Abstract
Severe lower extremity wounds have an increased risk of complications and limb loss. The aim of this study was to evaluate and compare the microbiological profile and antibiotic resistance of wounds in diabetic and nondiabetic patients. A cross-sectional comparative study was carried out at a public hospital including 111 patients with moderate to severe wound infections. Tissue samples were collected during a surgical procedure. One hundred and four patients (94%) had positive cultures and 88 (79%) had a Gram-negative microorganism. Among the 185 cultured microorganisms, 133 (72%) were Gram-negative species. Pseudomonas aeruginosa (23 cases) was the most isolated Gram-negative species, and Enterococcus faecalis (26 cases) was the most prevalent Gram-positive species. Among 185 isolated species, 45 (24%) were extended-spectrum beta-lactamase producers, 23 (12%) were carbapenem-resistant, and 5 (3%) were methicillin-resistant Staphylococcus aureus. Findings revealed that there was no significant difference in the microbiological profile and antibiotic resistance among patients with lower extremity wounds whether they were diabetic or nondiabetic.
Diabetes mellitus (DM) and its complications are responsible for about 80% of lower limb amputations worldwide. 1 Neuropathy is the main cause of injuries and ulcerations in diabetics, and half of the patients with neuropathic ulcers may develop an infection, with an amputation rate up to 20%. 1 In addition to neuropathy, peripheral vascular disease also contributes to severe foot lesions in diabetics, increasing the risk of major amputations. 2 Furthermore, DM is a risk factor for atherosclerosis, the main cause of cardiovascular and peripheral artery disease (PAD).3,4 Deep lower extremity wounds, when followed by infection and ischemia, carry a high risk of limb loss.2,5,6 Although DM is the leading cause of chronic lower extremity wounds, other common etiologies are prolonged unrelieved pressure, chronic venous disease, and PAD. 7
Chronic wound healing can be affected by ulcer location, size, and depth. 8 Classification systems for diabetic foot wounds consider size and depth, local or systemic infection, and absence of pulses as relevant criteria for a severity score. 9 The wound, ischemia, and foot infection (WIfI) Classification System developed by the Society for Vascular Surgery is based on 3 categories (WIfI) with 4 grades of severity. 10 Infection is an extrinsic factor that contributes to nonhealing wounds, even among patients who have undergone prior revascularization for critical limb ischemia.7,11,12 Foot lesions associated with infection and ischemia have a more complex treatment and an increased risk of lower limb amputation.2,4,5
Empirical antibiotic therapy is an initial measure in cases of infected wounds. The use of clinical protocols and knowledge of the microbiological profile may help in the choice of empirical antibiotic therapy. 13 In the absence of knowledge about the local microbiological profile, published research studies and guidelines served as the basis for choosing the appropriate antibiotics. 2 In severe infections, deep tissue cultures are indicated and the antibiogram provides the options for treatment of the causative pathogens. 14 Excessive antibiotic use has a significant impact on the treatment costs and on the resistant microorganisms selection, being one of the responsible factors for the growing bacterial resistance rates in different countries.13,15,16
In several regions of the world, including Latin America, the number of complications of diabetic foot infections has been increasing in recent years. 1 Hospitalization secondary to diabetic foot wounds has high costs, prolonged hospital stay, and high morbidity and mortality. 17 The knowledge about the microbiological profile can help in choosing the most appropriate antibiotic therapy, increasing the effectiveness of the treatment, reducing costs, and avoiding the selection of multiresistant bacteria. The aim of this study was to evaluate and compare the microbiological profile and antibiotic resistance in diabetic and nondiabetic patients with deep lower extremity wounds.
Patients and Methods
Patients
A single-center, observational, cross-sectional, retrospective, and comparative study was conducted at Complexo Hospitalar Universitário Professor Edgard Santos, Federal University of Bahia, Salvador, Brazil. The present study included 111 patients consecutively treated for lower extremity wound lesions with moderate and severe infection degrees in a vascular surgery service of a tertiary referral hospital, between December 2014 and January 2018. Only patients with deep foot wounds that required some surgical interventions were included. All patients had deep wound lesions grade ≥3 according to the Wagner system, Category 5 or 6 from Rutherford's classification, moderate to severe infection in a clinical severity Infectious Diseases Society of America (IDSA) score, and wound grade 2 or 3 in WIfI classification. Patients were excluded from the study if they did not have signs of infection neither surgical procedure at the foot was needed. This study was conducted at a tertiary care reference center and all patients received antibiotics in the trimester before hospitalization. Data collection from medical records and the microbiological registers from the Clinical Microbiology Laboratory, at the same institution, were performed. All cases had tissue samples collected in the operating room, during the surgical procedure. Following tissue collection, specimens were placed in sterile transport containers and sent to the Microbiology Laboratory for microbiological analysis and antibiogram.
