Abstract
Chronic foot ulcers are the leading cause of prolonged hospitalization and loss of social participation in people with diabetes. Conventional management of diabetic foot ulcers (DFU) is associated with slow healing, high cost, and recurrent visits to the hospital. Currently, the application of autologous lipotransfer is more popular, as the regenerative and reparative effects of fat are well established. Herein we report the efficacy of minimally manipulated extracellular matrix (MA-ECM) prepared from autologous homologous adipose tissue by using 3D bioprinting in DFU (test group) in comparison to the standard wound care (control group). A total of 40 subjects were screened and randomly divided into test and control groups. In the test group, the customized MA-ECM was printed as a scaffold from the patient autologous fat using a 3D bioprinter device and applied to the wound directly. The control group received standard wound care and weekly follow-up was done for all the patients. We evaluated the efficacy of this novel technology by assessing the reduction in wound size and attainment of epithelialization. The patients in the test group (n = 17) showed complete wound closure with re-epithelialization approximately within a period of 4 weeks. On the other hand, most of the patients in the control group (n = 16) who received standard wound dressings care showed a delay in wound healing in comparison to the test group. This technique can be employed as a personalized therapeutic method to accelerate diabetic wound healing and may provide a promising potential alternative approach to protect against lower foot amputation a most common complication in diabetes.
Keywords
Introduction
The prevalence of diabetes mellitus is growing rapidly to epidemic proportions worldwide. According to International Diabetes Federation (IDF) in 2019, approximately 463 million adults (20-79 years) were living with diabetes and it is projected to increase to 700 million by 2045 owing to urbanization, decreased physical activity, and a change in the diet. 1 A diabetic foot ulcer is the most common pathological complication of diabetes and up to 34% of all people with diabetes are estimated to have a foot ulcer during their life period. In India, the numbers of diabetic foot patients are increasing in both urban and rural settings, with 85% of amputations preceded by foot ulcers. 2 Diabetes decreases the wound healing capability through prolonged inflammation, reduced production of growth factors, and impaired angiogenesis. The characteristic feature of non-healing wounds in diabetes is due to a decrease in the ability to regrow microvasculature through the process of angiogenesis. 3 This interruption of the normal healing process contributes to a defective healing response and becomes a major therapeutic target for generating novel treatments. Regardless of current clinical care protocols for diabetic foot ulcer management, a high amputation rate is observed. This presents a major problem to patients’ health and happiness in addition to the cost of health care systems. Currently, there is an urgent need for a novel treatment strategy to enhance diabetic wound healing, increase the ulcer healing rate, and thereby preventing amputation. To provide a better treatment strategy and to accelerate the wound healing process, the authors explored the application of a minimally manipulated extracellular matrix (MA-ECM) prepared from autologous homologous adipose tissue using a 3D bio-printing system for the treatment of diabetic foot ulcers in this pilot study. Adipose-derived stem cells (ADSC) are adult multipotent, mesenchymal stem cells situated in adipose tissue that has been widely used in the field of regenerative medicine for cell-based therapies. 4 ADSC are more abundant compared to other forms of mesenchymal stem cells which have the potential for self-renewal, differentiate into cell types involved in skin repair. 5 MA-ECM is derived from the autologous adipose tissue which retains hundreds of proteins, growth factors, and cytokines that play a vital role in chronic wound healing and provide a close environment to the normal, non-diabetic wound without immunological reactions. With the help of 3D bioprinting technology, MA-ECM can be customized according to the patient's need which is a significant advantage of this technique. The efficacy of MA-ECM prepared from the lipid-aspirate of the participating subjects as a form of micronized adipose tissue was evaluated in this study. The primary objective was to explore the efficacy of MA-ECM in the process of healing the wound and the secondary outcome is to observe the decrease in the size of the wound. Here the authors present the data of the study conducted in our wound care center to explicate the efficacy of the technique.
