Abstract
This study compared subjective screening modalities recommended in diabetic foot screening guidelines for the detection of diabetic peripheral neuropathy (DPN) with an objective measure, the NC-Stat DPNCheck®. We assessed 63 participants (mean age 54.5 years ± 10.5) utilising subjective screening tools (Semmes-Weinstein 10-g monofilament, 128-Hz traditional tuning fork, neurothesiometer, O’Brien 128-Hz electronic tuning fork) and compared results with the objective automated sural nerve conduction test NC-Stat DPNCheck®. A significant difference was found in the number of limbs classified with DPN between all screening tools (P < .05). Therefore, this suggests that some screening modalities are more sensitive in diagnosing DPN than others, highlighting the importance of using multiple screening tools to a comprehensive understanding of the patient's neurological status. The findings also emphasize the need to incorporate objective measures in diabetic foot screening and encourage future research to establish a gold standard tool for DPN diagnosis.
Introduction
Diabetic peripheral neuropathy (DPN) is a prevalent complication of diabetes mellitus, often eluding timely diagnosis. Despite its high prevalence, there is a critical need for advancements in screening methods to facilitate early detection, especially since research indicates that up to 50% of DPN cases initially present with subclinical signs.1,2 The insidious nature of DPN underscores the importance of proactive measures for identification and management, aiming to prevent potential complications and improve the overall quality of life for individuals with diabetes. 3 Addressing the gaps in screening processes remains pivotal in mitigating the impact of DPN, as well as in promoting more timely and effective outcomes. 4
In an endeavour to improve screening, the NC-Stat DPNCheck® device has been developed for automated sural nerve conduction study. 5 This offers a non-invasive, objective test of sural nerve conduction with high sensitivity and specificity in identifying DPN. It may improve diagnosis and staging of DPN6,7 and shows good correlation with gold standard nerve conduction studies. 8 In contrast to subjective tools, the NC-Stat device is not influenced by clinician interpretation or patient co-operation. 6
Therefore, this study aimed to evaluate and compare different subjective screening modalities, currently recommended in diabetic foot screening guidelines for the detecting of peripheral neuropathy, with the objective NC-Stat DPNCheck®.
Materials and Methods
A prospective non-experimental study was conducted in 63 participants (126 limbs). The study was approved by the University Research Ethics Committee and all participants provided informed consent. All investigations were carried out in accordance with the principles of the Declaration of Helsinki as revised in 2000.
We included 63 participants. In each participant, the right and left foot were assessed (126 limbs). Inclusion criteria were: age 18-65 years, type 2 diabetes mellitus (T2DM) of at least 10 years duration, medical history or no history of DPN.9,10 Exclusion criteria were: type 1 diabetes mellitus, pregnancy, other causes of peripheral neuropathy or dysautonomia, thyroid disease alcohol abuse, liver disease, systematic infection or inflammation, implanted electronic devices, current ulceration and severe oedema.10–12
Study Protocol
Prior to subjects’ examination, the room was acclimatised between 25-30 °C. Maintaining a consistent room temperature was important, given that low temperatures could provide different readings due to its effect on nerve conduction. 6 All examinations were performed with the participant in a static and non-weight-bearing position. Participants were asked to expose their limbs up to the knee and lay supine on the clinical couch with their knee extended. Skin preparation utilising 70% alcohol was conducted. This method was adopted during data collection when utilising all the 5 different screening modalities for DPN which are described below. These subjective tools were selected for being widely used, user-friendly, readily available and recommended by diabetic foot screening guidelines. Furthermore, to minimize the risk of systematic bias, the subjective tests were conducted in a randomized order for each participant.
