Abstract
Only ultraviolet-C (UV-C) from UV lights, which are emitted by the sun and absorbed by the atmosphere's ozone layer, does not reach the Earth's surface. UV-C is a powerful disinfection method that is commonly used to sterilize fluids, air, and surfaces. There is a little knowledge of the effects of UV-C radiation on living bodies. The purpose of this study is to examine the ameliorative effect of UV-C on skin lesions in mice that have been experimentally created and infected with Staphylococcus aureus, Staphylococcus epidermidis, and Streptococcus sp. In total, 32 mice were used, and 4 mm skin defects were created and lesions infected with bacteria. Half of the mice in each group were treated with 254 nm UV-C twice a day for 4 days before being euthanatized. Blood samples were collected for hematological analysis, while skin samples were collected for microbiological, pathological, and immunohistochemical examinations. In addition, pathological examinations were performed on visceral organ samples. UV-C treatment caused rapid healing and complete or significant disinfection of skin lesions. Moreover, UV-C treatment reduced caspase-3 expressions in lesioned areas, according to immunochemistry. There were no pathological findings in visceral organs as a result of UV-C treatment. This study found that UV-C can be used to treat and disinfect infected skin lesions in short period and repeated doses.
Introduction
Ultraviolet (UV) light is a type of electromagnetic radiation that is classified into 3 wavelength groups: ultraviolet A (315-400 nm), B (280-315 nm), and C (200-280 nm). 1 Sunlight, also known as solar radiation, is the primary source of UV radiation (UVR). Because ultraviolet-C (UV-C) from the sun is almost completely absorbed by the Earth's atmosphere, it has few negative health effects. Ultraviolet-B (UV-B) has been accused of causing skin lesions, increasing the risk of skin cancer, and inducing immunosuppression. Ultraviolet-A (UV-A), on the other hand, contributes to skin aging and has recently been linked to the development of skin cancers in animals and immunosuppression in humans. 2
Bacterial agents can cause a wide range of diseases and problems in living organisms. To destroy bacteria in the surrounding environment, various disinfection techniques have been developed and are widely used. 3 UV-C irradiation is one of the most frequently used and effective techniques for this. 4 It exhibits this effect by interacting with nucleic acid components. 5 UV-C in the 240 to 280 nm range, in particular, is highly germicidal. 6 According to Ritter et al, 7 irradiating surgical wounds with germicidal UV-C during orthopedic surgical procedures reduces the rate of infection after the procedures. However, it has been reported that long-term exposure of human cells to 254-nm UV-C may result in the formation of mutagenic and cytotoxic DNA lesions that can cause skin cancer. 8 Furthermore, UV-C lights with short wavelengths, such as 200 to 230 nm, are clearly absorbed by proteins. 9 Therefore, even in fine tissues of 0.3 μm, short-wavelength UV-C light is reduced by half. 10 In contrast to conventional 254-nm UV-C, 222-nm UV-C has recently been shown to harmless to mammalian cells. 11
Until now, data on the rate of lowering the risk of UV-C wound healing and infection in living bodies has been contentious. The use of UV-C irradiation has been studied at an early stage, primarily with in vitro and ex vivo models.12,13 The purpose of this study was to assess the bactericidal and wound healing effects of UV-C on the twice-daily application for 4 days in infected skin wounds created in a mouse model.
Materials and Methods
Animals
All experiments were performed in accordance with the ARRIVE (Animal Research: Reporting in Vivo Experiments) guidelines in 2.0, and study was approved by the Local Ethical Committee on Animal Research of Burdur Mehmet Akif Ersoy University (712-13.01.2021). All animals were given unlimited food and water. They were kept at room temperature and subjected to a 12-h light/dark cycle.
Experimental Procedure
Methicillin-resistant Staphylococcus aureus (ATCC 43300); Staphylococcus epidermidis strain YT-169a, which had previously been used as a biofilm-forming bacteria in a study 14 ; and beta-hemolytic Streptococcus sp. strain isolated from bovine mastitis were used to produce wound infections in mice. These strains were cultured on blood agar (Condalab) that contained 5% defibrinated sheep blood. For 24 to 48 h, the plates were incubated in an aerobic atmosphere at 37°C. Bacterial suspensions were prepared in sterile normal saline from fresh colonies of strains, and the turbidity was adjusted to 0.5 McFarland standard (∼108 colony-forming unit [CFU]/mL) using a densitometer (Biosan, EU). The bacterial suspensions were further diluted 1: 10 in normal saline to obtain final inocula of 107 CFU/mL.
