Abstract
The venous leg ulcers are debilitating, painful, and often unresponsive to advanced dressing treatments, so drugs used locally and systematically are essential adjuvant therapy—pentoxifylline (PTX) whose anti-inflammatory effects may offer a promising avenue to treat venous leg ulcers. However, the current results are controversial. To further evaluate the efficacy and safety of PTX, we performed an updated meta-analysis of randomized placebo-controlled trials of PTX in the treatment of venous leg ulcers. We systematically searched multiple electronic databases PubMed, Web of Science, Embase, the Cochrane Library, the Cochrane Central Register of Controlled Trials, China Science and Technology Journal Database, WanFang Data, China National Knowledge Infrastructure, and the Chinese Biomedical Literature Database to identify eligible studies. Randomized clinical trials of pentoxifylline versus placebo treatment in patients with venous leg ulcers were considered for inclusion. The primary outcomes included ulcer healing rate and the incidence of adverse events after treatment. The secondary outcomes were the ulcer significant improvement (the ulcer size shrank by more than 60% after treatment) rate, mean duration of complete wound healing and changes in mean ulcer size. A meta-analysis and qualitative analysis were conducted to estimate endpoints. A total of 13 randomized clinical trials, including 921 individuals, were finally included. Compared with placebo, pentoxifylline significantly improved the ulcer healing rate (RR = 1.59, 95%CI 1.22 to 2.07, P < .001) and significant improvement rate (RR = 2.36, 95%CI 1.31 to 4.24, P = .004) while increased the incidence of gastrointestinal disturbances (RR = 2.29, 95%CI 1.04 to 5.03, P = .04) at the same time. Moreover, pentoxifylline also shortened mean duration of complete wound healing (P = .007) and shrank ulcer size (P = .02). Currently available evidence suggests that pentoxifylline could help venous leg ulcers heal more quickly and effectively. However, the evidence is insufficient to prove the results due to moderate-certainty evidence. Large-scale, well-designed randomized clinical trials are warranted.
Introduction
Venous leg ulcers (VLUs) occur with increasing prevalence, and their prevalence in the United States is approximately 10% to 30%.1,2 The available treatment options on VLUs include compression therapy, surgical debridement, hyperbaric oxygen therapy, growth factors, skin grafting, and oral drugs. 2 However, VLUs still cannot be solved successfully. VLUs result from a complex process secondary to increased pressure (venous hypertension) and inflammation within the venous circulation, vein wall, and valve leaflet with extravasation of inflammatory cells and molecules into the interstitium. 3 The treatment of ambulatory venous hypertension is either through the removal of obstruction via anticoagulation or ablation of superficial reflux, which is the key of VLUs management.4,5
Pentoxifylline (PTX) is a dimethylxanthine-derivative, which increases cyclic adenosine monophosphate (cAMP) levels in the smooth muscle of blood vessels. There is little evidence that PTX is an effective and convenient adjunct for treating venous ulcers with lower costs and mild adverse events. 3 The probable mechanisms of PTX promoting VLUs healing may be included but are not limited to the following: (i) PTX increases erythrocyte flexibility and improves blood flow in the vessels to ameliorate microcirculation and oxygen delivery, especially in the ischemic states; 6 (ii) PTX is related to the induction of prostacyclin synthesis and inhibition of phosphodiesterase E enzyme (PDE). PTX is a vasodilator and also a platelet aggregation inhibitor; 6 (iii) PTX decreases blood viscosity via its fibrinolytic effect; 6 (iv) PTX inhibits synthesis of inflammatory mediators, suppresses cytokine release, and restrains leukocyte function;6,7 (v) PTX can decrease oxygen-free radical production to reduce oxidative stress, and so on. 6
Some studies investigated that the combination of PTX and conventional treatment (compression, irrigation, and dressing, etc) increased ulcer healing rates and shortened ulcer healing time for VLUs.8,9 Other studies reported conflicting results.10–13 In a meta-analysis involving eleven randomized clinical trials (RCTs), PTX contributed to the higher ulcer healing or significant improvement rate than placebo (RR = 1.70, 95% CI 1.30 to 2.24). 14 Of which a trial published in 1999 was republished in 2007 after adding 20 patients to PTX group and 25 patients to placebo group.11,13 Nevertheless, the article republished in 2007 revealing that PTX had no better efficacy than placebo was not included in the previous meta-analysis. Moreover, a new study exploring the efficacy of PTX for venous ulcers was published in 2012. 15 Therefore, PTX treating patients with VLUs remains a matter of controversy. To further evaluate the efficacy and safety of PTX, we performed an updated meta-analysis of randomized placebo-controlled trials of PTX in the treatment of venous leg ulcers.
