Abstract
Diabetic foot infections (DFIs) are a common and costly complication of diabetes. Soft tissue and bone infections in DFIs frequently lead to amputation and/or sepsis which can be costly for both the patient and the healthcare system. Staphylococcus aureus is the most commonly identified causative agent in DFIs, and people with diabetes may have an increased risk of infection with methicillin-resistant Staphylococcus aureus (MRSA). In addition to increased susceptibility to severe infection, MRSA in DFIs is associated with high rates of treatment failure, morbidity, and hospitalization costs meaning appropriate treatment is a high priority. While hospitalized patients are usually treated with intravenous (IV) vancomycin, this can be costly in terms of inpatient stays, staffing costs, and adverse events. For example, vancomycin-associated acute kidney injury not only delays hospital discharge and increases costs but is also a particular concern for patients with diabetes who already have an increased risk of kidney problems. Vancomycin-resistant strains of S. aureus have also been identified, which means that alternative treatment options may need to be explored. Treatment alternatives to IV vancomycin, including oral antibiotics, have been shown to provide similar efficacy, with reduced costs, outpatient or home-based administration, and with fewer serious adverse effects. Although infectious disease specialists often use IV vancomycin alone, or in combination, as a first-line therapeutic option, they are increasingly seeing the value of outpatient or at-home oral antibiotics as an alternative. This manuscript reviews the evidence for true costs of vancomycin therapy for MRSA-associated DFIs and examines the alternatives.
Introduction
Diabetic foot ulcers (DFUs) are common in people with diabetes, with a global prevalence of 6.3%. 1 It is estimated that 9.1 to 26.1 million individuals with diabetes develop foot ulcers every year. 2 Approximately 60% of DFUs are complicated by infection, 3 and over two-thirds of amputations in people with diabetes arise from diabetic foot infections (DFIs). Diabetic foot infections are categorized as mild, moderate, or severe and involved a variety of presentations, including cellulitis, abscesses, necrotizing fasciitis, wet gangrene, septic arthritis, and osteomyelitis, and may lead to bacteremia and/or sepsis (Figure 1). 4 Diabetic foot infections increase the risk of major amputations, 5 which greatly impact the patient's quality of life (QoL), as well as impacting their families and caretakers. Furthermore, the complications associated with DFIs and amputations can be costly for both the health system and individual patients.