Methods
Lower extremity tissue samples were processed in <2 h after collected and cultivated in blood agar, MacConkey agar, and brain heart infusion broth and oven incubated at 35 °C ± 1 °C, for 18 to 24 h. After primary isolation, positive samples were subjected to identification and susceptibility testing to antibiotics, using the automated system Vitek 2 (bioMerieux—Marcy I’Etoile), following the manufacturer processing recommendations for inoculation and incubation of the cards. The interpretation of susceptibility test results was carried out following the recommendations of the Clinical and Laboratory Standards Institute. The strains American Type Culture Collection, Escherichia coli 25922, Staphylococcus aureus 29213, Enterococcus faecalis 29212, and Pseudomonas aeruginosa 27853 were used as an identification quality control and susceptibility test.
Studied Variables
Demographic characteristics of the patients (age and sex), comorbidities, DM, systemic arterial hypertension (SAH), PAD, chronic kidney disease (CKD), and 3 different classifications were studied for diabetic and nondiabetic patients. DM, SAH, and CKD patients had a previous diagnosis and were undergoing treatment at the same hospital. All patients diagnosed with PAD had critical limb ischemia, with the absence of both foot pulses (posterior tibial and pedal pulses) confirmed by digital-subtraction angiography or angiotomography (chronically occluded segments). Lower extremity wound lesions were classified according to the Wagner system, Rutherford's classification, IDSA 2012 severity score, and WIfI Classification System.2,4,9,10
Isolated microorganisms were identified according to the Gram-staining method, bacterial species, and antibiogram results. In this study, we performed a comparative analysis of the presence of antibiotic-resistant bacteria in diabetic and nondiabetic patients. We compared two groups with respect to the presence of methicillin-resistant S aureus (MRSA), vancomycin-resistant Enterococcus (VRE), vancomycin-resistant S aureus (VRSA), and Gram-positive linezolid-resistant. Regarding Gram-negative bacteria, the study analyzed the presence of extended-spectrum beta-lactamase (ESBL)-producing species, carbapenem-resistant isolates, and multiresistant P aeruginosa, according to the IDSA guidance. 18
Statistical Analysis
Statistical analysis was performed using EPI INFO™ version 7.2.2.6 and descriptive statistics were used to study the microbiological profile. Qualitative variables had their frequencies described and comparatively analyzed by the chi-square test. Fisher's exact test was used to compare multidrug-resistant organisms between diabetics and nondiabetics. Continuous variables had their means and standard deviation described and comparative analysis was performed by analysis of variance. Statistical significance was assumed at P values <.05.
Ethical Approval
The study protocol received full approval from the Research Ethics Committee of the Complexo Hospitalar Universitário Professor Edgard Santos, Federal University of Bahia, project approval number 1.390.740/33051514.0.0000.0049.
Results
Population of Patients
A total of 111 patients were included in the study population. Among the 111 patients, 57 (51%) had neuroischemic foot wounds (DM and PAD), 23 (21%) had neuropathic foot ulcers (only DM), 22 (20%) had ischemic foot wounds (only PAD), and 9 (8%) cases had autoimmune or vasculitic etiologies. Patients’ mean age was 63.6(±12.8) years. The median duration of the wounds was 60 days. Eighty patients were diabetic (72%) and 79 patients had PAD (71%). According to the IDSA severity score, 91 patients (82%) had severely infected lesions and 20 (18%) had moderate ones. Regarding the WIfI classification, all patients had grades 2 or 3 for wound along with grade 2 or 3 for foot infection and 62 patients (56%) had grade 3 for ischemia. The study population characteristics are detailed in Table 1.
Demographic and Clinical Characteristics of the Study Population (N = 111 Patients).
