Abstract
Saliva packing can be one of the most severe life-threatening and challenging behaviours to treat. A 9-year-old male with autism spectrum disorder and intellectual disability had 100% nasogastric (NG) feeding tube dependence and significant adaptive behaviour regression (in speaking, using the toilet and his hands, school attendance, sleep). He participated in an intensive behaviour-analytic paediatric feeding treatment programme. Saliva packing, as an automatically-maintained undifferentiated behaviour that persisted in all waking contexts despite high engagement in activities, warranted an additional outside of meal approach. He began swallowing, met 100% of his 21 goals, gained weight, and eliminated tube dependence. He reached a variety of 94 across all food groups, drinks, and supplements. Caregivers were trained and reported high social validity, and gains generalised and maintained in 1-month follow-up. This is the first case to our awareness in Australia of an in-home solely behaviour-analytic intervention to eliminate tube dependence, and it was conducted without hunger provocation, weight loss, or limited nutritional variety.
Keywords
1 Theoretical and Research Basis for Treatment
Paediatric feeding problems can manifest in a wide variety of inappropriate mealtime behaviours (IMB) such as crying, head turning, covering mouth, batting at the feeder or utensil, throwing meal materials, clenching teeth, gagging, and spitting out foods or liquids. Feeding problems may also encompass skill deficits in chewing, cup drinking, and using utensils. Fortunately, behaviour-analytic treatments are highly effective in increasing appropriate and decreasing IMB, and teaching mealtime skills (Kerwin, 1999; Sharp et al., 2010; Sharp et al., 2017; S. A. Taylor & T. Taylor, 2021; Volkert & Piazza, 2012). Though, one of the most challenging feeding problems to treat is packing, involving holding food or liquid in the mouth past a specified timeframe (i.e., 30 s) rather than swallowing. In its most extreme form, an individual may pack or pool even their own saliva, a seemingly biological reflexive behaviour, and refuse to swallow at all. Although it may be less outwardly apparent compared to overt aggression or self-injury, saliva packing can be one of the most severe life-threatening problem behaviours, with significant consequences including dehydration, rapid weight loss, adaptive behaviour regression, and hospitalisation, requiring feeding tube placement.
With acute onset packing not due to skill deficits or lack of experience, the original aetiology may be medical in nature (e.g., gastrointestinal issues; Hagopian et al., 1996; Williams et al., 2011) or a triggering event (e.g., choking episode, allergic reaction, major life changes such as in caregivers or schools; Lenz et al., 2018; Lesser et al., 2022; Seiverling et al., 2016; Tang et al., 2011). However, even once the original aetiology has resolved, packing may persist due to behavioural reasons (e.g., to gain interaction or preferred items or activities, to avoid nonpreferred tasks) (Girolami & Scotti, 2001; González et al., 2014; Piazza et al., 2003; Rivero & Borrero, 2019). Individuals may avoid swallowing that in the past may have been associated with discomfort, anxiety, or high response effort (e.g., Hagopian et al., 1996; Lenz et al., 2018; Lesser et al., 2022; Seiverling et al., 2016; Williams et al., 2011). Thus, successful treatment involves graduated exposure (the active ingredient, i.e., escape extinction of IMB) to associate eating and drinking with pleasant experiences and increased success (i.e., differential or noncontingent reinforcement to increase appropriate mealtime behaviour; rewards/incentives, praise; Kerwin, 1999; Volkert & Piazza, 2012).
Specialised behaviour-analytic paediatric feeding programmes have encountered saliva packing or pooling clinically, but few cases have been detailed in the literature. With exposure-based treatment and differential attention (DA), Williams et al. (2011) eliminated gastrostomy tube dependence in a girl with typical development who had stopped swallowing saliva due to emetophobia (fear of vomiting) following a gastrointestinal illness. The authors did not report the extent of saliva packing or if she expelled it. In a descriptive case report, Tang et al. (2011) reported treatment (differential reinforcement and escape extinction in the form of nonremoval) for a case with tube dependence and saliva packing and spitting into napkins. Although, tube dependence was not eliminated due to psychosocial complications and data were not presented. In a group outcomes article, Lesser et al. (2022) mentioned as an example of interventions, for participants who expelled saliva into a cup, treatment involved removing the cup and providing differential reinforcement of saliva swallowing. The researchers did not provide further details and it is unclear how many of the participants in the study engaged in this behaviour.
Yet to our knowledge, no prior research studies have reported presentations requiring combined approaches for saliva packing as an undifferentiated automatically-maintained behaviour outside of meals without anxiety or saliva expulsion in addition to a feeding problem. Additionally, presentation of experimentally-controlled data and comprehensive outcome data on eliminating tube dependency is lacking. Further, this is the first report to our awareness in Australia of an in-home solely behaviour-analytic intervention to eliminate tube dependence, and it was conducted without hunger provocation, weight loss, or limited nutritional variety.
2 Case Introduction
Éloi was a 9-year-old Chinese Australian male with autism spectrum disorder (level 3), intellectual disability, paediatric feeding disorder, and avoidant/restrictive food intake disorder (ARFID). He was 100% nasogastric (NG) tube dependent, including for hydration and medication. He also had constipation, laxative dependence, under-average growth, toothbrushing resistance, and sleep disturbance. Éloi spoke in short phrases (primarily mands) and followed 1–2 step instructions. He did not have problem behaviour (e.g., aggression, self-injury, property destruction), medical issues, dysphagia, or motor deficits. He was cooperative with daily routines and tasks, and had a range of items and activities he engaged in including social interaction and affection.
3 Presenting Complaints
Ten weeks prior to admission, Éloi stopped swallowing, required an NG tube, and had a significant regression in adaptive behaviour. When his mouth was significantly full of saliva, he was no longer speaking (he would not open his mouth to prevent saliva leaking out; instead he used gestures, leading, or made unintelligible noises with his mouth closed) or using his hands (he held his lips together with one hand to prevent saliva leakage and a napkin in the other). When his mouth was not filled to maximum capacity with saliva and he spoke, intelligibility was decreased due to presence of saliva. Caregivers attempted to prompt him to swallow and speak clearly before honouring his mands (i.e., requests), but he still would not swallow. He was no longer going to the toilet independently and required diapers. Éloi was no longer able to attend school. Caregivers reported decreased receptive communication (e.g., following instructions, responding to communication) and engagement with previously preferred leisure activities, and increased stereotypic behaviour. His sleep was also significantly impacted. Éloi resisted expelling the saliva to clear his mouth, and at times caregivers reported having to syringe saliva out. He had brief/mild negative vocalisations and made attempts to wipe or change if the saliva leaked out or wet his clothing. He did not engage/’play’ with the saliva (e.g., swoosh in mouth, manipulate with fingers). Caregivers reported the only context he swallowed saliva in was involuntarily during sleep.