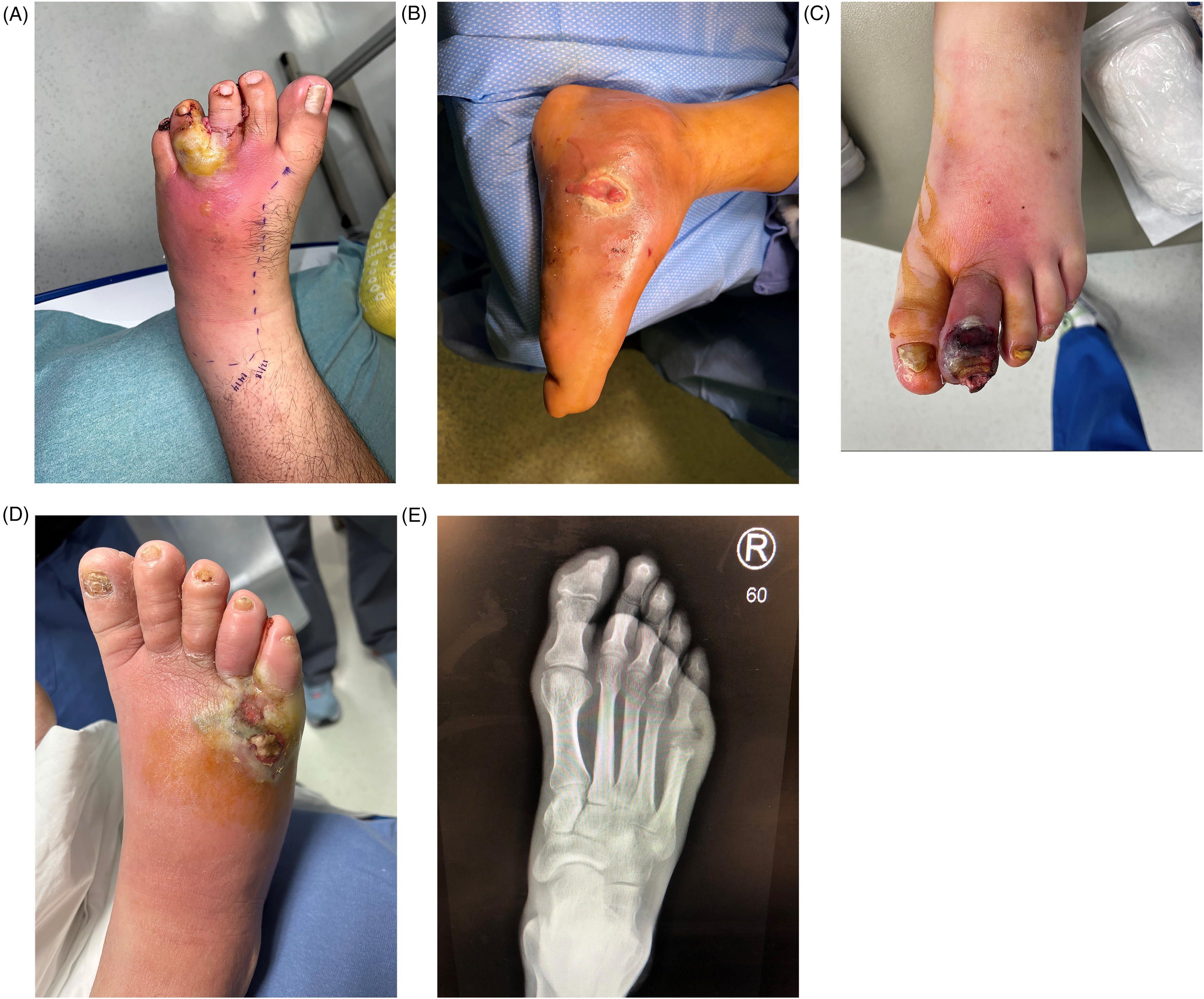
Examples of diabetic foot infections. (A) Cellulitis extending several centimeters around an interdigital abscess. IDSA Moderate Diabetic Foot Infection. (B) Diabetic foot infection with draining purulence from the medial foot. Bone was palpable through the wound creating a high probability of osteomyelitis. IDSA Moderate Diabetic Foot Infection. (C) Necrotic and infected second toe with ascending cellulitis on the foot. This is commonly referred to as “wet gangrene.” Typically these are polymicrobial infections and contain gram-negative bacteria and anaerobes. This patient met SIRS criteria and was thus classified as an IDSA Severe Diabetic Foot Infection. (D) A clinical image of a “blow out” lesion. This is caused by underlying osteomyelitis and abscess that escapes through the skin. This patient met SIRS criteria and was classified as an IDSA Severe Diabetic Foot Infection. (E) An AP foot x-ray image from the same patient in (D). There is erosion of the fifth metatarsal head, subluxation of the fifth metatarsophalangeal joint, and subcutaneous emphysema present, indicative of anaerobic bacteria. This is commonly referred to as “gas gangrene” even when there are no superficial signs of necrosis since the anaerobic bacteria cause tissue necrosis in the deeper tissue. AP, anterior-posterior; IDSA, Infectious Diseases Society of America; SIRS, systemic inflammatory response syndrome. Images provided by Lee C. Rogers.
Many DFIs are polymicrobial, with a broad range of causative pathogens implicated, 6 including both Gram-positive or -negative bacteria or fungi, with a small number of pathogens in DFI studies being unidentifiable. 7 An important factor in the causative pathogen identified may be geographic location. In North America and Western Europe, it has been reported that Gram-positive pathogens are the most commonly identified organisms in DFIs.7,8 However, in developing countries, such as India, 9 Malaysia, 10 and Cameroon, 11 the prevalence of Gram-negative organisms may be higher than Gram-positives.