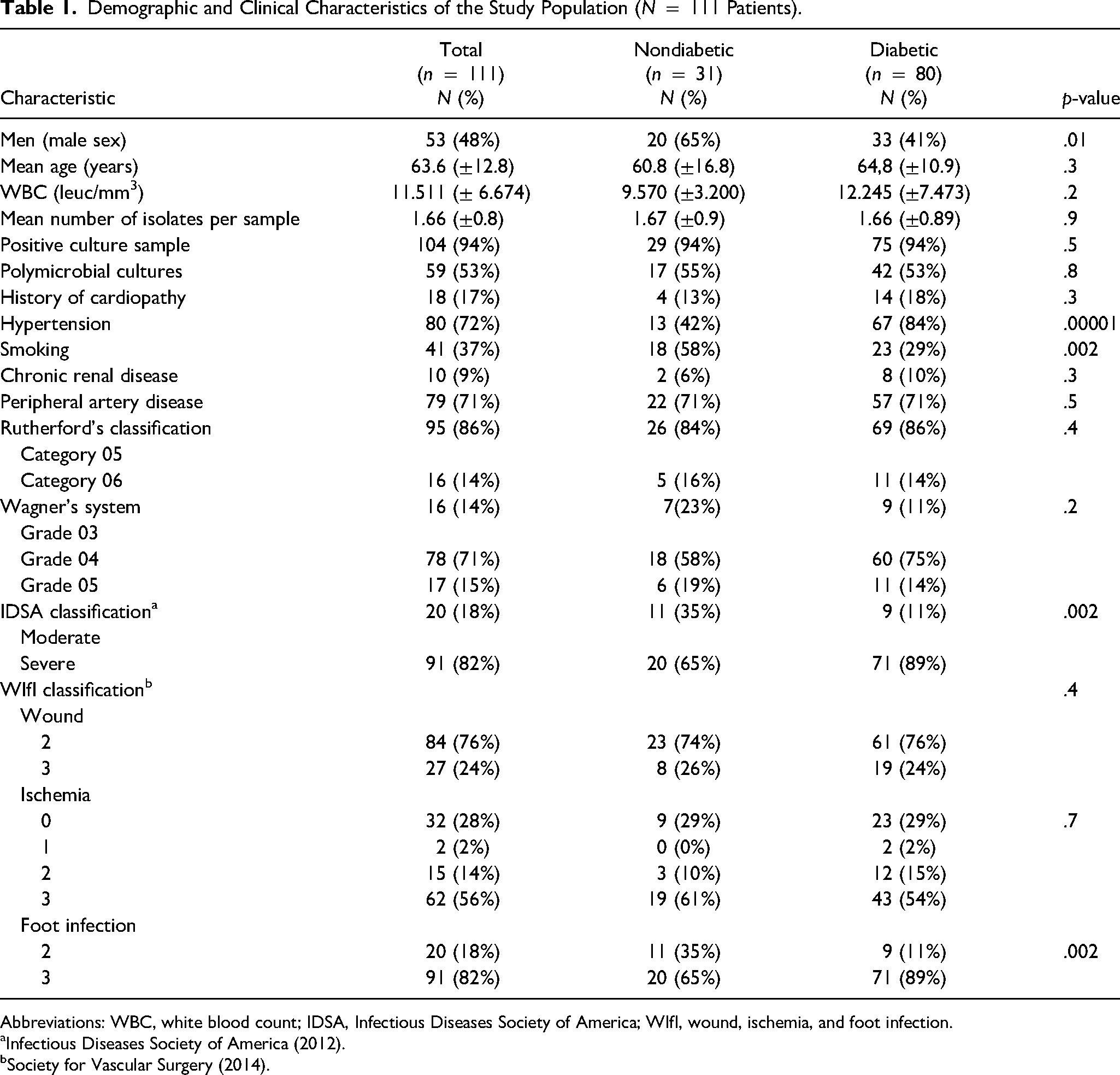
Abbreviations: WBC, white blood count; IDSA, Infectious Diseases Society of America; WIfI, wound, ischemia, and foot infection.
Infectious Diseases Society of America (2012).
Society for Vascular Surgery (2014).
