Abstract
This study aims to explore the effect in each stage of chronic kidney disease (CKD) on the major adverse cardiovascular events (MACE) in diabetes mellitus (DM) patients with peripheral arterial disease (PAD). A total of 246 DM patients with diagnosed PAD were enrolled in this study. Of these, 86 patients (35%) died and 34 patients had non-fatal cardiovascular events occurred at the last 7 years follow-up. The baseline eGFR obtained from the first quantified eGFR value within 6 months from the date of enrollment estimated using the Chronic Kidney Disease Epidemiology Collaboration (CKD-EPI). Then, based on eGFR at entry, we defined CKD as an eGFR < 60 mL/min/1.73 m2, and stratified all patients into four groups: eGFR-1, normal eGFR (≥90 mL/min/1.73 m2); eGFR-2, mildly decreased eGFR (60-89 mL/min/1.73 m2); eGFR-3, moderately decreased eGFR (30-59 mL/min/1.73 m2); and eGFR-4, severely decreased eGFR (<30 mL/min/1.73 m2). The mean eGFR was 54.4 ± 28.9 mL/min/1.73m2, and more than 30% of all patients had CKD (eGFR <60 mL/min/1.73m2). The seven-year cumulative incidence of MACE was 29.8% (95% confident interval [95% CI] 15.5-35.7) for eGFR-1 group, 40.4% (95% CI 27.4-45.2) for eGFR-2group, 66.2% (95% CI 47.6-71.4) for eGFR-3 group, and 94% (95% CI 75.0-99.0) for eGFR-4 group. In addition, after adjustment, hazard ratio (HR) for MACE was 2.36 (95% CI 1.26-4.40) in the eGFR-3 group and 7.62 (95% CI 3.71-15.66) in the eGFR-4 group. Restricted mean survival time (RMST) for survival analysis was consistent with HR in this study. After adjusting confounders, relative to eGFR-1 group, an association between the eGFR group and MACE outcome was found only in eGFR-3 group and eGFR-4 group. The moderate to severe reduction in eGFR, was an independent risk factor for MACE among DM patients with PAD throughout a 7-year follow-up duration. Thus, early CKD screening might be essential in the management of diabetic patients with PAD.
Keywords
The incidence of chronic kidney disease (CKD) in patients with peripheral arterial disease (PAD) is becoming higher and higher.1,2 This combination is a deadline combination, especially, in patients with the advanced stage of PAD (critical limb ischemia [CLI]). There are many reasons. Firstly, the chance of saving limb or successful revascularization is low.3,4 This is partly due to high calcification in the artery, so the successful chance of percutaneous transluminal angioplasty and even bypass is low. Secondly, this combination is associated with very high major adverse cardiovascular events (MACE) and death.5,6 Furthermore, diabetes is also associated with a high rate of mortality because diabetic patients tend to have premature atherosclerosis, including coronary artery disease and cerebrovascular disease.7–9 However, few studies have been performed in order to identify the risk factors of MACE in such combination in diabetic patients. We, therefore, explore further to explain the effect of each stage of CKD on the MACE in diabetic patients with PAD in our 7 years prospective database.
Material and Methods
Study Design
Diabetes patients from diabetic outpatient clinics across three tertiary hospitals, including Chiang Mai, Ching Rai, and Lamphun hospitals in northern Thailand who were diagnosed type 2 diabetes mellitus (T2DM) with first diagnosed with PAD were invited to conduct a long-term prospective observational study to evaluate the association between estimated glomerular filtration rate (eGFR) and MACE risk. The information on study design, patient eligibility (inclusion and exclusion criteria), schedule of appointed follow-up, and outcome measurements were described in our previous report. 10 In brief, patients eligible for the study included the following: those with T2DM with diagnosed PAD; aged ≥ 45 years; treating any hyperglycemic control ≥ 6 months before enrollment; diagnosed PAD by clinical presentation including chronic intermittent claudication (positive for simple questionnaire asking to have pain in the leg during usual walking and disappear when standing still), rest pain, and leg ulcer; or abnormal resting ankle-brachial index (ABI, VaSera VS-1500-Fukuda Denshi, Japan) ≤ 0.9 or with imaging study (ie, duplex ultrasound, digital subtraction/CT/MR angiography) showed vascular stenosis regardless of ABI was diagnosed PAD. 11 This study limits the definition of PAD to lower-extremity PAD.
