Abstract
Diabetic foot infection (DFI) is a common problem in patients with diabetic foot disease. Amputations and other complications of DFI lead to significant morbidity and mortality. The Society for Vascular Surgery (SVS)-WIfI (wound, ischemia, and foot infection) classification system can evaluate the benefit from revascularization and the risk of amputation in 1 and 3 years. We aimed to evaluate SVS-WIfI and Wagner-Meggitt (WM) prediction of DFI outcome, and to determine factors associated with major amputation and mortality rate. The patients with diabetes who presented between June 2018 and May 2020 with characteristics suggesting a more serious DFI or potential indications for hospitalization were reviewed in this cohort study. Demographic data, clinical characteristics, and type of revascularization were evaluated. One-year and 3-year amputation and mortality rates were the main outcomes. The grading of WM classification and the SVS-WIfI score were compared between amputation and nonamputation groups. Association between mortality and comorbidity were analyzed. One hundred and thirty-one patients admitted with DFI were included in study. And 73.28% had peripheral arterial disease (PAD). The 1-year and 3-year major amputation rates were 16.03% and 26.23%, respectively. Seventy-eight (59.54%) patients required minor amputation to control infection before revascularization. PAD (risk ratio [RR] 1.47: 95% confidence interval [CI] 1.29-1.67, P = .032), benefit from revascularization clinical stage 3 on SVS-WIfI score (RR 4.56: 95%CI 1.21-17.21, P = 0.007), and high WM classification score (RR 9.46: 95% CI 5.65-15.82, P < 0.001) were associated by multivariate analysis with high amputation rates. 1-year & 3-year amputation risk on SVS-WIfI score were not associated with amputation rates in DFI (P = .263 and .496). Only 9 (6.8%) patients were lost to follow up during the 3-year period. WM classification score, SVS-WIfI score on benefit from revascularization, and PAD were strongly associated with major amputation rates in patients with DFI.
Keywords
Introduction
The global prevalence of diabetes mellitus (DM) is increasing. The International Diabetes Federation reports that 463 million people have DM, which accounts for an estimated 10% of global health expenditure. And 79% of patients with DM live in lower to middle income countries. 1 In Thailand, the prevalence of DM is estimated to be 8.69%, or 5 million people. 2 One-fourth of patients with DM develop a diabetic foot ulcer (DFU), which is a severe complication of DM.2–4 The mortality rate from diagnosed DFU is 31.9%. Peripheral neuropathy induced foot deformities, loss of protective sensation, anhidrosis, and arteriovenous shunt are risk factors for recalcitrant DFU. Arteriovenous shunting was found to be directly related to autonomic neuropathy which lead to venous distension and unhealed ulcer.4,5 Microvascular and macrovascular complications of DM cause lower extremity angiopathy, namely peripheral arterial disease (PAD), which leads to poor tissue oxygenation at a DFU.5,6 DM has been associated with a 4-fold increase in the prevalence of PAD. 7 If angiopathy progresses to the critical stenotic level, patients have a risk for limb loss or amputation.4,5,8,9 Chronic limb-threatening ischemia (CLTI) is the end stage of PAD, which has highest risk for amputation (30%) and mortality (25%).10,11 In addition, other vascular complications, including cerebrovascular disease (CVD) and coronary artery disease (CAD), are common comorbidities in patients with DM. These comorbidities increase mortality rates for patients who need urgent operations to treat DFU, such as debridement with or without revascularization.8,12–14
Diabetic foot infection (DFI) is a common presentation of DFU. The chemotaxis dysfunction and poor blood supply to the foot lead to the local infection process which can progress to systemic infection and stimulates systemic inflammatory response syndrome (SIRS). CAD is the most common cause of death in patients with DFU with PAD.12,15,16 However, DFI is an important factor increasing amputation and mortality rates.4,17–19
The Wagner-Meggitt (WM) classification is a world-wide traditional classification system to predict amputation of lower extremities in diabetic patients. It is easy to use in clinical practice, and does not require adjunctive devices.20,21 Risk factors of amputation, including wound extension, severity of infection, and gangrene are taken into account in the WM classification, but systemic infection and hemodynamic parameters to evaluate the severity of ischemia are not.20,21
In 2014, the Society for Vascular Surgery (SVS) published the SVS-WIfI (wound, ischemia, and foot infection) classification system which can evaluate the benefit of revascularization and the risk of amputation within 1 year.11,22 The new SVS-WIfI classification system, which documents the wound, ischemia, and infection dominants objectively, is the most recent classification of DFI. The severity of ischemia is evaluated by ankle pressure, ankle brachial index (ABI), toe pressure (TP), and transcutaneous pressure oxygen measurement (TcPO2). The wound part of SVS-WIfI is separated into a gangrene and an ulcer of lower extremity. However, limitations in technique for measuring the severity of ischemia and the dynamic pattern of infection and ischemia after debridement, minor amputation, and revascularization cause a difficult interpretation of amputation risk.4,20,23 The aim of this study was to determine the predictive factors associated major amputation and mortality rate. SVS-WIfI score, and WM classification were evaluated for prediction of risk of amputation. The incidence of amputation and mortality rates in patients with DFI were explored.
Materials and Methods
Study Design
The patients with DM who presented between June 2018 and May 2020 with characteristics suggesting serious DFI or indications for potential hospitalization due to DFI were included in this study consecutively. Baseline demographic data, clinical characteristics, predictive factors, SVS-WIfI classification, WM classification, amputation, and mortality data were collected from the electronic medical database retrospectively. There are a lot of literatures of the validation of the SVS-WIfI and WM classification. The studies included the patients in setting of both primary hospital and tertiary care. So, the use of WIfI can apply not only vascular center but also primary care to evaluate the patients to early referral or continuing treatment in the primary hospital.11,17,22,24
Data Sources and Study Cohort
We assessed all patients who were admitted between June 2018 and May 2020 with DFU by the electronic medical database of Thammasat University Hospital. Inclusion criteria were (1) new onset DFI including DFU or gangrene with evidence of infection8,11,17,22,24,25 or (2) chronic infected foot or leg ulcer for more than 2 weeks, with new diagnosis of DM in the same admission.4,17–19 We excluded patients with DFI who (1) had recurrent ulcer, (2) had previous revascularization due to new onset DFU before admission,8,9,11 or (3) were unfit for debridement and/or revascularization due to poor performance status or comorbidities, such as decompensated heart failure or other active cardiac conditions, acute cerebrovascular events, or uncorrected end stage disease, for example, advance stage cancer patients. 14 In-hospital DFU treatment criteria included wound grade of SVS-WIfI score or WM classification score ≥ 2, moderate to severe DFI (the International Working Group on Diabetic Foot [IWGDF]/The Infectious Diseases Society of America [IDSA]), concomitant PAD, and any characteristics suggesting a more serious DFI or potential indications for hospitalization.