4 History
Caregivers reported Éloi previously had typical mealtime and drinking skills, feeding behaviour, and consumption of a variety across food groups and flavours. However, at approximately age 6 he had food selectivity (ate primarily noodles) for which he received early intensive behavioural intervention. In the past when packing saliva or food, they reported he would swallow when prompted. He had a history of brief 1–3-day hospitalisations due to saliva pooling and lack of swallowing for a few days, resolving without requiring a feeding tube. Also, on one occasion he packed saliva continuously daily for approximately a month while continuing eating and drinking in meals within his typical routine. Per caregivers, he had a history of lactose intolerance and positive allergy test results to kiwi and strawberry, but he regularly consumed these fruits and had no clinical reactions except for diarrhoea with lactose in the distant past. We did not target these fruits.
More recently for approximately a month prior to tube placement, caregivers reported decreased appetite and long meal durations. During a trip overseas, he had diarrhoea, stopped swallowing, and was hospitalised for a week requiring intravenous fluids and a feeding tube. He was then hospitalised for 10 days upon returning home to Australia. We reviewed the hospital discharge summary. Éloi had extensive medical workup (e.g., magnetic resonance imaging [MRI], laryngobronchoscopy and endoscopy with biopsies, bloodwork, faecal and urine cultures, transthoratic echocardiogram, computed tomography [CT] scans). Éloi had consults with multiple disciplines including a speech pathologist, dietitian, multiple physicians with various specialties (e.g., ear nose and throat, gastroenterology, dental, endocrinology), and his behavioural therapists. He had five teeth extracted and prescription of fibre and laxatives for constipation, as well as elevated prolactin levels. The definitive initial cause remained undetermined (e.g., dental pain and chewing difficulty, constipation, food poisoning/gastrointestinal illness, stress or adjustment to a significant environmental change in travelling overseas, sore throat).
5 Assessment
Éloi’s physician provided written clearance to participate in the intensive feeding programme including a review of systems focused on feeding relevant areas. Caregivers provided historic videos of him eating and drinking on social occasions. For the entirety of the initial appointment (3 hr), Éloi packed large volumes of saliva with protruding cheeks and held a napkin. We did not observe (or hypothesise based on caregiver interview and record review) any social or environmental maintaining factors (e.g., attention, tangible, non-meal-related escape).
We conducted paired-stimulus preference assessments for tangibles (i.e., items or activities that could potentially be provided as reinforcement; rewards/incentives to increase motivation) and a variety of feeding edible mealtime targets (foods, drinks, and supplements). Éloi engaged with a variety of sensory items, toys, iPad, an indoor playground (trampoline, exercise ball, tunnel, monkey bars, climbing grid, swing), interactive dancing and sing-along videos, and cooking videos. We provided a small tan plastic cup caregivers used for saliva expelling to clear his mouth. He did not use the cup independently and initially refused to expel saliva in our sessions when prompted. We reserved napkins for contingent access (during his earned reinforcement interval).