A 2021 meta-analysis on the microbiology of DFIs (including 112 studies and around 16 000 patients) reported that 41% of culture-positive patients had monomicrobial infections and 59% had polymicrobial infections. 7 The main causative agent identified in DFIs was Staphylococcus aureus (23%), including methicillin-resistant S. aureus (MRSA). Furthermore, the most commonly identified Gram-negative organisms were Escherichia coli (12%) and Pseudomonas spp. (11%), 7 but empiric therapy directed at Pseudomonas aeruginosa is usually unnecessary with guidelines stating that it may be considered if P aeruginosa has been isolated from cultures of the affected site within the previous few weeks, or if the patient resides within tropical/subtropical climates. 12
With S. aureus being the most common pathogen identified in DFIs, MRSA is an increasing problem for people with diabetes, and these patients may be more at risk of MRSA infection. A 2019 meta-analysis reported that people with diabetes had a 4.75% greater colonization rate (P < .0001) compared to people who did not have diabetes. 8 Another study suggested that people with poor glycemic control (glycated hemoglobin [HB1Ac] > 8.5%] have heightened risk for the development of severe MRSA skin and soft tissue infections, so greater time spent in hyperglycemia may be driving susceptibility to infection. 13
The rate of MRSA reported in patients with diabetes and DFIs is variable, which may reflect the differences in geographic region, or the populations involved. For example, MacDonald's 2021 meta-analysis was international, with the majority of studies identified from Europe and Asia, and reported that 18% of the identified S. aureus isolates were MRSA. 12 Another meta-analysis involving 10 994 patients with DFIs reported an MRSA prevalence of 17%. These results are aligned with 2 US studies; one study of 318 patients from a university hospital reported that 46% of culture-positive DFIs were caused by S. aureus, with 15% being MRSA 14 ; and another study of DFIs (N = 151) from a veteran's hospital reported incidence of MRSA in a DFI cohort to be 15%. 15 A review from India reported that MRSA was present in 10% to 32% of diabetic infections. 16 In addition to increased susceptibility to severe MRSA infection, MRSA in DFIs is associated with high rates of treatment failure, morbidity, and hospitalization costs, 16 meaning appropriate treatment is paramount.
Antibiotic treatments for DFIs
A meta-analysis found that empiric therapy in DFIs often targets aerobic Gram-positive pathogens (S. aureus and beta-hemolytic streptococci). When MRSA is suspected or identified, the recommended antibiotics are glycopeptides (such as vancomycin), linezolid, daptomycin, trimethoprim/sulfamethoxazole (with or without rifampin), and doxycycline. 12
The overall treatment plan for DFI requires a comprehensive approach, with consideration of surgical incision and drainage and debridement of necrotic, nonviable, and infected tissue. Systemic antibiotics are not sufficient alone in many DFIs. Necrotic tissue may be an indicator of concomitant peripheral artery disease, or it may be a result of the infection. Surgical management may be required, depending on the severity of the infection.
Antibiotic treatment of DFIs was previously covered under the Food and Drug Administration (FDA) guidance for the treatment of complicated skin and skin structure infections (cSSSIs). As such, treatment had to cover a broad range of pathogens, and skin and structure infection types. In 2013, the FDA introduced the acute bacterial skin and skin structure infections (ABSSSI) terminology to replace the prior categories of complicated and uncomplicated SSSI, which allowed treatment to become more specific. 17 However, this led to a number of drugs no longer having indications for cSSSIs, including DFI), regardless of their efficacy against commonly identified DFI pathogens, such as S. aureus and MRSA. To date, only 3 antibiotics have an FDA indication for the treatment of DFIs: ertapenem, 18 linezolid, 19 and piperacillin/tazobactam. 20 Diabetic foot infections are often treated with piperacillin/tazobactam plus vancomycin due to their wide availability and low cost, as well as the misconception that Pseudomonas spp. needs to be treated.18,19,21
Despite the lack of FDA Guidance, the International Working Group on the Diabetic Foot has published recommendations for antibiotic selection in DFI. Two specific recommendations, their numbers 12a and 13, state that any systemic antibiotic regimen that have been shown to be effective in randomized controlled trials can be used to treat a patient with DFI and a soft tissue infection of the foot. They clarify that the antibiotic should be chosen on the likely, or proven pathogen, antibiotic susceptibility of those organisms, the clinical severity of the infection and other factors including efficacy, risk of adverse reactions, potential for drug–drug interaction, agent availability, and cost. 21
Vancomycin as a Popular Standard of Care for DFIs
Vancomycin, a glycopeptide antibiotic, is used to treat a wide range of bacterial infections, including bacteremia, cellulitis, osteomyelitis, and other infections due to MRSA. 22 Vancomycin has long been considered a key treatment for MRSA infection because it is well-known by clinicians and has a relatively low drug acquisition costs when compared to newer antibiotics. However, vancomycin is less effective than beta-lactams against methicillin-sensitive S. aureus and requires therapeutic drug monitoring for toxicity and efficacy.23,24 Vancomycin minimum inhibitory concentration (MIC) creep has been observed in clinical isolates of MRSA. This means that although these isolates were not deemed resistant, the effective concentration of vancomycin required to treat was increasing over time which was associated with increased clinical failure, particularly in patients with bacteremia. 25 As such, guidelines recommend against using vancomycin as a treatment for MRSA when the vancomycin MIC is 2 µg/mL or greater. 26 Lastly, at least 52 strains of vancomycin-resistant S. aureus have been isolated across the US, India, Iran, Pakistan, Brazil, and Portugal. 27 This indicates that vancomycin resistance may be a growing problem, requiring alternative therapeutic options.