Materials and Methods
Study Design
This randomized controlled single-center clinical trial was approved. The approved protocol was implemented as per the regulatory guidelines. The subjects were screened based on the inclusion and exclusion criteria as summarized in Table 1. The protocol was explained in vernacular language to all the subjects in both groups. The patients signed the informed consent and were randomly assigned to control (standard wound care) or test group (application MA-ECM) based on simple randomization sampling. In our study, we selected the subjects with foot ulcers located below the malleoli on the plantar or dorsal surface of the foot, and ulcer size was between 1 cm2 to 5 cm2. All the subjects included in the study had neuropathy with loss of protective sensation but PVD was not observed. The subjects in the test group were treated with MA-ECM one time by using the 3D bioprinting system and subjects in the control group received the standard wound care with Mepitel-one®, (Molnlycke Health Care, Sweden) the dressing. All subjects were observed weekly to evaluate the wound size and epithelialization as in Figure 1.
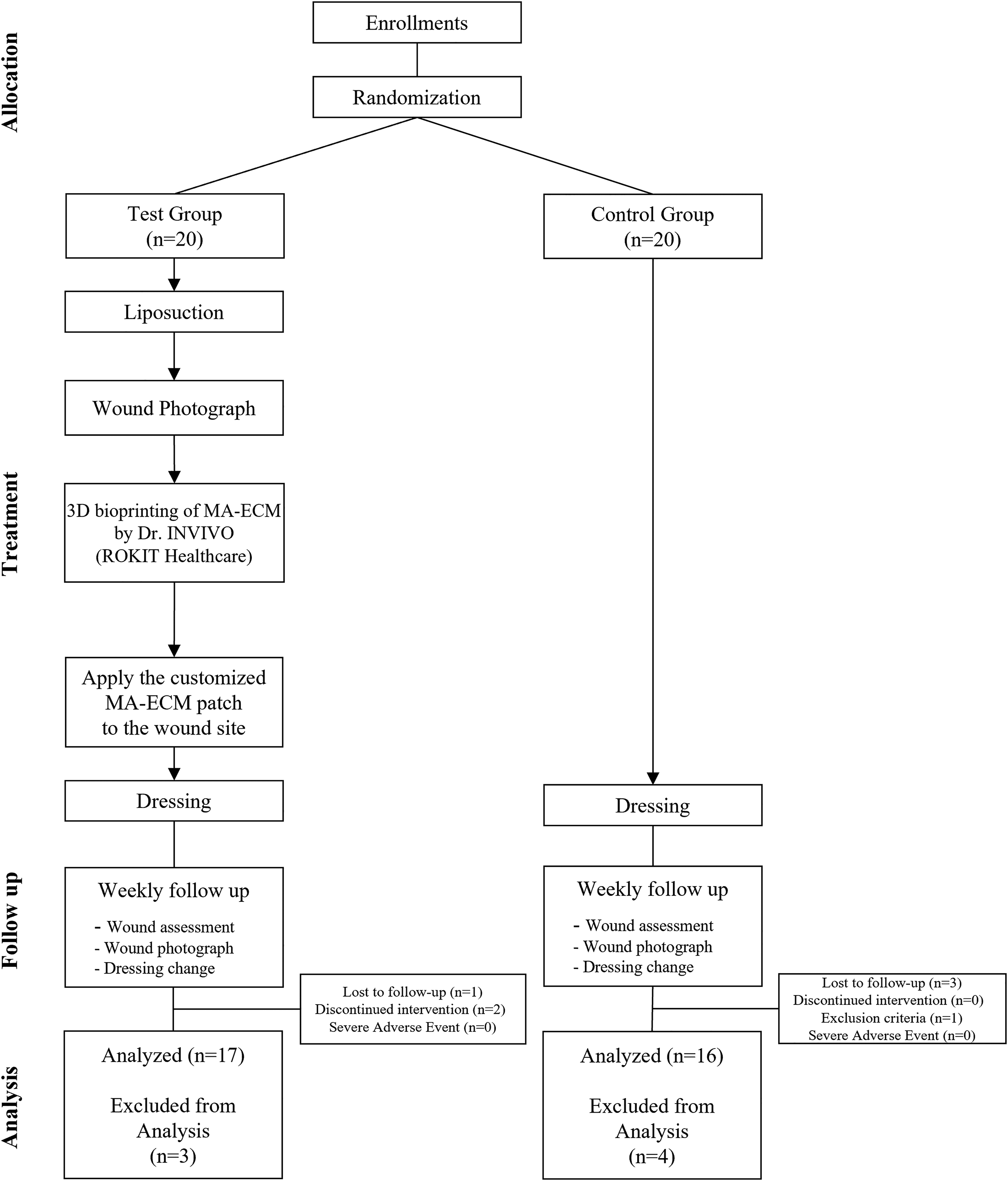
Schematic diagram of pilot clinical study process. Flow chart consists of following phases: Allocation, Treatment, Follow up, and Analysis. After assessing for eligibility of 40 subjects, patients were randomized and divided into two groups: test (n = 20) and control (n = 20) group. 17 and 16 patients in test and control group were analyzed respectively. 3 subjects in the test group, and 4 subjects in the control group were excluded from the analysis due to lost to follow up.