The exclusion included patients who had a history of diagnosis with cerebrovascular diseases or coronary diseases and other comorbidities that affected life expectancies or vasculopathy such as advanced stage malignancy or acquired immunodeficiency syndrome (AIDS) which is the most advanced stage of HIV infection, fungal arteritis, and abdominal aortic aneurysm who had planned for surgery. Cases with noncompressible artery (ABI ≥1.4) and without clinical symptoms indicative of PAD were excluded.
The human experimentation ethics committee of the Faculty of Medicine, Chiang Mai University approved the study protocol in accordance with the Declaration of Helsinki (ethics reference number 2564-08149). All study participants gave written informed consent.
Clinical Outcomes
The primary outcome was MACE. MACE following a diagnosis of PAD was defined as a composite outcome including acute myocardial infarction, heart failure, stroke, and all-cause death. 12 The outcomes were evaluated every 6 months along with routine healthcare or by semiannual telephone follow-up by attending nurses. Patients who were uncontactable were censored at the date of last contact.
Covariates
Baseline characteristics, vital signs, physical examination, ABI testing, and family history of cardiovascular disease were obtained in the outpatient department at entry. Medical history and laboratory results were collected from medical records. The patient outcome and the primary cause of death were obtained from the hospital record and the death certificate from the death registration data of hospital and from governmental national registration department in case of changing hospital or loss to follow-up.
The baseline eGFR obtained from the first quantified eGFR value within 6 months from the date of enrollment estimated using the Chronic Kidney Disease Epidemiology Collaboration (CKD-EPI). Then, based on eGFR at entry, we defined CKD as an eGFR < 60 mL/min/1.73m2, and stratified all patients into four groups: eGFR-1, normal eGFR (≥90 mL/min/1.73m2); eGFR-2, mildly decreased eGFR (60-89 mL/min/1.73m2); eGFR-3, moderately decreased eGFR (30-59 mL/min/1.73m2); and eGFR-4, severely decreased eGFR (<30 mL/min/1.73m2).
Statistical Analysis
Descriptive analysis was carried out to characterize patients in the different eGFR groups. Categorical variables were presented as percentage and frequency, and continuous variables as means ± standard deviation (SD). The Shapiro–Wilk test was used to determine data normality for all variables. Fisher's exact and one-way ANOVA were used to compare among eGFR groups for categorical and continuous data, respectively.
The incidence of each outcome event was independently estimated as a hazard. The Kaplan–Meier approach was used to assess the cumulative proportion of patients suffering any of the MACE incidences over time. To assess the impact of eGFR groups on the rate of outcome events, Cox proportional hazard analyses for multi-event data by total time-restricted model were conducted. The proportional hazard was tested using Schoenfeld residuals. A multivariable Cox model was used to explanatory the independent effect of different eGFR categories on MACE, and subgroup analyses were used to determine whether the effect of eGFR was increased or decreased by the control of glucose level, blood pressure and blood lipid level at baseline according to the optimal target in T2DM with PAD. 13 Model was adjusted for age 60, sex, and prognostic variable for cardiovascular mortality based on previous research, including hypertension, dyslipidemia, history of smoking, body mass index (BMI), and PAD classification symptoms.14,15
We employed the restricted mean survival time (RMST) analysis is considered as a supplement to traditional Cox proportional hazards analysis. We pre-specified the time point at 7 years based on the maximum study time and estimated the area under the survival curve up to that point. Finally, we presented the changes in RMST and the difference in RMST as a function of time to realize how the negative effect of CKD changes over time.
Results
We examined 246 patients who arrived at our diabetic clinic from January 2014 to February 2015 (Table 1). The average age was 66.4 ± 10.1 years, with 90 males (36.6%) and 156 females (63.4%). The mean eGFR was 54.4±28.9 mL/min/1.73 m2, and more than 30% of all patients had CKD (eGFR <60 mL/min/1.73 m2). There were 79 patients in eGFR-1 group (eGFR ≥ 90 mL/min/1.73 m2), 91 patients in eGFR-2 (eGFR 60-89 mL/min/1.73 m2), 60 patients in eGFR-3 (eGFR 30-59 mL/min/1.73 m2), and 16 patients in eGFR-4 (eGFR < 30 mL/min/1.73 m2). 13 cases were starting on hemodialysis therapy at time of enrollment, and all were in eGFR-4 group.