Materials and Methods
This systematic review and meta-analysis of RCTs has followed the recommendations from the Cochrane Collaboration and was conducted according to PRISMA (Preferred Reporting Items for Systematic Reviews and Meta-Analyses) guidelines. 16
Search Strategies
A systematic search of databases [PubMed/MEDLINE (1993 to 12 June 2021), Web of Science (1992 to 12 June 2021), Embase (1974 to 12 June 2021), the Cochrane Library (1990 to 12 June 2021), the Cochrane Central Register of Controlled Trials (CENTRAL) (1990 to 12 June 2021), China Science and Technology Journal Database (CQVIP) (1990 to 12 June 2021), WanFang Data (1999 to 12 June 2021), China National Knowledge Infrastructure(CNKI) (1987 to 12 June 2021), and the Chinese Biomedical Literature Database (SinoMed) (1990 to 12 June 2021)] was conducted to identify eligible studies. Keywords and MeSH terms pertinent to the exposure of interest were used in relevant combinations: “PTX”, “TRENTAL”, “pentoxifylline”,” oxpentifylline”, “varicose ulcer”, “leg ulcer”, “foot ulcer”. We also searched ClinicalTrials.gov and the World Health Organization International Clinical Trials Registry Platform and reviewed the reference lists of all relevant studies and abstract books in research proceedings.
Eligibility Criteria
We decided on inclusion criteria based on PICOS principles. Study population: Patients with venous leg ulcer; Interventions: PTX; Control group: placebo or no treatment; Primary outcomes: the ulcer healing rate and the incidence of adverse events after treatment; Secondary outcomes: the ulcer significant improvement (the ulcer size shrank by more than 60% after treatment) rate, mean duration of complete wound healing and changes in mean ulcer size; Study design: RCT. No restrictions on sample size, languages, and follow-up periods were imposed. Studies were removed from the analysis if they met one of the following exclusion criteria: (i) case reports, reviews, animal and in vitro experiments, conference abstracts, and single-arm trials; (ii) studies with incomplete important information.
Study Screening and Data Collection
Titles and abstracts were screened independently by two authors, and the full text of potentially eligible articles was sequentially assessed to determine the final roster of included studies. From included studies, we extracted data including study characteristics (authors’ name, year of publication, country, initial number in experimental and control groups), baseline characteristics (sex ratio, intervention, treatment paths, basic treatment, dose with a frequency of PTX, follow-up period, mean age, duration of reference ulcers) and outcomes. We extract data by independent author. Discrepancies were resolved through discussion or, if required, adjudication by a third author.
Risk of Bias Assessment
Two reviewers independently assessed the methodological quality of the RCTs according to the risk bias assessment tool in the Cochrane Handbook. 17 The risk bias assessment tool included seven items: random sequence generation, allocation concealment, blinding of patients and personnel, blinding of outcome assessment, incomplete outcome data, selective reporting, and other bias. Finally, we divided the literature according to the stratification of “low bias risk,” “high bias risk,” and “unclear bias risk.” The Egger's test was applied to assess publication bias. The bias was considered to be statistically significant if P ≤ .05. Any discrepancies were resolved by consensus.
Data Synthesis and Analysis
RevMan 5.2 software and Stata 12.0 were used for meta-analysis. Dichotomous outcome adopted risk ratio (RR) and 95% confidence intervals (CI) to pool the data. The synthesized results were summarized by the effect value with 95% CI derived from Inverse Variance and Mantel-Haenszel estimate and expressed by Forest plots. The I2 parameter and Chi-squared tests performed heterogeneity analysis. If I2 values<50%, there was no significant heterogeneity among the included studies, and the fixed-effect model was used for meta-analysis. If I2 values ≥50%, a random-effect model was used due to apparent heterogeneity. Sensitivity analysis was also performed to explore the robustness of outcomes using a “leave-one-out” approach. The GRADE profile was used to assess the certainty of the evidence for each outcome, which was graded as high, moderate, low, very low after all-around assessments of study limitations, inconsistency, imprecision, indirectness, and publication bias. 18
Results
Literatures Search and Study Selection
A total of 686 related literatures were retrieved. Five hundred and ninety-four records were screened after removing duplicate records. After reading titles and abstracts, 507 articles were excluded. Eighty-seven full-text articles were assessed for eligibility. A trial that compared PTX with defibrotide was not included in the present meta-analysis, because it was a positive control trial and might affect the assessment of the efficacy of PTX. 19 Finally, thirteen RCTs meeting the inclusion criteria were included (Figure 1). We also included the older study published in 1999 11 because of the lack of incidence of gastrointestinal disturbances in the same trial republished in 2007 13 although we contacted corresponding author for data but did not receive a reply. However, we were unable to contact experts in the field to identify further published and unpublished studies.