Microbiology
A total of 185 bacteria were isolated, resulting in an average of 1.66 (±0.8) microorganisms per sample. Among the 111 patients, 104 (94%) had positive tissue cultures. Gram-positive microorganisms were present in 46 (41%) of the 111 cases and 52 Gram-positive species were cultivated. Gram-negative microorganisms were present in 88 patients (79%) and 133 Gram-negative species were cultivated. Forty-five patients (41%) had only 1 organism isolated from the lower extremity wound lesion.
Among the 185 isolated species, 133 (72%) isolates were Gram-negatives and 52 (28%) were Gram-positives. Among 133 Gram-negative isolated species, members of the family of Enterobacteriaceae were the predominant group (104/78% microorganisms). Considering all tissue samples, the most common isolated species were E faecalis (26 tissue samples) and P aeruginosa (23 tissue samples). S aureus was cultivated in 10 patients (5.4% of 185 causative pathogens) (Table 2).
Culture Results of the 185 Organisms Isolated From 111 Deep Tissue Samples.

Antibiotic Resistance
Considering the 111 patients in the study population, 87 (78%) had at least 1 Gram-negative isolate that can resist some kind of antibiotic and 33 patients (30%) had at least 1 Gram-positive resistant organism in a tissue sample. Among the 185 isolated species, 45 (24%) were ESBL-producing microorganisms, 23 (12%) carbapenem-resistant Gram-negative, and 5 (3%) were MRSA. There was no Gram-positive resistance to vancomycin or linezolid. Considering the 133 species of Gram-negative isolates 45 (34%) were ESBL-producing bacteria and 23 (17%) carbapenem-resistant germs (Table 3). The most common ESBL-producing pathogens were Klebsiella pneumoniae (11 cases), followed by Proteus spp. (9 cases) and E coli (8 cases). Among the 23 carbapenem-resistant isolates, Proteus mirabilis were the most common species (7 cases) followed by Pseudomonas aeruginosa (4 cases). Among the four Acinetobacter baumannii isolates, 3 (75%) of them were carbapenem-resistant. From a total of 23 Pseudomonas aeruginosa isolates, 6 (26%) were multidrug-resistant.