Subjective Tool: Semmes Weinstein 10-g Monofilament Testing
Participants were familiarized with the light touch sensation by applying the monofilament to the wrist. Ten-point test sites on each foot were tested in a random order and the examination was repeated three times at each site. 13 The participant was asked to look away and to confirm each time the monofilament was felt. If a participant failed to identify a point more than twice, that site was considered to have loss of protective sensation. If all 10 sites were felt, the participant was classified as having normal sensation, while if at least one site was not felt, the foot was classified as having absent sensation. 14
Subjective Tool: 128-Hz TTF Testing
The timed assessment method was chosen where this evaluates the duration a patient can detect tuning fork vibration and compares it with the clinician's perception. 11 Before assessment, participants were familiarised with the vibration sensation by application to their hand. The base of the tuning fork was then placed on the apex of the hallux and participants were asked to state when the vibration ceased. If vibration persisted for at least 10 s after participants no longer felt it, they were categorised as having absent sensation. On the other hand, those who felt the vibration stimulus were categorised as having normal sensation. The procedure was applied bilaterally and repeated 3 times: the average value was used. 15
Subjective Tool: Neurothesiometer
Participants were familiarised with the expected sensation by applying the stylus to their hand prior to testing and were asked to avert their eyes during examination to eliminate visual cues. The stylus was placed on the distal plantar aspect of the hallux, starting from 0 V and gradually increasing at a rate of 1 Volt per second until the initial sensation of vibration was reported. 16 Three examinations were performed on both feet: mean voltage was calculated and recorded. Depending on the mean voltage obtained, those with 0 to 20.99 V were classified as low risk, 21 to 30.99 V were classified as medium risk and ≥31 V were classified as having high risk of neuropathy. 17
Subjective Tool: 128 Hz ETF Testing
The O’Brien 128-Hz ETF is an easily operated and portable modality assessing the vibration perception thresholds. It produces an almost silent vibration with constant amplitude and a frequency similar to that of a tuning fork. Both the descending mode and the constant mode were chosen for this study since both methods mimic the timed vibration tests and the on/off test of the TTF, respectively.18,19
Descending mode: To initiate the assessment, the round contact tip was placed on the dorsal aspect of the distal phalanx of the hallux, just proximal to the nail bed. The ETF generated a 25-s vibration that gradually decreased in amplitude and participants were instructed to indicate the moment the vibratory sensation was no longer felt. The elapsed time was recorded in seconds and compared to a reference scale to determine neuropathy risk. This test was repeated three times on each foot, and the average duration was calculated. Participants with a mean of 7 to 25 s were categorised as having low risk, 4 to 7 s were categorised as having moderate risk and below 4 s were categorised as having high neuropathy risk.18,20
Constant mode: Participants were asked to close their eyes during the duration of this examination. The contact tip was placed on the hallux in the same anatomical area as that of the descending mode. The contact was maintained for approximately a second and this process was repeated twice consecutively. 18
In the first application, the ETF device was activated to produce a vibration, while in the second application, there was no vibration. Participants had to distinguish between the two and indicate in which application they felt vibration. This process was repeated three times for both feet, with randomised ETF device activation in each set to prevent bias. If participants could not differentiate between the two applications in more than two trials or vibration was not felt, then the foot is categorised as having absent sensation. However, if participants are able to distinguish the stimulus, then they were classified as having present sensation. 18
Objective Tool: NC-Stat® DPNCheck® Testing
Participants were positioned in a lateral recumbent position with the leg to be tested on top. The biosensor tail was inserted into the connector port and attached to the adhesive foam. Following this, the researcher set the leg to be tested on the device and applied conductive gel to the probes. The anode was aligned to the lateral malleolus bone and the cathode was placed adjacent to the central prominence of the lateral malleolus. The device was aligned to the lower lateral calf, pushed firmly against the biosensor foam and pointed towards the posterior knee. Mild pulses were delivered, and results included sural nerve conduction velocity (m/s) and amplitude (μV), assessed using a guide. Normal readings were above 40 m/s for velocity and above 4μV for amplitude. Participants were categorised based as “normal”, “mild neuropathy”, “moderate neuropathy” or “severe neuropathy”. Both limbs were tested.21,22
Statistical Analysis
All statistical tests were conducted using the IBM Corp SPSS (Statistical Package for the Social Sciences, International Business Machines Corporation, Armonk, NY, US) software. The Chi-squared Test for independence (also known as the Pearson's chi- square test) was utilized to analyse data, and the Kappa test was used to investigate inter-instrument reliability since the evaluations (present or absent sensation) had a categorical scale. The Kappa value ranges from 0 to 1 where a value close to 1 indicates satisfactory inter-instrumental reliability. The null hypothesis specified poor inter-instrument reliability and accepted if the P-value exceeds the .05 level of significance. The alternative hypothesis specified satisfactory inter-instrument reliability and accepted if the P-value is smaller than the .05 criterion.