In total, 32 CD-1 female mice were used in the experiment. On the dorsum of all animals, skin defects with a diameter of 4 mm were created. The mice were divided into 4 groups of 8 animals each, and these 4 groups were further divided into 2 subgroups with and without UV-C treatment. The first group served as the control group, with a skin defect that was not infected with bacteria. However, skin defects of mice in the second group were infected with Streptococcus sp. In the third group, skin defects of mice were infected with S aureus. On the other hand, skin defects of mice in the fourth group were infected with S epidermidis. Each wound received a 100-μL bacterial suspension. The gauze was used to bandage the wound. After 24 h, wound areas were examined for the presence of any local abscess or inflammatory reaction. While 4 mice in each group were allowed to heal normally, another 4 mice in each group were exposed to UV-C for 4 min in the morning and evening. For this procedure, mice were exposed to a total of 8 sessions of UV-C for 4 days at a wavelength range of 240 to 257 nm from a distance of 1 m with the energy density of UV-C 120 mw/cm2 using a programmable UV radiation generator with sensor (Qzuradiation [nonionizer] generator, Qzu QSS Technology Burdur). All mice were sacrificed 2 days after the last UV application under intraperitoneal injection of 90 mg/kg ketamine (Alfamin, Alfasan IBV) and 10 mg/kg xylazine (Alfazin, Alfasan IBV) anesthesia. Pathological and microbiological examinations were performed on blood and lesioned skin samples. Moreover, internal organs were also sampled for pathological examinations.
Hematological Evaluations
Before euthanasia, cardiac blood samples were collected in EDTA tubes for hematological analysis. For the complete blood count, Abacus Junior Vet equipment was used. Further, an automatic biochemistry analyzer (Gesan Chem 200 autoanalyzer) was used to perform biochemical analyses.
Histopathological Method
During the necropsy, skin and visceral organ samples were fixed in a 10% neutral formalin solution. After a 2-day fixation period, samples were subjected to routine tissue processing for histopathological examination, embedding in paraffin, and sections of 5-µm thickness were taken, stained with hematoxylin–eosin (HE), and examined under a light microscope.
Immunohistochemical Method
In addition, a series of sections drawn on poly-L-lysine-coated slides were immunohistochemically stained for caspase-3 (anti-caspase-3 antibody [ab4051], Abcam) expression by streptavidin-biotin technique according to manufacturer instructions. The sections were incubated with the primary antibody for 60 min before incubation with a biotinylated secondary antibody and a streptavidin-alkaline phosphatase conjugate. As a secondary antibody, the EXPOSE Mouse and Rabbit Specific HRP/DAB IHC Detection Kit (ab80436) (Abcam) was used. Moreover, diaminobenzidine (DAB) was used as the chromogen. Instead of primary antibody, antigen dilution solution was used as a negative control. All examinations were carried out on blinded samples.
Quantitation of Bacteria on the Wound
Skin samples taken from the defects were used for bacterial reisolation and microbiological examinations. At the end of the treatment period, the number of bacteria on the wound surface was calculated using the previously described procedure. 15 In brief, for each group, half of the wound created areas were taken, weighed, added to normal saline at a concentration of 0.01 g/mL, and homogenized. For bacterial counting, the sample was diluted 6 fold in normal saline. Furthermore, 10 µL of homogenate of each dilution was spread on 2 Plate Count Agar (Biokar). Streptococcus sp. colonies were counted using Plate Count Agar supplemented with 5% sheep blood. Petri dishes were incubated at 37°C for 24 to 48 h under aerobic conditions. At the end of the incubation period, the colonies were counted as CFU, and then the average CFU/g value was calculated.
Statistical Analysis
For statistical analysis of microbiological isolations, the UV-C applied and non-UV-C applied groups were compared, and one-way ANOVA and paired sample t-test were used. Furthermore, SPSS-15.00 software package was used. The differences in the width of the wounds between the groups were evaluated by Duncan test. The level of significance was considered at P < .05.
Results
Hematological Findings
Statistical analysis was carried out by comparing the hematology results of UV-C treated and non-UV-C treated groups. Only the granulocyte count and percentage were found to differ significantly between groups. The granulocyte count was reduced after exposure to UV light. Table 1 displays the results.
Statistical Analysis Results of the Granulocyte Count and Percentage Between the Groups.