We began with small bolus sizes and a variety of previously highly preferred foods, drinks, and vitamins and textures and consistencies as a self-feeder/drinker. Intervention included choice (Éloi could choose which bites or drinks to take) and demand fading (starting with only one small bite or drink required). For Éloi to contact and experience the high-quality reinforcement available and the rules of the sessions, we also conducted modelling and practice with empty spoons and a clean mouth check (feeder prompts to “open ah” to check mouth for saliva, food, or liquid) immediately after clearing his mouth of saliva or administering over-the-counter sore throat spray. The feeder conducted clean mouth checks every 30 s by verbally prompting “show me ah.” If Éloi did not open, we modelled an open mouth with the verbal prompt and gently touched his chin with the tip of our index finger. If he still did not open, we gently touched a flipped (180°) rubber coated tip metal infant spoon to his lips along with the verbal and model prompts. If he packed (bolus remained in mouth; not swallowing) when the timecap (maximum session duration) elapsed, we removed the bite using a rubber coated tip metal infant spoon and napkin.
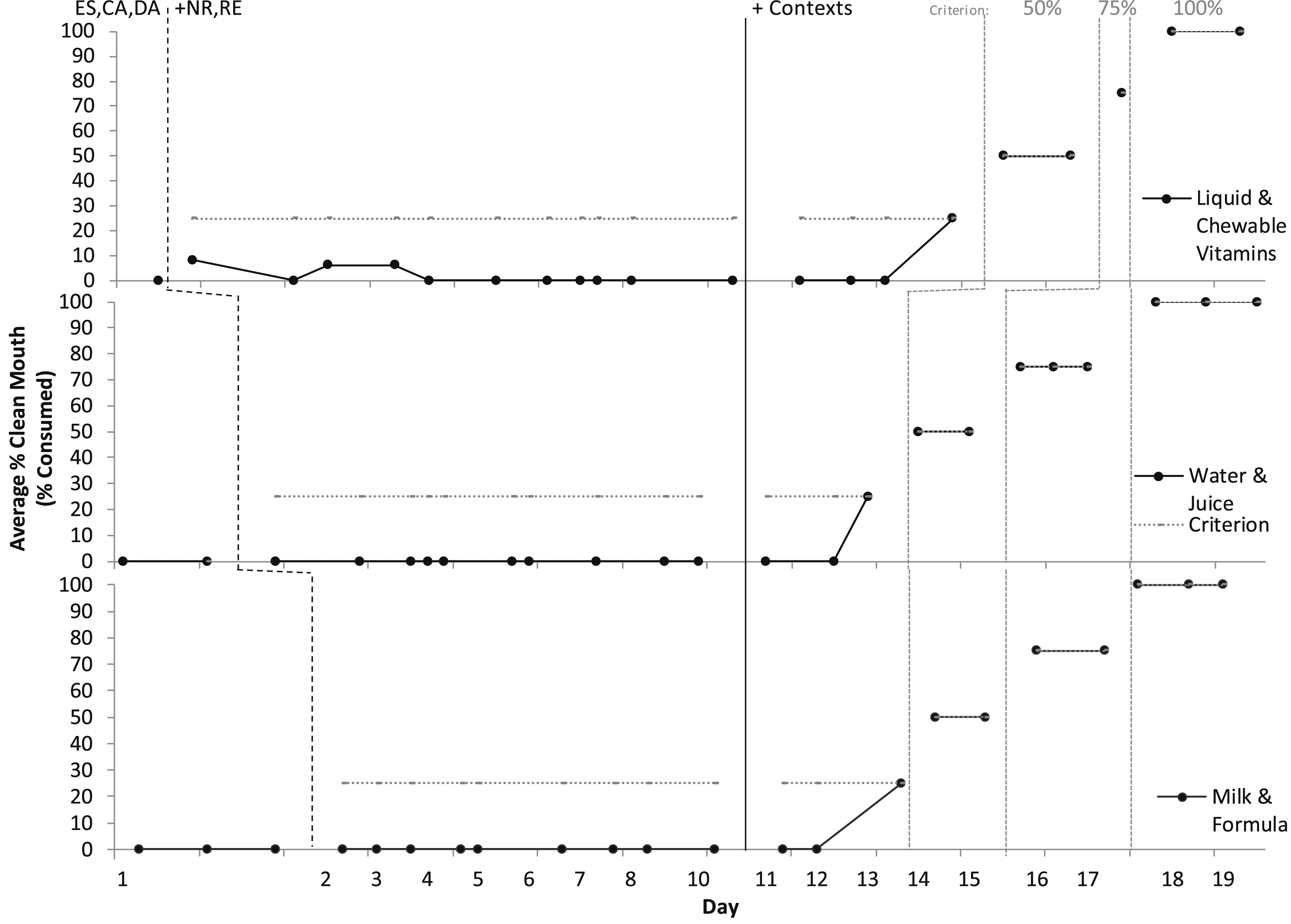
In paediatric feeding treatment evaluations (Figures 1 and 2), baseline consisted of escape (ES) in the form of a break for IMB(head turning, mouth covering, batting at the utensils or feeder), refusal, or expulsion (spitting out). Baseline also consisted of differential reinforcement for attention (DA for appropriate mealtime behaviour in the form of descriptive praise and no attentive reactions for IMB) and tangibles (contingent access to preferred items and activities [CA] for independent acceptance and swallowing); and continuous noncontingent preferred music. These reinforcement components were in place for the entire treatment evaluation. In baseline, Éloi accepted zero bites, drinks, or supplements into his mouth. We did not observe any indicators of anxiety (e.g., trembling, irregular breathing, tensing). We had zero starting points for swallowing (not even empty utensils and saliva or a ‘dry’ swallow), so an empirical packing assessment was not applicable (i.e., Rivero & Borrero, 2019). Percentage of swallowing in meal sessions for solids. Note. ES, Escape; CA, Contingent Access; DA, Differential Attention; NR, Nonremoval; RE, Re-presentation; REDIS/SF, Redistribution/Swallow Facilitation (Lower Texture only). Percentage of swallowing in meal sessions for medications and liquids. Note. ES, Escape; CA, Contingent Access; DA, Differential Attention; NR, Nonremoval; RE, Re-presentation.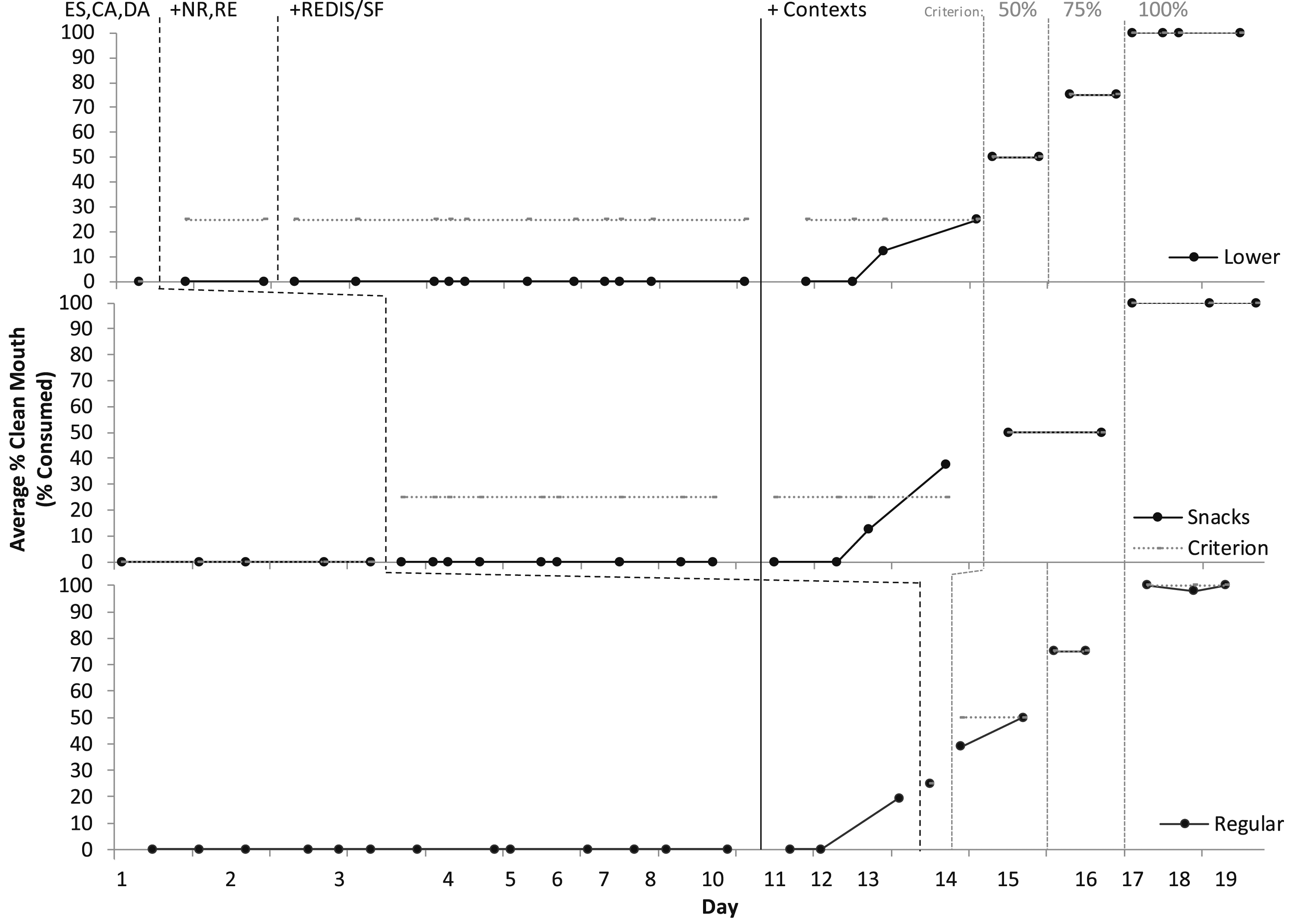
We observed continuous saliva packing regardless of context, including when engaged in participating in highly preferred items and activities with 1:1 high quality attention without demands. As this was a clinical feeding programme admission targeting food and liquid refusal (as described in Taylor et al., 2020), we did not conduct an analogue functional analysis of saliva packing outside of meals. However, information gathered via direct behavioural observations and caregiver interview suggested results would be undifferentiated across all conditions including the control (toy play) condition (noncontingent access to attention and tangibles with no demands), suggesting undifferentiated automatic reinforcement not maintained by environmental social consequences. Nor did we conduct a formal competing stimulus assessment to identify stimuli associated with high levels of engagement and low levels of saliva packing, but information gathered suggested that despite high engagement with items and activities, saliva packing would remain high across all conditions. Further, the only response incompatible with saliva packing is eating and drinking (i.e., an individual can still engage in almost all activities while still packing saliva), except for speaking intelligibly when his mouth was overfull.