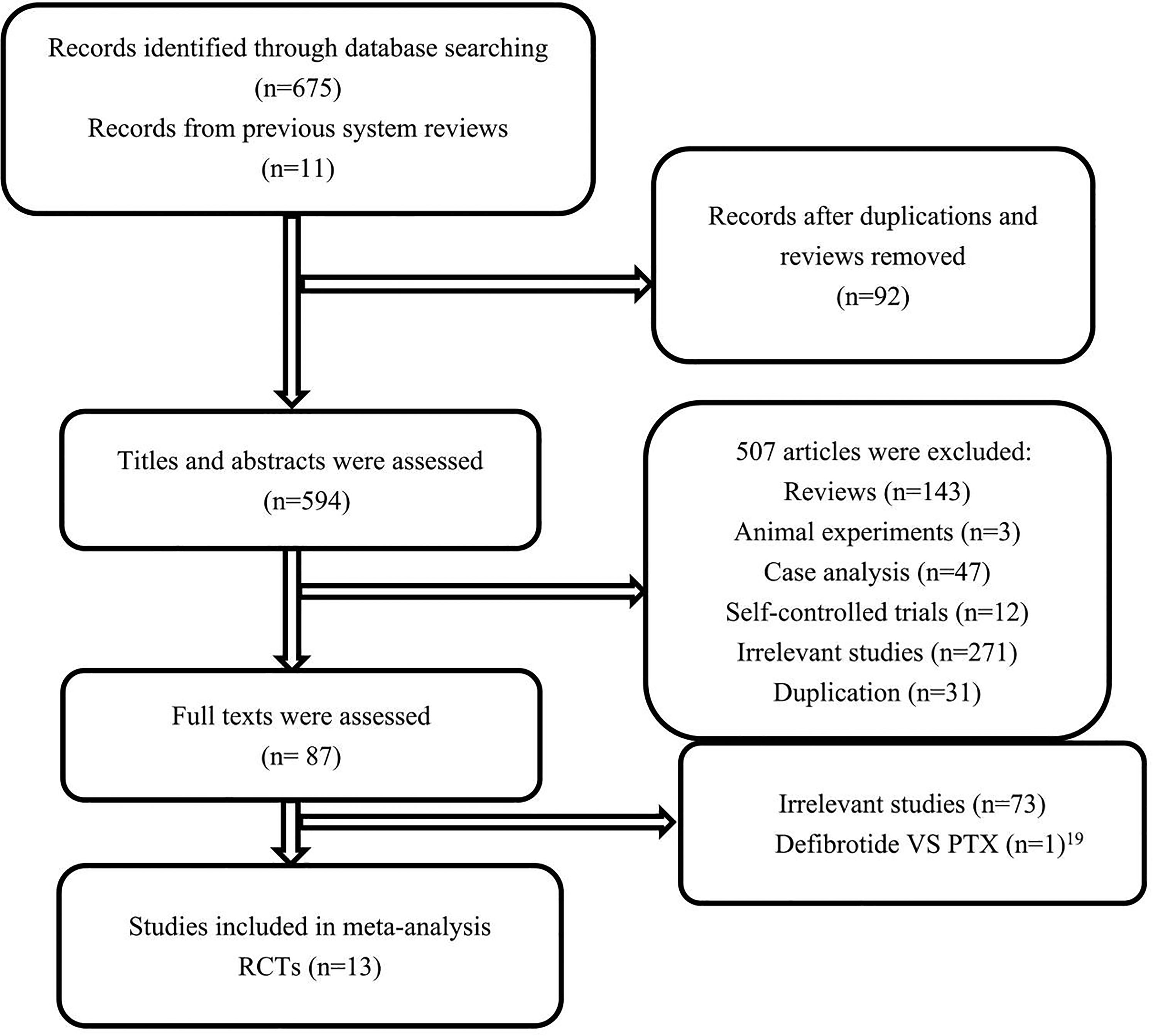
Flow chart of selection of studies.
Study Characteristics
Finally, a total of 13 RCTs with sample sizes ranging from 12 to 245 were eligible for inclusion8–13,15,20–25. All RCTs were published from 1983 to 2012. There were 921 patients aged 26 to 83 years old enrolled in RCTs, 481 in the intervention group, and 440 in the control group. Twelve studies employed oral treatment while one RCT combine oral with intravenous therapy. 10 All trials treated patients with 400 mg three times a day (TID), and one study also used 800 mg TID. 12 The baseline characteristics of these RCTs included were presented in Table 1. The information of the control group and the intervention group mentioned in Table 1 were comparable (no statistical difference).
Characteristics of the RCTs Included in the Systematic Review and meta-Analysis.
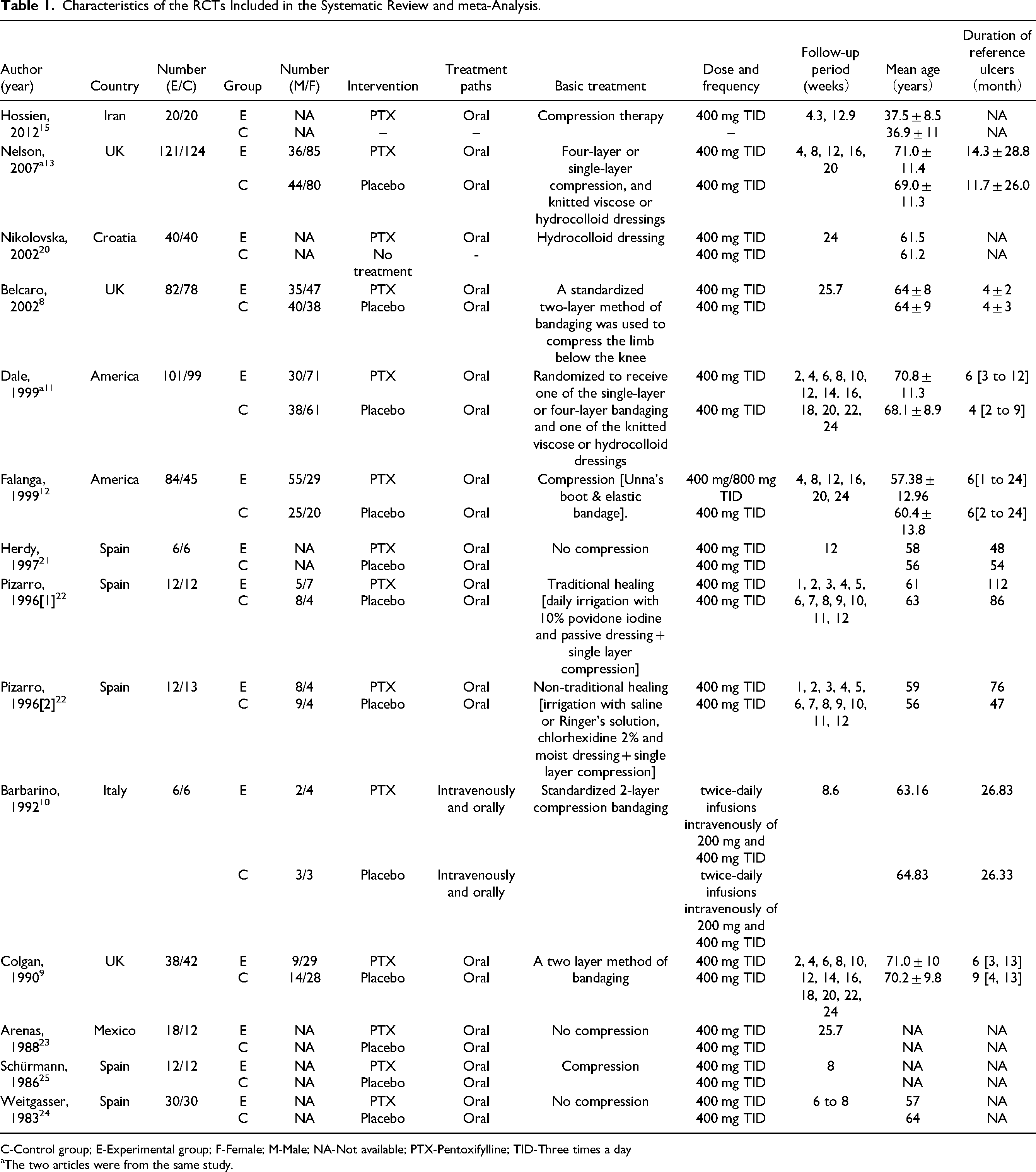