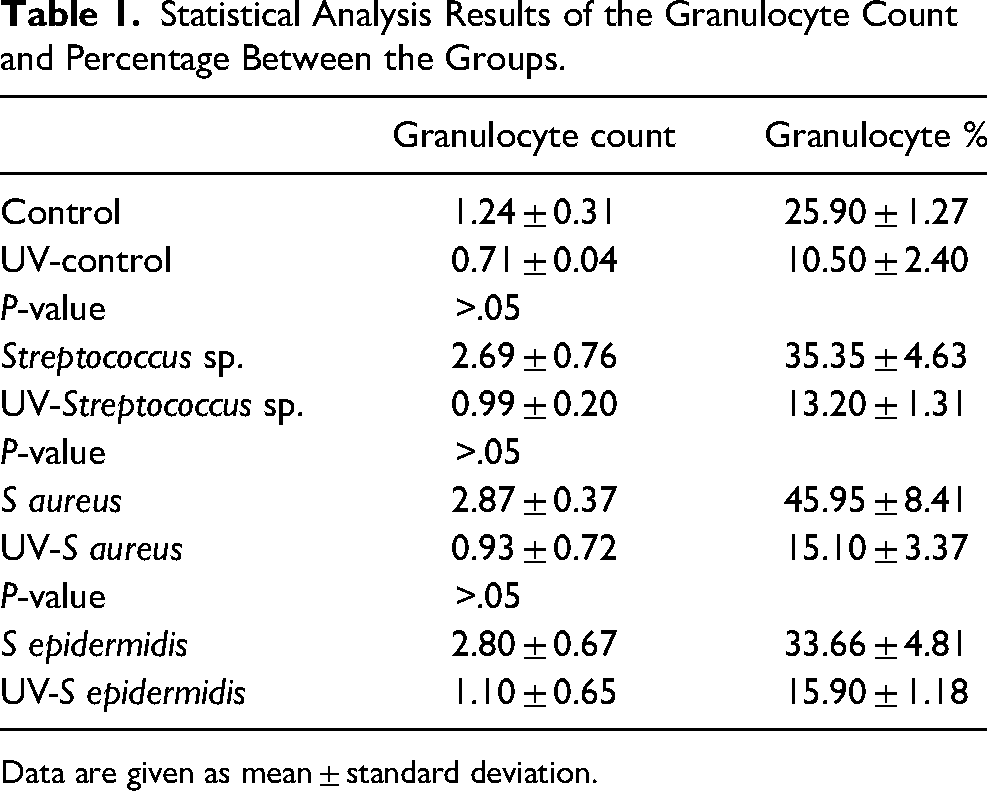
Data are given as mean ± standard deviation.
Gross Findings
During the study, no mortality was observed in any of the UV-C treated groups. One mouse died in each of the non-UV-C treated S aureus and non-UV-C treated Streptococcus sp. groups. When wound healing was compared between subgroups, it was discovered that healing was delayed and defects were larger in the non-UV-C treated groups than in the UV-C treated groups. Wounds infected with Streptococcus sp., S aureus, and S epidermidis and treated with UV-C healed faster and had smaller defect areas than those of non-UV-C treated subgroups. Figure 1 shows the macroscopic findings of the groups. During the necropsy width of the wound was measured and results were statistically analyzed. Figure 2 illustrates the mean width of the wounds at the end of the study.