6 Case Conceptualization
Caregivers did not wish to decrease tube feeding volume prior to Éloi’s oral intake increasing (see Figure 3). They shifted feed times apart from mealtimes as much as possible to minimise potential fullness decreasing motivation to eat or drink prior to sessions (e.g., earlier in the morning, evenings). Initially, our approach was from a paediatric feeding perspective using well-established empirically-supported treatments (Kerwin, 1999; Volkert & Piazza, 2012) including escape extinction for IMB and expulsion and differential reinforcement (DA, CA) of appropriate mealtime behaviour. In escape extinction, IMB and expulsion no longer resulted in escape. For nonremoval (NR) of the spoon/bite/cup, if Éloi did not accept the bite/drink within 5 s of the verbal prompt, we did hand-over-hand (placed our hand over his hand with the spoon/bite/cup) and guided the spoon/bite/cup to his upper lip (e.g., Hoch et al., 1994; Kerwin et al., 1995). The feeder followed his mouth with head turns, and blocked disruptions and mouth covering. The bite/drink remained at his upper lip until acceptance or the timecap (10 min) elapsed. If he opened his mouth, we inserted the spoon/bite/cup with the exception of gags, coughs, yawns, or emesis (vomiting). For re-presentation (RE) of expulsions, we scooped up expulsions and re-presented immediately, but if the bite/drink could not be re-presented (e.g., emesis), we re-presented a new fresh bite/drink of the same food/liquid/medication. These interventions were effective in increasing acceptance and independence with a wide variety of targets. However, packing of saliva and feeding targets persisted (Figure 1) even when we added highly specialised components such as NUK® (infant gum brush) redistribution and swallow facilitation [REDIS/SF] with lower textures (Gulotta et al., 2005; Hoch et al., 1995; Sevin et al., 2002), involving holding the NUK® brush horizontally and perpendicular to the tongue, and brushing the bolus directly onto his tongue by rolling the brush down and out with downward pressure to deposit it in place (midline for redistribution; posterior of tongue for swallow facilitation (as described in Taylor, 2021, 2022). Once added to the treatment package, these escape extinction and differential reinforcement components remained in place for the entire remainder of the treatment evaluation. Volume and weight per day.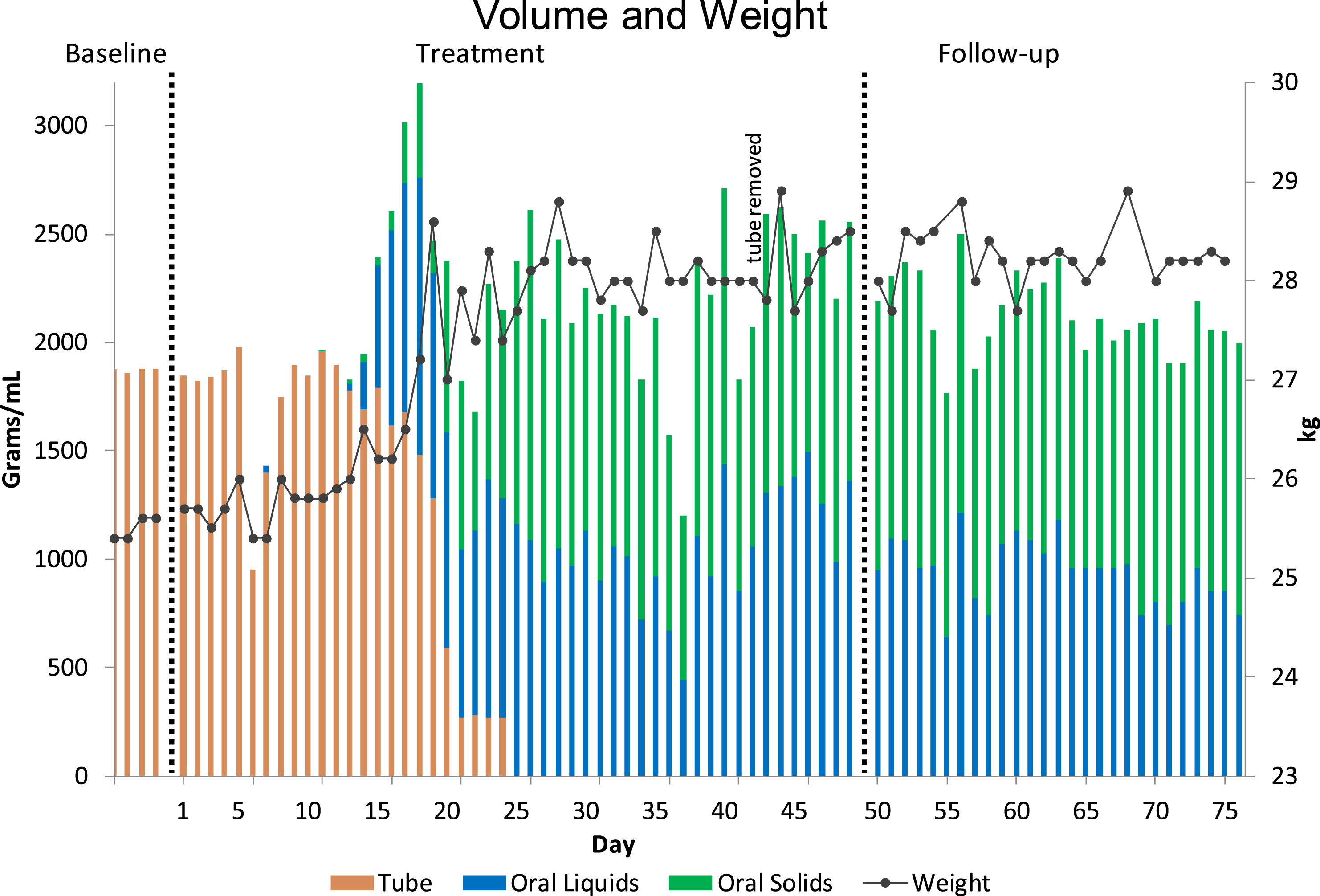
In the United States, intervention is often conducted with children with gastrostomy tubes rather than NG tubes. We were unaware if the NG tube’s presence in Éloi’s throat was contributing to lack of swallowing due to physical discomfort or novelty, or behaviourally (e.g., stimulus control, classical conditioning). Therefore, under the same treatment conditions (DA, CA, NR, RE; REDIS/SF in lower textures only) we used a wide variety of targets with the aim of pairing swallowing with optimal physical comfort and decreasing response effort such as ice cream, smooth yogurt, avocado, coconut oil, and medication/pill swallowing lubricant products (Gloup® Forte and Original). We also probed ‘sampling’ of the reinforcement available and pausing to wait for a swallow, removing the expulsion cup (not clearing his mouth prior to session), reclined seating with a chin prompt, full-cup sipping (larger boluses), and choice arrangements for negative reinforcement (e.g., he could swallow a small preferred bite/drink to avoid presentations of larger boluses of novel or potentially nonpreferred foods). In the initial full-cup session, Éloi had a few swallows of water with the full cup presentation with no aversive reaction (e.g., grimacing, anxiety responses). However, he packed the remainder of the bolus during the reinforcement interval, and then in all subsequent sessions consistently packed the entire cup bolus. This was the only swallowing we had observed and suggested he had the ability to swallow without apparent difficulty. No swallowing occurred in any other probes or across a wide variety of targets (e.g., textures, consistencies, flavours, food group or type, utensils, presentation styles, bolus sizes; Rivero & Borrero, 2019) with potential high quality, magnitude, and rate immediate reinforcement, choice, and demand fading, or in probes of free access choice meals in dining room without prompting (Taylor & Taylor, 2024).