C-Control group; E-Experimental group; F-Female; M-Male; NA-Not available; PTX-Pentoxifylline; TID-Three times a day
The two articles were from the same study.
Risk of Bias
The methodological quality of the RCTs included was depended on evaluation for seven items. Because blinding did not affect performance bias and detection bias for ulcer healing and significant improvement rates, twelve studies were regarded as low risk of bias.9–13,15,20–25 Only one trial was classified as high risk of bias, because of the large difference between PTX group and control group in the number of lost follow-up (Table 2). 8 There was no publication bias in the ulcer healing rate (P = .075) and the incidence of gastrointestinal disturbances (P = .244).
The Methodological Quality of the RCTs Included.
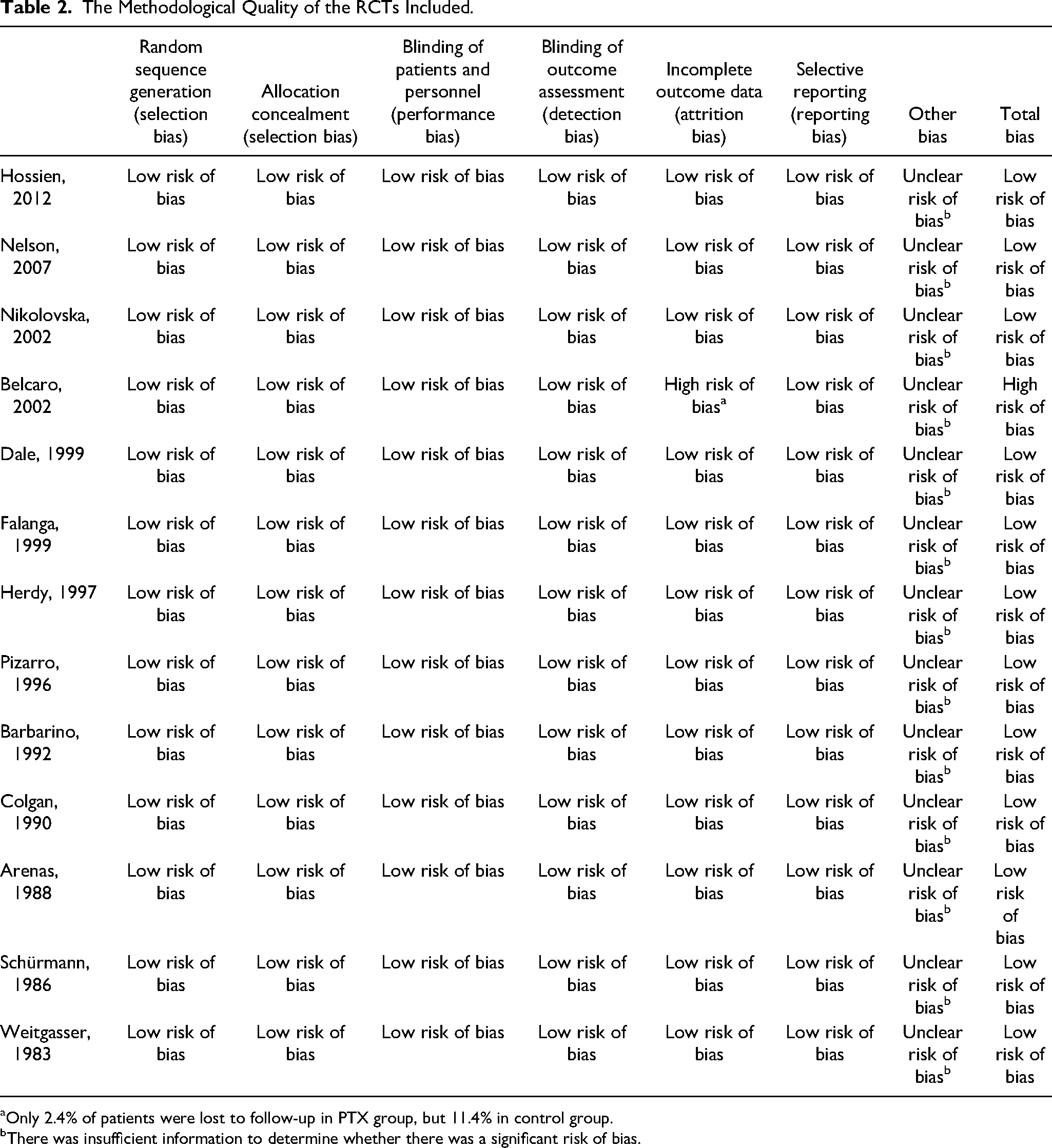
Only 2.4% of patients were lost to follow-up in PTX group, but 11.4% in control group.
There was insufficient information to determine whether there was a significant risk of bias.