Baseline characteristics of participants with diabetes and PAD in different eGFR groups.
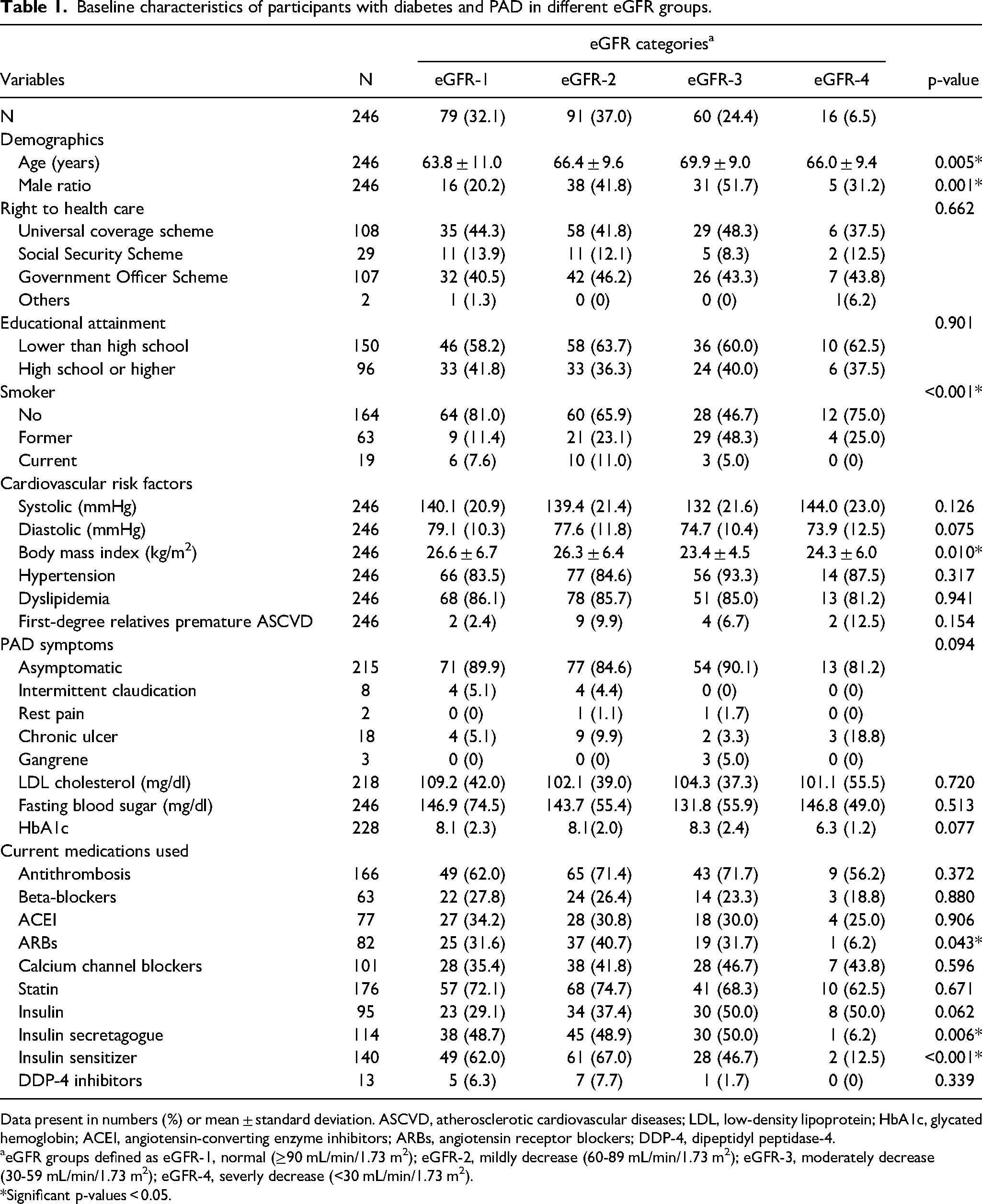
Data present in numbers (%) or mean ± standard deviation. ASCVD, atherosclerotic cardiovascular diseases; LDL, low-density lipoprotein; HbA1c, glycated hemoglobin; ACEI, angiotensin-converting enzyme inhibitors; ARBs, angiotensin receptor blockers; DDP-4, dipeptidyl peptidase-4.