Major Inclusion and Exclusion Criteria

3D Bioprinting Preparation
To harvest autologous adipose tissue, the standard liposuction technique with a cannula was performed in the abdomen of each patient in the test group by the plastic surgeon in the operating room.6,7 Collected autologous fat tissue was mechanically disaggregated into specific sizes using a fat separator to produce the MA-ECM which is then transferred in the dispenser chamber of 3D Bioprinter Dr INVIVO (ROKIT Healthcare, Seoul, Korea). After cleaning the wound site, photograph image files of each patient were prepared. In the test group, the image files were converted to G-code files by using the software, New Creator K (ROKIT Healthcare, Seoul, Korea) and fabricated to customize the shape of the wound patch with MA-ECM. The wound patch was directly applied to the wound site in the test patient. The Mepitel-one®, (Molnlycke Health Care, Sweden) dressing was used as primary and a foam dressing as the secondary to prevent pressure and infection. Subjects randomized in the control group were provided with standard wound care, weekly follow-up was done as the test group. All subjects were off-loaded using a foam dressing and they were asked to wear the footwear with cushioned insoles.
Analysis
Digital photographs of the wound area of all subjects were taken to observe wound healing progress during each visit. Wound size was measured with image files by means of ImageJ (National Institutes of Health, Rockville, MA). The result was tabulated and the reduction in wound size of each patient during every visit for a period of 24 weeks was calculated. The time taken to accomplish the wound healing was analyzed with the Kaplan-Meier analysis method using Prism software.
Results
A total of 40 subjects were screened and were randomly divided into test and control groups to compare the wound healing efficacy of MA-ECM. In the test group, out of 20 subjects allotted, 17 subjects (7 females, 10 males) were analyzed as 3 subjects were lost to follow-up. Correspondingly 20 subjects were allocated in the control group, but 3 subjects were lost to follow-up (COVID-19 pandemic) and one was excluded by the criteria eligibility as shown in Figure 1. Demographic data and blood investigations were performed for screening and summarized in Table 2. In both groups, the age range of all subjects diagnosed with diabetes was 37 ∼ 79 years and the mean age was 60.4 years in the test group and 54.9 years in the control group. There were no adverse events reported during the study. To evaluate the wound healing efficacy of MA-ECM in the test and control group, foot ulcers at the toe, plantar, medial plantar, dorsal plantar, and heel for both groups were selected and visualized by the photograph at the baseline and 4 weeks after the surgery. The wound size reduction and epithelization in each group were analyzed. Subjects in the test group displayed complete wound healing and re-epithelialization at the wound site approximately about 4 weeks of the treatment (Figure 2) in comparison to the control group (Figure 3). Figure 4 depicts the rate of complete wound healing of the total 33 subjects under study within a period of 12 weeks. The graph denotes the number of subjects who completed the wound healing process within a period of 12 weeks in terms of percentage. With the help of our innovative treatment in the test group irrespective of wound size and location of the ulcer, we observed a complete wound healing when compared to the control group. In the control group, only 50% of subjects showed wound healing by 12 weeks period.