The clinical study protocol was accepted and gets the certificate of approval by Human Research Ethics Committee of Thammasat University (Medicine) (No. 065/2023). The study process and report followed the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) statements in Reports of Cohort Studies (Figure 1).26,27 After admission, a multidisciplinary team planned treatment. All patients had individual plans with sequential monitoring of the results of the treatments. First, the infection was treated by broad spectrum intravenous antibiotic, debridement, and minor amputation of necrotic tissue; all superficial and deep spaces of foot abscesses were drained by standard incision.4,17,28 Vascular examination and ABI screening were performed in all DFI. All patients who were documented with CLTI by clinical and hemodynamic criteria were preoperatively prepared and optimized by a cardiologist and an endocrinologist. A computed tomographic angiography of the lower extremity was performed in all patients with CLTI who were candidates for revascularization.8,11 Endovascular therapy (EVT) or open surgical bypass were selected considering preoperative risk, comorbidities, vein graft conduit availability, anatomic lesion, degree of tissue loss, and surgeon preference.8,9,11,15 Definite closure of the wound depended on the area and location of the defect. Split thickness skin graft or flap was performed for large defects after wound bed preparation by plastic and reconstructive surgeons.4,29 Offloading techniques were performed by physical medicine and rehabilitation doctors after wound completely healed. For minor amputation patients, offloading shoes was performed under foot pressure analysis and mapping. The prosthesis was offered for all major amputations who candidate for continuing the ambulatory status. For the surgical offloading, the patients were planned to do the surgical correction by foot and ankle orthopedic doctors in patients with well-vascularized feet and good risk for surgery in ambulatory patients. The DM and other underlying diseases were minimized by internal medicine specialist doctors. 4 The patients were followed up until the wounds or stumps were healed.
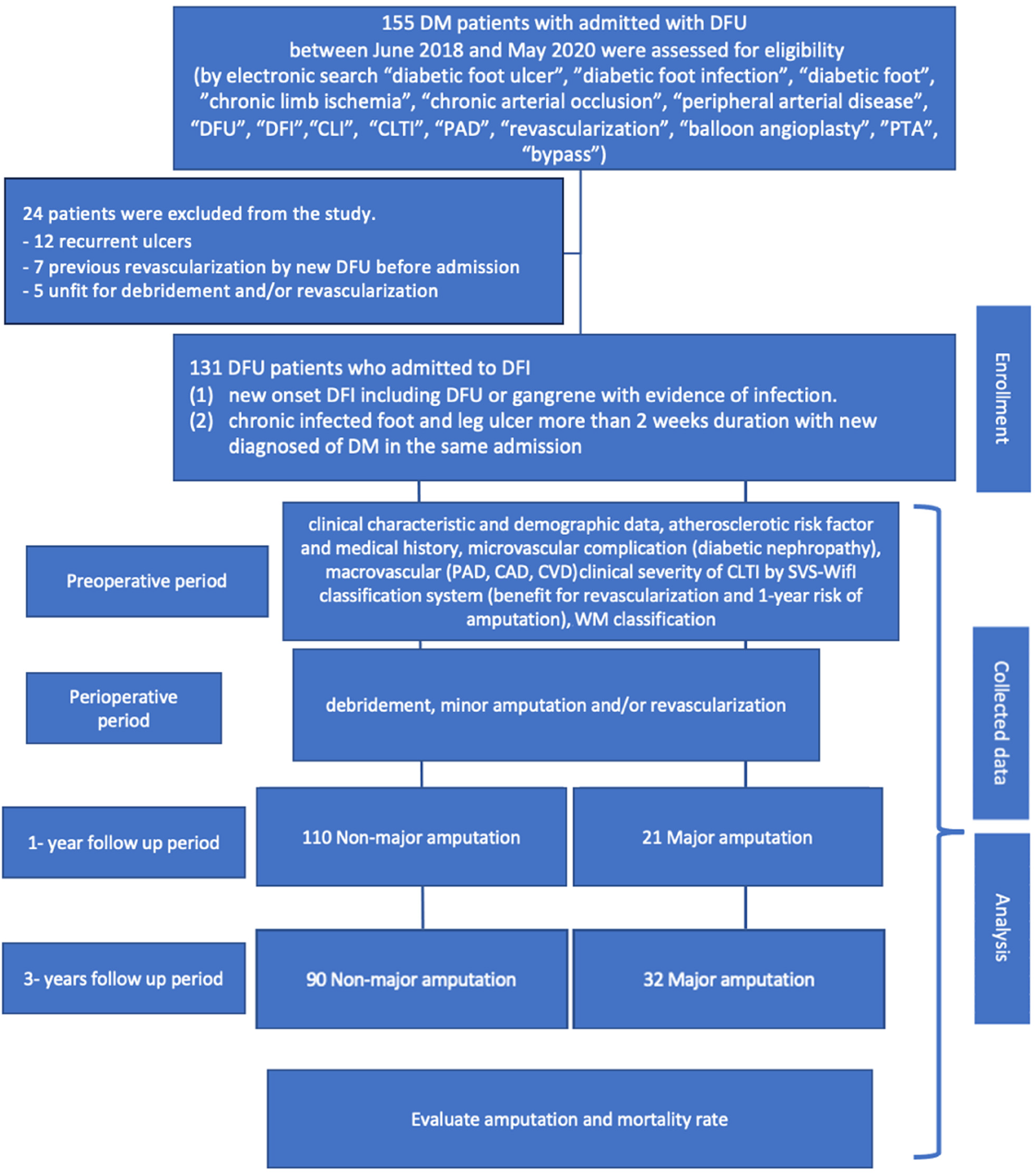
The study flow diagram of cohort study (STROBE Statement, 2008). tStatistically significant at P < .05. Abbreviations: CAD, coronary artery disease; CVD, cerebrovascular disease, CLI, critical limb ischemia; CLTI, chronic limb-threatening ischemia; DFI, diabetic foot infection; DFU, diabetic foot ulcer; PAD, peripheral arterial disease; PTA, percutaneous transluminal angioplasty; SVS, Society for Vascular Surgery; WIfI, wound, ischemia, and foot infection; WM, Wagner-Meggitt classification.
