Abstract
Parkinson’s disease (PD) is characterized by degeneration of dopaminergic neurons in the substantia nigra (SN), leading to motor impairments, while hippocampal dysfunction contributes to memory deficits. Botulinum neurotoxin (BoNT), a therapeutic modulator of acetylcholine (ACh) release, its cognitive effects remain underexplored. We investigated the effect of BoNT on spatial learning, memory, microglia and hippocampal neurogenesis in 1-methyl-4-phenyl-1,2,3,6-tetrahydropyridine (MPTP)-induced mouse model of PD. Behavioral tests, including the open field, novel object recognition, and Morris water maze, demonstrated significant improvements in locomotion, learning, and memory with BoNT treatment. BoNT increased the number of doublecortin (DCX)-positive immature neurons and percentage of bromodeoxyuridine (BrdU)/neuronal nuclei (NeuN) double-positive cells, while the reduced number of microglia was evident in the hippocampal dentate gyrus (DG). Additionally, histological analyses revealed BoNT-mediated protection of pyramidal neurons in hippocampal cornu ammonis (CA)-1 and CA3 regions. These findings suggest that BoNT mitigates memory deficits by promoting neurogenesis in experimental PD.
Introduction
Parkinson’s disease (PD) is an adult onset-progressive neurodegenerative disorder that affects 1%-2% of the population over the age of 60. 1 Given the increased risk of sporadic or genetic changes leading to neurodegeneration during aging, the global prevalence of PD continues to increase each year.1,2 The selective loss of dopaminergic neurons in the substantia nigra (SN), resulting in dopamine deficiency is strongly associated with clinical symptoms of PD. 3 The pathogenic events in PD deteriorate neuroplasticity and contribute to various movement disorders, including tremors, bradykinesia, rigidity, and postural instability. 4 As the severity of PD intensifies, progressive cognitive deficits arising from hippocampal dysfunction, often lead to dementia. 5 Despite prominent motor deficits, cognitive decline is also considered one of the most common and debilitating non-motor symptoms in PD. 6 The hippocampus is an important portion of the limbic system that plays a crucial role in encoding and retrieving memory. 7 In PD, hippocampal neurons are also highly vulnerable to microglia-mediated neuroinflammation and mitochondrial oxidative stress. 8 The dentate gyrus (DG) of the hippocampus retains the ability to produce new neurons in adulthood, reflecting the neuroregenerative capacity of the brain to counteract neuronal loss and facilitate neuroplasticity of memory. 9 Ample experimental evidence links impaired hippocampal neurogenesis to cognitive deficits observed in PD. 10 Notably, abnormal proliferation and activation of microglia have been implicated in pathogenic changes associated with neuroregenerative failure and neuronal loss.11,12 Therefore, pharmacological interventions that boost neural regenerative plasticity and promote neuroprotection in the hippocampus by attenuating pathogenic mechanisms at the level of microglia have been disseminated as effective therapeutics to mitigate memory loss in PD.