Analyses of Outcomes
The Ulcer Healing Rate and Significant Improvement Rate
Eight trials (n = 779, not include Dale 1999) reported the ulcer healing rate at the endpoint. 8–10,12,13,20,22,25 A random-effect model was used for meta-analysis because high heterogeneity was detected among studies (Chi2 = 18.12, P = .02, I2 = 56%). As shown in Figure 2, PTX was significantly associated with increased ulcer healing rates, compared with control (RR = 1.59, 95%CI 1.22 to 2.07, P < .001). Three RCTs (n = 101) reported the ulcer significant improvement rate.21,23,24 We concluded that the ulcer significant improvement rate was significantly higher for the PTX group versus the placebo group (RR = 2.36, 95%CI 1.31 to 4.24, P = .004, I2 = 0%). (Figure S1)
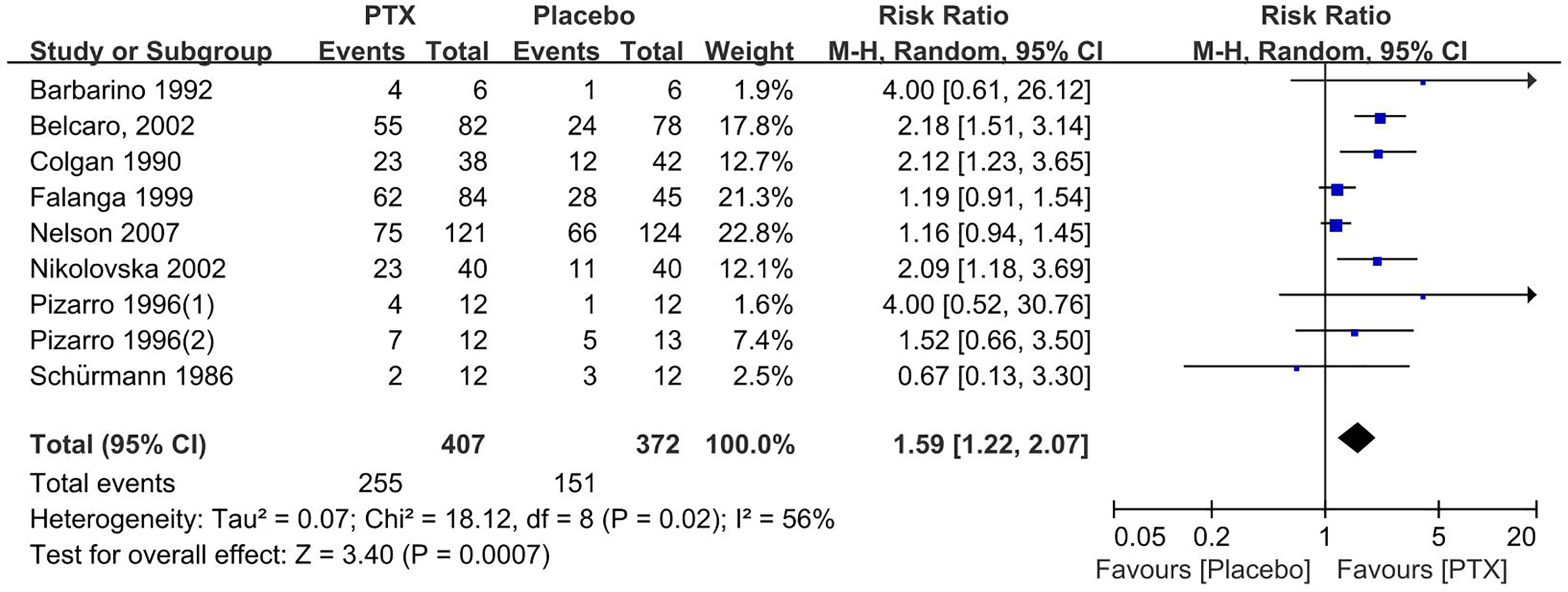
A meta-analysis of the effect of pentoxifylline (PTX) treatment compared with placebo (control group) on the ulcer healing rate.