Study Outcomes
Data including clinical characteristics and demographic data, atherosclerotic risk factor, medical history (hypertension, dyslipidemia, and smoking),9,15 microvascular complication (diabetes nephropathy), macrovascular complication (PAD, CAD, and CVD),4,12,15–19,30 clinical severity of CLTI by SVS-WIfI classification system (benefit from revascularization and 1-year risk of amputation),11,22 WM classification,24,31 revascularization,10,11 minor amputation, 12-month amputation, and mortality12,16,32–34 were collected retrospectively from the electronic medical database at preoperative, perioperative, and postoperative follow-up periods.
Statistical Analysis
Associations between clinical characteristics and demographic data, atherosclerotic risk factor, SVS-WIfI classification by 1-year risk of amputation and benefit from revascularization, WM classification, revascularization status, and other predictive factors were analyzed by univariate 2-sample t-test for continuous data and chi-squared (χ2) test for categorical data. Parametric data were presented as the mean with standard deviation (SD). Nonparametric data were presented as the median value with interquartile range (IQR). Multivariate analysis used logistic regression analysis. The relationship between predictive factors, amputation, and mortality rate were also analyzed. The P-value <.05 was considered statistically significant. The power was 0.80. All the statistical analyses were performed with STATA/SE 16.0 for Mac (StataCorp, TX, USA).
Sample Size Calculation
PAD was the most important factor to determine the risk of amputation in patients with DFU.7,23 Previous data demonstrated that concomitant DM and PAD were associated with a 4-fold increase in amputation risk. Amputation rates of 25% to 90% in overall patients with DM were reported in previous literature.7,23,31 The sample size was determined from previous literature that reported 34% of amputations within 1 year were related to SVS-WIfI stage 4,31,35,36 and from retrospective data of patients with DFI who were admitted to Thammasat University Hospital (TUH) during the study period, which reported 66% risk of amputation within 1 year.
From the electronic medical database, after exclusion, 131 patients with DFI were included for analysis. The ratio of amputation to limb salvage patients in our study was 1/5.23. Under the 2-sample comparison of proportions under the 0.05 of type I error (α) and 0.2 of type II error (ℬ), 131 patients with DFI were required to be included in the study. One hundred and ten limbs of the limb salvage group and 21 limbs of major amputation group were needed for analysis for our study.
Result
From retrospective chart review of the electronic medical database, 155 patients with DFU were considered. And 131 patients with DFI between June 2018 and May 2020 were included in the study after evaluating by inclusion and exclusion criteria. Baseline characteristics and demographic data are shown in Table 1. The mean age of the patients was 68.4 ± 11.2 years, with 52% female. And 80.2% of patients with DFI had hypertension; and 73.3% had concomitant PAD. There is no difference of gender between major amputation and nonmajor amputation group in the result part already. Most patients were categorized in class 4 of the SVS-WIfI classification system by 1-year risk of amputation (48.1%) and benefit from revascularization (49.6%). WM classification 4 was the most common presentation (48%). Previous studies estimated the amputation rate in non-PAD patients with DFU at 5%, and concomitant DM with PAD was estimated at 30%.31,35 In our study, 73.3% of patients with DFI had concomitant PAD. Thus, the estimated amputation rate in our study = [5 × (proportion of non-PAD) + 30 × (proportion of concomitant PAD)] = 23.3%. The amputation rate in our study was 16%. The 3-year major amputation rate was 26.23%; however, 9 (6.8%) patients failed to follow up after 1 year.
Baseline Characteristics and Demographic Data by Major Amputation During 1-Year Follow Up in Hospitalized Patients With DFI.
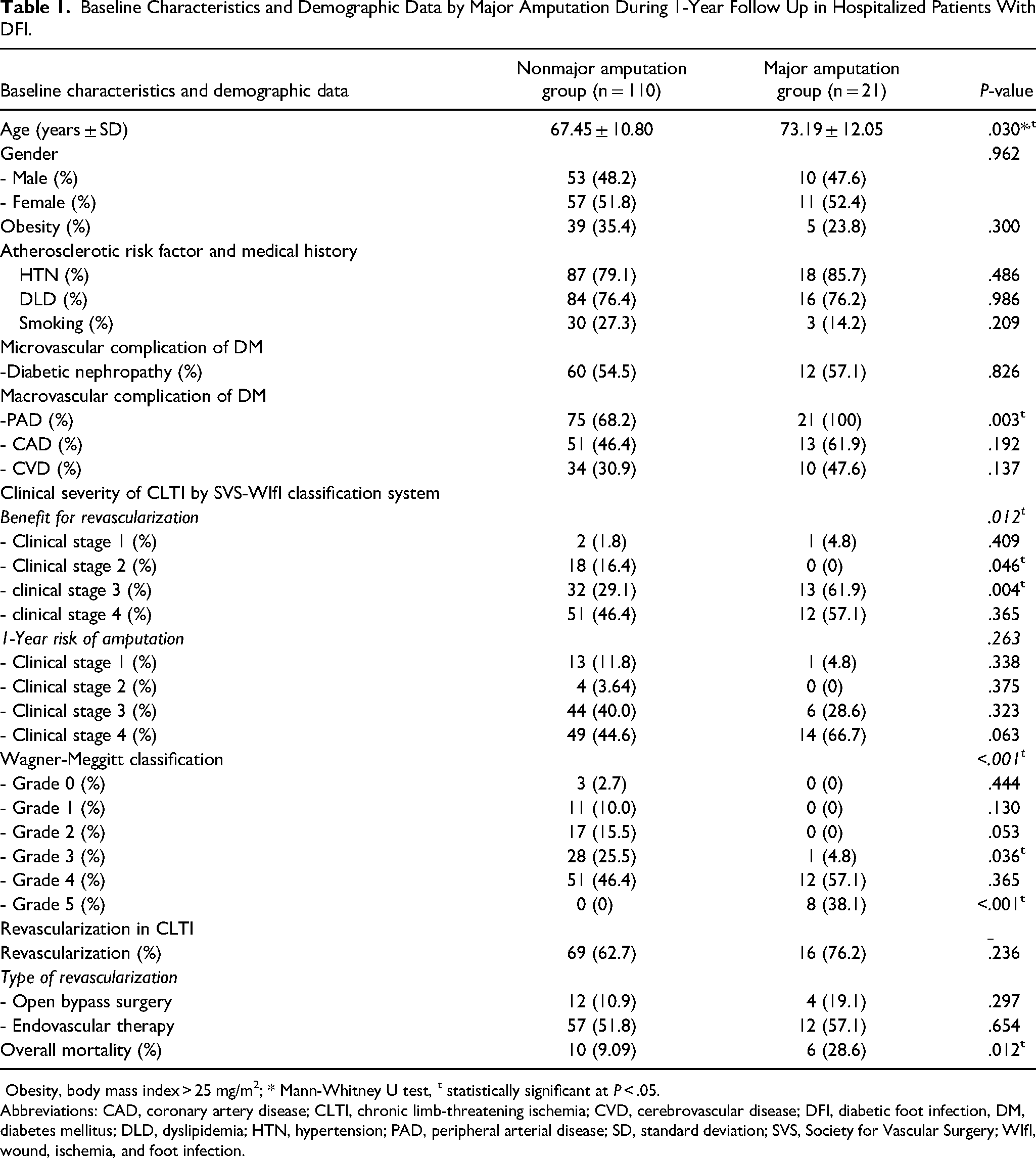
Obesity, body mass index > 25 mg/m2; * Mann-Whitney U test, t statistically significant at P < .05.