Botulinum neurotoxin (BoNT), produced by the bacterium Clostridium botulinum, causes botulism, a dreadful disease that affects the respiratory system and nervous system of humans and animals, leading to death.13,14 However, its therapeutic value has been realized in minimal doses to treat various diseases, including cervical dystonia, blepharospasm, sialorrhea, hyperhidrosis, and overactive bladder. 15 BoNT inhibits the release of acetylcholine (ACh) at the neuromuscular junction by cleaving soluble N-ethylmaleimide-sensitive factor attachment protein receptors (SNARE), essential for fusion of synaptic vesicles. 16 This blockade prevents muscle contraction, leading to temporary paralysis of the targeted muscles. The paralytic effect of BoNT appears to be dose-dependent and reversible, making its pharmacological form useful for treating various hyperactive neuromuscular conditions and movement disorders, including PD.13,14 Although the pathogenic roles of abnormal ACh levels in PD remain unclear, BoNT has emerged as a promising therapeutic agent for managing motor symptoms of PD, suggesting that it may act directly on ACh pathways or through mechanisms independent of ACh.13,17,18 Recently, we investigated the effects of BoNT on behavioral and biochemical changes in aging mice. The results showed that BoNT enhances cognition, increases platelet count in circulation, and provides neuroprotection in the hippocampus. 19 Further, we identified that the improvement of memory functions mediated by BoNT is associated with enhanced neurogenic processes accompanied by reduced microglial activation in the hippocampus of aging experimental mice. 20 Regardless of its primary mechanism of action through the regulation of ACh release, the therapeutic effects of BoNT have also been reported to be associated with cell cycle regulation, cellular differentiation, and neurotrophic functions.21,22 Considering these facts, we hypothesize that BoNT treatment could reverse cognitive deficits in PD by enhancing the differentiation of neural stem cells (NSCs) into new neurons and providing neuroprotection in the hippocampus. In this study, we investigated the effects of BoNT treatment on locomotion, and cognitive behavior, focusing on spatial learning and memory in an 1-methyl-4-phenyl-1,2,3,6-tetrahydropyridine (MPTP)-induced mouse model of PD using the open field test (OFT), novel object recognition (NOR) test, and Morris water maze (MWM) test. To assess its role in regulating the neurogenic process in the hippocampus, we quantified doublecortin (DCX)-positive immature neurons and assessed the neurogenic fate of newborn cells using bromodeoxyuridine (BrdU) and neuronal nuclei (NeuN) double-positive cells using immunohistochemistry followed by confocal microscopy assessment. In addition, the number of pyramidal neurons in the cornu ammonis (CA)-1 and CA3 regions of the hippocampus was quantified using Nissl staining. Additionally, we evaluated the number of ionized calcium-binding adaptor molecule (Iba)-1-positive microglia using immunohistochemistry.
Materials and Methods
Animals and Experimental Design
C57BL/6J male mice of age 3-4 months old were procured from Biogen Laboratories, Bangalore, India. They were maintained in the central animal facility of the institute under a 12-12 h light-dark cycle at a controlled temperature of 25 ± 2°C with free access to feed and drinking water. Experimental mice (N = 24) were randomly divided into 4 groups, namely, Group 1: Control, Group 2: BoNT, Group 3: MPTP, and Group 4: MPTP + BoNT. The experimental mice in groups 3 and 4 received an intraperitoneal injection of 30 milligram (mg) of MPTP (Sigma-Aldrich, USA) per kilogram body weight (Kg BW) for five consecutive days (Day 1 to 5). The effective dose of BoNT was selected based on a previously published study, in which no signs of toxicity were reported. 19 The mice in groups 2 and 4 were intramuscularly injected with 1U of BoNT (Allergan, Ireland) per Kg BW after 48 h of the last MPTP injection (Day 8). After 14 days of BoNT injection, experimental animals of all groups received intraperitoneal injections of 50 mg of BrdU (Sigma-Aldrich, USA) per Kg BW for five consecutive days to label the dividing cells. Then animals were subjected to behavioral examinations. Activities of animals during the behavioral experiments were tracked using the SMART 3.0 module (Panlab, Spain). After completion of behavior tests mice were perfused and the brains were collected and subjected to histological and immunohistochemical procedures. All experimental procedures were conducted in accordance with the approval granted by the Institutional Animal Ethical Committee (IAEC), Bharathidasan University, under the regulations of the Committee for the Purpose of Control and Supervision of Experiments on Animals (CPCSEA), India (Ref. No: BDU/IAEC/P26/2021).