eGFR groups defined as eGFR-1, normal (≥90 mL/min/1.73 m2); eGFR-2, mildly decrease (60-89 mL/min/1.73 m2); eGFR-3, moderately decrease (30-59 mL/min/1.73 m2); eGFR-4, severly decrease (<30 mL/min/1.73 m2).
*Significant p-values < 0.05.
Patients with the normal eGFR group were younger and more likely to be female than those with decreased eGFR groups. A decrease in eGFR was associated with smoking and lower BMI. In the severe eGFR group, there was a low proportion of using angiotensin receptor blockers, an insulin secretagogue, and insulin sensitizers.
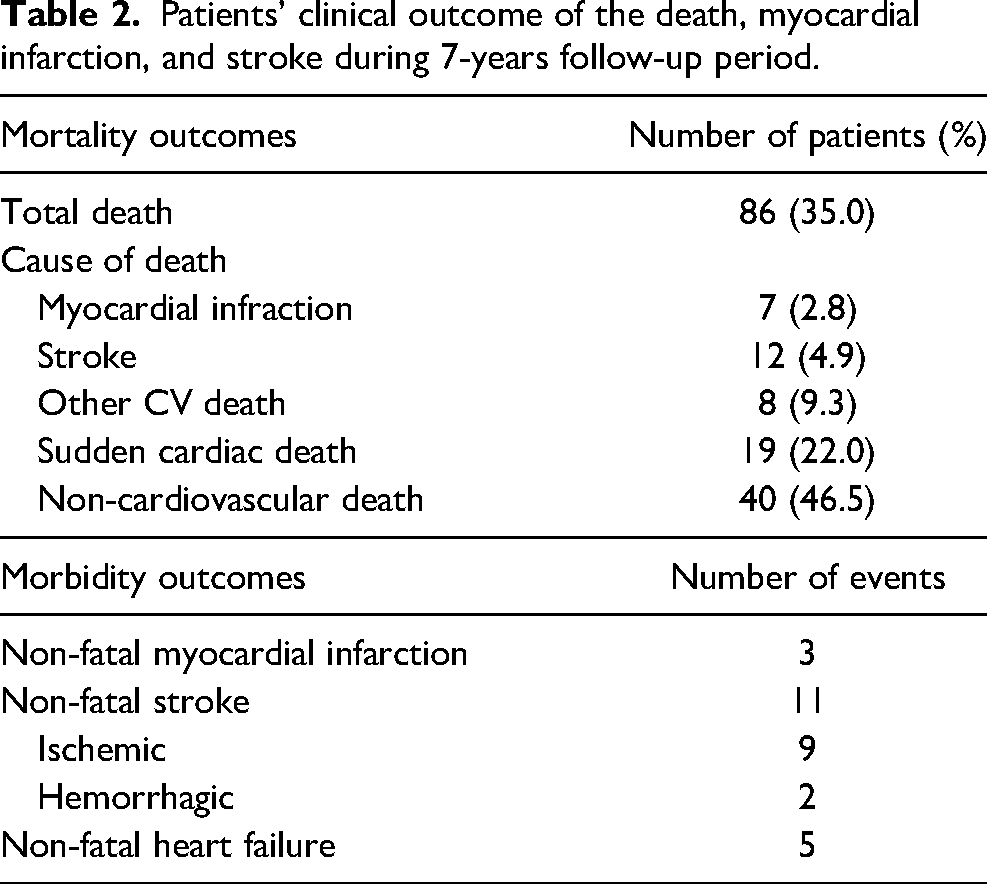
Eighty-six (35%) of these patients died, and 34 non-fatal cardiovascular events occurred at the last follow-up on February 28, 2021 (Table 2). We determined the MACE risk of the different eGFR groups using Kaplan-Meire analysis (Figure 1). The median time of follow-up period was 6.5 years. The 7-year cumulative incidence risk and 95% confidence interval (CI) for eGFR-1, eGFR-2, eGFR-3, and eGFR-4 were 29.8% (95%CI 15.5-35.7), 40.4% (95%CI 27.4-45.2), 66.2% (95%CI 47.6-71.4), and 94% (95%CI 75.0-99.0), respectively. For pair-wise comparison, they indicated a significant difference in survival of eGFR-1 versus eGFR-3 and eGFR-1 versus eGFR-4 groups (all p < 0.001). Another comparison for eGFR-2 versus eGFR-3 and eGFR-2 versus eGFR-4 showed marginally significant differences (p = 0.063 and 0.081, respectively). However, all other pair-wise comparisons indicated no significant difference in 7-year survival.