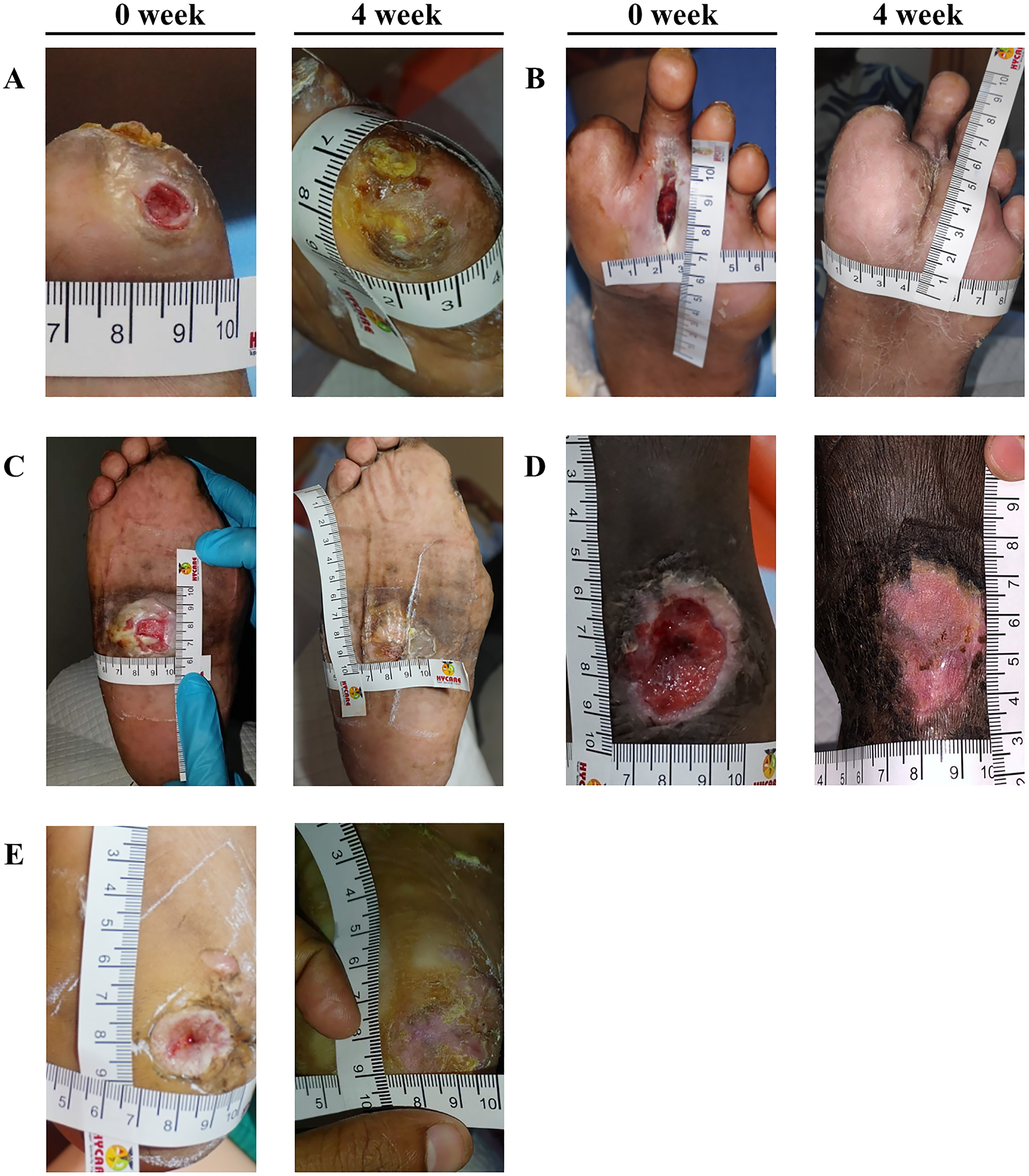
Wouund healing in the test group. Epithelialization and wound healing were completed at 4 weeks after the new treatment in this test group (0 week; at baseline, 4 week; 4 weeks from the treatment). The location of DFU: first toe (A); Plantar fore foot (B); Plantar mid foot (C), Dorsal mid foot (D); Heel (E).