Behavioral Experiments
Open Field Test
Open field test (OFT) is a widely used behavioral test to assess locomotion-based explorative behaviors in experimental mice. The OFT apparatus is a square-shaped arena with the size of 120 cm × 120 cm sub-divided into 16 equal sections. After allowing the animals to acclimate to the apparatus, the test was initiated by placing the mouse at the center of the arena and the uninterrupted activities of each mouse were captured for 5 min. Three trials for 3 consecutive days were done. At the end of each trial, the apparatus was cleaned and wiped with 70% ethanol. The number of grids crossed and the distance travelled by the animals were evaluated and tracked using SMART 3.0 module. 23
Novel Object Recognition Test
Novel object recognition (NOR) is a widely used behavioral paradigm for assessing recognition memory in experimental mice. This test is based on the natural tendency of animals to explore novel objects over familiar ones as the reflection of their innate exploratory behavior. The procedure of NOR test involves three stages: habituation, training, and testing phase. During the habituation phase, each mouse was allowed to explore an empty arena (30 × 15 × 30 cm). The test arena was digitally divided into 2 equal portions, zone 1 (red) and zone 2 (blue) using SMART 3.0, and 2 yellow-colored cylindrical wooden objects of identical size and shape were positioned in the middle of each zone. Using SMART 3.0 software, two circular inner zones designated as zone 3 (brown) and zone 4 (green) were digitally inserted around each object. During the training phase, each mouse was released in the center of the arena and was allowed to freely explore the objects for 5 min for 3 consecutive trials. After 2 h, the test trial was conducted, in which the object kept in zone 3 was substituted by a novel, green-colored rectangular-shaped wooden object. During the test trial, the mouse was released into the arena and was allowed to explore the objects for 5 min. The overall activities of each mouse were tracked using SMART 3.0 software. The time spent by the mouse in both zones and the ability of the mouse to recognize the novel object were evaluated. The percentage of the discrimination index was calculated using the formula, Discrimination index % = (Exploration time in novel object − exploration time in familiar object)/(total exploration time) × 100. 19
Morris Water Maze Test
The Morris water maze (MWM) test is widely applied to determine the ability of spatial navigation and retention of memory in rodents. It consists of a circular tank of diameter 150 cm and a depth of 50 cm filled with water and made opaque using milk powder. The temperature of the water was maintained at 20-22°C. The entire MWM apparatus was digitally divided into 4 quadrants namely, zone 1, zone 2, zone 3, and zone 4. A circular platform was placed in the middle of the zone 4 approximately 1 cm below the water surface and marked as zone 5 using SMART 3.0. Additionally, 4 visual cues were placed on each wall of the behavioral room for spatial orientation. Briefly, the test consisted of a learning phase for 8 consecutive days and a probe trial after 24 h of the learning phase. In the learning phase, 4 trials per day with an inter-trial interval of 30 min were conducted. During trial, each mouse was released systematically from different sides and was given 1 min to find the platform. On the first few days, if the platform was not found within 1 min, the mouse was gently guided to recognize the platform. If the mouse found the platform within 1 min, the tracking software was programmed to stop automatically and the mouse was taken back to its home cage. During the learning phase, the swimming path and time spent by the mouse in each quadrant were recorded. The time taken by each mouse to find the platform was used to plot the learning curve which represents the escape latency. During the probe test to assess the retention of memory, the platform was removed and the mouse was allowed to freely explore the maze. The activity of each mouse was recorded for 1 min and the time spent by the mouse in the target zone, where the platform was previously located was taken into account to evaluate the degree of memory retention. 19