Aside from antimicrobial resistance, there are other issues with the use of vancomycin as a therapeutic agent. For example, vancomycin is associated with several adverse events (AEs) including nephrotoxicity, hypotension, phlebitis, ototoxicity, hypersensitivity reactions, vancomycin infusion reaction (also known as red man syndrome 28 ), neutropenia, chills, fever, and interstitial nephritis. 29
In the treatment of DFIs, significant AEs associated with vancomycin use is nephrotoxicity and acute kidney injury (AKI). 30 A systemic review and meta-analysis by Ray et al (including 4033 patients) reported that vancomycin administration leads to a 2.5-fold increase in the risk of AKI. 31 Vancomycin-associated AKI often develops after 4 to 17 days of vancomycin administration and improves after prompt discontinuation of treatment, 32 although patients with critical illness may not experience full renal function recovery.32,33 A combination of vancomycin and other antibacterial agents is increasingly being used to treat potential polymicrobial infections, for example, vancomycin plus piperacillin-tazobactam. However, this combination synergistically increases the risk of nephrotoxicity.34–37 In hospitalized patients, AKI results in increased length of stay and excess hospital costs, especially if dialysis is required. 38 Overall, this makes vancomycin particularly unsuitable for the treatment of DFIs, because people with diabetes are already at increased risk for kidney injury. 39
The use of vancomycin is further limited by other issues such as the lack of activity against Gram-negative bacilli, 22 the necessity for patient-specific Bayesian dosing, 40 and requirement of intravenous (IV) administration and PICC line use, leading to IV-associated complications. 30 These issues with vancomycin mean that alternative antibiotic options are required. For hospitalized patients with complex skin and soft tissue infections, IDSA guidelines recommend other antibiotics including oral (PO) or IV linezolid, daptomycin, telavancin, and clindamycin. Beta-lactam antibiotics such as cefazolin may be considered. Oral antibiotics are recommended for outpatients, including clindamycin, trimethoprim/sulfamethoxazole, tetracycline (ie, doxycycline or minocycline), and linezolid 41 (Table 1). As discussed above, recent guidelines from The International Working Group on the Diabetic Foot recommend a similar list of antibiotics with further stratification by infection severity and additional factors. 21
IDSA Recommended Antibiotics for Treatment of MRSA Skin Infections.