Abbreviations: CAD, coronary artery disease; CLTI, chronic limb-threatening ischemia; CVD, cerebrovascular disease; DFI, diabetic foot infection, DM, diabetes mellitus; DLD, dyslipidemia; HTN, hypertension; PAD, peripheral arterial disease; SD, standard deviation; SVS, Society for Vascular Surgery; WIfI, wound, ischemia, and foot infection.
The univariate analysis to compare between the limb salvage and the amputation patients revealed statistical significance for age (67.45 ± 10.8 vs 73.19 ± 12.0 years, P = .030), PAD (P = .003), SVS-WIfI classification for benefit from revascularization (P = .012), WM classification 5 (P < .001), and mortality rate (P = .012) (Table 1). For the multivariable model (Table 2), PAD (relative risk [RR] 1.47: 95% confidence interval [CI] 1.29-1.67, P = .032), SVS-WIfI classification for benefit from revascularization (RR 4.56: 95% CI 1.21-17.21, P = .007), and WM classification 5 (RR 9.46: 95% CI 5.65-15.82, P < .001) were significant. All major amputation patients had ABI values <0.9, which defined PAD. Age of patients was not associated risk factor of major amputation by multivariate analysis. All non-PAD patients were categorized in the limb salvage group (Figure 2a). An important cause of major amputation in our study was dominant ischemia. In all cases of DFI without concomitant PAD, the infection could be controlled by debridement or minor amputation. In Figure 3a, the SVS-WIfI classification for benefit from revascularization shows high risk for amputation because most patients in our study were comorbid with PAD. Patients with CLTI were at a clinical stage of benefit from revascularization, especially those with severe wounds and dominant infections. The SVS-WIfI classification system for 1-year amputation risk was not significantly associated with amputation rate. However, the stacked bar chart demonstrates the concordant trend of the high amputation rate in high clinical stages (Figure 3b). And 22% of patients in clinical stage 4 of the SVS-WIfI classification system for 1-year amputation risk were classified in the amputation group (P = .063).
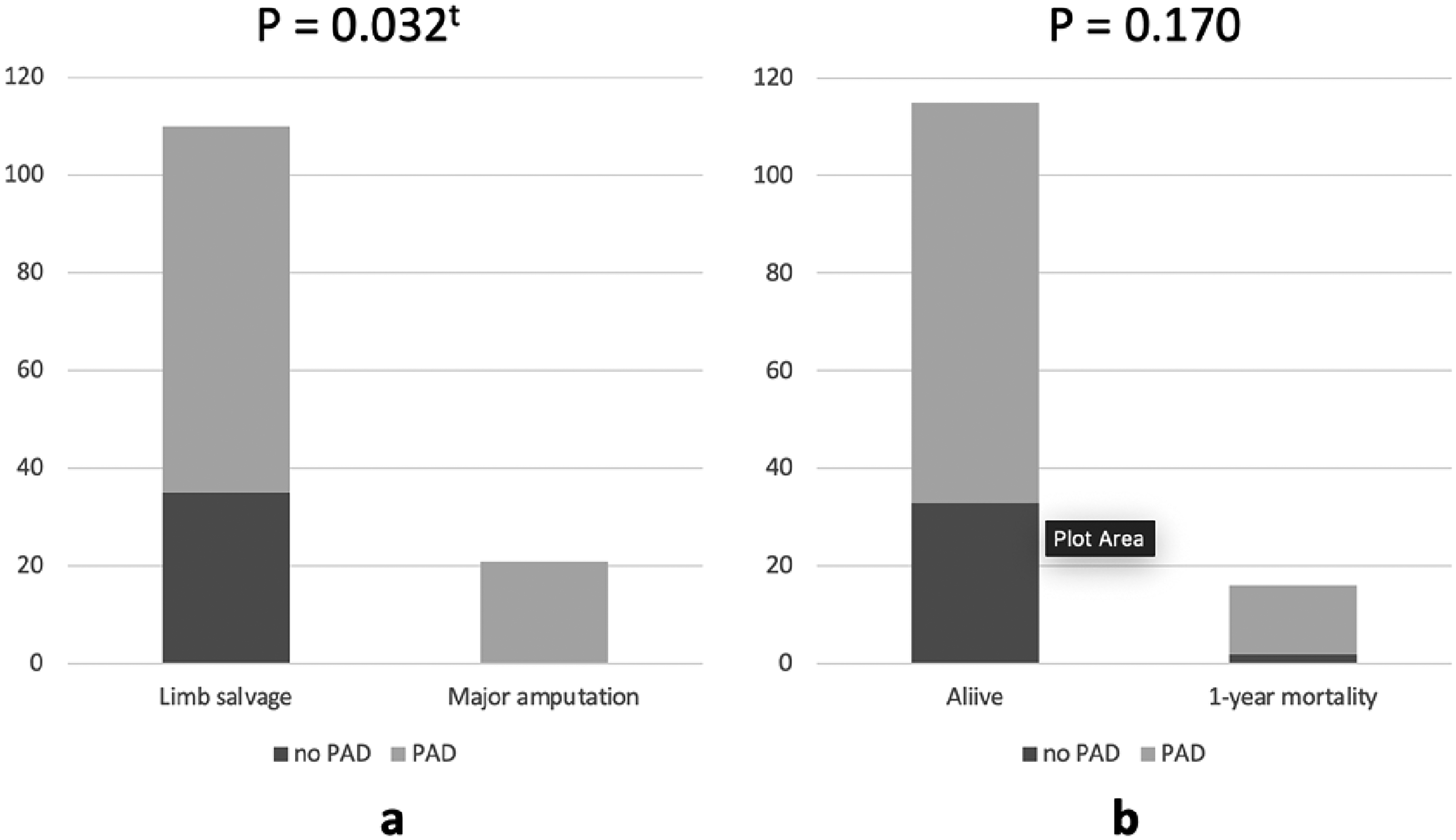
The stacked bar charts demonstrated the major amputation (a) and 1-year mortality rate (b) between non-PAD and PAD group in patients with DFI. tStatistically significant at P < .05. Abbreviations: DFI, diabetic foot infection; PAD, peripheral arterial disease.