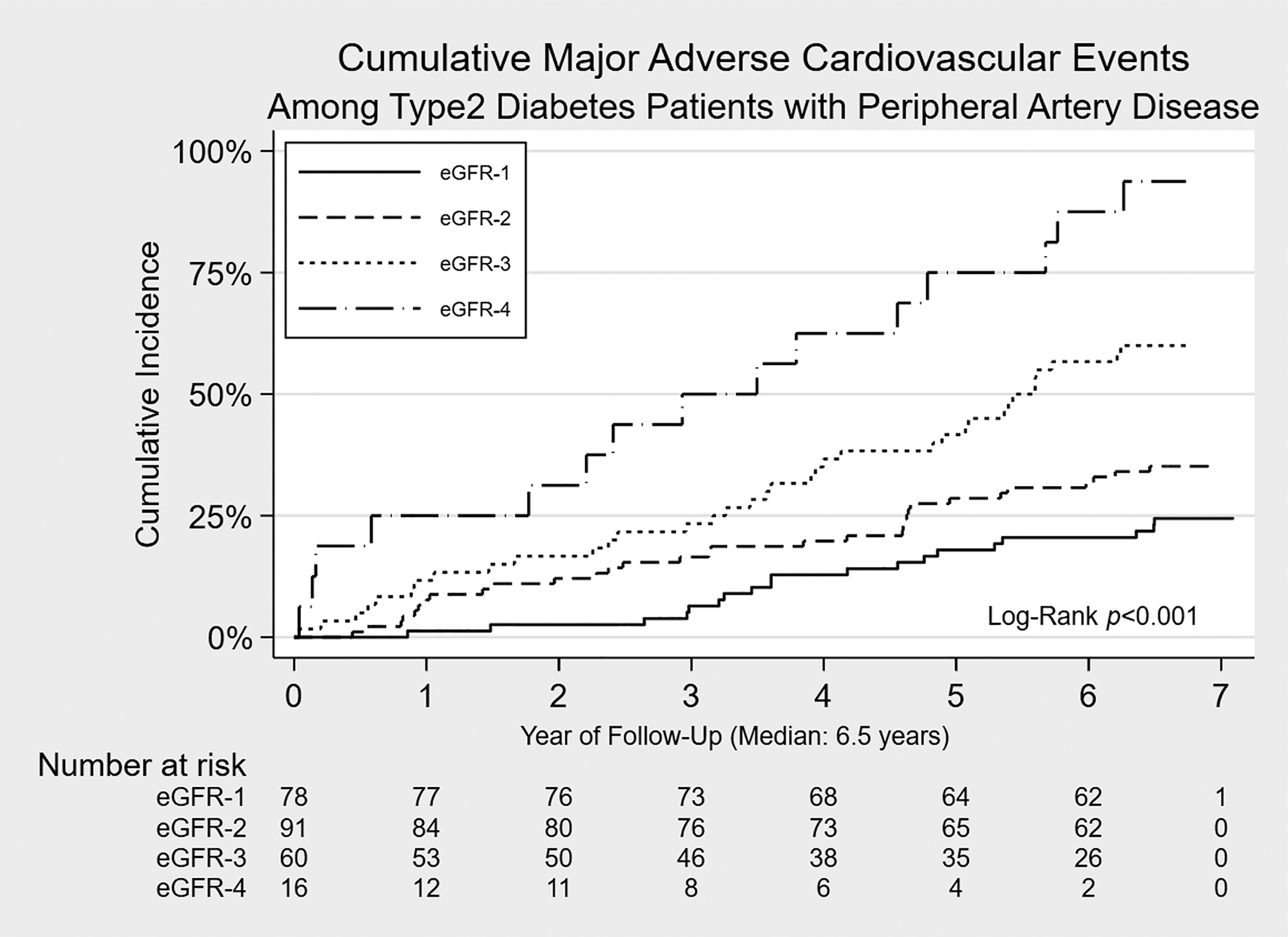
Kaplan–Meier analysis of MACE in the different eGFR groups. Abbreviation and eGFR groups are defined in Table 1.