Perfusion and Tissue Processing
On completion of the behavioral experiments, all mice were deeply anesthetized and transcardially perfused with 0.9% saline and 4% paraformaldehyde (PFA) (HiMedia, India). Brains were removed and post-fixed overnight in PFA at 4°C and then transferred to 30% sucrose solution for 24 h. After a week, brains were frozen in PolyFreeze medium (Sigma-Aldrich, USA) and 30 μm thick sagittal serial sections were made using a sliding microtome (Weswox, India) and stored in cryoprotectant at −20°C for further histological and immunohistochemical analysis as described earlier.19,24
Immunohistochemistry Analysis of Neurogenesis and Microglia in the Hippocampus
To estimate the degree of neurogenesis in the hippocampus, the number of DCX-positive immature neurons was quantified. The fate of NSCs was assessed using BrdU incorporation over 4 weeks, combined with co-labelling for the mature neuronal marker NeuN. 1 out of 12 sagittal brain sections with 360 μm distance apart from the left hemisphere of each brain was taken in 12 well plates (Tarson, India) and washed with 1× tris-buffered saline (TBS) (Tris HCl (HiMedia, India) and NaCl (SRL, India), pH 7.4) for three times using a shaker (Tarson, India) for 10 min each at room temperature (RT). To unmask the antigen, the brain sections were incubated with sodium citrate buffer (10 millimolar (mM) sodium citrate dihydrate (Thermo Fisher Scientific, USA) and 0.05% Triton X (HiMedia, India, pH 6.0), for 2 h at 65°C in a water bath (Kemi, India). Next, the brain sections were washed three times in 1× TBS for 10 min each at RT. Then, sections were blocked with 3% bovine serum albumin (BSA) (HiMedia, India) in 0.1% Triton-X (HiMedia, India) in TBS for 1 h on a shaker at RT. After blocking, the primary antibodies were mixed in the blocking solution, followed by incubation for 48 h at 4°C. To label the DCX-expressing immature neurons, the brain sections were incubated with rabbit anti-DCX antibody (Cell Signaling Technology, USA; 1:250 dilution) for 48 h at 4°C. Similarly, an additional set of brain sections was processed for immunohistological labelling of microglia and incubated in a solution constituted with a rabbit anti-Iba-1 antibody (Cell Signaling Technology, USA, 1:250 dilution) and incubated for 48 h at 4°C. After 48 h, the solution containing primary antibodies were removed and washed thrice in 1× TBS for 10 min each at RT. Then, the brain sections were treated with a fluorescent conjugated secondary antibody, namely, goat anti-rabbit Dylight 594 (Novus Biologicals, USA; 1:500 dilution) for 24 h at 4°C. The next day, the solution containing secondary antibodies was removed, and the brain sections were washed three times in 1× TBS for 10 min each at RT. To label the cell nuclei, the brain sections were treated with 0.1 mg/mL of 4′,6-diamidino-2-phenylindole (DAPI) (HiMedia, India) in TBST for 10 min. Further, the brain sections were washed in 1× TBS for 10 min and placed on the double-frosted microscopic slides (Borosil, India), and allowed to dry overnight in a lightproof slide tray. Upon complete drying, the ProLong™ glass antifade mountant (Thermo Fisher Scientific, USA) solution was applied to the brain sections on the microscopic slides and sealed with coverslips. Upon complete drying, slides were blind-coded and the immunolabelled brain sections were visualized under a fluorescence microscope (DM750, Leica Microsystems, Germany). The number of DCX- and Iba-1-positive cells was quantified in five non-overlapping microscopic fields of the hippocampal DG regions using ImageJ software with the cell counter plugin. The average numbers of DCX- and Iba-1-positive cells per microscopic field were estimated and represented as bar graphs.
