitations related to the conventional punch grafting procedure, conditioning its use on some DFUs. First, the plantar location restricts punch grafting adherence to the wound bed. Second, the need to combine with Negative Wound Therapy Pressure (NWTP) in complicated anatomical locations to promote graft acceptance increases costs. 19 Additionally, the size of the donor site will be larger depending on the baseline wound area, potentially increasing morbidity and further complications. Finally, the usual punch grafting procedure is not homogeneous, depending on the specialist's preferences and experience. 18 Standardizing the process through the novel medical device presented in this study could offer more homogeneous results. No adverse events were associated to this procedure on the current literature. 16 Complications like wound infection of the ulcer or the donor site specially in patients with concomitant comorbidities were described as the most frequent complication in traditional punch grafting, 18 and should be also considered.
For this reason, skin micro-fragment therapy appears as an emerging alternative to consider in non-healing DFUs where other treatments are not indicated or have failed. Therefore, this study aims to evaluate the clinical outcomes of skin micro-fragment therapy in managing hard-to-heal DFUs.
Materials and Methods
Study Design
A prospective case series was conducted involving a total of ten patients with hard-to-heal DFUs treated with HT-MG in a specialized diabetic foot unit from October 2021 to July 2024.
The inclusion criteria included having a confirmed diagnosis of type 1 or type 2 DM, being over 18 years of age, presenting with a non-infected, neuropathic or neuroischemic chronic DFU classified as Texas grade IA, IC- IIIA, or IIIC, 20 that has not responded to conventional or advanced treatments such as Negative Wound Pressure Therapy (NWPT), topical oxygen, or silver products, and has had at least 4 weeks of evolution.
The exclusion criteria included patients with a clinical infection as defined by the Infectious Diseases Society of America criteria, 21 untreated osteomyelitis, and critical limb ischemia characterized by an ankle-brachial index (ABI) ≤ 0.4, an ankle systolic blood pressure less than 70 mm Hg, or a toe systolic blood pressure less than 50 mm Hg. 22
All patients provided their written informed consent before inclusion, adhering to the principles listed in the Declaration of Helsinki. 23
Skin Micro-Fragmentation Procedure
The HT-MG Technology (Fidia Farmaceutici S.p.A., Italy), a sterile, single-use medical device, was used to fragment tissue biopsies. Full-thickness skin punch biopsies were obtained from a healthy area of the patient's skin using 5 mm punch biopsy tools. Topical local anesthesia with Mepivacaine (2%) was applied to the donor area, which in all cases was the anterolateral aspect of the thigh.
After disinfecting the donor site with 0.5% chlorhexidine and marking it with a dermographic pen, four skin biopsies were taken from a single treatment using a 5 mm biopsy punch. The biopsies were placed on sterile gauze soaked in 0.9% saline until processing. 18
The HT-MG device was prepared per the manufacturer's instructions. Biopsy skin samples were situated on the device's grid area, and a saline solution (15.5 mL) was injected into the lower chamber to saturate the biopsies. The top section of the device, furnished with a rotating cutting surface, was then fastened onto the lower base. Fragmentation was achieved by rotating the top portion using a micromotor at 150 rpm for one minute. We obtained tissue fragments measuring less than 400 μm, which passed through the grid and were collected in the lower chamber. Lastly, we withdrew a suspension of micrografts from the lower chamber using a 20 mL syringe without a needle (Figure 1a–f).