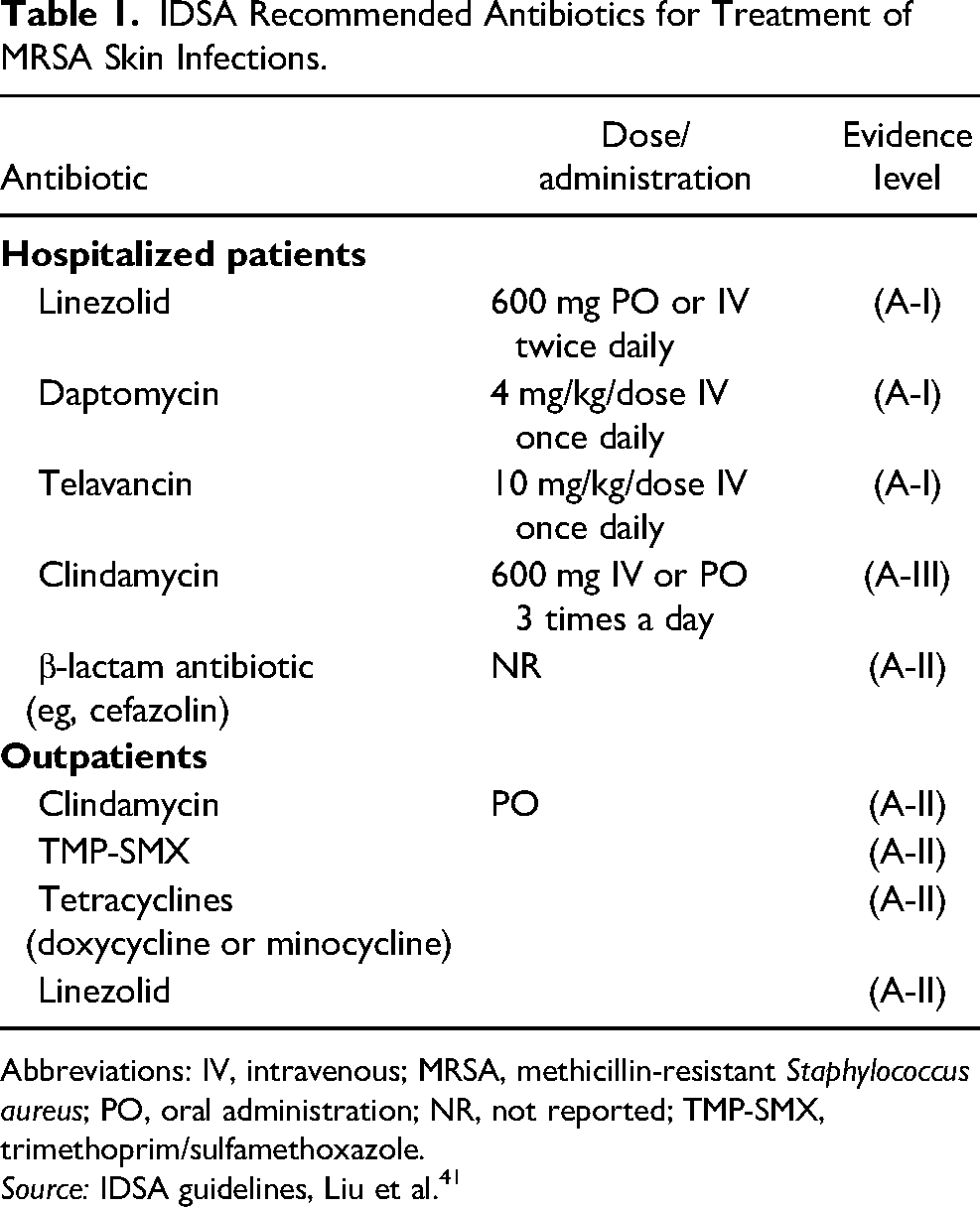
Abbreviations: IV, intravenous; MRSA, methicillin-resistant Staphylococcus aureus; PO, oral administration; NR, not reported; TMP-SMX, trimethoprim/sulfamethoxazole.
Source: IDSA guidelines, Liu et al. 41
Intravenous to PO Switch
The management of complex DFIs and the infections that extend into the bone usually necessitate a prolonged course of IV antibiotics. 42 However, parenteral therapy typically requires an inpatient setting or increased health care resource utilization due to the requirement for IV lines and patient monitoring. This can result in higher costs for an IV antibiotic compared to an oral alternative.42,43 Intravenous administration is also associated with substantial risks such as bleeding, local and systemic infections, and venous thrombosis 44 and poses a greater inconvenience to the patient compared with oral therapy, particularly if they must remain hospitalized. 45 An alternative is outpatient parenteral antibiotic therapy (OPAT), 46 which offers benefits to the healthcare system including avoiding or shortening length of stay and prevention of nosocomial infections, which can result in cost savings. 47 Outpatient parenteral antibiotic therapy is also advantageous to patients, allowing them to return to school or work and to resume the general activities of daily life. 47
Oral therapies also provide an alternative, and perhaps improved, treatment option but may not be familiar to healthcare providers. Intravenous antibiotic therapies have been used for decades, reflecting a broadly held belief that parenteral therapy is inherently superior to oral antibiotic treatments. 48 This belief is not based upon superiority of IV drugs, but rather due to inadequate drug levels of some oral antibiotics at the site of infection for some pathogens. If adequate concentrations of an oral drug reach the site of infection, the effects are similar to that observed with IV-administered therapy regardless of the drug administration method. As such, drugs with higher bioavailability or bioequivalent oral formulations are well-positioned for treatment success. This has been demonstrated with fluoroquinolones (FQs). However, current use of FQs is limited by unreliable activity against MRSA (with the exception of delafloxacin), antimicrobial resistance, 49 and black box warnings from the FDA. 50 The FDA warning noted that patients may develop serious AEs from FQ treatments, including tendon rupture, convulsions, hallucinations, depression, prolonged QTc (corrected QT interval), and torsades de pointes arrhythmia, as well as Clostridioides difficile-associated diarrhea. 50 The FDA also warned that FQs should be not be used to treat patients who have an aortic aneurysm or are at risk for an aortic aneurysm and should only be prescribed if no other treatment options are available. 51
Several antibiotics, such as linezolid, 52 omadacycline, 53 delafloxacin, 54 and tedizolid, 55 are available in bioequivalent oral formulations and have demonstrated efficacy for the treatment of bacterial infections (summarized in Table 2). The benefits of these new therapies include coverage against common pathogens and low microbial resistance. Oral administration also allows patients to be treated at home without an IV line, which is beneficial for patients in terms of QoL and convenience, as well as being cost-saving for the healthcare system. 56