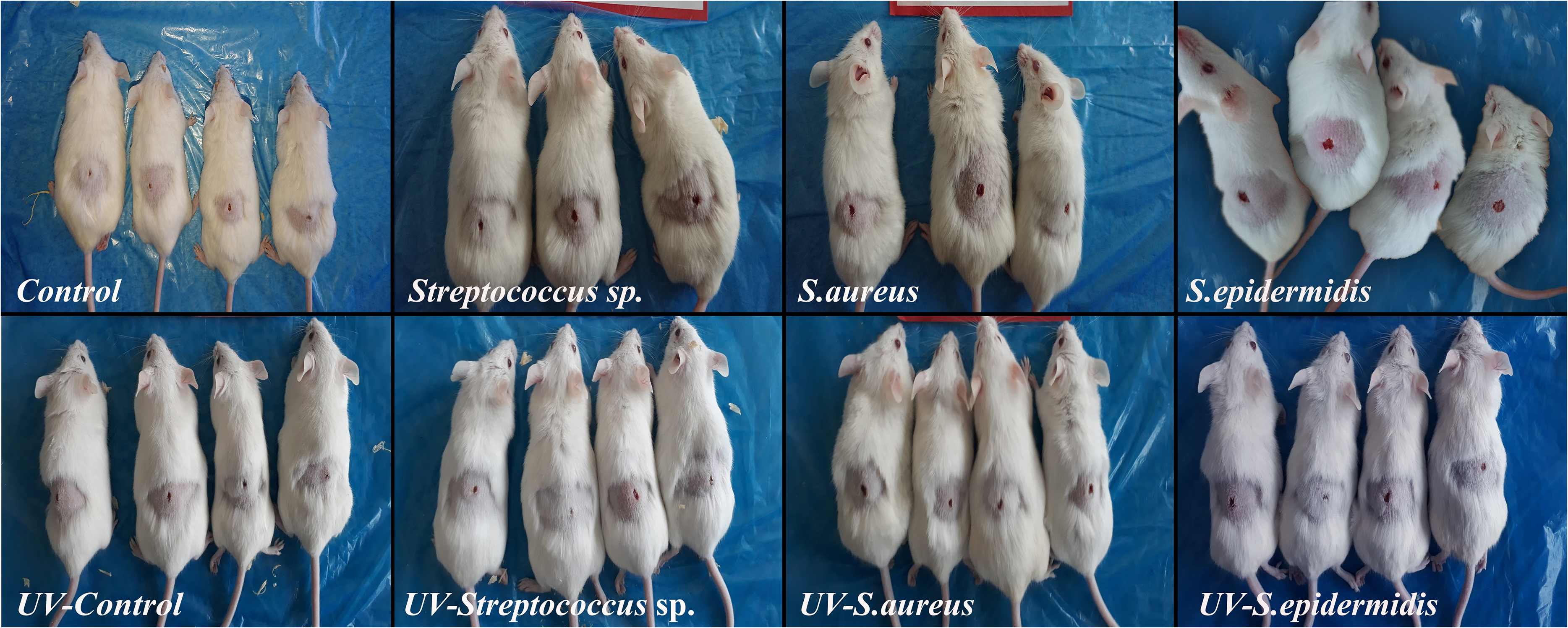
Gross findings of the defect created control and infected groups mice with and without ultraviolet-C (UV-C) treatment.
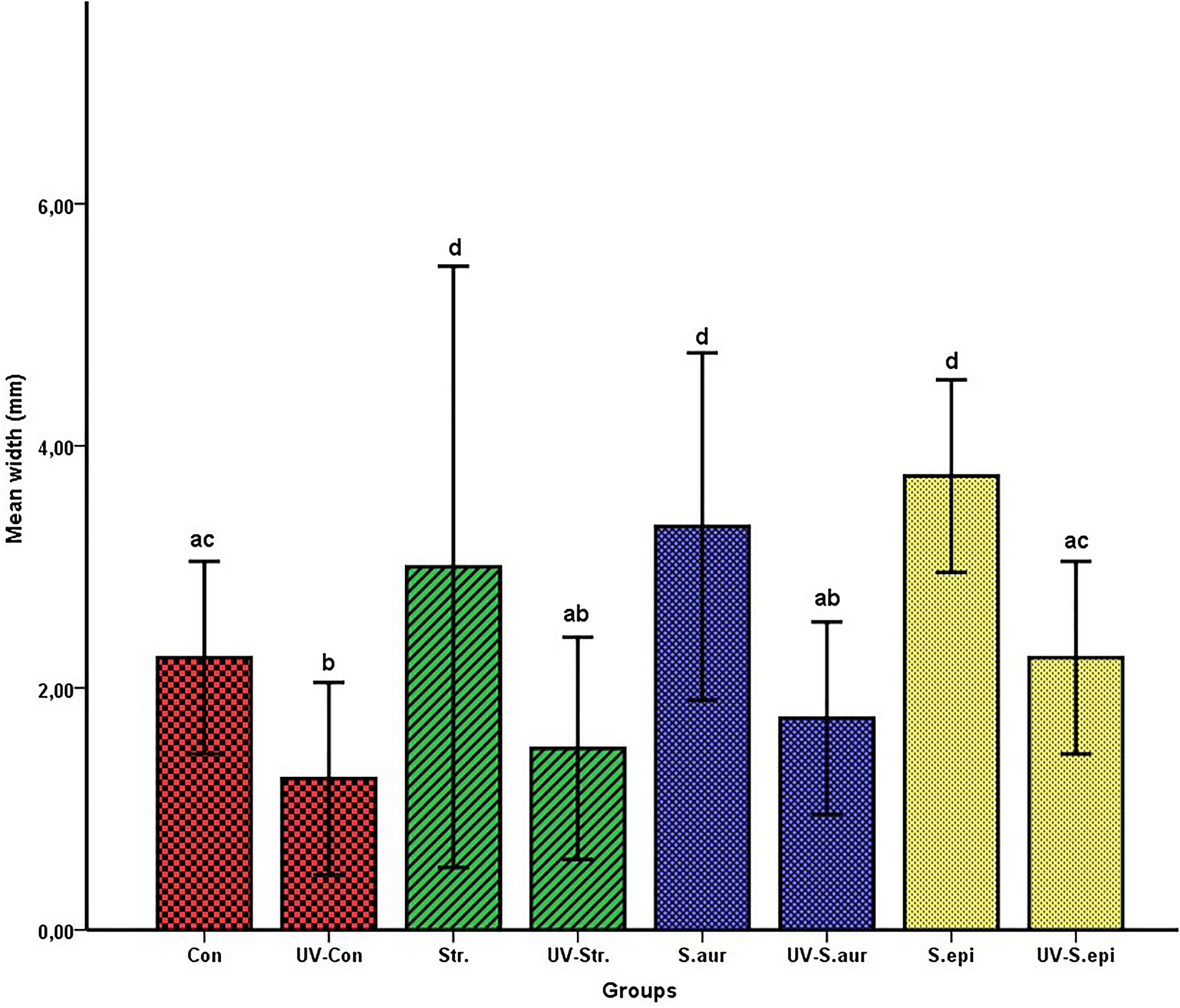
Statistical analysis results of the mean wound width between the group, ultraviolet-C (UV-C) treated and nontreated groups shown in the same color. Values represent mean ± standard deviation and the differences between the means of groups carrying different letters in the same column are statistically significant.
Microscopical Findings
During the histopathological evaluation, UV-C treatment significantly reduced the lesion size and necrotic crust in the defect area. In addition, UV-C treatment also reduced the