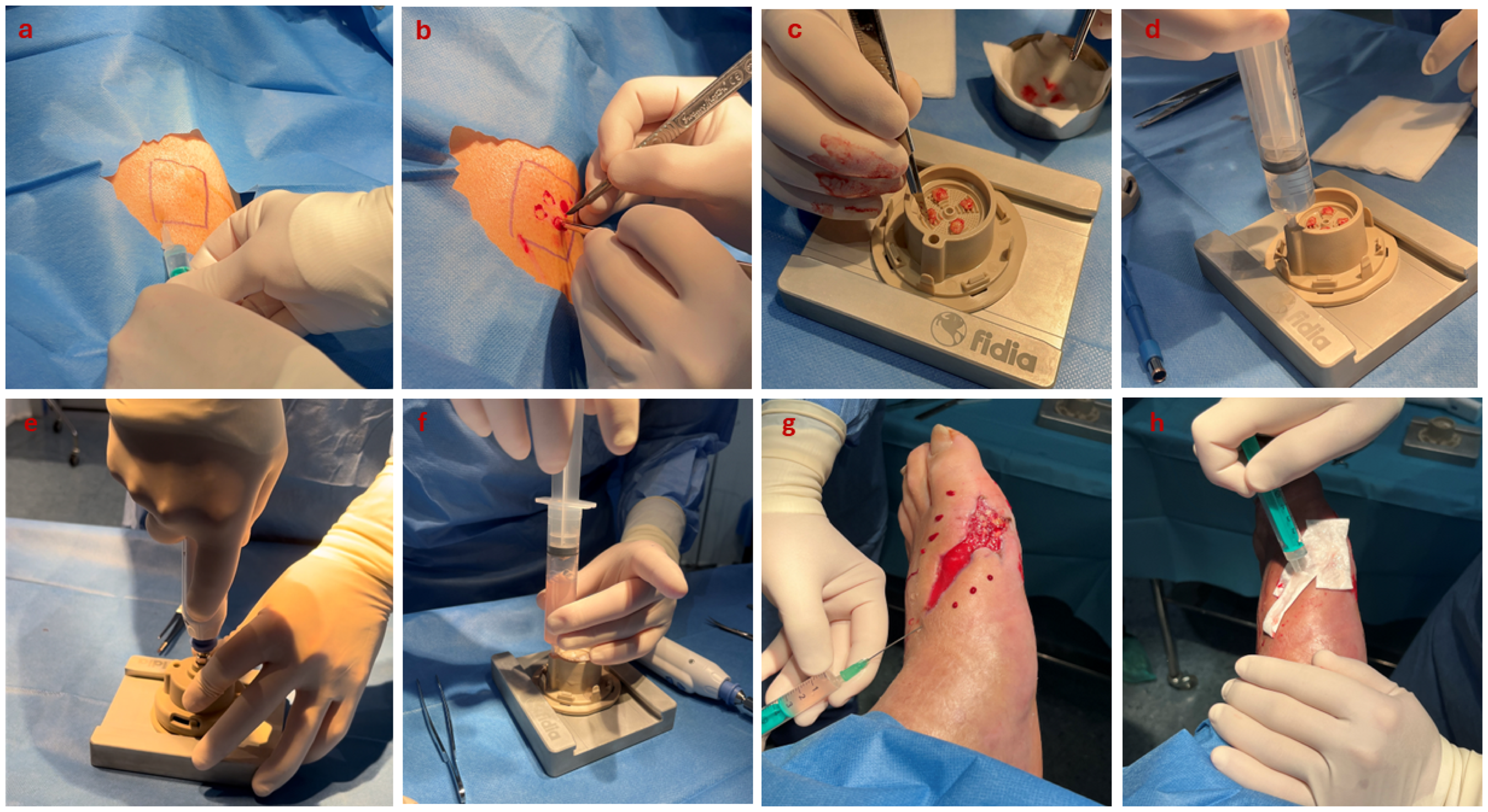
Skin micro-fragmentation procedure. (a) Anesthetic infiltration in the marked donor site; (b) graft collection with scalpel and forceps; (c) positioning of biopsied skin samples on the device's grid area; (d) saline solution injection into the lower chamber; (e) fragmentation using a micromotor at 150 rpm for one minute; (f) extraction of the micrograft suspension from the lower chamber using a 20 mL syringe; (g) injection of the micro-fragments perilesionally around the DFU; (h) placement of bioactive dressing with any remaining suspension.
The micro-fragment suspension was injected perilesionally around the ulcer using a 21-gauge needle. Each injection point, spaced 1 cm apart, received approximately 0.5 mL of the micrograft suspension. The remaining suspension was applied to a Hyalo4Regen Pad (Fidia Farmaceutici S.p.A., Italy). This bioactive dressing consists of heterologous type I horse collagen and hyaluronic acid in a lyophilized, sterile, spongy pad form. The pad, serving as a scaffold for the micro-fragments, was positioned over the wound bed to ensure optimal contact with the ulcer surface (Figure 1g and h). The wound was subsequently covered with a compressive gauze dressing to secure the Hyalo4Regen Pad.
Patients received a single skin micro-fragment treatment, followed by standard wound care, which included weekly changes of dressings and bandages. All patients were prescribed appropriate off-loading devices in accordance with the International Working Group on Diabetic Foot (IWGDF) guidelines. 24
Clinical Evaluation and Follow-up
All patients were monitored weekly until wound healing was accomplished. Wound healing was characterized as complete epithelialization without any discharge, verified at least 10 days post-closure. 25 The initial dressing change took place 7 days following the HT-MG procedure. Subsequently, patients visited the outpatient clinic twice a week for dressing care until their wounds healed. This schedule was based on the characteristics and needs of the DFU.