Antibiotic Resistance Among 133 Gram-Negatives Isolated (111 Tissue Samples).

Abbreviation: ESBL: extended-spectrum beta-lactamase.
ESBL-producing species.
45 ESBL-producing species from 35 patients/tissue samples.
23 Carbapenem-resistant isolates from 20 patients/tissue samples.
Among 52 isolated Gram-positive microorganisms, 10 (19%) were S aureus and half of them were methicillin-resistant (5 MRSA). No linezolid or vancomycin resistance was found for Staphylococcus spp. or Enterococcus spp. in our samples.
Comparative Analysis Between Diabetic and Nondiabetic Patients
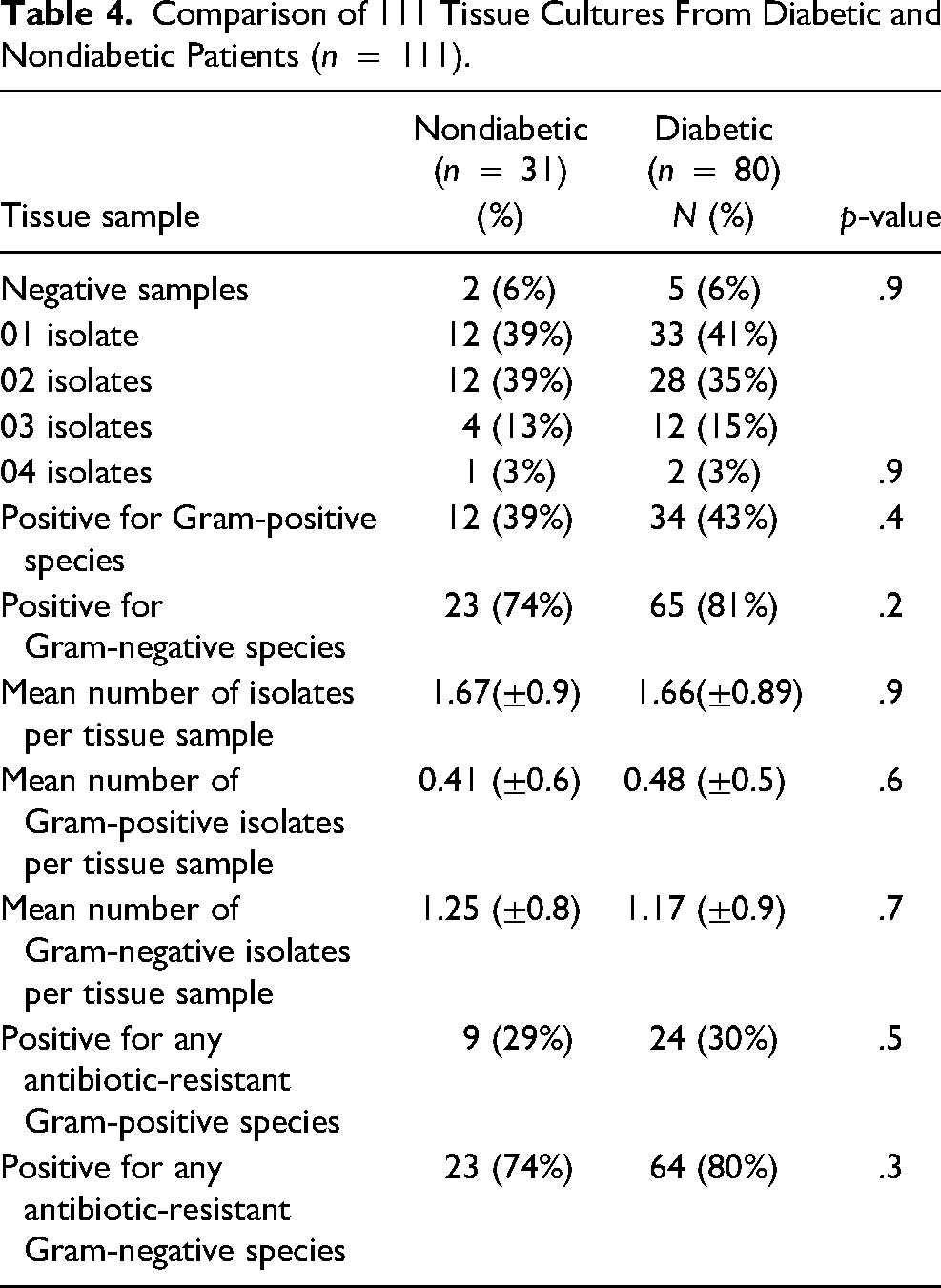
Comparing the results of the microbiological profile between diabetic and nondiabetic patients no significant differences were found regarding the number of microorganisms or the predominance of Gram-negative species (Table 4). Considering the antimicrobial resistance the average of ESBL-producing per tissue sample among diabetics was 0.45 (±0.7) versus 0.29 (±0.6) among nondiabetics (p = .3). In qualitative analyses, although diabetics had a greater presence of ESBL-producing Gram-negative species (35% vs. 23%) the difference was not significant (p = .2). The results showed no significant differences in the presence of MRSA or carbapenem-resistant microorganisms between the two groups (diabetics and nondiabetics) (Table 5).
Comparison of 111 Tissue Cultures From Diabetic and Nondiabetic Patients (n = 111).
Comparison of Diabetic and Nondiabetic Patients regarding Antibiotic-Resistant Isolated bacteria in the Tissue Sample (N = 111 Cases).