Subgroup Analysis About the Ulcer Healing Rate
Seven trials8–10,12,13,22,25 adopted bandaging to improve venous drainage in PTX and placebo group but one 20 not. We performed subgroup analysis to evaluate the influence of compression by bandaging on the ulcer healing rate. From Figure S2, we concluded that compression did not influence the result that PTX treatment had better efficacy than placebo (RR = 1.53, 95% CI 1.15 to 2.03, P = .003; RR = 2.09, 95% CI 1.18 to 3.69, P = .01). In addition, we explored the effect of the course of treatment on the ulcer healing rate. The result revealed that compared with the placebo group, the PTX group showed better efficacy after eight weeks of treatment (RR = 1.40, 95% CI 1.03 to 1.91, P = .03, I2 = 5%), and the superiority persisted for 12, 20, and 24 weeks. However, this superiority was not observed at 4 and 16 weeks (RR = 1.14, 95% CI 0.65 to 1.98, P = .65, I2 = 0%; RR = 1.19, 95% CI 0.99 to 1.44, P = .07, I2 = 0%) (Figure 3).
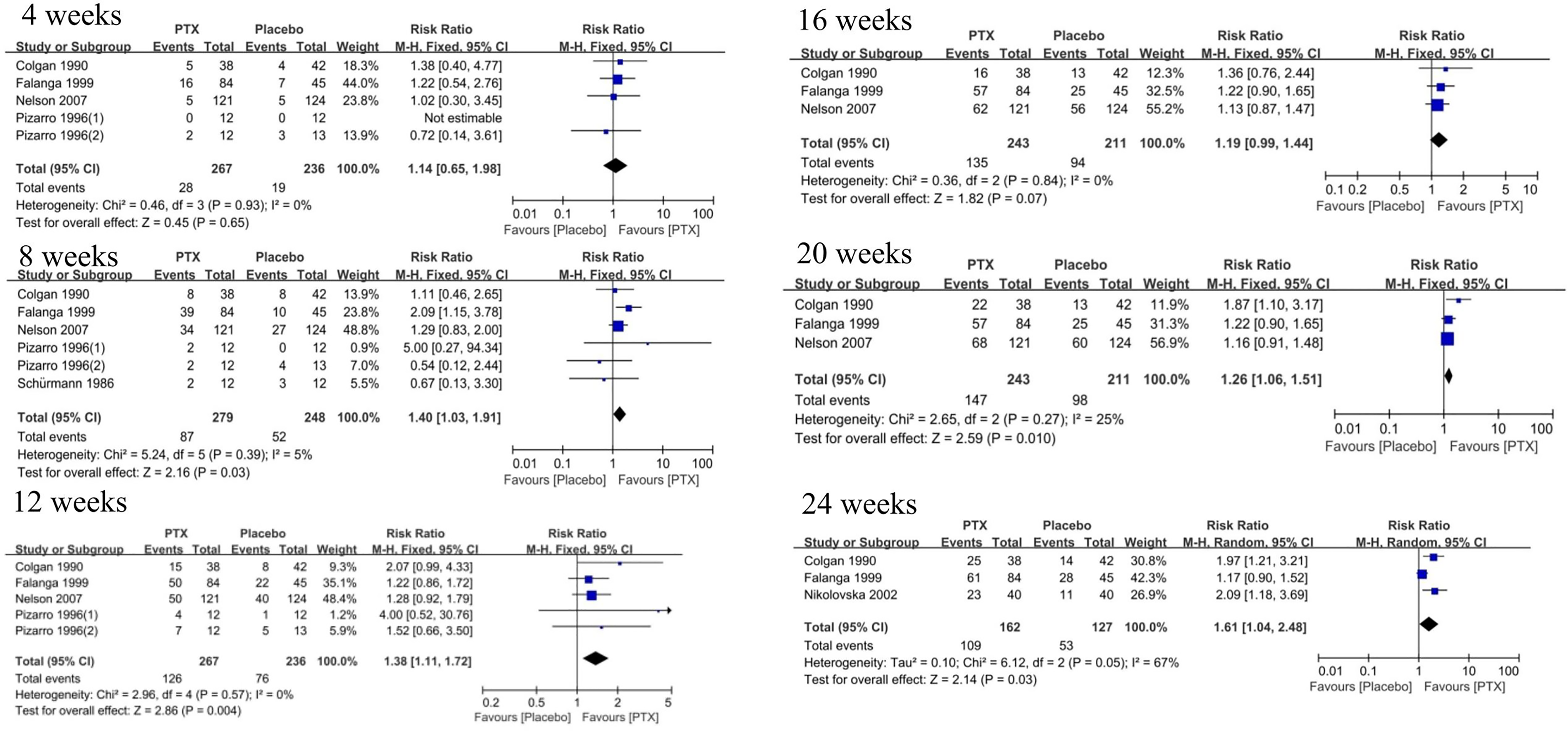
Subgroup analysis of the ulcer healing rate according to different treatment periods.