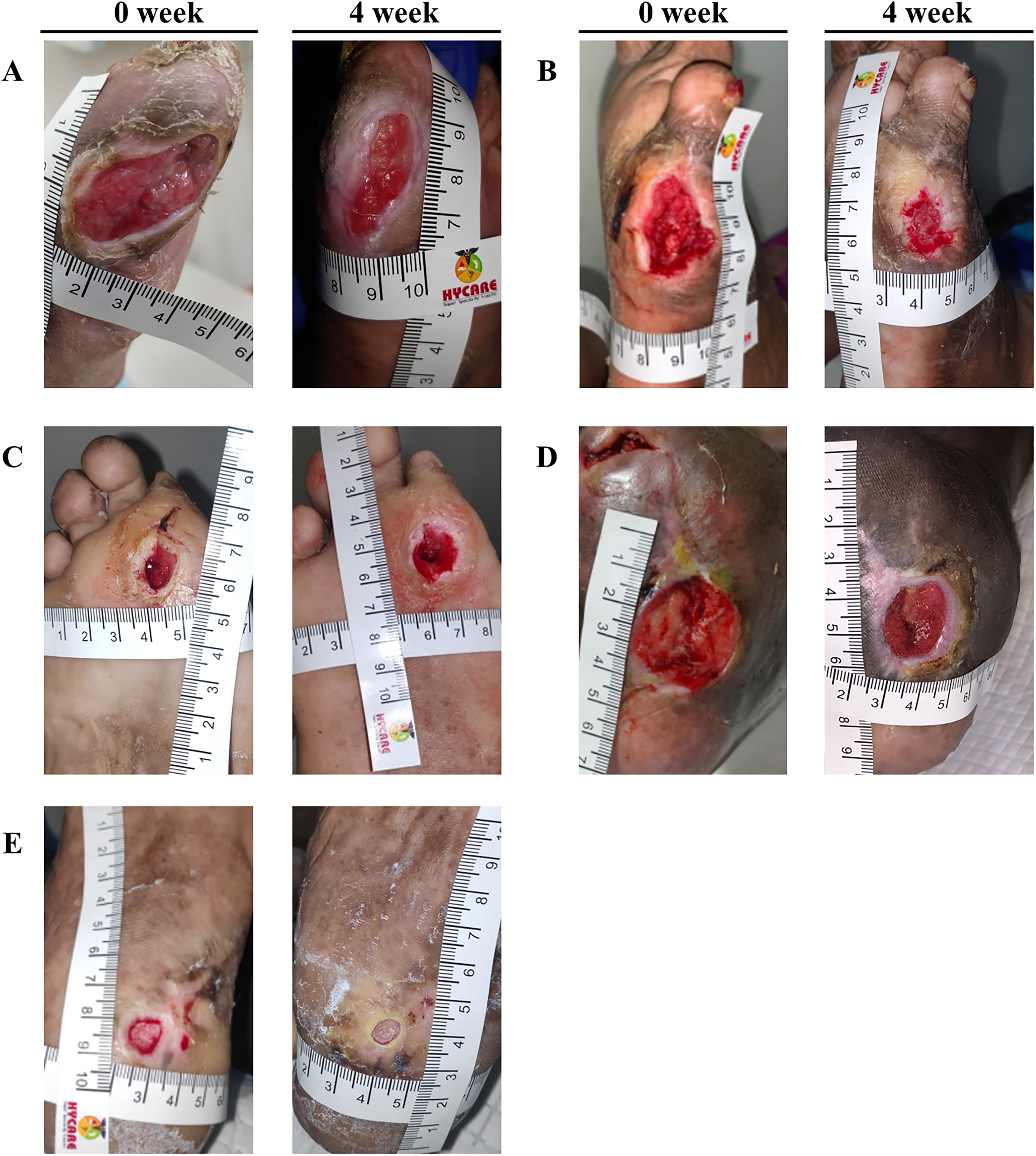
Wound healing in the control group. Non-healing wound was observed at 4 weeks in the control group. Time to wound closure in the subject A and B were initiated at 11 weeks, but the subject C, D and E were not closed within 12 weeks. The location of DFU: first toe (medial) (A); Lateral fore foot (B); Plantar fore foot (C); Amputation site (D); Plantar hind foot (E).