Patients’ clinical outcome of the death, myocardial infarction, and stroke during 7-years follow-up period.
We also performed a Cox proportional hazard model to compare survival times in the different eGFR groups (Table 3). After adjustment, HR for MACE was 2.36 (95% CI 1.26-4.40, p = 0.007) in the eGFR-3 group and 7.62 (95% CI 3.71-15.66, p < 0.001) in the eGFR-4 group. However, no significant difference was in MACE in eGFR-1 and eGFR-2 (HR 1.27, 95%CI 0.71-2.27, p = 0.419, respectively).
Results of proportional Cox regression analysis of risk factors associated with MACE in type 2 diabetic patients and peripheral arterial disease (PAD) at 7 years follow-up.
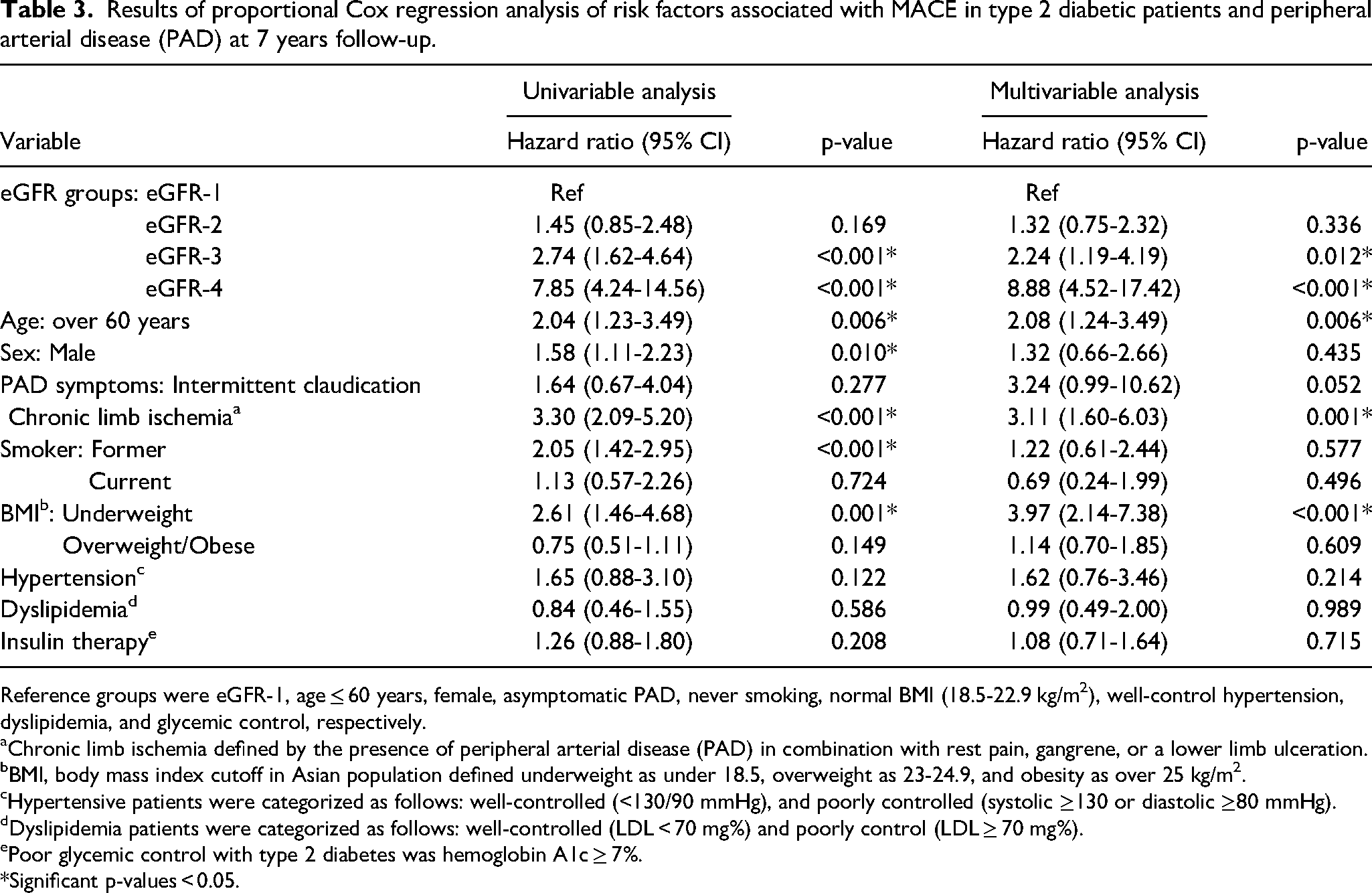
Reference groups were eGFR-1, age ≤ 60 years, female, asymptomatic PAD, never smoking, normal BMI (18.5-22.9 kg/m2), well-control hypertension, dyslipidemia, and glycemic control, respectively.