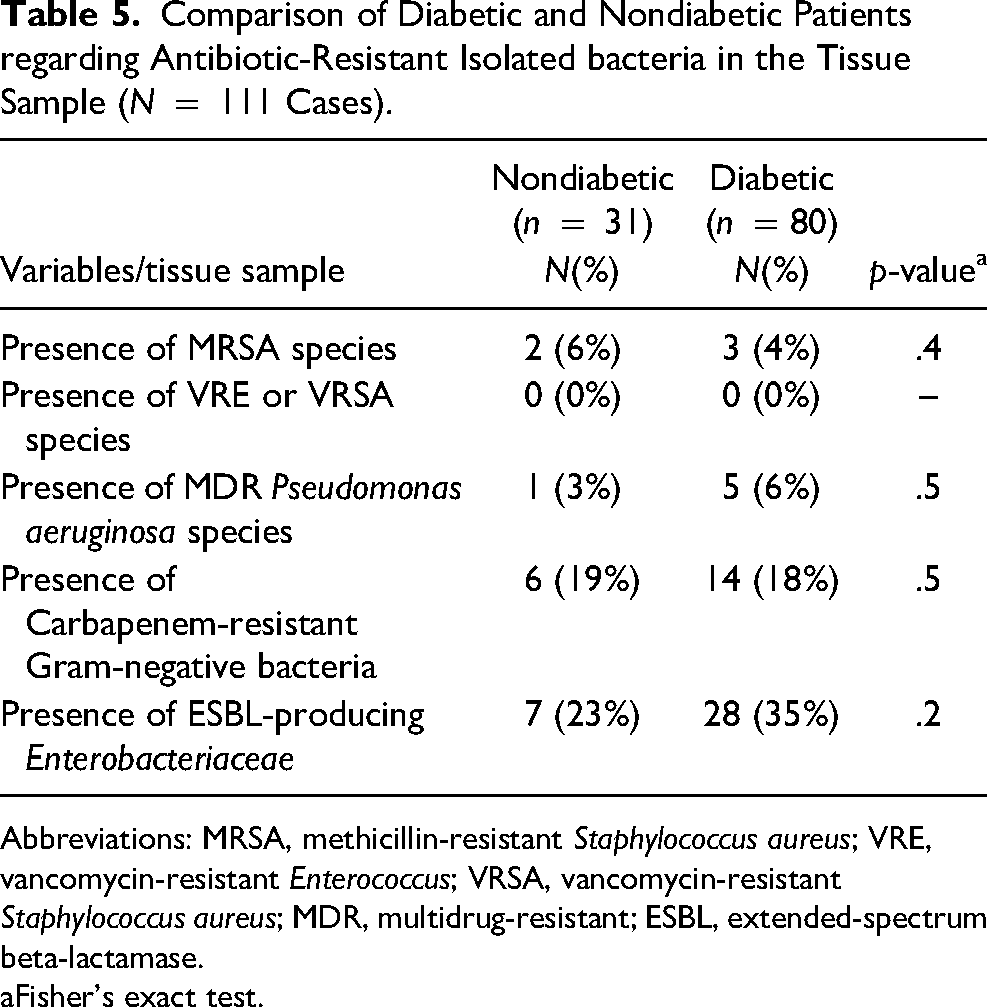
Abbreviations: MRSA, methicillin-resistant Staphylococcus aureus; VRE, vancomycin-resistant Enterococcus; VRSA, vancomycin-resistant Staphylococcus aureus; MDR, multidrug-resistant; ESBL, extended-spectrum beta-lactamase.
Fisher's exact test.
Discussion
The results of this study have demonstrated that in severe lower extremities wound of diabetic and nondiabetic patients there is a predominance of Gram-negative microorganisms, notably of the family Enterobacteriaceae. The most frequently isolated bacteria were Enterococcus faecalis followed by Pseudomonas aeruginosa. More than 70% of the study's population had at least 1 Gram-negative pathogen in the tissue sample. About a third of the Gram-negative isolates were ESBL producers. Klebsiella pneumoniae and Citrobacter freundii had a high percentage of ESBL-producing and Acinetobacter baumannii showed a high rate of carbapenem-resistance. We found no significant difference between diabetics and nondiabetics regarding the microbiological profile or the pattern of antibiotic resistance.