DFU cleansing was performed with 0.9% saline, and all patients received a SoC for their wounds with off-loading. 24 This, along with sharp or mechanical debridement to remove non-viable skin, included peri-wound skin if required. Detailed wound assessments were conducted weekly at each follow-up visit until wound healing. These assessments included measuring the wound area surface, taking before and after debridement photographs, and evaluating local DFU characteristics (coverage of deep structures, wound bed tissue, exudate level, and signs of infection). Planimetric measurements of wound size were performed by tracing the perimeter on an acetate grid with a fine-nibbed pen and using Visitrak (Smith & Nephew, UK). The wound bed tissue was evaluated for the presence of granulation, slough, or necrotic tissue.
Outcome Measures
The primary outcome measure of the study was the rate of wound healing at 12 weeks. Secondary outcomes included time taken to reach complete epithelialization and wound area reduction (WAR - %) at 4 and 12 weeks. The nature, incidence, and severity of adverse events were documented and categorized during the study by the principal investigator in relation to the product.
Statistical Analysis
All statistical analyses were conducted using GraphPad Prism version 10.0.0 for Windows, provided by GraphPad Software, Boston, Massachusetts, USA. The assumption of normality for all continuous variables was confirmed through the Shapiro–Wilk test. Categorical variables were represented as numbers and percentages. Continuous variables were delineated by their means and standard deviations (SD, for parametric distribution) or by their median and interquartile range (IQR, for nonparametric distribution).
Results
Patient Demographics
A total of ten patients, with 11 chronic DFUs, were included in the current study. The average patient age was 59.8 ± 4.5 years, and 90% of the participants were male. Most patients had type 2 diabetes (90% of them), with an average duration of 19.2 ± 10.2 years. Prevalent comorbidities in the cohort included cardiopathy (50%), retinopathy (30%), and nephropathy (30%) (Table 1).
Demographic and clinical aspects of the patients at baseline.
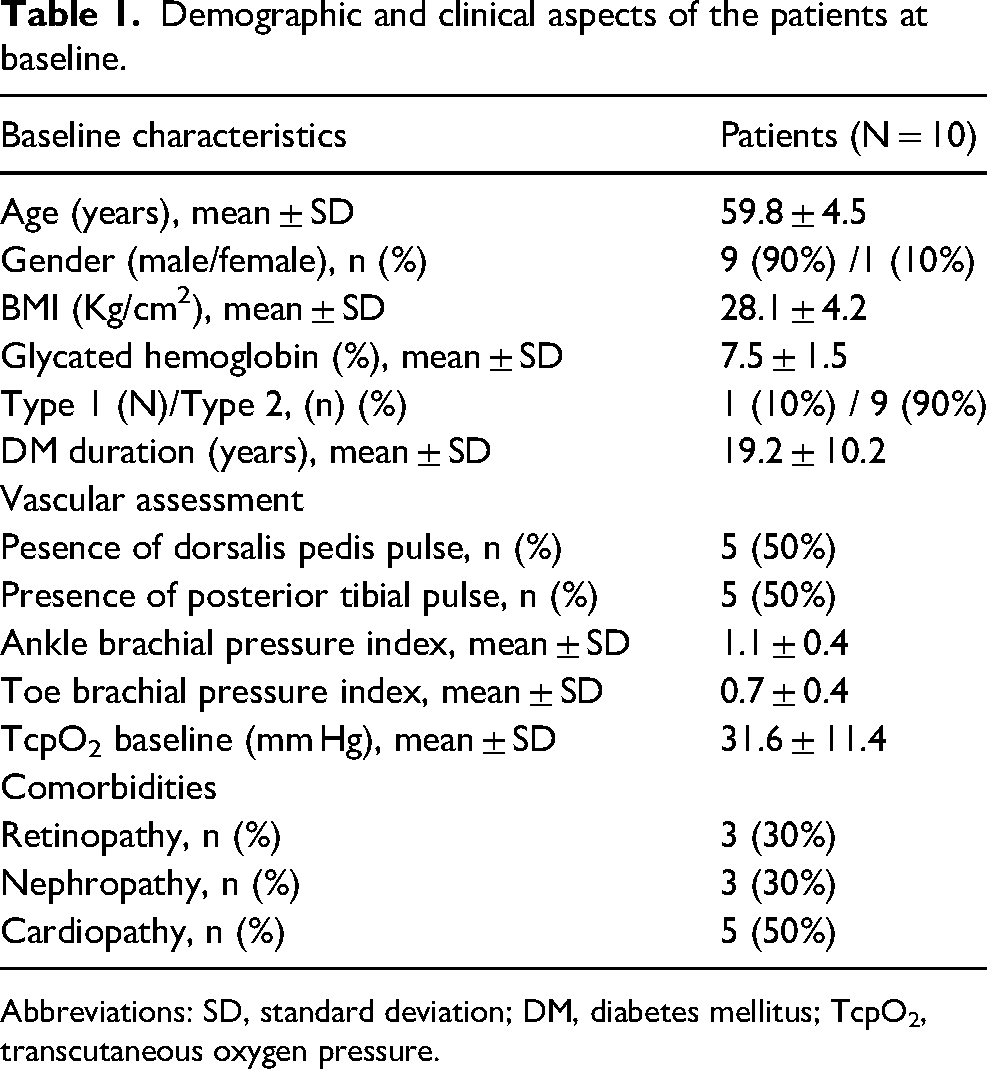
Abbreviations: SD, standard deviation; DM, diabetes mellitus; TcpO2, transcutaneous oxygen pressure.
Baseline DFU Characteristics
The initial characteristics of the DFU are summarized in Table 2. The average wound area at baseline was 3.1 ± 2.2 cm2. The location of DFUs was distributed as follows: 4 (36.4%) cases on the heel, 1 (9%) case on the midfoot, 4 (36.4%) cases on the metatarsal heads (including concurrent wounds in one patient at the second and fourth metatarsal heads), and finally, 2 (18.2%) cases at the metatarsophalangeal joints.
Diabetic foot ulcer characteristics at baseline.
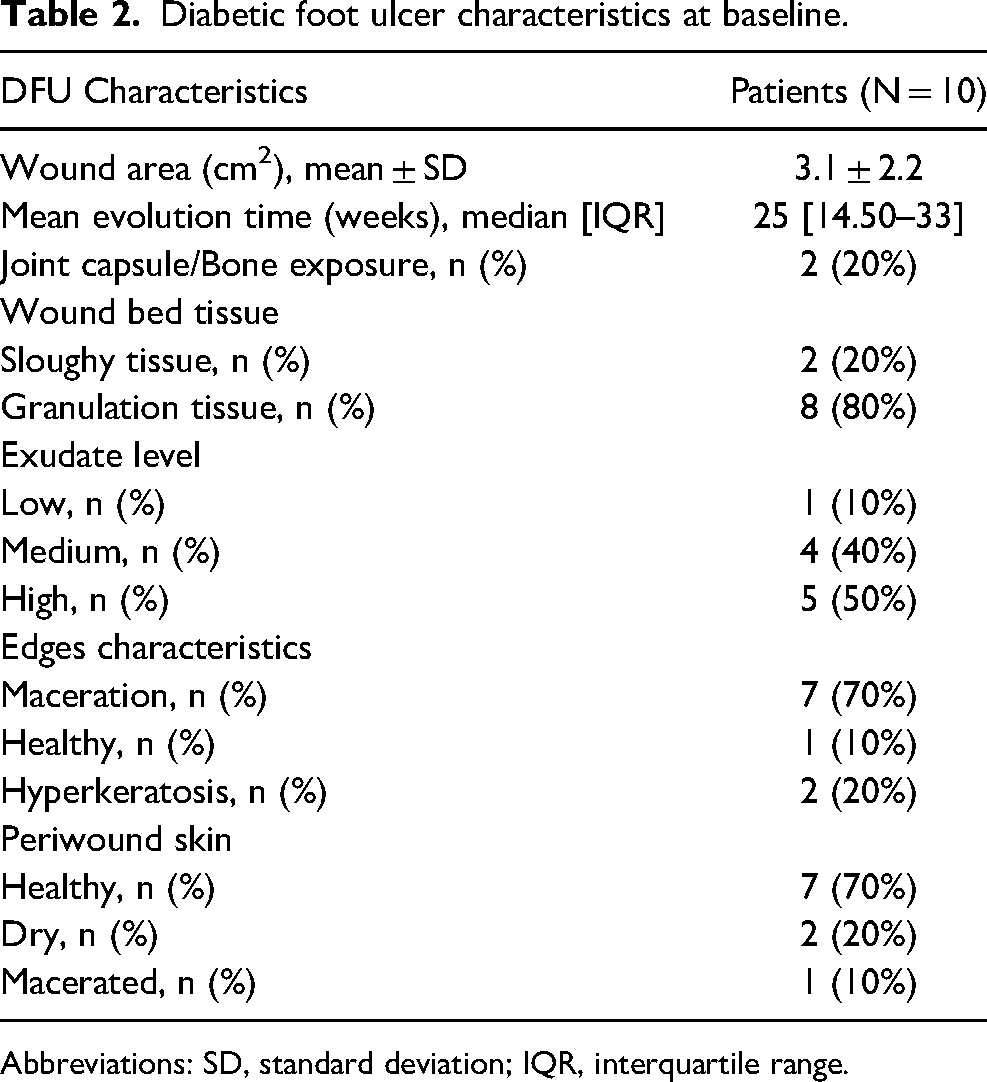
Abbreviations: SD, standard deviation; IQR, interquartile range.