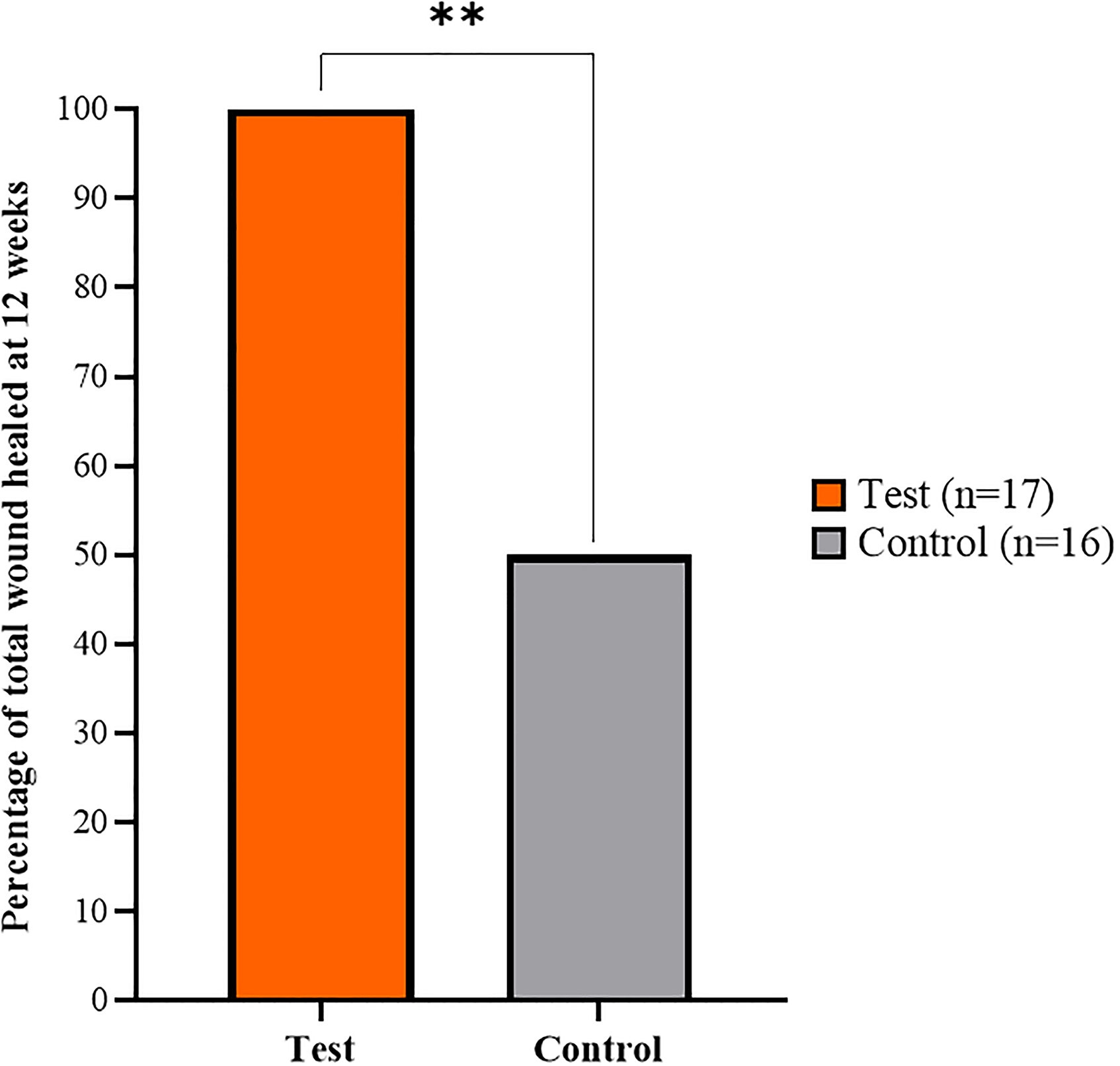
Percentage of subjects presented complete epithelialization at 12 weeks. The bar represents the number of subjects who completed the wound healing process within a period of 12 weeks in percentage. With the help of our innovative treatment in test group, irrespective of wound size and location, we observed all the subjects in the test