Paediatric feeding treatments are typically rapidly highly effective, but packing continued even with swallow facilitation and after the occurrence of unintentional swallowing with the full water cup. Hence after 8 days without swallowing results, we began conceptualisation of outside of meal behaviour approaches. Given an undifferentiated automatically-maintained behaviour that persisted even with high engagement with highly preferred items and activities and 1:1 attention/interaction, we were unaware of established assessments from prior literature to use. We conducted outside of meal observations and created a shared data collection log with caregivers to list any potential mands (or observed potential ‘desires’) or refusals/noncooperative behaviour that occurred. We removed napkin access with caregivers. On Day 11, we began outside of meal sessions with caregivers in different contexts (i.e., academics, domestics, structured leisure, unstructured leisure) and a structured schedule. We scheduled bathroom trips to clear mouth (to interrupt packing) and use the toilet. An additional rationale was to regain both engagement in activities and independence in areas that had regressed (e.g., pulling pants up and down for toileting). We also placed unintelligible speech attempts (due to saliva packing) and arm dragging of/hanging on caregivers on extinction, and any mand granting (e.g., tangible, attention) of requests contingent dependent on swallowing. We trained caregivers on 3-step prompting (verbal, gestural, physical) and physical safety for transitions. The shared log did not reveal a pattern of consistent behaviours, except some resisting expelling saliva/mouth clearing. Éloi participated in the scheduled activities, did not engage in problem behaviour, and displayed indices of happiness (e.g., smiling). Yet, no swallowing occurred, and saliva packing remained continuous both in and outside of meals with us and caregivers. We developed plans to do preference assessments to get a hierarchy of preference for the contexts (including lower texture meal sessions, bathroom trips, and transitions) and place preferred contexts contingent on swallowing. Though, outside of sessions, we observed a high frequency of Éloi remaining next to his mother in the kitchen to watch her cook and clean, and participate in dish washing and food preparation. Interestingly, some of Éloi’s preferred activities were food related including watching cooking shows, watching others eat, toy foods and drinks, and assisting with meal-related activities such as food preparation and serving.
7 Course of Treatment and Assessment of Progress
On Day 12, we prompted Éloi to swallow saliva to enter the kitchen (blocked entry and waited for a swallow) and for the first time, he finally swallowed saliva several times. Though, in meals, packing continued. During contingent access (reinforcement interval) for empty spoon practice in meals, he began following prompts to swallow saliva to continue the activity longer (we paused the activity and waited for a swallow). Outside of meals, we prompted Éloi to sit at the far end of the dining room alone without sight of the kitchen, and he swallowed saliva to enter and remain in the kitchen. His caregivers continued this in the evening alone, and he began consistently following prompts to swallow to access to other items and activities also. The next day (Day 13), he swallowed saliva outside of meals including a large full mouth bolus in the morning, but initially continued to pack in meals; however, he later finally chewed and swallowed a regular texture bite of orange and then some other fruit bites, drinks, and snacks. This was the last day Éloi required any mouth clearing of saliva (e.g., expel cup, wipe or rinse out), except for on Day 26 his father reported at night during bedtime, approximately after 1 hr in bed his mouth was full of saliva, and he prompted him to expel into the cup. We added water drink presentations in between outside of meal sessions with caregivers and he swallowed the water as well.
Figure 1 depicts meal treatment evaluations for solids and Figure 2 depicts liquids and medication. Graphs depict average per day of clean mouth, a product measure of swallowing/consumption at any time, where no food or liquid larger than the size of rice was observed in his mouth at a 30-s check with the feeder prompting him to “show me ah.” We organised treatment evaluations aiming to employ multiple-baseline with embedded changing criterion single-case experimental designs with paediatric feeding interventions. However, results could be conceptualised with baseline as extended probes and response to treatment occurring following the addition of contingent access for saliva swallowing in outside of meal session contexts. Consumption and acceptance increased from zero to 100% in all targets (food, drinks, supplements) within 19 days. Éloi had a decrease in latency to acceptance and swallowing (how long it took him to take and swallow bites/drinks), IMB, expulsion (spitting out), and negative vocalisations (e.g., crying, screaming, negative statements about the meal). He had an increase in indices of happiness and independent acceptance (taking bites/drinks by himself without hand-over-hand guidance and assistance) and swallowing (without using the NUK® brush for redistribution/swallow facilitation). He began speaking more frequently and clearly.
Figure 3 depicts tube and oral volume and weight per day over 80 days (4 days of baseline prior to starting the programme, 49 days during the 7-week intensive programme with 44 full days of treatment, and 27 days of follow-up including one final full day of treatment 1-week after the intensive programme). After 23 days of treatment, Éloi was no longer using the NG tube because he was meeting all his needs orally. Caregivers removed the tube after 39 days of treatment. Oral daily volume increased from 0 to up to a maximum of 1525 g of solids and 1493 mL of liquids. Éloi’s weight increased from 24.5 kg to over 28 kg and he did not lose any weight during the programme, but rather, gained weight.