Outcomes
A total of six patients (60%) with seven DFUs achieved complete epithelialization within a 12-week follow-up period. The mean time to complete wound healing was 7.8 ± 2.6 weeks. One more patient reached full healing at 14 weeks. The overall WAR was 67.2 ± 23.5% at 4 weeks, and 87.5 ± 24% at 12 weeks, respectively. All the wounds in this study required only one single treatment of skin micro-fragments. The donor site healed in all patients without complications, within an average of 12 ± 3.63 days. Lastly, two patients (20%) with initial bone and capsule exposure achieved full coverage of these structures within 4 weeks. Table 3 shows an overview of patients included in the cases series.
Overview of patient wound characteristics: DFU location, evolution time, and previous treatment (local and offloading device).
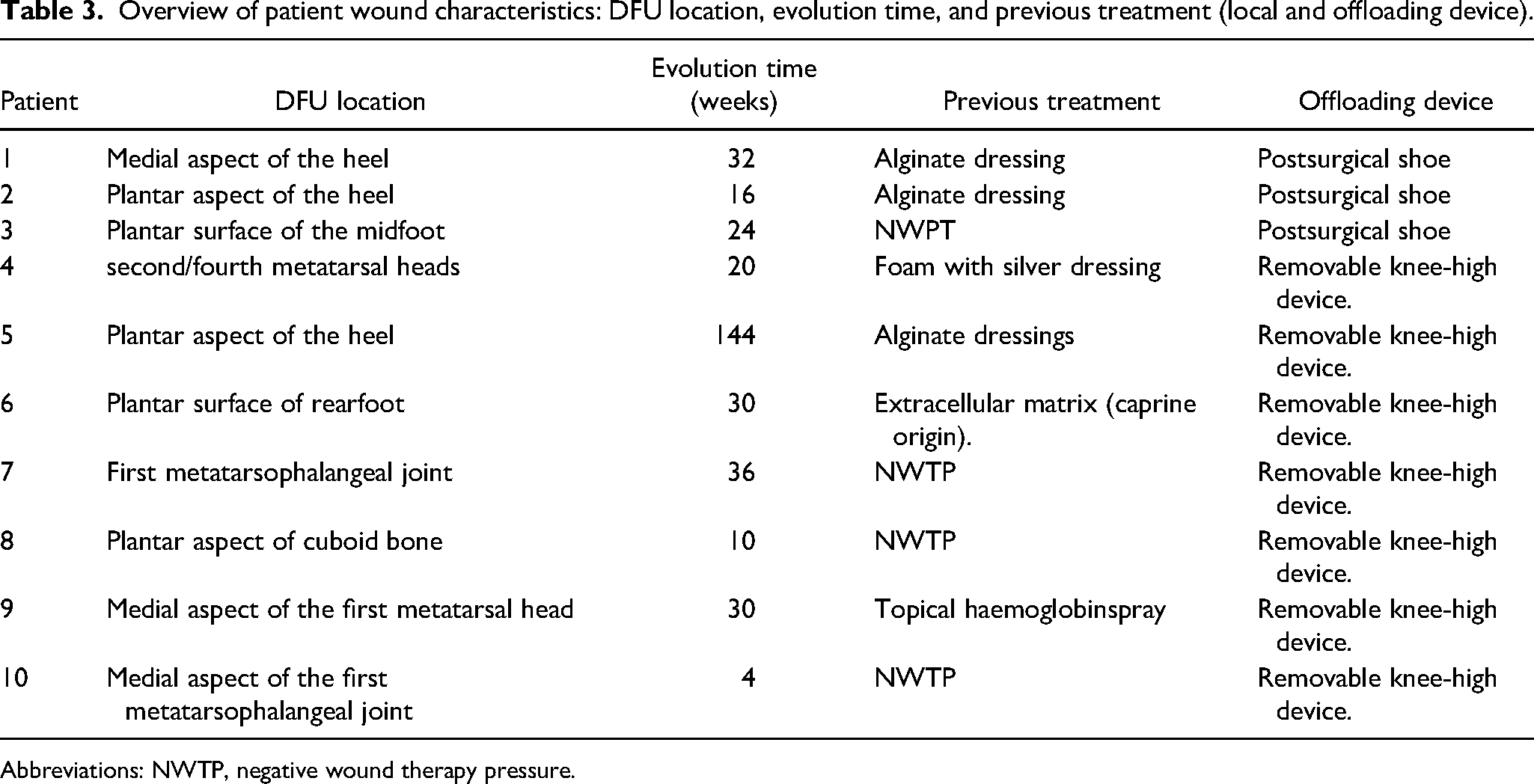
Abbreviations: NWTP, negative wound therapy pressure.