Similarly, other reports have shown a predominance of Gram-negative species, notably Enterobacateriaceae, in diabetic foot infections.19–23 A multicentric study found a higher prevalence of Gram-negative microorganisms (56.1%) in moderate infections. 20 Another study performed at 35 centers, including patients with infected wound lesions, showed that 60.02% of the 387 microorganisms were Gram-negative isolates and the most frequent were E. coli, P. aeruginosa, and Proteus spp. 19 In a reference tertiary hospital in India, 289 bacteria were obtained from 178 foot tissue samples and 165 were Gram-negative pathogens with 102 (61.8%) belonging to the Enterobacteriaceae family. 22
In contrast to earlier findings, several studies suggest that Gram-positive cocci, notably S aureus, are the predominant pathogens in diabetic foot infections.24–26 A multicentric study that cultured 454 specimens (1.145 aerobic pathogens) found that 80.3% were Gram-positive bacteria, the most commonly encountered were Staphylococcus spp. and 19.6% were Gram-negatives. 21 However, different methods were used to collect the specimens, besides exclusion of patients with critical limb ischemia or that had received recently antibiotic therapy. 26 In a retrospective study, other authors analyzed deep cultures of 92 diabetic patients and also find a predominance of Gram-positive bacteria, particularly S. aureus (55.38% of tissue cultures) with 17.30% of MRSA and 4.34% of VRE. 27 A study that includes 405 consecutive diabetic patients (56.5% mild infections) observed a predominance of Gram-positive strains (59%), being S aureus the most common isolates. 28 Among pathogens isolated from bone samples in a study that included 76 diabetic patients with osteomyelitis (28.9% had PAD and 14.4% required surgical debridements), other authors found a predominance of Staphylococci (52%). 25 A single-center study that included 21 outpatients with diabetic foot ulcer (excluded critical limb ischemia and moderate/severe infections) also found a predominance of Gram-positive, with 36.8% Gram-negative (21% Enterobacteriaceae group). 29
The observed differences in microbiology and antibiotic resistance could be due to the study population characteristics. Our study included inpatients in a tertiary care hospital with deep wounds, mostly with severe infection and a high prevalence of critical limb ischemia. These patients often had ambulatory treatment and received previous antibiotic therapy. Also, the differences in the microbiological profile can be due to the analyses of deep tissue samples, collected during surgical procedures. In the literature, other authors have already demonstrated the difference between the results of superficial and deep cultures,25,30 and in the current study, all tissue samples were collected in the operating room. There are, however, other possible explanations. Another possibility is that Gram-negative causative pathogens are more commonly isolated in patients residing in a tropical/subtropical climate zone than in those living in temperate climates. 14
Regarding antibiotic resistance, the results of this study show that a third of the Gram-negative isolates are ESBL-producing Enterobacteriales and 3 patients have carbapenem-resistant Acinetobacter baumannii (CRAB) in tissue samples. This finding is consistent with a multicentric study that included 477 diabetic foot patients and found 58.8% of Ceftriaxone susceptibility and about 90% of carbapenem susceptibility among Gram-negative isolates. 14 It was also shown that only 30% and 27,7% of Acinetobacter ssp. had susceptibility for Imipenem and Meropenem, respectively. 19 The present study found that only 25% of the A baumanii isolates had carbapenems susceptibility. A prospective study conducted in Egypt analyzed 124 aerobic isolates from 98 patients with diabetic foot and found 98 (79%) Gram-negative pathogens, with a higher proportion of Gram-negative as the severity of infection increases (18, 36, and 44 were isolated from mild, moderate, and severe diabetic foot infections, respectively). 31 Among Gram-negatives, the authors also found 48 (49%) ESBL producers and 40 (40.8%) carbapenemase producers (77.8% of Klebsiella spp. were ESBL-producing and 83.3% of Acinetobacter spp. were carbapenem-resistant). 31
The literature emphasizes that antibiotic resistance is a global health problem.32,33 In Portugal, a review presented findings of increased antibiotic resistance of the Enterobacteriaceae family (especially K pneumoniae), P aeruginosa, and A baumannii species. 34 In Holland, a study population of 346 residents of long-term care facilities had a prevalence of 18.2% of multidrug-resistant Gram-negative colonization and 14.5% of ESBL-producing Enterobacteriales (0.8% MRSA and 0% VRE) among the 12 long-term institutions (prevalence of ESBL-producing bacteria varied from 0% to 34%). 35 In 2019, the Centers for Disease Control and Prevention reported a decrease in the number of infections caused by VRSA (41%), MRSA (21%), and CRAB (33%), while the number of carbapenem-resistant enterobacterales (CRE) infections remained stable and the number of ESBL-producing Enterobacteriaceae infections increased 50% in the United States. 36
In our sample, we observed a low prevalence of S aureus among cultivated microorganisms, half of which were MRSA. The low prevalence of MRSA in diabetic foot infections was found by other authors, who verified 1.8% MRSA among all 387 microorganisms isolated in the cultures of the 447 patients included in the study. 19 In Greece, a study included 113 diabetic patients with infected ulcers and found S aureus in 24 cases, nine of them being MRSA. 37 Several reports have shown the predominance of S aureus in diabetic foot infections and differences in the prevalence of MRSA.21,26,27,38 A survey of 185 patients diagnosed with osteomyelitis, most of them without associated soft tissue infection, revealed that S aureus was the most prevalent, cultivated in 95 bone cultures with 35 (36.8%) MRSA. 39 The predominance of S aureus was also reported in a randomized comparative study about diabetic foot osteomyelitis. 40 Again, the difference in this study findings may be due to the predominance of severe infected and ischemic wounds in patients hospitalized in a tertiary care center and all specimens were collected during surgery.