Equivalency Data for Antibiotics Approved for Intravenous and Oral Administration.

Abbreviations: ABSSSI, acute bacteria skin and skin structure infection; AE, adverse event; FDA, US Food and Drug Administration; IV, intravenous; RCT, randomized controlled trial.
Although IV therapies are frequently considered for initial empirical therapy in severe infections as recommended by the IWGDF, oral therapies should be considered where available and appropriate.21,45 There are 3 main treatment approaches when transitioning from IV therapy to oral therapy (IV to PO): sequential therapy, switch therapy, and step-down therapy
45
:
Sequential therapy refers to the replacement of the parenteral version of a medication with its oral counterpart of the same compound at the same dosage, Switch therapy is the replacement of an IV medication to an oral equivalent at a similar potency, but a different compound within the same drug class, Step-down therapy refers to the conversion from an IV medication to an oral agent in another class, or to a different medication within the same class where the frequency, dose, or spectrum of activity may not be exactly the same.
Barriers to Oral Antibiotic Therapies for DFI
The Lower Drug Acquisition Cost of Vancomycin
Vancomycin remains a popular choice for the treatment of DFI and particularly when MRSA is involved, due to low drug acquisition costs. However, the total costs of IV antibiotic administration are formed not only by the costs of the drugs themselves but also by staffing time and associated costs, additional disposable materials involved in IV administration (eg, bags, tubes, and IV solutions), and overheads such as inpatient stay times. The use of IV antibiotics is associated with considerable workload and additional costs that can exceed the acquisition costs of the medications themselves. 68 The increased cost may be unjustifiable, as indicated by the results of the OVIVA study. OVIVA compared oral versus IV antibiotic treatment in bone and joint infections, reporting that oral therapy was noninferior to IV in terms of efficacy. If patients were assigned to PO therapy, they received FQs (43.8%), combination (14.1%), penicillins (13.9%), macrolides/lincosamides (11.4%), tetracyclines (8.9%), or other antibiotics (7.9%). Of these drugs, only penicillins favored IV therapy over oral therapy, with an odds ratio of 1.59 (0.65, 3.89). All other drugs either favored oral over IV or with similar outcomes between oral and IV administration. 42 There were also significant cost savings, driven by shorter median inpatient stay as well as decreased indwelling IV catheter days. Quality-adjusted life-years were numerically higher in the oral treatment arm but this did not reach significance. 69
Economic modeling studies indicate that the avoidance or reduction of inpatient stay is a major driver in the high cost of treating skin infections, particularly for DFI and bone and joint infections. A UK study compared inpatient stay for each infection type against 100% oral outpatient care, and 3 differing ratios of oral and IV outpatient therapy: 25%, 50%, and 75% oral therapy, with the remaining administration being IV. All outpatient administration models were over 70% cost-saving compared to inpatient stay (Table 3). 70
Comparative Costs of Inpatient and Outpatient Administration of Antibiotics.