Chronic limb ischemia defined by the presence of peripheral arterial disease (PAD) in combination with rest pain, gangrene, or a lower limb ulceration.
BMI, body mass index cutoff in Asian population defined underweight as under 18.5, overweight as 23-24.9, and obesity as over 25 kg/m2.
Hypertensive patients were categorized as follows: well-controlled (<130/90 mmHg), and poorly controlled (systolic ≥130 or diastolic ≥80 mmHg).
Dyslipidemia patients were categorized as follows: well-controlled (LDL < 70 mg%) and poorly control (LDL ≥ 70 mg%).
Poor glycemic control with type 2 diabetes was hemoglobin A1c ≥ 7%.
*Significant p-values < 0.05.
The second analysis of RMST is shown in Table 4. At the end of the follow-up, the direction of the RMST estimate was consistent with HR from Cox modeling. After adjusting confounders, relative to eGFR-1, an association between the eGFR group and MACE outcome was found only in eGFR-3 and eGFR-4. Patient free of MACE was significantly shortest in eGFR-3 and eGFR-4 than eGFR-1 group (−1.0 year, 95% CI −1.6, −0.4, p = 0.002 and −3.0 years, 95% CI −2.2, −3.9, p < 0.001, respectively). No significant difference in MACE-free survival among patients with eGFR-1 and eGFR-2 (p = 0.166).
Event-free time (RMST) with 95% confidence interval among type 2 diabetes with peripheral arterial disease by eGFR status on MACE outcome.

Model were adjusted for age > 60 years, sex, and other prognostic variables, including PAD symptoms (asymptomatic, claudication, chronic limb ischemia), BMI categories (underweight, overweight/obesity), hypertension, dyslipidemia, and insulin therapy. *Significant p-values < 0.05.
Since our participant was associated with high percentage of history of hypertension. We then performed a subgroup analysis to determine whether specific risk factor management differences increased or decreased the effect of eGFR on MACE. Stratified by hypertension, management at baseline, after adjustment, indicate that relative to the eGFR-1 group, poorly control hypertension in eGFR-2 to eGFR-4 and well-control hypertension in eGFR-2 to eGFR-4 had significantly increased MACE (all p < 0.05). The HR (95% CI) in eGFR-2, eGFR-3, and e GFR-4 in poorly controlled hypertension group was 2.25 (1.02-5.00), 2.60 (1.10-6.23), and 10.63 (4.19-27.02) respectively. The same figures in the well-controlled hypertension group were 0.58 (0.19-1.70), 2.50 (0.97-6.40), and 6.28 (2.43-16.19), respectively.
Discussion
Our study illustrated that reduction in eGFR, ranging from moderate to severe, was an independent risk factor for MACE among T2DM patients with PAD throughout a 7-year follow-up duration. This trend is similar to other studies, which focus only CKD in PAD patients.1,5 Additionally, the influence of this moderate to severe eGFR decline on MACE within these patients exhibited a sustained elevation, even after adjustments for covariates linked to cardiovascular mortality. We also found that making risk factors modifications to glycemic control, and hypertension management could impact on the outcomes of these patients. Individuals with poorly controlled hypertension appeared to experience adverse consequences concerning MACE across all stages of eGFR decline.