group were completely healed. In control group, only 50% of the subjects showed healed wound at 12 weeks period. There was statistically significant difference in the test subjects when compared to the contol. (P < 0.0001, **).
Patients Variables Between the Test and Control Groups
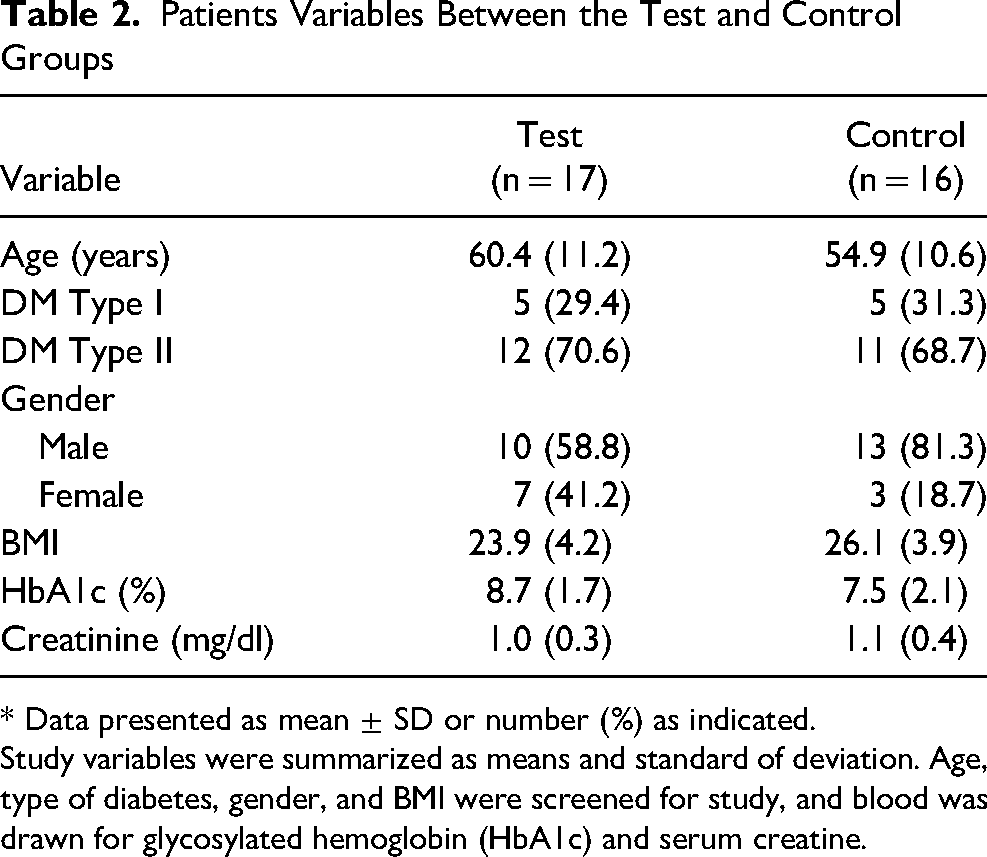
* Data presented as mean ± SD or number (%) as indicated.