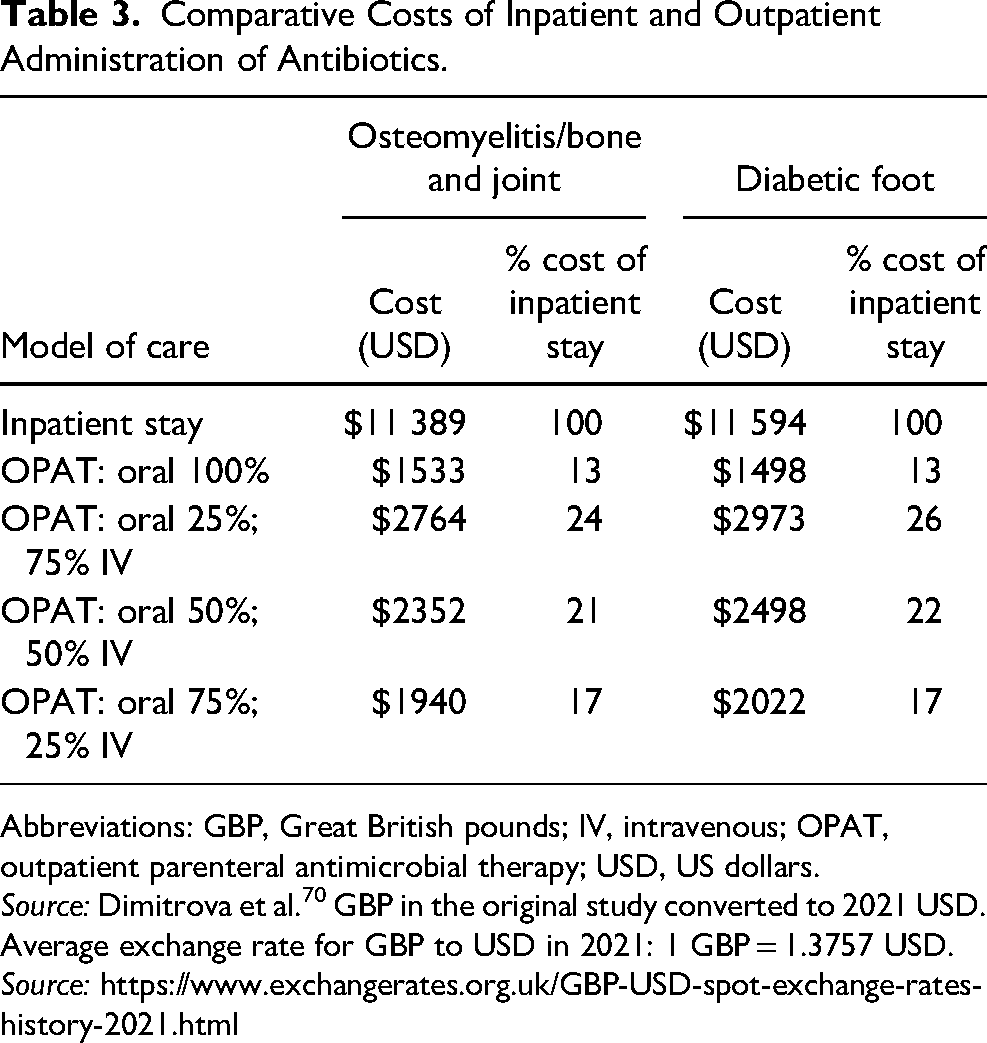
Abbreviations: GBP, Great British pounds; IV, intravenous; OPAT, outpatient parenteral antimicrobial therapy; USD, US dollars.