Sensitivity Analysis for the Ulcer Healing Rate
The result was robust after the sensitivity analysis was performed using a “leave-one-out” approach (Figure S3). However, heterogeneity significantly reduced (from 56% to 37%) after the poor-quality study (Belcaro 2002) 8 was removed, suggesting that high risk of bias was the primary source of heterogeneity.
Mean Duration of Complete Wound Healing and Changes in Mean Ulcer Size
One article published in 2012 explored the changes in mean ulcer size at 1 and 3 months between PTX and control group, and the result showed a significant difference after 3 months (E vs. C: 1.41 ± 1.77 cm2 vs. 3.78 ± 3.78 cm2, P = .02) 15 . The trial also revealed that PTX could shorten ulcer healing time (E vs. C: 4 ± 1.298 months vs. 6.25 ± 3.143 months, P = .007).
Adverse Events
Gastrointestinal disturbances, such as nausea, vomiting, abdominal pain, etc, were the most common adverse events. Compared with placebo, PTX treatment increased the occurrence of gastrointestinal disturbances (RR = 2.29, 95% CI 1.04 to 5.03, P = .04, I2 = 0%) (Figure 4, include Dale 1999). In addition, other adverse events included sleep disturbances(E vs. C: 1/101 vs. 0/99), hot flushes(E vs. C: 1/101 vs. 0/99), and bad effects on genitourinary(E vs. C: 1/38 vs. 0/42). 11 Serious adverse events included thrombocytopenia(E vs. C: 1/121 vs. 0/124), gastrointestinal bleeding(E vs. C: 1/121 vs. 0/124) and damage to the central nervous(E vs. C: 7/38 vs. 5/42), respiratory(E vs. C: 3/38 vs. 0/42) and cardiovascular system(E vs. C: 2/38 vs. 1/42).9,13,20
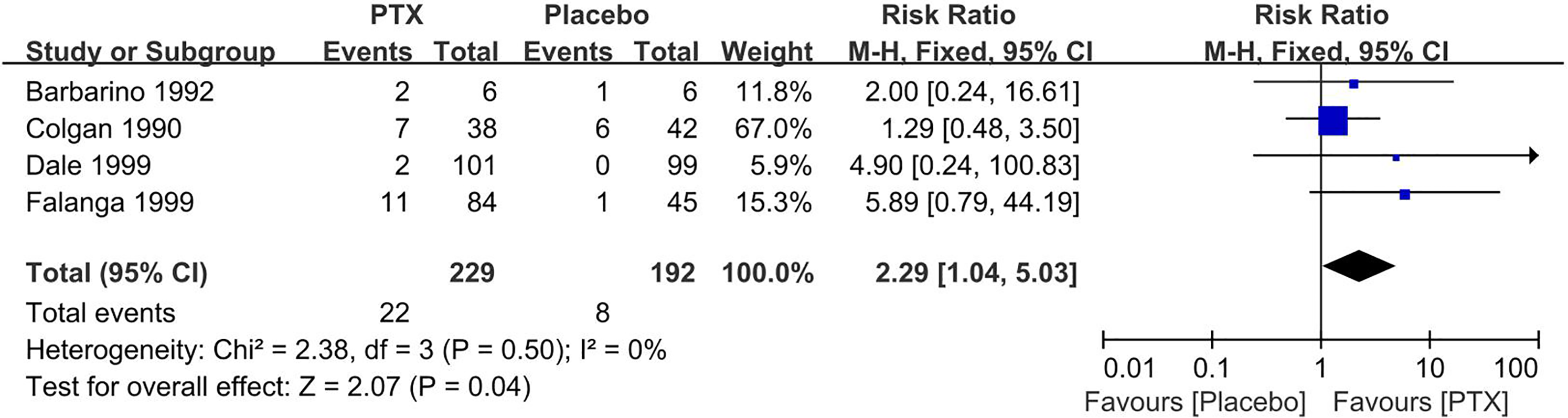
A meta-analysis of the effect of pentoxifylline (PTX) treatment compared with placebo (control group) on the incidence of gastrointestinal disturbances.
Certainty of Evidence
GRADE assessment of the evidence for the ulcer healing rate was predominantly judged moderate certainty due to high heterogeneity. The certainty of the evidence for the incidence of gastrointestinal disturbance was high (Table S1).