Figure 4 depicts pictures of meals consumed. Éloi reached a variety of 94 across all food groups (protein, starch, vegetable, fruit, combinations) and drinks and supplements. He met 100% of goals (n = 21). Both parents participated in behavioural skills training (BST) and were trained to procedural integrity informally based on behavioural observations. In full plate/cup meals, we made modifications aimed at increasing independence and decreasing meal duration. For example, we added beat the clock in which Éloi had a visual timer set with approximately 5–10 min longer in duration than an above average meal. If he completed the meal before the timer ran out, he earned the remaining time on the timer as the reinforcement interval. He also then was able to choose his foods from a ‘buffet’ and make his own meal plate for the subsequent free access/choice meal and eat socially at dining room table (and watch others eat and the activities in the kitchen) versus alone at the far end of the room. We also added a levels system (González et al., 2013) in between meals for beating the timer with a bracelet signally high-quality attention and mand honouring until the next meal compared to independent leisure or potentially less preferred contexts or tasks. Caregivers were trained on using the NUK® brush for swallow facilitation and infant spoon redistribution (Taylor, 2020). They rarely needed to implement the procedures, but when implemented they were effective and Éloi would swallow more quickly. Sample photographs of meals consumed.
Figure 5 depicts outside of meal context sessions. Éloi initially consistently had zero saliva swallowing, but when he began swallowing, it remained consistently high and stable at 100% for a week and generalised across settings and people, so we paused this data collection. However, he still was not swallowing saliva independently without prompting, so on Day 38 we started self-monitoring sessions with a timer and token board and preferred video and praise as reinforcement. It took several modifications and multiple sessions, but Éloi made some progress with self-monitoring (e.g., started looking at the timer, swallowing, and showing assistant or parent an open mouth). We began fading assistant and parent proximity and probed self-monitoring in other contexts. Percentage of swallowing saliva outside of meals.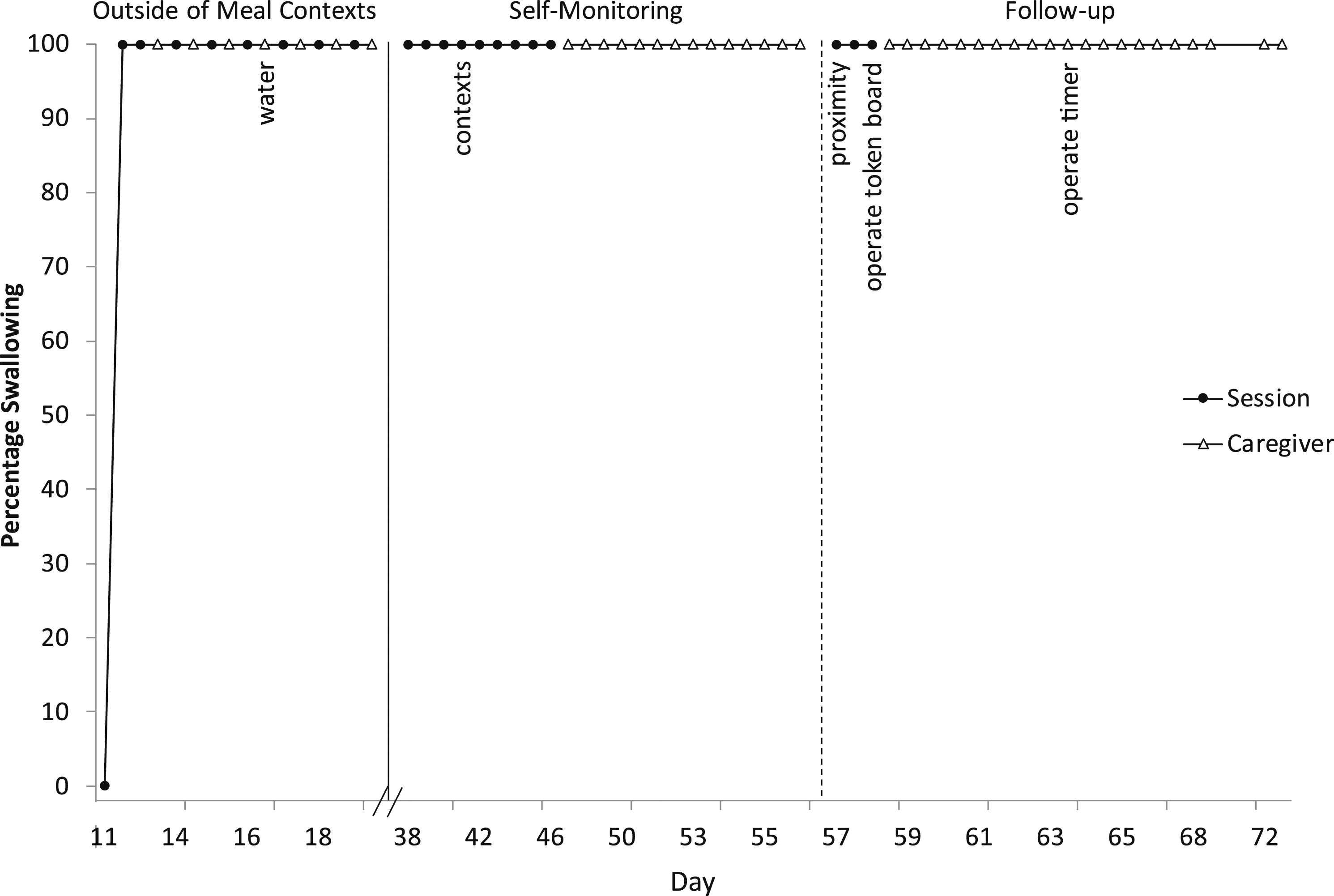
Caregivers reported maximum social validity and satisfaction (M = 7 out of 7, range 7; 280 out of 280, 100%) in written discharge surveys based on Anderson et al. (2021) and Hoch et al. (1994), and treatment acceptability (7 out of 7 for all items) on the separate treatment approaches (based on Taylor & Taylor, 2022) except for slightly lower ratings on earning money as reinforcement (5 out of 7) and move-on to the next bite/drink as a treatment component for packing (6 out of 7). Full text surveys are available in Taylor et al. (2024).