Individual case assessments revealed varied healing trajectories based on ulcer location and baseline condition which are shown in Figure 2a and b.
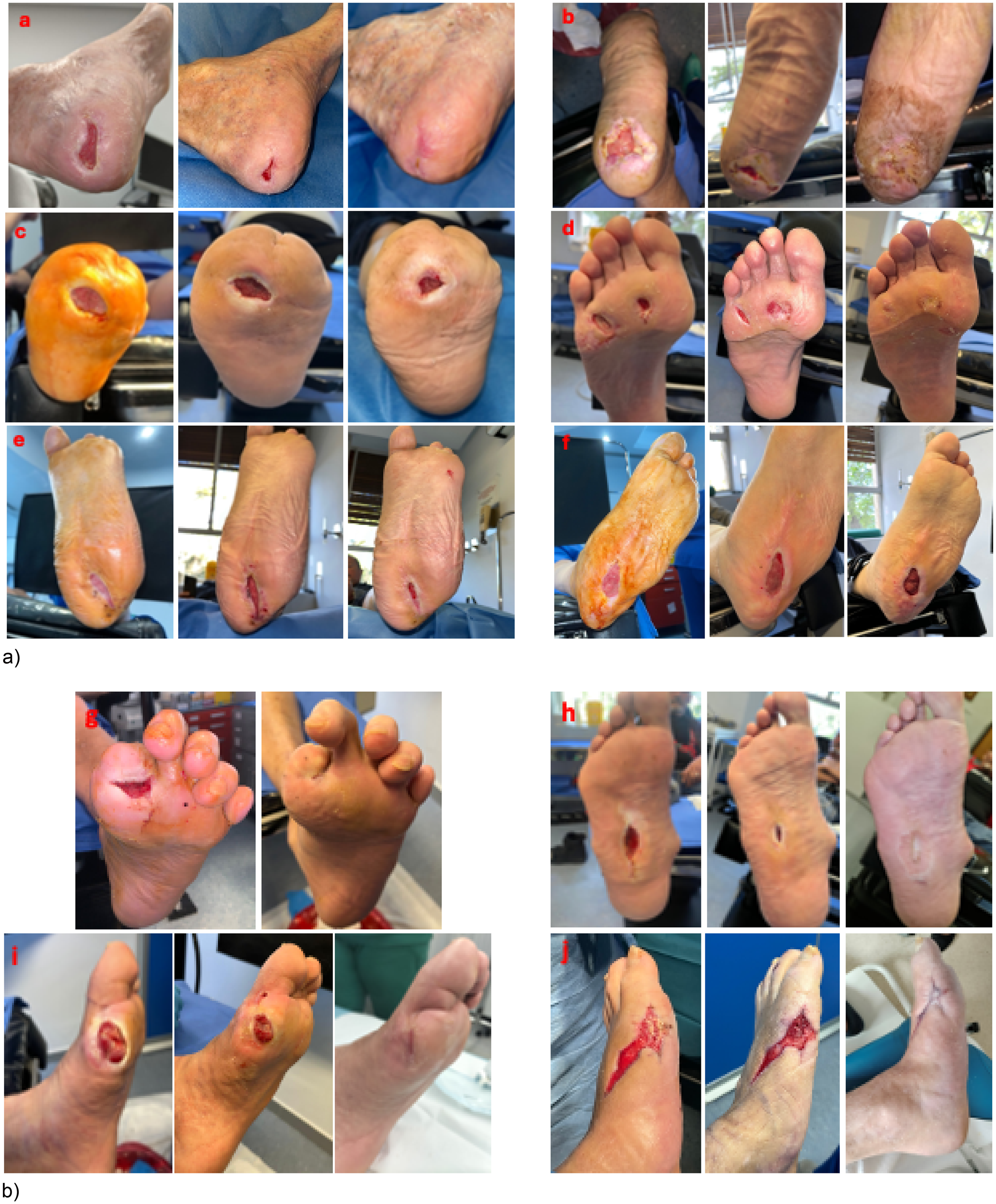
a. Clinical outcomes of DFU treated with HT-Micrograft at day 0, week 4 and week 12 or wound healing if occurred previously; (a) DFU located in the medial aspect of the heel (initial wound area: 4.1 cm2), complete epithelialization achieved in 10.7 weeks; (b) DFU located in the aspect of the plantar heel (initial wound area: 8.1 cm2) complete epithelialization achieved in 7 weeks; (c) DFU located in the plantar surface of the midfoot post-Lisfranc amputation (initial wound area: 2.3 cm2) showed 78% WAR at 12 weeks but was lost to follow-up afterward; (d) DFU located on the second metatarsal head (initial size: 0.5 cm2) achieved complete epithelialization in 4 weeks, while a concurrent ulcer on the fourth metatarsal head (initial wound area: 1.1 cm2) achieved complete epithelialization in 10 weeks; (e) DFU on the plantar aspect of the heel (initial size: 1.3 cm2) showed 84.64% reduction at 12 weeks but remains under treatment; (f) DFU on the plantar surface of the rearfoot (initial size: 4.8 cm2) showed 18.75% reduction at 12 weeks and remains under treatment. b. Clinical outcomes of DFU treated with HT-Micrograft at day 0, week 4 and wound healing; (g) DFU located at the plantar aspect of first metatarsophalangeal joint (initial wound area: 1.8 cm2) achieved complete epithelialization in 5 weeks; (h) DFU on the plantar aspect of the cuboid bone associated to Charcot foot (initial wound area: 1.8 cm2) achieved complete epithelialization in 8 weeks; (i) DFU on the medial aspect of the first metatarsal head (initial wound area: 3.0 cm2) achieved complete epithelialization in 14 weeks; (j) DFU on the medial aspect of the first metatarsophalangeal joint (initial wound area: 5.6 cm2) achieved complete epithelialization in 10 weeks.