Source: Dimitrova et al. 70 GBP in the original study converted to 2021 USD. Average exchange rate for GBP to USD in 2021: 1 GBP = 1.3757 USD. Source: https://www.exchangerates.org.uk/GBP-USD-spot-exchange-rates-history-2021.html
In a network meta-analysis and cost evaluation, IV vancomycin was compared to oral linezolid, tedizolid, daptomycin, and ceftaroline. For the clinical cure of ABSSSI due to MRSA, linezolid was superior to vancomycin, while tedizolid was similar to vancomycin, with no significant safety differences between any 2 interventions. The cost per life-year saved when comparing vancomycin to linezolid, tedizolid, daptomycin, and ceftaroline were US$2833; US$5523; US$6200, and US$9057; respectively. The authors note that they applied a Willingness-To-Pay threshold of US$50 000 and these 4 alternative antibiotics are all within this threshold, with the oral therapies being the least costly. 59 Therefore, any of these agents could be potential alternatives for the treatment of ABSSSI due to MRSA in health technology assessments in the US 71 or UK. 72
There are also additional costs from complications associated with the vancomycin PICC line, and the treatment of vancomycin-associated AEs from the use of vancomycin itself.30,73,74 For example, vancomycin-associated AKI is associated with a prolonged hospital stay, hospital readmission rate, and patient mortality. 75 Guidelines suggest additional monitoring for patients at risk of vancomycin-associated AKI (eg, patients with diabetes), further adding to the costs involved. 40
Accessibility
With increasing levels of antimicrobial resistance and few new antibiotics in the developmental pipeline, many institutions have antimicrobial stewardship programs in place to ensure the appropriate use of antibiotics. 76 Antimicrobial stewardship programs include formulary restriction for inpatient use, but there may be a misalignment between the best therapy in an inpatient setting where monitoring and nursing care is readily available, compared to the at-home treatment setting. For example, explorations in the hospital setting to support stewardship have led to conclusions counterintuitive to optimal at-home therapy, for example, narrow-spectrum OPAT with multiple daily doses. 77 This is counterintuitive when studies have consistently demonstrated that patients have better outcomes for drugs that have less frequent dosing schedules.78,79 Furthermore, restricted testing of formulary options has limited the scope for step-down or alternative therapies, in the case of side effects.
Clinical Specialists’ Opinions
Specialists may prescribe IV therapies because they work in the hospital setting, where IV equipment and consumables are readily available. They may also prefer IV therapy over oral due to the risk of bacteremia, which typically requires 2 to 6 weeks of IV therapy.41,80
Evidence is changing the perception of specialists. A 2020 survey of 665 IDSA physicians reported that 88% would transition patients with Gram-negative bacteremia to complete oral antibiotic course, 71% would transition patients with Gram-positive bacteremia to oral agents, and only 12% would not use oral agents to treat any patient with baceteremia. 81 It should be noted that the survey did not report the opinions for oral therapy for DFI and osteomyelitis. However, given the large burden of disease, infection severity, and risk of mortality, it may be reasonable to extrapolate similar assumptions.
Conclusion
Diabetic foot infections are a common complication of diabetes and can lead to amputation, with a severe impact on patient QoL. One of the most commonly identified microorganisms in DFIs is S. aureus, including MRSA, and as such, is typically targeted in empiric therapy. Evidence suggests that people with diabetes may be at an increased risk of MRSA infection, which typically means a long course of treatment and the possibility of severe infection. Current FDA guidance has narrowed the possible therapeutic options for DFIs, particularly where MRSA is a risk, leading to the widespread reliance on vancomycin to treat DFIs. However, the use of vancomycin is associated with a number of issues that warrant the investigation into the use of alternative therapies for these infections. Vancomycin-associated issues include higher risks of AEs such as nephrotoxicity and AKI, increased length of stay, higher rates of readmission, and high costs to the healthcare system, patients, and carers. RCTs, systematic reviews, and meta-analyses indicate that oral therapies are noninferior to IV therapies, and there are numerous alternative oral counterparts which are effective, tolerable, and lower costing than their IV counterparts. The IWGDF lists “what is the place of various new antibiotics in the management of DFI” as one of their “Key Controversies.” This review attempts to answer that question by demonstrating that many of these newer antibiotics can play a significant role in the treatment of DFI while avoiding complication associated with some of the more established antibiotics. 21 Where hospital admission or OPAT can be avoided with oral therapy, this can benefit patients in terms of their QoL and avoidance of AEs, as well as allowing substantial cost savings for the healthcare system.
Footnotes
Acknowledgments
Images provided by Lee C. Rogers. Medical Writing assistance was provided by Meridian HealthComms Ltd, Manchester, UK, funded by Paratek Pharmaceuticals, Inc. according to Good Publication Practice (GPP4).
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Dr Joseph receives honoraria from and is on the Speakers Bureau for Paratek Pharmaceuticals, Inc. Dr Kosinski is listed as Additional Study Personnel as part of the Investigator Initiated Research (IIR) program for Paratek Pharmaceuticals, Inc.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