8 Complicating Factors
The NG tube was a complicating factor (more common for longer durations in Australia) compared to gastrostomy tubes. The tube caused nose bleeding and mucus, and irritation to his nostrils and facial skin. The facial tape became soiled requiring tape changes. Per caregiver report, during a tape change with less experienced nurses, the tube came out. Éloi had to go to the hospital to have it reinserted. The NG tube also may potentially cause throat irritation and swallowing discomfort particularly with solids and higher textures. Conceptually, we were unaware if the NG tube’s presence in Éloi’s throat was contributing to lack of swallowing physically and/or behaviourally. Additionally, lack of hunger as an abolishing operation could potentially pose a complicating factor as his caregivers did not reduce tube feedings prior to oral intake increases (Roth et al., 2010). However, these factors do not seem likely as packing continued on the days the tube was accidentally removed. Further, once Éloi started swallowing, he swallowed a wide variety of textures and large boluses with the NG tube and feeds still in place with no adverse reactions.
Packing saliva as a target behaviour poses challenges as it is essentially the absence of a behaviour (i.e., swallowing), and saliva is colourless and builds up gradually, requiring an arbitrary cut-off in time (e.g., 30 s) or volume to operationalise. An additional challenge is delivering immediate consequences for a behaviour that is not a discrete occurrence. Volume of saliva present in Éloi’s mouth was variable. Packing was readily apparent when a large volume of saliva was present due to protruding cheeks, but less so with smaller volumes even if his mouth was open. It was initially difficult to assess if his mouth was fully clear of all saliva during outside of meal contexts. Initially, Éloi refused to open for clean mouth checks. We had to use a finger prompt (straight index finger inserted along the upper gum line) (Borrero et al., 2013) and rubber-coated tip metal infant spoon (to check under and over the tongue). When Éloi opened his mouth for the check, he initially would not put his tongue up or down. Then, when he appeared to put his tongue down, he would continue packing on the far back of it. We attempted to observe and touch for throat movements (as employed in Hagopian et al., 1996), but Éloi would engage in mouth and throat movements which appeared to be swallowing, but were actually aimed at continuing packing. Éloi was significantly skilled at packing large volumes and refraining from swallowing or expelling for long durations. We used a headtorch lamp, and videoed all sessions and reviewed and discussed the footage between the behaviour analyst (first author), assistant (second author), and his mother. Swallowing was also difficult initially to assess in meal sessions due to the small boluses. Hence, in Figure 2 the few (3) recorded consumptions of liquid vitamins prior to outside of meal sessions commencing were most likely in error (packing the bolus on the high back of his tongue). During outside of meal sessions, his mother reported that she was able to assess full saliva swallowing via speech clarity. She prompted speech to confirm his mouth was completely clear and we were able to hear the difference in clarity. A concern with prompting speech is as with swallowing, it cannot be physically prompted; however, Éloi always complied with the prompt to speak. Fortunately, once Éloi began swallowing saliva upon prompting, he did so consistently (high and stable at 100%) and necessity of mouth clearing of saliva (expulsion or removal of saliva) ceased, although at times he required more than one prompt to fully clear his mouth. Additionally, once he began swallowing in meals, we were able to rapidly increase bolus size, he independently opened for clean mouth checks and put his tongue down, packing was infrequent, and swallowing was clearly apparent. Therefore, we have high confidence in the reliability and validity of the large majority of the data except for the three liquid vitamin sessions in meals prior to Éloi beginning to swallow; though, we agreed in hindsight that likely no swallowing occurred.
Initially, Éloi also refused to clear his mouth (expel) of saliva and mealtime boluses he had accepted. We used the finger prompt and infant spoon, and had to wipe the inside of his mouth to clear it. Independence in expelling into the cup when prompted (the cup was held to his mouth by the feeder) eventually increased. The presence of saliva in frequent amounts and large volumes was challenging to manage in meals, and we frequently had to clear saliva to properly implement redistribution and swallow facilitation. Additionally, we were initially unable to successfully implement redistribution or swallow facilitation due to tight clenching and lip pursing (even with the finger and NUK® brush prompts), tightly retracting lips, blocking the NUK® brush with his tongue, and excess saliva. Outside of sessions to increase cooperation and independence with these tasks, we informally did modelling, empty utensil practices with prompting sequences and reinforcement, and fading (of distance and pressure with prompts and utensils; from redistribution at midline to swallow facilitation at posterior tongue; from NUK® brush prompt to back to finger prompt). We were then able to successfully implement the procedures with decreased effort and prompting (e.g., without the finger or NUK® brush prompts or infant spoon) and obtain proper bolus placement with the headtorch. This process targeting redistribution/swallow facilitation cooperation and independence and fading to our awareness has not yet been reported in the literature, although we did not systematically evaluate or collect data on it being a clinical case with time constraints.
Several additional complicating factors included communication as caregivers spoke English as a second language. Éloi had a history of prompt dependency in prior services and therapies. Although it was a positive factor that Éloi did not have outside of meal problem behaviour, he also happily entertained himself alone (e.g., looking at and moving his hands, humming, rubbing his fingers on surfaces, rocking) and did not respond (e.g., attempts to leave the area or access his mother, negative vocalisations, repeated mands) to many of the treatment modification attempts made. Finally, he did not have consistent mands and his potential ‘desires’ were variable and appeared ritualistic rather than to have ‘his way’ or ‘control’ others.
9 Access and Barriers to Care
Implementing this treatment programme in Australia posed several barriers to care. Funding was a significant barrier to care and stressor for caregivers and us as providers. The assistant conducted the final 5 weeks of the programme in person alone with caregivers with the behaviour analyst on call remotely to extend treatment time within funding constraints. We did not have enough time to complete self-monitoring steps to achieve independent saliva swallowing or full integration back into his daily routines prior to the tube. We were not able to conduct self-monitoring as systematically as we would have preferred, and did not obtain data on independence with the steps. We also did not present latency to swallowing data, but this significantly decreased (initially it took long durations for him to swallow). On a daily basis, Éloi’s mother had to continue feeding almost all of the protocol and free access/choice meals and self-monitoring sessions and contexts in the structured schedule. His father worked full time and assisted in caring for Éloi’s sibling, and they did not have funding for further assistance.
Additionally, we did not have access to a multidisciplinary team or other disciplines with the required paediatric feeding or behaviour-analytic expertise. Given the public health system in Australia, during the admission there were waiting lists and cancellations in obtaining appointments with physicians and a dietitian. We used free Australian online calculators (Australian Government National Health and Medical Research Council, 2024) for caloric and liquid needs, Éloi’s pretube portion sizes and volumes per caregiver report, and caregiver monitoring of weight, urine, and bowel movements. Caregivers handled all tube feeds, modifications, and reductions, and removed the physical feeding tube on their own.