Adverse Events
No adverse events or complications related to HT-MG therapy were observed throughout the study period, confirming a favorable safety profile for this treatment approach.
Discussion
The results of this case series study revealed a 60% wound healing rate at a 12-week follow-up period for patients with hard-to-heal DFU, averaging a healing time of 7.8 ± 2.6 weeks to achieve complete epithelization. We also noted a WAR of 67.2 ± 23.5% and 87.5 ± 24% after 4 and 12 weeks, respectively. Moreover, an additional DFU showed wound healing at 14 weeks, while three patients were still undergoing treatment at the end of the study follow-up period. These outcomes are promising, particularly given the complex nature of non-healing DFUs with an average evolution time of 25 [14.50–33] weeks – an ongoing challenge for clinicians and healthcare systems. 11
Several studies have demonstrated the potential benefits of using the conventional punch grafting technique on various wound etiologies.26,27 Our healing rate and time align with those reported in our previous research: 50% at 12 weeks in DFU with 172 [44–276] weeks of evolution time. 18 However, despite the DFUs having a longer evolution, most of them were superficial. In contrast, in the present study, 2 (20%) patients presented with bone and joint capsule exposure, achieving coverage at 4 weeks. This result stands out, as it opens up a new opportunity in the treatment of non-healing ulcers. These are typically managed with NWTP or dermo-epidermal skin substitutes, both of which are more expensive and require more specialized skills for their application on the foot.