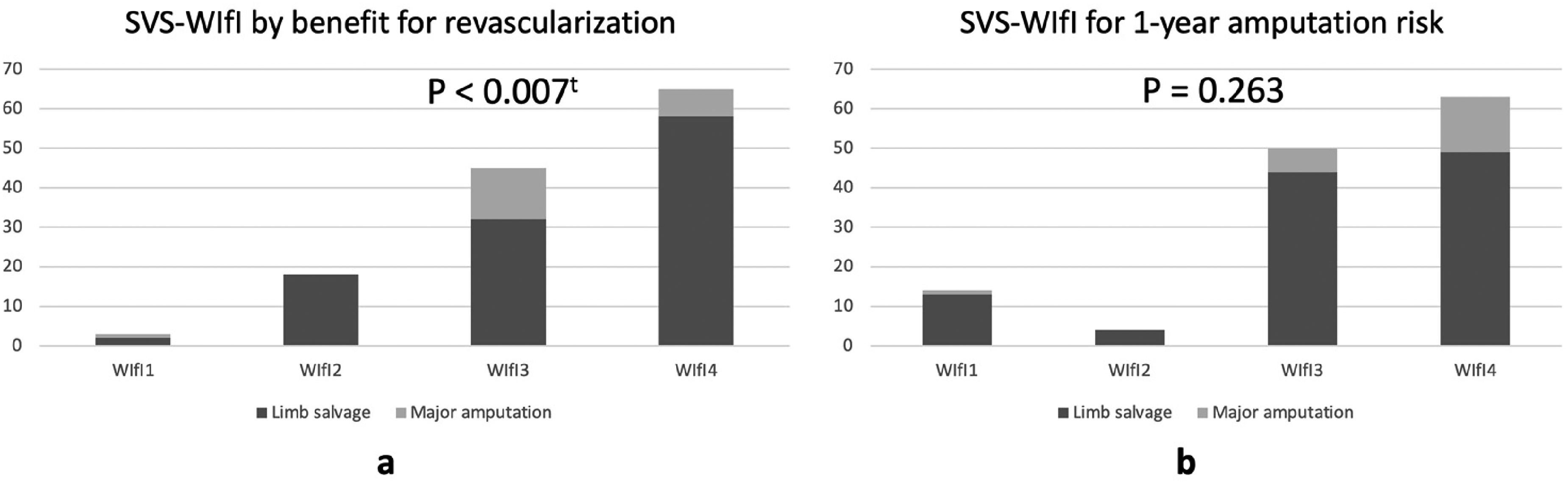
The stacked bar charts demonstrate the SVS-WIfI classification system by benefit from revascularization (a) and 1-year amputation risk (b) between limb salvage and major amputation in patients with DFI. tStatistically significant at P < .05. Abbreviations: DFI, diabetic foot infection; PAD, peripheral arterial disease; SVS, Society for Vascular Surgery; WIfI, wound, ischemia, and foot infection.
Multivariate Analysis of Predictive Factors of Major Amputation in Hospitalized Patients With DFI.
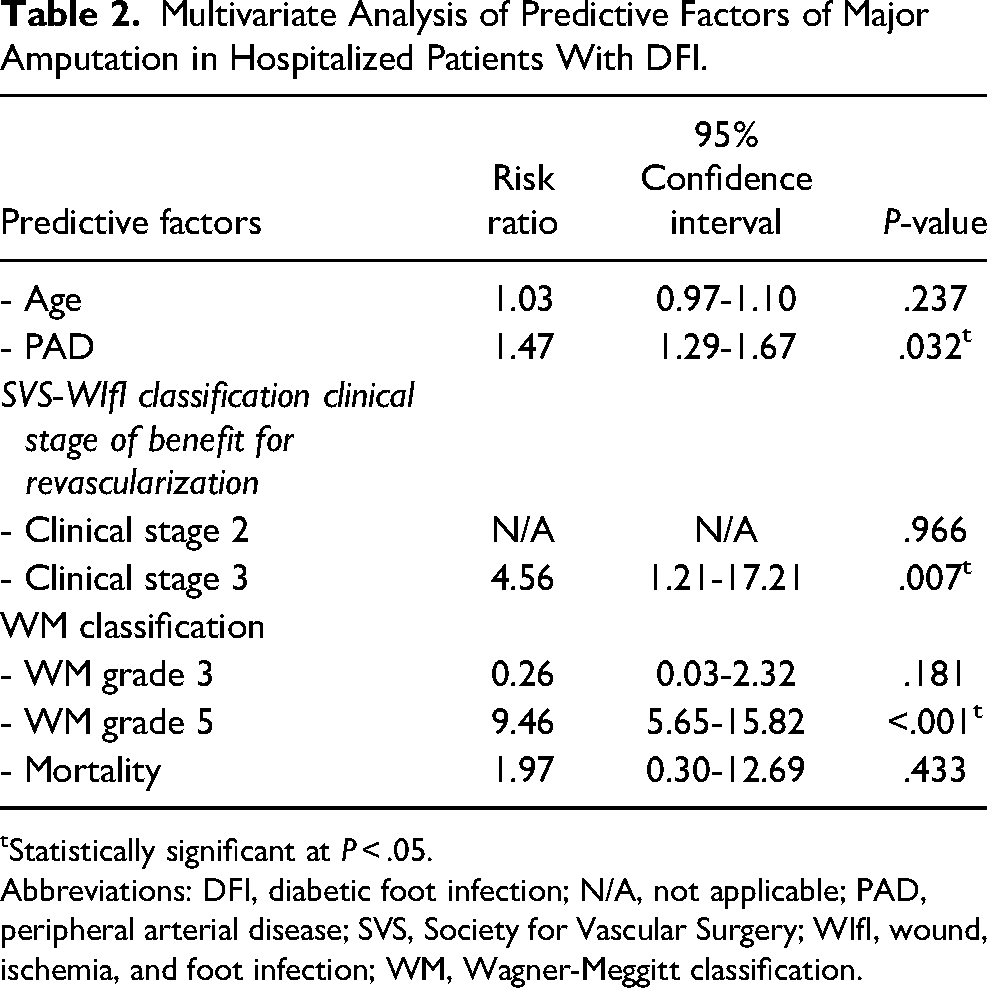
Statistically significant at P < .05.
Abbreviations: DFI, diabetic foot infection; N/A, not applicable; PAD, peripheral arterial disease; SVS, Society for Vascular Surgery; WIfI, wound, ischemia, and foot infection; WM, Wagner-Meggitt classification.