Fortunately, Éloi had an adequate medical work-up prior to treatment, and was medically stable and healthy. Another positive aspect of access to care for the family was outside of the United States in travelling distance the availability of a behaviour analyst specialised in paediatric feeding with additional expertise in automatically-maintained severe problem behaviour (e.g., pica) and equipment (e.g., NUK® brushes) from the United States. These treatments originated from a specialised hospital setting in Baltimore, Maryland, United States (Babbitt et al., 1994) where the first author trained. However, this access is rare outside of the United States in countries such as Australia (S. A. Taylor & T. Taylor, 2021) where there may be a lack of early intervention access and high-quality behaviour-analytic training and specialised intensive services, in addition to the barriers in lack of adequate funding and intensity for treatment.
10 Follow-Up
Gains in oral solid and liquid intake, weight, and swallowing saliva outside of meals upon prompting maintained up to 1-mo follow-up. We conducted a full day in-person follow-up visit 1-week following the intensive programme and weekly phone calls. We will continue to follow-up including a written survey at scheduled intervals (every 3 months then yearly). Caregivers fed 107 meals alone on days off and the month immediately following the intensive feeding programme. They took meal plate pictures, weighed grams and mLs, videoed meals, and recorded this data, his weight, and self-monitoring sessions on shared electronic spreadsheets where we communicated. They uploaded pictures and videos in a shared folder. Average meal rating was 6.8 out of 10 (range 5–10), on a scale from 1 (terrible/worse meal) to 10 (great/perfect meal). Consumption was 100% for all meals. Caregivers continued free access/choice meals; however, Éloi rarely ate or drank independently and thus still required a feeder and mealtime protocol. Éloi had not yet independently swallowed saliva. Parents reported when outside of home, when prompted he would consistently drink water and formula and swallow saliva.
We provided caregivers written next steps for self-monitoring. In self-monitoring (Figure 5), parents began having Éloi operate the token board and timer. Via comments on the shared electronic data sheet, for some sessions parents reported it ‘went well’ and he ‘responded quickly,’ and for some sessions he missed varying numbers of tokens and had to repeat them. Future steps planned included having him do the entire session independently via video camera (showing ‘ah’ for a clean mouth check to the camera instead of a person conducting the check) and then bringing the completed token board to the person after it is completed with a final clean mouth check including assessing vocal clarity, regularly conducting self-monitoring in all contexts (e.g., academics, domestics, structured and unstructured leisure), and gradually increasing the timeframe from 2 min.
11 Treatment Implications of the Case
Automatically-maintained problem behaviours may be the most difficult type to treat because the reinforcement is not in control of the environment or others, and even more so when undifferentiated across contexts. In Éloi’s case, the behaviour of saliva packing persisted despite high engagement in other activities, and the only potential incompatible response was eating and drinking. Therefore, even recent advancements in assessment and treatment technology for treatment-resistant behaviours (Hagopian et al., 2020) would not be adequate for this case. Similarly, in paediatric feeding, packing is one of the most difficult behaviours to treat because the bolus is in the mouth out of control of the feeder and swallowing cannot always be physically prompted (even with swallow facilitation). Further, saliva packing is its most extreme form. Therefore, this combination in saliva packing as both a feeding and outside of meal problem behaviour was significantly challenging to treat.
As a sole practitioner in Australia a paediatric feeding programme conducted this as a clinical case with time constraints, we employed single-case experimental designs for the paediatric feeding intervention, but did not systematically evaluate the intervention from an outside of meal perspective, and it was based on arbitrary reinforcement. We do not know if the feeding intervention alone would have been effective with more time without outside of meal intervention. However, based on the knowledge we have including Éloi continuing to pack saliva outside of meals while eating and drinking orally, we hypothesise that the combination approach was necessary. Further, Éloi required a feeder for meals and we did not achieve high consistent independence in eating or drinking during the intervention timeframe we had. Therefore, had the treatment been conducted in reverse order (outside of meal approach conducted first), we also hypothesise the feeding intervention still would have been necessary to establish oral intake and feeding tube elimination. Researchers with more resources could use this case example to address the limitations in existing assessment and treatment technologies we have outlined and hopefully produce novel advancements to more systematically approach this type of target behaviour.
In contrast to other cases of saliva packing, there was no anxiety or expulsion, although Éloi also did not engage with the saliva. Based on other cases from an anxiety perspective, we anticipated that after Éloi initially experienced swallowing without adverse consequences, treatment would rapidly progress, and he would swallow saliva and eat and drink more independently. However, this was not the case with independence. Moreover, the current case did not have some of the positive advantages compared to some other cases in the literature. In Hagopian et al. (1996), the participant’s repertoire included an empty swallow, although he did have significant ongoing gastrointestinal issues in contrast to Éloi. In Williams et al. (2011), the child swallowed 2 cc of water in the intake appointment, albeit accompanied by a 40-min tantrum. In Lesser et al. (2022), expulsion opportunities were removed as part of the intervention. Finally, Éloi had intellectual disability and autism.
Hunger provocation approaches (decreasing tube feeds) are commonly employed in Australia, but do not address skill deficits or IMB, and pose health risks (e.g., weight loss, nutritional deficiencies; Taylor et al., 2019). Even without skill deficits, this approach was not appropriate and would have been ineffective for Éloi, and caregivers did not prefer this. Behaviour-analytic treatments are the only well-established empirically-supported treatment for paediatric feeding, and were effective for Éloi without hunger provocation. Conversely, Éloi gained rather than loss weight, and consumed a wide and increased variety of foods, drinks, and supplements to ensure adequate nutrition and hydration. Tube reductions were gradual following increases in oral volume. Regrettably in Australia, the large majority of children receive paediatric feeding interventions other than these empirically-supported behaviour-analytic treatments (S. A. Taylor & T. Taylor, 2021).
12 Recommendations to Clinicians and Students
Saliva packing may not receive the attention it warrants compared to problem behaviours such as aggression or self-injury or other IMBs (e.g., throwing, thrashing, spitting). However, akin to pica, saliva packing is one of the most extreme and life-threatening behaviours. In Éloi’s case, in addition to the hospitalisations and the feeding tube, his entire broader life and adaptive behaviour was significantly disrupted (e.g., no longer speaking, in diapers, not using his hands, not going to school, sleep). Determining the severity of paediatric feeding problems, adequate multidisciplinary and medical work-up prior to intervention, and adequate treatment outcomes and procedures requires highly specialised training, expertise, and competency (Piazza et al., 2020; Taylor & Taylor, 2023; Williams & Seiverling, 2022). Children should be referred as early as possible to receive expert empirically-supported treatments at the individualised intensity warranted for case severity (S. A. Taylor & T. Taylor, 2021; T. Taylor & S. A. Taylor, 2021).
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