Furthermore, the literature has explored various types of skin micrografts across different wound settings, with the level of success varying due to process heterogeneity. 28 Baglioni et al 29 reported a 75% healing rate by day 90 from using autologous micrografts derived from adipose tissue or dermis. This study involved a total of 14 patients with postsurgical dehiscence, with only two presenting DM. The main finding revealed that immediate efficacy resulted from fat-only applications, while a combination of dermis and fat prolonged benefits. Boggio et al 30 achieved a 90% wound healing rate using a manual minced micrograft technique on chronic vascular leg ulcers. This approach used a full-thickness skin specimen from a total of 20 patients.
Comparing our results with other studies on skin micrograft fragmentation procedures is challenging due to variations in the grinding process, application techniques,29–31 anatomical locations, and the types of chronic wounds evaluated. These include different etiologies to DFU where factors such as ischemia and neuropathy may delay wound healing.17,32
In addition to clinical efficacy, the cost-effectiveness of this type of procedure must also be considered. 33 One-third of the direct costs related to DM were attributed to the care for diabetic foot disease, with costs ranging from £3456 for an ulcer, to £9477 for an associated amputation per patient per year. 34 The situation worsens when DFU does not progress in a timely and orderly manner, with costs becoming 2.8 times higher in the second year of stagnation. 35
In this regard, though HT-MG may entail a relatively larger initial investment, it holds the potential for significant cost savings due to its expedited healing rates. Over 12 weeks, the average total cost of HT-MG treatment in an outpatient context is around 2200 €, which covers consumables and staff costs, whereas in a surgical context, including operating room expenses, the cost averages approximately 2600 €. This swifter healing trajectory not only eases the long-term financial strain on healthcare systems but also lessens the need for extended wound care treatments. The application of a standardized HT-MG process ensures consistent micrograft applications, which can lead to more dependable outcomes and lessen the cost of additional interventions compared to non-standardized procedures.
Additionally, HT-MG offers several advantages that are particularly relevant for the common characteristics of DFU, compared to other similar techniques such as punch grafting. First, HT-MG enables the treatment of plantar wounds where punch grafting is impractical due to the high risk of non-adherence. Moreover, HT-MG is faster and simpler than punch grafting, which necessitates precise dermal alignment for graft adherence and thereby extends the procedure time. In contrast, grinding and infiltration with HT-MG can be applied directly to the wound, streamlining the treatment process.
Other limitations related to DFU characteristics may potentially benefit from the use of HT-MG. A single application of HT-MG maximizes coverage with fewer biopsies. Only four 5 mm punches per cycle can cover up to 50 cm2, as DFUs do not typically exceed 30 cm2 in wound size. 36 In contrast, the baseline wound area determines the amount of punch grafting required, which can be more aggressive for larger ulcers, thereby increasing donor site morbidity. It is also mainly limited to superficial ulcers. Finally, the presence of ischemia in more than half of the patients with DFU and various degrees of vascular impairment restricts the application of punch grafting due to the need for subsequent management with a compressive bandage.
In addition to its methodological aspects, NWPT has demonstrated efficacy in promoting graft adhesion, specifically in complex anatomical locations and in the presence of high exudate levels. 18 Traditionally, NWPT is used following surgical debridement to encourage wound healing by augmenting granulation tissue formation. 14 However, DFUs treated with NWPT tend to stagnate after its removal, necessitating further interventions. The shift from NWPT to neutral dressings often fails to stimulate closure, potentially hindering progress. 14 HT-MG acts as a slow-release natural scaffold, continually delivering growth factors and other modulatory information in support of the regenerative process. 16 For this reason, it presents a promising strategy for promoting further healing, especially when incorporated after NPWT in stationary wounds.
Despite the favorable clinical results, the existing literature is sparse and the evidence remains unclear, with only one study specifically addressing conventional punch grafting on DFU, 18 and no studies explicitly evaluating skin micro-fragmentation in this population. Recently, innovative strategies rooted in regenerative medicine have drawn significant attention in the scientific community due to their potential to expedite wound healing. 16 Consequently, this is the first study in the literature to assess clinical outcomes resulting from the use of skin micrografts in hard-to-heal DFU.
Our results also present a few limitations that must be considered. As a case series, this study lacks a control group and randomization, which limits the strength of our conclusions. This design also increases the risk of selection bias, and higher-level studies are needed to confirm effectiveness.
Conclusion
Autologous micro-fragmented skin grafts have shown promising clinical outcomes in healing hard-to-treat DFUs. This approach could be considered a promising therapeutic alternative, and future studies should focus on increasing the evidence level of this innovative strategy.
Footnotes
Acknowledgments
The authors gratefully acknowledge the support of the staff and patients of the study center.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