inflammatory cell infiltrations at the defects. During the immunohistochemical examination, UV-C treatment reduced caspase-3 expressions in the defect area. The decrease was observed in both the number of cells and the intensity of the expression. Figures 3 and 4 show the histopathological and immunohistochemical findings in the defect area, respectively. There was no evidence of UV-C-induced tissue damage in any visceral organ samples taken from mice.
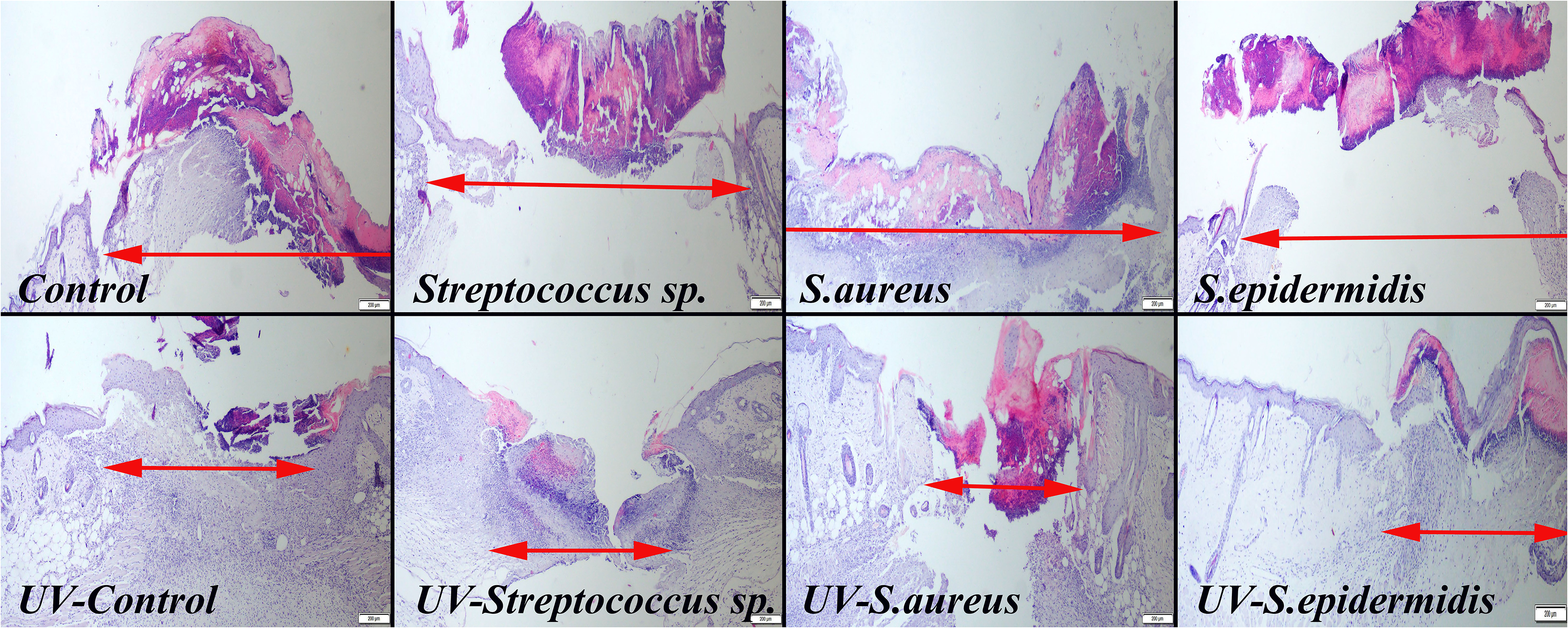
Histopathological appearance of the defect area among the groups. UV-C treatment on reduced crest formation, inflammatory reaction, and wound width. The 2-headed red arrows show the wound size, HE, scale bars = 200 µm.