The major amputation rate at Thammasat University Hospital, categorized by clinical stage of SVS-WIfI score for 1-year risk of amputation, was lower than data from SVS. Twelve percent and 22.2% of patients were categorized in the amputation group by SVS-WIfI stages 3 and 4, respectively (Figure 4). High ulcer grade of WM classification was associated with high risk of major amputation in patients with DFI, especially in WM classification grade 5. 57.1% and 38.1% of patients were categorized in WM classification grades 4 and 5, respectively. All patients in WM classification grade 5, which was defined by mid to hind foot gangrene, underwent major amputation (P < .001; Figure 5).
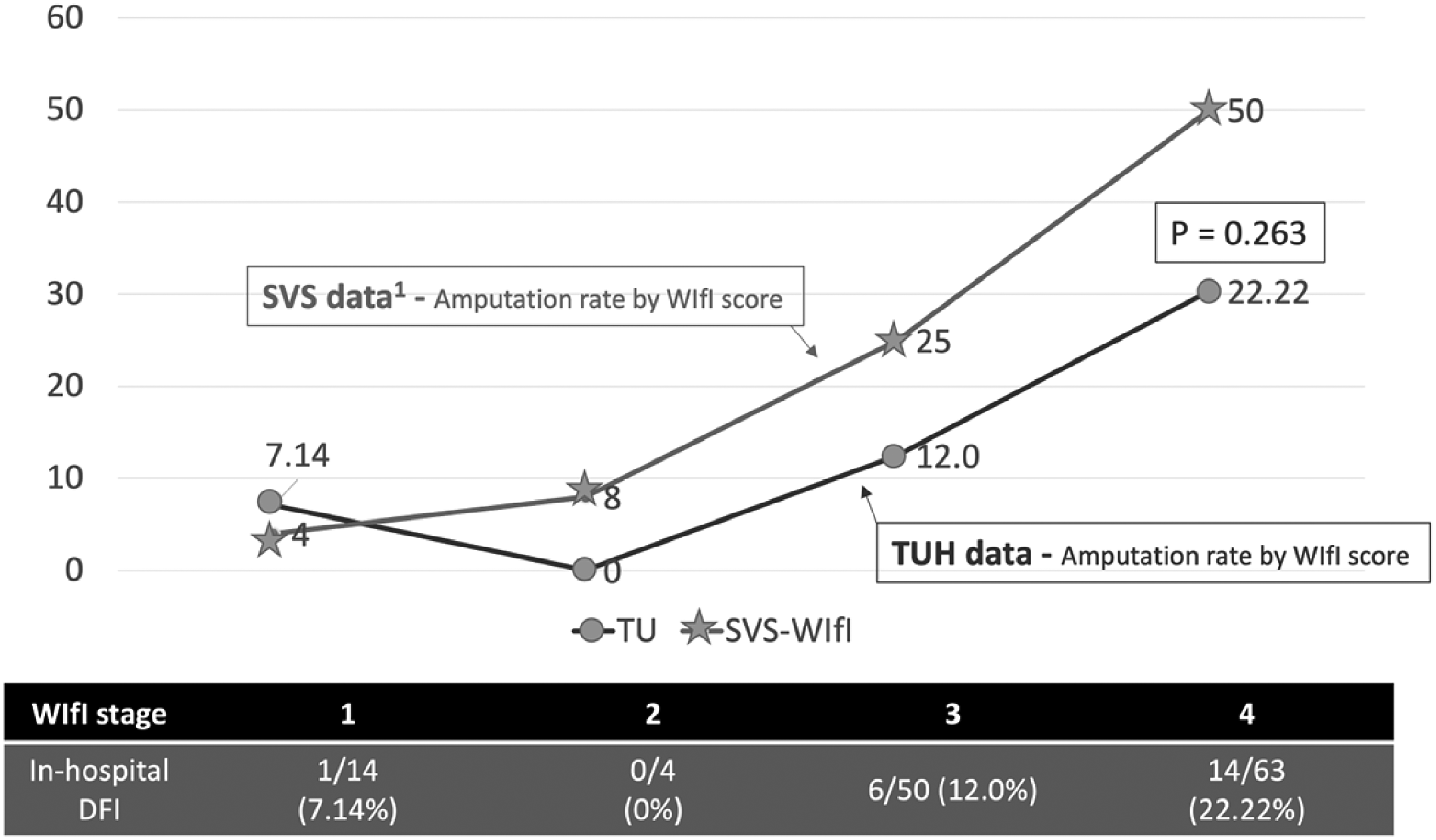
The line graph demonstrates the major amputation rate associated with the clinical stage of SVS-WIfI score for 1-year risk of amputation between Thammasat University Hospital and SVS data. tStatistically significant at P < .05. Abbreviations: DFI, diabetic foot infection; SVS, Society for Vascular Surgery; TUH, Thammasat University Hospital; WIfI, wound, ischemia, and foot infection.