Study variables were summarized as means and standard of deviation. Age, type of diabetes, gender, and BMI were screened for study, and blood was drawn for glycosylated hemoglobin (HbA1c) and serum creatine.
Discussion
Wound healing is a complex dynamic and tightly regulated process, linking complex ECM-dependent molecular and cellular interactions. 8 In the case of diabetes, high glucose content and other metabolic changes can lead to modifications in ECM, like decreased collagen deposition, increased production of MMPs, and the prolonged presence of abundant matricellular proteins. These alterations can have a negative impact on cellular functions, deteriorating the pathological state of ECM, resulting in chronic wound healing. 9 Autologous fat grafting has been described as a procedure of tissue augmentation for over a century. Fat grafts contain adipose-derived stem cells (ADSC) adult multipotent, mesenchymal stem cells located in adipose tissue that has recently become popular in the field of regenerative medicine. 10 Our unique procedure generates a MA –ECM from nano-fat autologous in origin, using a 3D bioprinter which comprises various proteins, growth factors, and cytokines. Cytokines and growth factors are well known to play a critical role during the various phases of wound healing. The present clinical study was implemented to compare the autologous micro-fragmented fat (test group) as a scaffold at the wound site with standard care (control group), predominantly targeting the efficacy of improving the wound healing rate in diabetic foot ulcers. The authors observed a significant rate in wound size reduction over 50% at 4 weeks, which is a clinically appreciated indicator in the prognosis of healing, and attainment of 90% area reduction at a period of 8 weeks is significant to predict the wound closure. Collagen constitutes 70% of skin ECM and its expression, deposition, and remodeling are critical in repair. 11 Neosynthesis of collagen stimulated by autologous fat grafting fibers and improved vascularization and thickening of the dermis and subcutaneous tissue is demonstrated by Mojallal et al 12 These events are vital for the acceleration of wound healing. The adipose tissue obtained through lipoaspiration is a rich, ubiquitous, and easily accessible source for mesenchymal stromal cells (MSCs). ADSCs are multipotent precursor cells that can differentiate into cell lineages associated with the regeneration of tissues such as fibroblasts, keratinocytes, and endothelial cells. 13 Specific growth factors accelerating wound healing, anti-inflammatory cytokines, proangiogenic factors, and healing-related peptides present in the autologous fat may also have a positive effect on the wound healing process. 14 Many clinical studies have revealed that local injection of autologous micro-fragmented adipose tissue improved healing rate in DFU patients and fat grafting can also be used to treat hypertensive leg ulcers and thermal injury. 15 It can be concluded that the test patients treated with MA-ECM have established almost 100% healing of ulcer in our study, which indicates the potent role of various factors present in our primary wound dressing material in the enhancement of communication for the acceleration of the wound healing process.
Conclusion
Our experience with autologous MA-ECM has proven to be an effective therapeutic option that can significantly accelerate the healing process in diabetic foot ulcers. The extracellular matrix (ECM) derived from Nano-fat consisting of supportive proteins, growth factors, and cytokines have been reported as an important player to provide efficient 3D environments during cell proliferation and differentiation. It could be concluded that treatment with MA-ECM prepared from the autologous fat loaded with adipocytes and growth factors could have played a vital role in the healing of wounds. Also since the MA-ECM is autologous in nature possibility of immune rejection can be ruled out. Finally, it could be concluded that 3D bioprinting technique is a customized, therapeutic, and a potential alternative approach for the management of diabetic foot ulcers.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship and/or publication of this article.
Ethical Approval
The clinical study was approved by the Ripon Independent Ethics committee registered under CDSCO (ECR/299/Indt/TN/2018) and the study was conducted as per the regulatory guidelines in the site Hycare Super Speciality Hospital, a tertiary wound care center in South India.