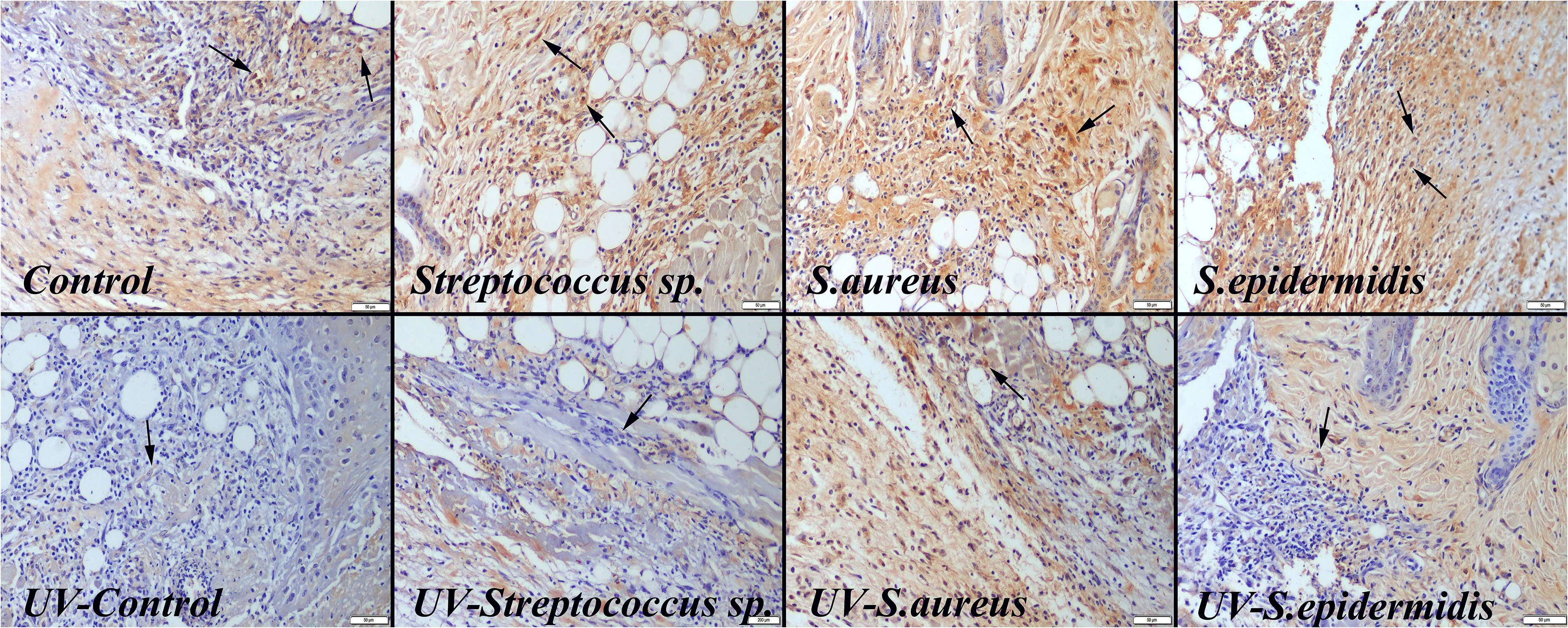
Caspase-3 immunohistochemical findings between the groups. Ultraviolet-C (UV-C) treatments caused a decrease in expression compared to the nontreated groups, Streptavidin-biotin peroxidase method, scale bars = 50 µm.