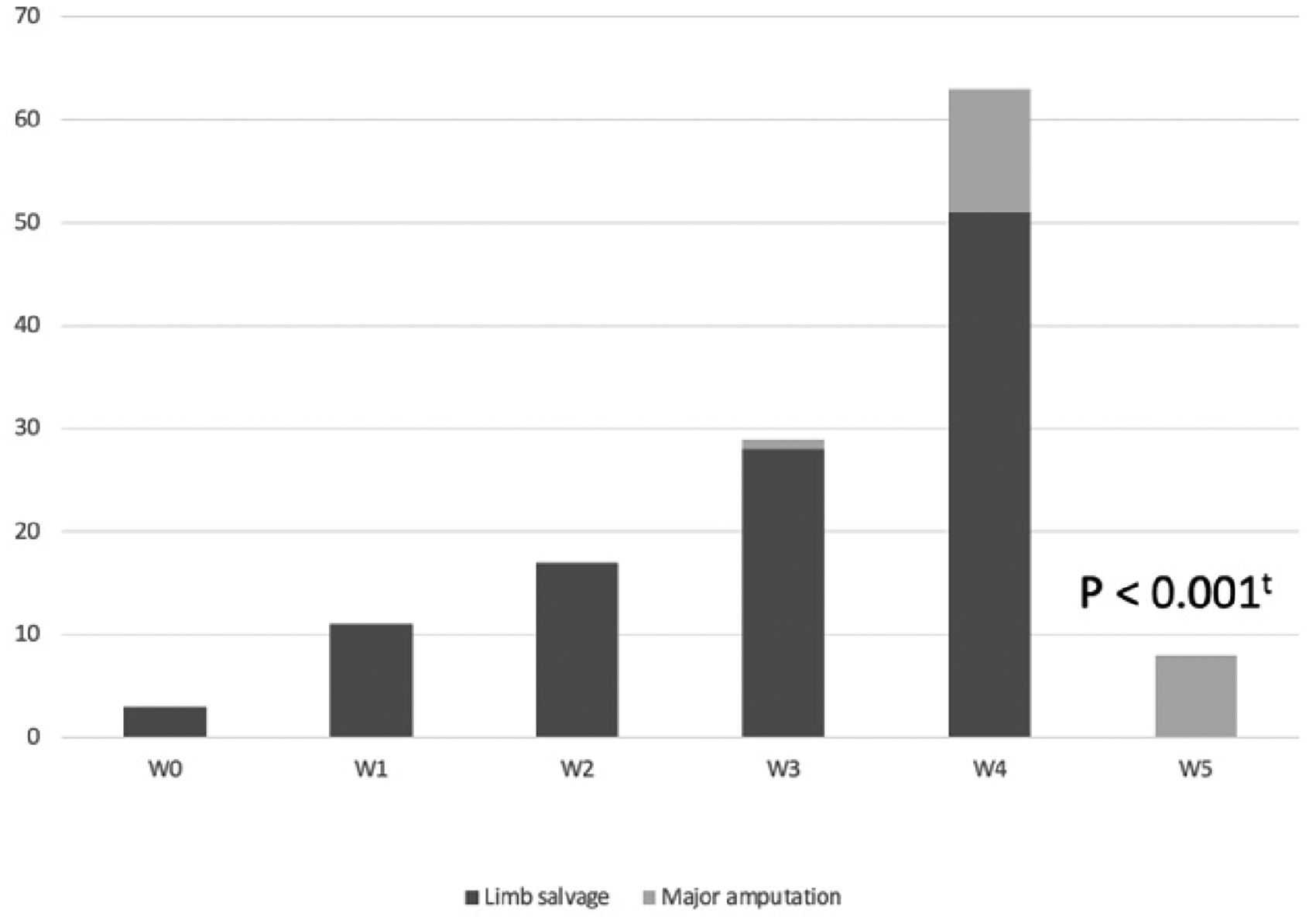
The stacked bar chart demonstrates the WM classification system between limb salvage and major amputation in patients with DFI. tStatistically significant at P < .05. Abbreviations: DFI, diabetic foot infection; WM, Wagner-Meggitt classification.
Our study demonstrated CAD and CVD comorbidity estimated at 75% and 62.5% of all mortality patients, respectively. However, the multivariate model of mortality rate in patients with DFI was not associated with CAD (RR 1.93: 95% CI 0.43-8.68, P = .391) or CVD (RR 2.43: 95% CI 0.61-9.69, P = .208; Table 3). In addition, PAD was not associated with mortality rate (Figure 2b). One-year mortality rate was 12.2%. The subgroup analysis in the revascularization group demonstrated comparable outcomes of amputation and mortality rate between EVT and open surgery (17.4% vs 25%, P = .721 and 13.0% vs 2.5%, P = .816, respectively; Figure 6).
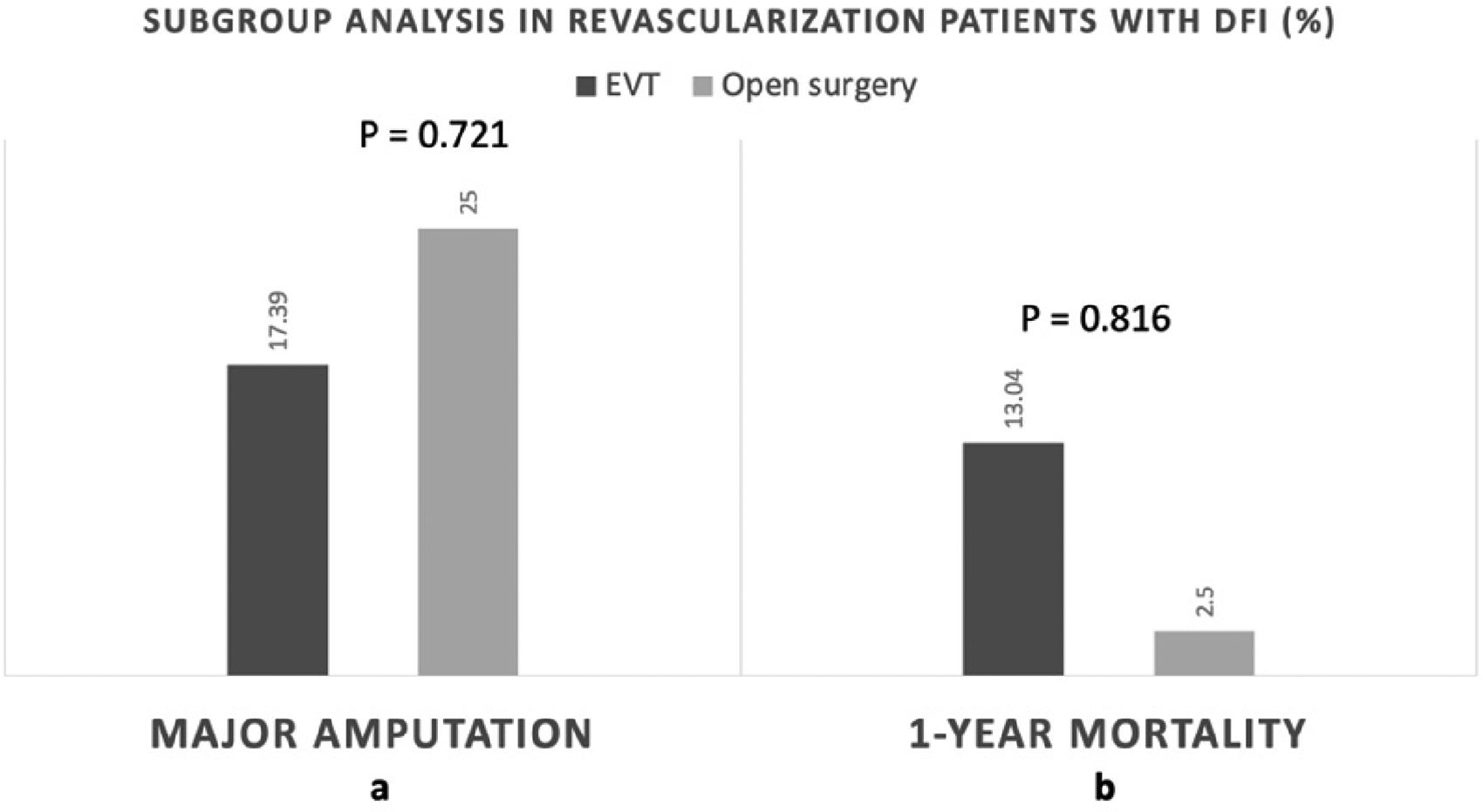
The stacked bar chart demonstrates major amputation (a) and 1-year mortality rate (b) between EVT and open surgery groups in revascularized patients with DFI. tStatistically significant at P < .05. Abbreviations: DFI, diabetic foot infection; EVT, endovascular therapy.