Our finding is consistent with many previous studies. Fallon et al studied based on a registry of 689 hemodialysis patients undergoing lower extremity revascularization. The overall survival at 1, 2, and 5 years survival rate was low at 60%, 43%, and 21%, respectively. 6 This study identified another five independent risk factors (age > 80 years, coronary artery disease, COPD, low ambulation status, and rest pain/tissue loss). The predicted risk of death at one-year survival after revascularization in 0, 1, 2 and at least three factors is 29%, 33%, 40%, and 64%. This might be a very useful information to discuss risk/benefit in selecting the type of treatment in HD patients with CLI. Overall, the prognosis of hemodialysis patients with PAD is even worse than most malignant diseases. 16
The mechanism of high mortality in PAD patients with CKD might be due to highly associated comorbidity. In our study, the history of hypertension in eGFR-1, eGFR-2, eGFR-3, and eGFR-4 were 83.5%, 84.6%, 93.3%, and 87.5%, respectively. These factors can make systemic atherosclerotic plaque in an unstable stage and prone to rupture and eventually thrombosis. Albert et al. presented that 3-year survival of HD patients underwent bypass surgery for CLI (41%) was similar to HD patients underwent coronary artery bypass surgery (42%).3,17 These findings seem illustrated that clinically severe atherosclerotic disease is a major factor of low survival in HD patients.
Early referral in HD patients with PAD to expert is of important. Even referral to expert, studies including our study showed high mortality after treatment. Therefore, before the decision to make for any invasive intervention namely revascularization, clinicians, interventionists, and surgeons should discuss with patients and relatives by taking high mortality risk into account. Perhaps, according to 5 factors in Fallon et al. works, might be helpful in discussion. 6 Although the regular PAD screening protocol in the CKD population is not available. Based on our findings, this should be developed. The early PAD detection might benefit in initiation of proper PAD management including regular surveillance, proper self-foot care, proper exercise therapy, adequate medical treatment and revascularization. Also, this protocol might help in prompt assessments in systemic conditions, such as cardiac ischemia, and cerebrovascular ischemia. As our study, the cause of 7 years follow-up death due to cardiovascular problem is 53.5% (46/86). Of course, this guideline should include intensive medical treatment for atherosclerosis according to Global vascular guideline. Currently, a global vascular guideline on the management of CLI in 2019 suggests the application of metabolic-lowering agents such as antihypertensive, antithrombotic, glycemic control, or lipid-lowering agents in CLI patients to decrease rates of major amputation, increase patency after revascularization and increase survival. 18 Our study was also found in patients with hypertension, well-controlled hypertension group had lower HR of MACE than those with poor controlled hypertension group.
PAD is a strong predictor of cardiovascular mortality in the general and dialysis patients.19,20 According to the KDOQI clinical practice guidelines for cardiovascular disease in dialysis patients, all patients should undergo an evaluation for the presence of PAD at the initiation of dialysis. This evaluation should include a physical examination, which involves assessing arterial pulse and skin integrity. Patients with PAD should receive treatment that addresses smoking cessation, lipid-lowering therapy, glycemic control, blood pressure management, and the use of ACE inhibitors and antiplatelet agents to improve cardiovascular survival in dialysis patients. Furthermore, supervised exercise regimens and medications to enhance vasodilation should be considered for patients with claudication but without critical leg ischemia. 21
Limitation
Very poor survival was found in the CKD stage 4-5 (eGFR-4 group), but the number of these patients is very small in our study (16 cases). Thus, further large studies are needed. Also, in the large studies, this might be able to identify the subgroups of patients with good reasonably good life expectancy and who might obtain benefit from revascularization procedure. Our study based on tertiary hospital, so this data might not generalize all level of medical cares. Further studies are needed to confirm our finding.
Conclusion
Our study found that reduction in eGFR, ranging from moderate to severe, was an independent risk factor for MACE among T2DM patients with PAD throughout a 7-year follow-up duration. The early PAD detection in CKD patients might benefit in initiation of proper PAD management. More studies are needed to explore subgroup that be benefit to identify proper treatment in each patient.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This project was partially supported by Chiang Mai University.