Results
Among 63 participants, 39 (61.9%) were males and 24 (38.1%) were females. Mean age was 59.16 (44-65) years. Mean T2DM duration was 16.19 (10-38) years. Mean HBA1c was 7.89%.
The screening modalities used in this study had different ways in categorising the presence of neuropathy. Therefore, for the purpose of comparing all screening tools together, those with more than two categories were grouped as either having present sensation (in cases where the result suggested is normal or low risk of neuropathy), or absent sensation (for any other classification which ranges from mild to high-risk neuropathy). This was implemented to analyse and compare the results from all screening modalities used in this study (Figure 1). The NC-Stat® DPNCheck® had the highest percentage of limbs (32.5%) classified as having absent sensation. This was followed by the 128Hz-ETF constant (23.8%), 128Hz- ETF descending (23%), 128 Hz TTF (20.6%) and neurothesiometer (11.1%). The 10-g monofilament had the least percentage of limbs (4%) which were classified as having absent sensation. It can be noted that the percentage of absent sensation from the NC-Stat tool is significantly higher especially when compared with the neurothesiometer and the 10-g monofilament.
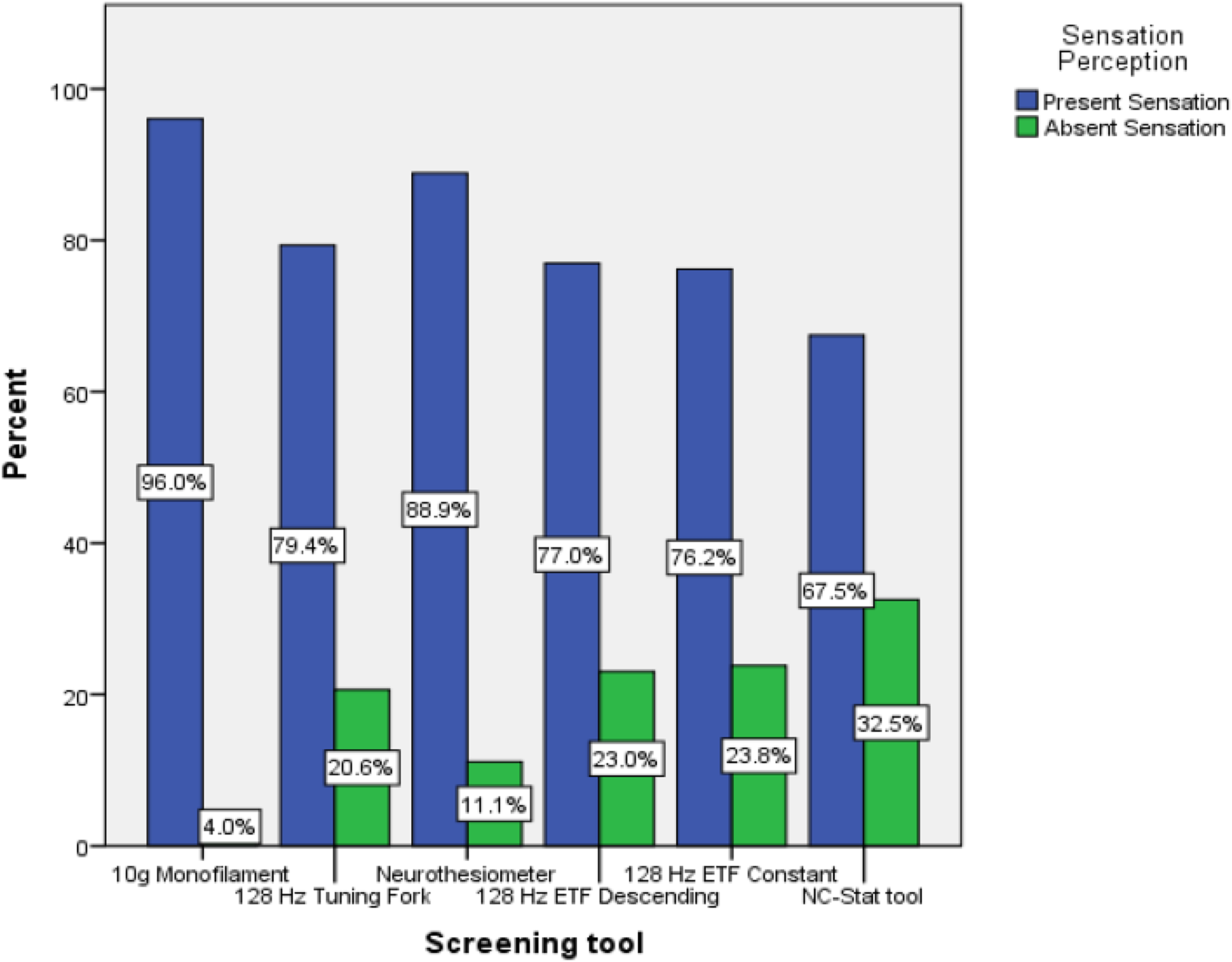
Percentage of limbs with present and absent sensation for each screening tool.