Multivariate Analysis of Predictive Factors of Mortality in Hospitalized Patients With DFI.

Statistically significant at P < .05.
Abbreviations: CAD, coronary artery disease; CVD, cerebrovascular disease; DFI, diabetic foot infection; WM, Wagner-Meggitt classification.
Discussion
The traditional ulcer classification of WM score is simple to use in clinical practice. 21 This classification does not require any noninvasive hemodynamic measurement. The WM score grade 5 is the most strongly associated with risk of amputation because the extensive gangrene involved with mid and hind foot is associated with severe infection that extends to deep tissue and/or PAD-induced gangrene. When the gangrene extends to calcaneal bone or hind foot, most surgeons prefer below or above knee major amputation rather than limb salvage. In addition, ischemia with co-infection in this area usually presented with SIRS. 17 However, our data cannot determine the cause of amputation because of the retrospective data collection process. Moreover, WM classification combined infection and ischemia. The extensive gangrene of some patients with WM grade 5 originated from severe infection with microvascular involvement. This did not involve PAD, which is ischemia dominant. Severe forms of infection, including abscess or osteomyelitis, are classified as clinical grade 3 of WM score. So, the degree of infection severity by WM classification may be misinterpreted. After debridement, the WM classification needs to be re-evaluated. Some patients who undergo drainage of WM clinical grade 3 may progress to clinical grade 5 if underlying CLTI is present.4,6,37
A high SVS-WIfI classification for benefit from revascularization, which is a most recent and updated scoring system, is strongly associated with major amputation in this study. The study demonstrated 12.0% and 22.2% of amputation rate of clinical stage of SVS-WIfI score for 1-year risk of amputation stages 3 and 4, respectively. Mills and colleagues reports the higher SVS-WIfI classification associated with higher estimated 1-year risk of amputation. Mills JL publication demonstrated 25% and 50% of amputation rate of clinical stage of SVS-WIfI score for 1-year risk of amputation stages 3 and 4, respectively. 22
However, the hemodynamic parameter measurement, which needs to be objectively documented, may underestimate the severity of ischemia. ABI applicability is limited in patients with DM due to medial calcinosis, especially in concomitant chronic kidney disease from diabetic nephropathy. More than half of patients with DM in this study had concomitant diabetic nephropathy. The clinical severity of SVS-WIfI score for benefit from revascularization and 1-year amputation risk increased in diabetic nephropathy patients. TP and TcPO2 are limited to evaluation in patients with minor amputation and DFI, respectively.8,11 So, the clinical severity of SVS-WIfI score by benefit from revascularization and 1-year amputation risk can underestimate the risk of amputation. PAD is the same issue as ischemia dominant of SVS-WIfI score; IWGDF recommended that clinical evaluation of ischemic dominant is still an important part of a physical examination. 5 The paucity of ischemic symptoms may be related to the presence of co-existing neuropathy and loss of pain sensation. The palpation of foot pulses should form a key part of the initial clinical examination. No single measure predicted healing accurately. 5
Similar to the WM score, the clinical severity of SVS-WIfI score must be re-evaluated after debridement or revascularization. The timing for debridement to remove infection and revascularization is very important, but not well established by previous publication or guidelines.3,8,11,15,17 Delayed revascularization in concomitant CLTI in patients with DFI is associated with risk of amputation. On the other hand, too early revascularization, or revascularization in moderate to severe DFI without eradication of infection, leads to catastrophic results of SIRS and amputation. Proper ordering and timing of a multidisciplinary treatment approach are important factors to determine amputation and mortality rates, which were not included in this study.3,11,17
CAD or CVD comorbidity is the common cause of mortality in patients with DFI. Perioperative major adverse cardiac event (MACE), including preoperative MI of patients with CAD and acute stroke in patients with CVD may be associated with perioperative mortality. In proper preoperative preparation, MACE can be prevented by a multidisciplinary team approach. However, determining the overall mortality rate in DFI would require a long-term follow up.10,11,13,30,38 Open surgery decreases major amputation rates by rapidly increasing the in-line blood flow to the foot.6,39 Patients with good operative risk may prefer open revascularization instead of EVT. The 1-year mortality rate was lower in an open revascularization group than in an EVT group. The EVT technique is preferred in high-risk patients with multiple comorbidities. MACE tend to occur more in EVT due to severe underlying disease of patients. 39 Although MACE increase the mortality rate of EVT, in our study, the mortality rate did not differ significantly between the EVT and the open surgery groups.3,8,11,15,17
The limited detail of causes of amputation in the retrospective data was the main limitation of this study. Patients’ data and causes of amputation at 3-year follow up were limited, and 6.8% of patients did not follow up. The SVS-WIfI score summarized clinical severity of ischemia, wound, and foot infection dominants without the assessment of the agreement between each measurement of scoring system.11,22 CAD and CVD comorbidity, WM classification and major amputation were not associated with mortality rate in multivariate analysis model. However, to conclude that the PAD, WM ulcer classification and clinical stage of SVS-WIfI score by benefit from revascularization are the major risk factors of amputation and to explore the strong evidence of the factors associated mortality rate in patients with DFI, a further large prospective study with long-term follow up is required.
The amputation risk factors in patients with DFI with characteristics suggesting a more serious DFI or potential indications for hospitalization include clinical grade 5 of the WM ulcer classification score, high clinical severity of the SVS-WIfI score, and PAD. In appropriate multidisciplinary treatment, the mortality rate of patients with DFI can be minimized. False negatives of noninvasive hemodynamic parameter measurements should be reduced. Skillful physicians should conduct dynamic evaluation of progression of DFI. WM classification and SVS-WIfI score should be reevaluated.
Conclusion
DFI is a high morbid and mortality disease. High WM classification and SVS-WIfI score on benefit from revascularization, and PAD were strongly associated with major amputation rates in patients with DFI which led to decreasing of the patients’ performance status and quality of life. Thus, the appropriate SVS-WIfI score evaluation and early detection of PAD with proper management are very important for limb salvage in patients with DFI.
Footnotes
Acknowledgments
This work was supported by the Research Group in Surgery, Faculty of Medicine, Thammasat University. Special thanks to the Clinical Research Center, Faculty of Medicine, Thammasat University for assistance in editing the English version of this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