Discussion
The present meta-analysis indicates that PTX treatment improving ulcer healing (RR = 1.59, 95%CI 1.22 to 2.07) and significant improvement rate (RR = 2.36, 95%CI 1.31 to 4.24) compared versus placebo may be an effective adjunct therapy. Moreover, PTX treatment may be superior to placebo after eight weeks of treatment, and the efficacy of PTX was significantly better than that of placebo control over the course of treatment. Our meta-analysis included two new RCTs13,15, and the updated results from 13 RCTs were consistent with the previous meta-analysis published on 2012. 26
Venous hypertension increases capillary permeability leading to the fibrinogen leakage and formation of the pericapillary fibrin cuff, whose development creates a diffusion barrier to oxygen leads to non-healing ulceration and plugging of capillaries by white blood cells. The pericapillary fibrin cuff is believed to be the primary mechanism in the formation of VLUs. An essay showed that PTX administration induced about a 50% decrease of local elastase in VLUs, indicating a local reduction of neutrophil activation and a decrease in fibrin deposition. 27 Chronic inflammation, hemodynamic disorders, and the deficiency of growth factors are important factors leading to chronic ulcers. 28 PTX whose anti-inflammatory effects may be related to the initiation of immunity, the inhibition of leukotriene synthesis and TNF-α, as well as a non-selective phosphodiesterase inhibition resulting in higher cAMP concentrations. Subsequently, cAMP can decrease the activation of macrophages and concentrations of superoxide anions and inhibit the release of lysosomal enzymes from polymorphonuclear cells. The anti-inflammatory effect of PTX is also related to increasing the production of prostaglandin E2 and transforming growth factor-beta, and inhibit natural killing cells. 29 In addition, PTX decreases the potential for platelet aggregation and blood clot formation and reduces blood viscosity. 30
Venous ulcer healing begins with conservative management, the pillars of leg elevation, compression therapy, and various dressings. 31 Interestingly, we found that compression did not influence the efficacy of PTX, which was similar to the meta-analysis published in 2012. 26 Compression promotes venous and lymphatic return, improves microcirculation, and reduces inflammation. PTX has a similar effect, which may lead to this result and consequently still be suitable for patients with venous and arterial ulcers or for whom compression is contraindicated or poorly tolerated. 1 We suggest that the course of treatment may have driven the efficacy of PTX. There was a significant difference in healing rates in favor of PTX when treatment was range from 8 weeks to 24 weeks except for 16 weeks. However, the ulcer healing rate was similar between PTX and placebo within 4 weeks. One article published in 2012 showed that there was a significant difference in changes in mean ulcer size after 3 months (P = .02), which was consistent with our conclusion. 15
In all, PTX led to more adverse events in the present meta-analysis, especially gastrointestinal disturbances. The safety of PTX has not been fully unraveled, and there are increasing concerns about severe adverse effects such as damage to sperm, ear and labyrinth disorders, and thrombocytopenia.32–34 However, the included RCTs did not report the increasing risk of some severe adverse effects above in the PTX group during the follow-up period in the present meta-analysis. Considering that some adverse events like damage to sperm and ear may need a more extended follow-up period to be observed, it is still necessary to collect and evaluate the safety of PTX in further studies, particularly RCTs.
Our study has several strengths. Firstly, we included two new RCTs in the present meta-analysis. Secondly, the most common gastrointestinal disturbances were analyzed separately in this meta-analysis, and all adverse events with their incidence were mentioned. However, all adverse events were analyzed in a meta-analysis, and other adverse events were unclear except for gastrointestinal disturbances in the meta-analyses published in 2012. Thirdly, we explored the effect of the course of treatment on the ulcer healing rate. Finally, compared with the meta-analyses published in 2012, our meta-analysis excluded a trial comparing PTX and defibrotide, because it was a positive control trial and might affect the assessment of the efficacy of PTX. 19 However, some limitations should be considered when interpreting our results. Firstly, among these RCTs, sample size, duration, and severity of chronic ulcer and basic treatment were significantly different, leading to high heterogeneity. However, the paucity of cases and data hampered subgroup analysis based on these differences. Secondly, the certainty of the evidence for the ulcer healing rate was moderate, and hence the results might be underpowered. Finally, due to the limited number and information of the included studies, the evidence for some critical safety outcomes was weak. Considering rare or long-term adverse effects are uneasy about being observed, the safety of PTX needs to be further collected and evaluated.
In conclusion, this meta-analysis suggests that PTX for VLUs achieved better ulcer healing rates than placebo, and the adverse events were mainly limited to gastrointestinal discomforts. PTX may offer a potential adjunct option for VLUs. However, the current evidence for the abovementioned conclusions is weak. More carefully designed and adequately powered RCTs are warranted to examine the efficacy and safety of PTX on both short-term and long-term essential outcomes.
Supplemental Material
sj-docx-1-ijlew-10.1177_15347346211050769 - Supplemental material for Efficacy and Safety of Pentoxifylline for Venous Leg Ulcers: An Updated Meta-Analysis
Supplemental material, sj-docx-1-ijlew-10.1177_15347346211050769 for Efficacy and Safety of Pentoxifylline for Venous Leg Ulcers: An Updated Meta-Analysis by Shi-Yi Sun, Yan Li, Yun-Yi Gao and Xing-Wu Ran in The International Journal of Lower Extremity Wounds
Footnotes
Acknowledgement
Thank the CSPC Ouyi Pharmaceutical Co., Ltd. for helping us find the full text of articles.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was partial supported by Science and Techology Bureau of Sichuan Province [Grant No. 2021JDKP004], 1.3.5 Project for disciplines of excellence, West China Hospital, Sichuan University [Grant No. ZYGD18025], West China Nursing Discipline Development Special Fund Project, Sichuan University [Grant No. HXHL20005], Health Medical Big Data Application and Innovation Project in Sichuan [Grant No. 2018gfgw001], Science and Technology Bureau of Chengdu city [Grant No. 2017-CY02 to 00028-GX].
Ethical Approval
Not applicable, because this article does not contain any studies with human or animal subjects.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