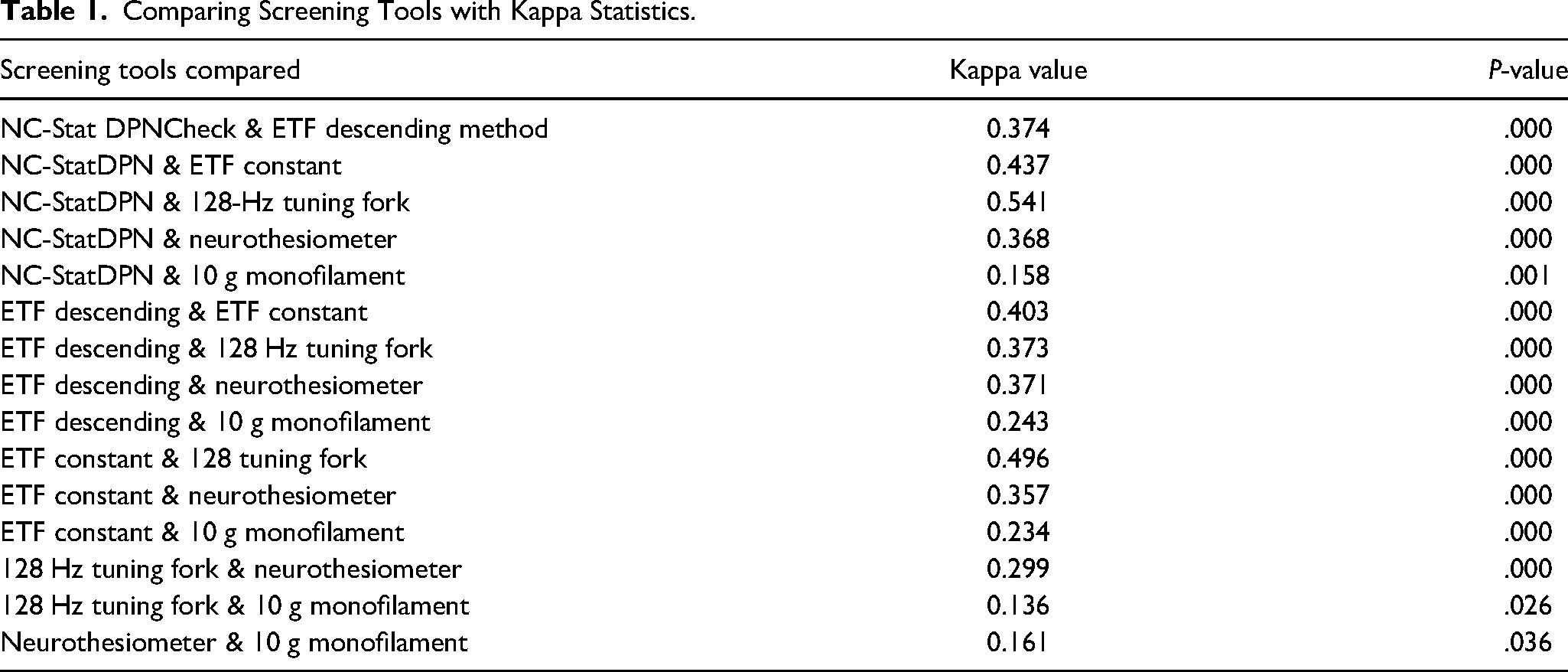
The Kappa test revealed that each of the screening tools yielded a P-value below .05 (Table 1), signifying acceptance of the alternative hypothesis and indicating adequate inter-instrument reliability. However, the 10-g monofilament exhibited the lowest level of agreement when compared with each screening tool, as indicated by the Kappa value obtained. Additionally, the remaining compared screening tools demonstrated only minimal to weak levels of agreement.