In our study, we found no vancomycin-resistant Enterococcus or VRSA. A literature review showed an overall VRSA prevalence of 1.5% among 5855 S aureus isolates, increasing over the years, being 1.2% in 2444 isolates from surveys prior to 2010, and 2.4% in 3411 isolates from surveys conducted between 2010 and 2019. 41 Vancomycin-resistant S aureus or other vancomycin-resistant Gram-positive organisms were not found in diabetic foot infections by other studies.21,26,31
In this study, a comparative analysis between diabetic and nondiabetic patients found no significant differences in the microbiological profile and antibiotic resistance, despite a higher prevalence of ESBL-producing Enterobacteriaceae among diabetics. Likewise, some authors compared the microorganisms responsible for urinary infections in diabetics and nondiabetics and found no difference in the microbiology (E coli was the most frequent infection cause in both groups: 71% vs. 69%) neither in the E coli antibiotic resistance. 42 Another study about urinary tract infection did not found either that DM was a significant risk factor for E coli fluoroquinolones resistance. 43 A retrospective study that included 115 patients (41 DM vs 74 non-DM) with different types of wound lesions also found that DM was not a risk factor for multidrug-resistant organisms (MDRO) infections (63.4% DM vs. 50% non-DM). 44 A possible explanation for the absence of significant difference between diabetic and nondiabetic microbiology is that other factors as infection severity, wound depth, previous antibiotic treatment, and inpatient tertiary care might have more impact on the microbiological profile.
The results of this investigation showed that hospitalized diabetic and nondiabetic patients, in tertiary care hospitals, with severe deep wound lesions and limb ischemia have a high prevalence of Gram-negative microorganisms and ESBL-producing Enterobacteriaceae. These findings suggest that Gram-negative rods are the most common causative pathogens for inpatients with severe foot infections. Considering that severe foot infections are initially treated empirically, these results may be helpful for the choice of the initial antibiotic regimen. After the causative organisms and their antibiotic susceptibilities had been identified, the first antimicrobial choice can be modified. A systematic review showed that 17.8% to 72.5% of diabetic foot infections are caused by MDRO and found a possible association between amputations and MDRO diabetic foot infections. 38 In diabetic foot infections, Gram-negative rods, persistent infection located in the bone, peripheral arterial disease, and ischemic heart disease were related to undergoing a major amputation after revision surgery, while MRSA infection was not associated. 45 Among patients with diabetic foot osteomyelitis, Gram-negative isolates were associated with more severe infections. 46 Although this study did not analyze diabetic foot infection outcomes, the high prevalence of Enterobacteriaceae suggests Gram-negative empirical antibiotic coverage for hospitalized patients with a severe foot infection, while waiting for the tissue culture and susceptibility testing results.
This study has several limitations. The main limitation of the present study is that the single-center and retrospective study design might have produced bias in the results. Also, the study population only included inpatients in a reference tertiary care hospital. Differences between populations' sizes may limit the statistical power of the tests. The hospital’s geographic location can influence the microbiological profile. Some classification systems were developed for diabetic foot ulcers. Anaerobic cultures were not performed. The absence of data about previous hospitalization and antibiotic therapy is a relevant limitation and may have biased the findings. Furthermore, antibiotic use before the specimen collection could affect the proportion of Gram-negative rods. Despite these limitations, this study contributes to the knowledge about the microbiological profile and antibiotic resistance in deep infected and ischemic wound lesions, a common health challenge that carries a risk of limb loss. Future studies are necessary to find the best empirical antibiotic treatment for mild, moderate, and severe foot infections.
In conclusion, diabetic and nondiabetic patients with severe and deep lower extremity wound lesions had most commonly Gram-negative infections, with a predominance of Enterobacteriaceae, and about one-third of them had an infection by ESBL-producing species.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