Microbiological Findings
In this study, we looked at how UV-C treatment affected the antibacterial activity of methicillin-resistant S aureus (ATCC 43300), S epidermidis strain YT-169a, and Streptococcus sp. in a mouse skin wound model. The table summarizes the findings. At the end of the experiment, there was a statistically significant (P <.05) decrease in the bacterial load of all the groups that received UV-C treatment compared to the control groups. This result indicates that UV-C treatment inhibits the growth of methicillin-resistant S aureus, S epidermidis, and Streptococcus sp. at infected skin wounds (Table 2).
Results of Methicillin-Resistant S aureus, S epidermidis, and Streptococcus sp. Growth on the Wounds (Units: 105 CFU/g).

Values are presented as mean standard deviation. P < .05.
Discussion
As harmful microorganisms in our environment develop resistance to disinfection methods, there is a greater need for new methods to combat them. Because of the increasing resistance to antibiotics and other antimicrobial agents, there has recently been a surge in interest in the use of ultraviolet lights for this purpose. 16 It has been reported that UV-C is particularly effective in areas such as operating rooms and hospitals and in destroying resistant agents such as methicillin-resistant S aureus. 17 It is well known that UV-C, in particular, significantly destroys this resistant bacterium in the early stages of bacterial growth and provides disinfection. 18 In this study, 3 different bacterial agents were used to infect mice with skin defects. Furthermore, UV-C disinfection of infected skin wounds was found to be very effective in reisolation studies. In addition, UV-C treatment reduced the inflammatory reaction of the defect area. It also decreased the risk of septicemia and mortality, because no mortality was observed in UV-C treated groups.
The sun is the most important source of UV-C, which has the shortest wavelength (100-280 nm) of any ultraviolet light. UV-C light has extremely potent bactericidal and antimicrobial properties. However, because of ozone layer absorption, these lights cannot reach the Earth's surface. 19 In this study, a UV-C generator and a lamp were used. A total of 8 sessions of UV-C treatment were used over a 4-day period, with daily improvement observed. In addition to having a strong antimicrobial effect, it has not resulted in any pathological findings in any organs.
The strong antimicrobial property of UV-C light has recently increased its use in the sterilization of gaseous media such as air, liquids such as water, foods, and solid surfaces.20,21 The mechanism of UV-C inactivation of microorganisms is to cause cellular damage by inducing changes in the chemical structure of DNA chains. 22 Many factors influence the efficacy of UV-C lights as a bactericidal, including the type and intensity of the agent, exposure time, lamp power, placement, and air movement. In addition, the wavelength range of 100 to 280 nm in which DNA absorbs the most light is also the wavelength range of UV-C light. This feature allows this light to have a bactericidal effect of this light. 23 Similar findings were found in this study, and we observed that UV-C has an antimicrobial and healing effect on skin wounds. UV-C treatment also reduced the inflammatory response and the increased rate of defect closure in infected skin wounds. It is also effective for reducing apoptotic activity in damaged areas. Moreover, antimicrobial and anti-inflammatory effects may be related to the mechanisms of all positive effects.
The goal of skin irradiation is to generate reactive oxygen species (ROS) at the application site. Disinfecting effect mechanisms of ROS are linked to direct cell death. This occurs through various pathways, necrosis, and/or autophagy, including apoptosis, depending on the type of cell treated, the amount of light supplied, the concentration of bacteria used, and the intracellular location of bacteria. 24 In this study, immunohistochemically evaluated apoptosis is one of the main mechanisms of the ameliorative effect of UV-C on examined skin. Although irradiation is effective against bacteria, skin cells showed decreased apoptotic activity.
Skin infections are one of the most common diseases in both humans and animals. This type of skin disease has a negative impact on the patient's quality of life. To treat most bacterial infections are difficult and take a long time. Although there are numerous methods for destroying microorganisms, and each method has some disadvantages. For this reason, UV-C draws attention due to its ease of use and lack of drawbacks. 25 Studies on the effects of UV-C on living bodies are very limited and generally negative. However, there have been studies in recent years on the use of UV-C in living beings, particularly on wound healing.7,11 These results were corroborated by the findings of this study.