Comparing Screening Tools with Kappa Statistics.
Discussion
This study compared the agreement of commonly used non-invasive subjective clinical modalities versus an objective measure utilized for the detection of DPN in subjects living with Type 2 diabetes mellitus. As shown in our previous publication 5 years ago, 23 a significant disparity in the detection of DPN is present when using different screening modalities. This may suggest that certain screening modalities are more sensitive in detecting DPN compared with others, further highlighting the need for careful consideration when selecting screening methods. Lack of agreement among results obtained from all 5 screening tools indicates the potential occurrence of false positive or false negative outcomes for certain individuals.
Among the screening tools used in this study, the NC-Stat® DPNCheck® demonstrated the highest percentage (32.5%) of limbs with peripheral neuropathy. This finding aligns with previous research comparing the NC-Stat device with gold standard tools in detecting peripheral neuropathy, further validating its efficacy. The NC-Stat device has shown a strong correlation with the established gold standard NCS, as evidenced by multiple studies highlighting its excellent reliability, good correlation, and acceptable accuracy in the early detection of peripheral neuropathy.5,8
Several studies have reported that the NC-Stat® DPNCheck® showed high sensitivity and good specificity for the diagnosis of peripheral diabetic neuropathy in individuals with type 2 diabetes mellitus were percentages ranged from 84.3% to 95% and 68.3% to 86.11% respectively.6,7,24,25 The findings of this study may also suggest the potential implementation of the NC-Stat® DPNCheck® as an objective measure for diabetic foot screening assessment for early detection of peripheral neuropathy.
A strength of this study was the use of the objective NC-stat device, recognized for its accuracy in diagnosing peripheral neuropathy particularly in its early stages. 7 However, there are some limitations. The first relates to the small participant series. Another limitation was that the screening modalities chosen are not considered “gold standard” tools. Moreover, their choice was not only on their recommendation in various diabetic foot screening guidelines but also on their availability in the clinics. Further research by including a longitudinal follow-up study to evaluate the prognostic value of each screening tool, can enhance the clinical relevance of these findings and potentially influence screening protocols in clinical practice.
The practical implications of our findings may be outlined as follows. Clinicians can enhance the sensitivity of peripheral neuropathy screening by utilising multiple screening tools. This approach increases the likelihood of identifying cases, even in its early stages, potentially missed by individual screening modalities. Moreover, employing multiple tools allows for result validation and cross-referencing, thereby increasing confidence in diagnosis accuracy, as supported by previous studies.23,26 However, it is important to consider that while objective tools are generally more accurate, they can be expensive and less accessible in certain settings. Although the use of multiple subjective screening tools may be more cost-effective over time, it presents challenges related to time constraints and resource allocation in clinical practice. Furthermore, discrepancies between tools indicate the need for further investigation and prompt referral for neurological testing. In primary care, this practical approach may prove useful, but further experience is needed.
Furthermore, this study highlights the importance of standardised diabetes foot screening guidelines to ensure healthcare providers are equipped in assessing patients’ neuropathy risk accurately. Regular monitoring and close reporting are crucial when clinical test results are inconclusive, with screening frequency determined by the patient's risk status severity. In this endeavour, the NC-Stat® DPNCheck® offers a reliable and non-invasive screening option, providing objective data on nerve conduction, unaffected by patient-reported symptoms or subjective clinician interpretation. 27
In conclusion, advancements in technology for DPN screening should be used in practice. The emergence of innovative and sensitive diagnostic tools, like the NC-Stat® DPNCheck®, emphasise the necessity of revisiting screening modalities to ensure accuracy and efficacy. It now remains to explore how these may be used in primary care to enable early DPN diagnosis and avert its complications.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