Although there have been some reports about the DNA-damaging effect of 254-nm UV-C light on normal human cells, recently, authors reported that 200 to 222-nm UV-C light has fewer side effects on skin cells while also providing effective wound disinfection. 12 In this study, both disinfective and ameliorative effects of 254-nm UV-C at a 1-m distance on mice were evaluated, and quick healing was observed. In addition, there is no discernible damage in skin cells.
The only small number and generally reversible side effects reported long-term UV-C treatment. Dai et al 26 reported that mild epidermal shrinkage 24 h after UV-C exposure in BALB/c mice. Sterenborg et al 27 reported a slight tumorigenic effect of UV-C than UV-B after long-term treatment. There is no reported marked side effect of UV-C in short duration usage. This study also showed that no side effects in mice model infected skin wounds.
Comparing to conventional 254-nm UV-C, 222-nm UV-C has recently been shown to harmless to mammalian cells. 11 But especially the range of 250 to 270 nm UV-C spectrum, is the most lethal range of wavelengths for microorganisms and known as the germicidal spectrum. 5 For that reason 254 nm UV-C was used and no harmful effect was observed in this study.
The role of caspase-3 in apoptosis is commonly studied in various tissue damages. Moreover, the role of caspase-3 in bacterial and viral pathogens has received increased attention in recent research. While inhibiting the caspase-3 reaction allowed microorganisms to survive intracellularly, it prevented them from destroying cell barriers in extracellular microorganisms. These damaged cells can also have a negative and dramatic impact on neighboring cells, resulting in necrosis. As a result, the damage to the lesioned area may disrupt host barriers or the host immune response, allowing bacteria to invade. For that reason, inhibiting caspase-3 in extracellular bacterial infections results in rapid healing. 28 The findings of this study also revealed that decreased caspase-3 expression in cells in the defect area caused rapid healing.
The mechanism of disinfective effect of UV-C is related to cause cell damage by the production of cyclobutane pyrimidine dimers of the DNA. These DNA changes cause problems in cell replication and lead to necrosis. Dai et al 26 reported that UV-C can selectively kill the microorganism when using appropriate doses without marked cellular damage in mammalian cells. This study also supported this idea and showed antibacterial, anti-inflammatory, and wound healing effects of UV-C in an infected skin wound model in mice.
UV-C has less damaging effects on tissues than UV-B. For that reason, it is commonly used for numerous cutaneous disorders in humans as a phototherapeutic option with an excellent safety profile. 6 The main drug for treating infected wounds is antibiotics but they have several disadvantages such as resistance, repeated dosages, important side effect for different organs, and high prices. In contrast, UV-C has numerous advantages compared to antibiotics. For example, no resistance, only short time usage, no side effects, only electric price after purchasing the device, and repeated using during the lifetime of the lamp. Because of the high regenerative capacity of the skin, possible degeneration of skin cells by high dosage may easily tolerate the skin. This may be one of the other advantages of UV-C usage in infected skin wounds. UV-C may be the best choice to prevent and treat surgical wounds against bacterial contamination before, during, and after an operation. While evaluating the efficacy and safety profile of UV-C in this study, it was observed that UV-C irradiation gave positive results when used as an antimicrobial agent on infected skin wounds, and also had a healing effect on uninfected skin wounds. According to the study results, UV-C was safe when used for a short time on both infected and noninfected skin wounds.
The main limitation of this study is only a small number were mice used. But the results were indicating marked differences between treated and nontreated groups. UV-C treatment may be the best choice in both medical and veterinary practitioners for infected skin lesions caused by antibiotic-resistant bacteria.
In a conclusion, UV-C irradiation of skin wounds that were experimentally created and infected with different bacteria had a positive effect on the 4-day healing process and accelerated tissue repair. The findings of this study show that UV-C can not only disinfect bacteria but also promote wound healing by inhibiting apoptotic activity in skin cells. According to the findings of this study, the effect of UV-C may be related to and vary depending on the species of the agents, and UV-C can be used safely in infected skin lesions for short periods of time. Further studies are needed to determine the dosage and duration of the various microbial agents.
Footnotes
Acknowledgments
The authors thank Mustafa KUZU and Ugur OZDEMIR from Qzu QSS Technology, Burdur, Turkey, for allowing them to use nonionizing UV-C radiation generators for this study and staff of the Burdur Mehmet Akif Ersoy University Experimental Animal Production and Experimental Research Center for their valuable supports during the experiment.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Ethical Approval
This study approved by the Local Ethical Committee on Animal Research of Burdur Mehmet Akif Ersoy University, Turkey (MAKU-HADYEK- 712-13.01.2021).