Next, to assess the frequency of neuronal differentiation, a next set of brain sections was taken, which were washed in 1× TBS three times for 10 min each at RT. After antigen retrieval, the brain sections were incubated in 2 normal (N) HCl (Finar, India) for 10 min at 37°C on a shaker. Then, the brain sections were neutralized with 0.1 molar (M) borate buffer (boric acid (HiMedia, India), pH 8.5) for 10 min. Following this, the brain sections were washed three times in 1× TBS for 10 min each at RT. Further, the brain sections were incubated in 3% BSA for 1 h on a shaker at RT. After blocking, two different primary antibodies, namely, mouse anti-BrdU (Novus Biologicals, USA; 1:250 dilution) and rabbit anti-NeuN (Novus Biologicals, USA; 1:250 dilution), were added and incubated for 48 h at 4°C. Next, the primary antibodies were removed, and the sections were extensively washed in 1× TBS. The brain sections were treated together with two different species-specific secondary antibodies such as sheep anti-mouse Dylight 488 (Novus Biologicals, USA; 1: 500 dilution) and goat anti-rabbit Dylight 594 (Novus Biologicals, USA; 1: 500 dilution) antibodies at 4°C for 24 h. After 24 h, the secondary antibodies were removed, and the sections were washed three times in 1× TBS for 10 min each. Finally, the brain sections were arranged on the double-frosted slides (Borosil, India) and allowed to dry overnight in the dark. After complete drying, the sections were preserved using the ProLong™ glass antifade mountant (Thermo Fisher Scientific, USA) and dried in the dark. The brain sections were subjected to the double immunofluorescence assessment using a laser-scanning confocal microscope (LSM 710, Carl Zeiss, Germany) at the central instrumentation facility of Bharathidasan University and the Z-stack optical images of hippocampal DG were obtained to assess the colocalization of the BrdU with NeuN. For the frequency of neuronal differentiation, 25 BrdU-incorporated cells (green) in hippocampal DG from each animal were assessed. The BrdU-positive cells that are co-labelled with NeuN (red) were considered double-positive cells (yellow) and multiplied by four to estimate the percentage of double-positive cells, representing the newborn neurons. 24
Nissl Staining
For the quantification of pyramidal neurons in the hippocampus, 1 out of 12 brain sections 360 μm distance were fixed on microscopic glass slides coated with 3-aminopropyl triethoxysilane (APTES) (SRL, India). 100 mg of cresyl violet acetate (SRL, India) was dissolved in 250 μL of glacial acetic acid (SRL, India) and 100 mL of distilled water at 60°C using a magnetic stirrer (Remi, India). After allowing it to reach ambient temperature, the cresyl violet solution was purified using Whatman®filter paper No.1 (Sigma, USA). The slides containing the brain sections were first immersed in an alcohol-chloroform (1:1) solution for three minutes. The brain sections were rehydrated with 100%, 95% ethanol, and double distilled water for three minutes. Following this, the sections were immersed in cresyl violet solution for 30 min. Then the brain sections were washed with double-distilled water and sequentially treated with 100%, 95% ethanol, and finally in xylene (Merck, Germany) each for three minutes. Upon air drying, the sections were mounted with dibutyl phthalate polystyrene xylene (DPX) (Merck, Germany). A microscope linked to a computer installed with the ImageJ along cell counter plugin was used to capture the Nissl-positive cells in cornu ammonis (CA)1 and CA3 regions of the hippocampus. The number of pyramidal neurons was quantified from five non-overlapping microscopic images of the CA regions in the hippocampus for each animal, and the average number of cells per microscopic field was calculated as previously described. 25
Results
BoNT Improves Locomotory Performance in the MPTP-Induced Mouse Model of PD
To determine the effect of BoNT locomotory behaviors, experimental mice were subjected to OFT. The total number of grids crossed by PD mice was found to be less when compared to control, BoNT, and PD + BoNT-treated mice. In addition, the distance travelled by the mice in the PD group was reduced than mice in the control, BoNT, and PD + BoNT groups. BoNT-treated mice crossed more grids than mice in control, PD, and PD + BoNT groups. Mice in the PD + BoNT treated groups crossed more grids than mice in the PD group (Control = 36 ± 10, BoNT = 68 ± 7, PD = 20 ± 7, PD + BoNT = 56 ± 12). Eventually, mice in the BoNT-treated group covered more distance than mice in the control, PD, and PD + BoNT groups. Notably, the distance travelled by mice in PD + BoNT was found to be increased than the PD group (Control = 33 ± 9, BoNT = 60 ± 6, PD = 18 ± 4, PD + BoNT = 51 ± 9) (Figure 1). BoNT Improves Locomotory Performance During OFT in the MPTP-Induced Mouse Model of PD. Representative Tracking Images of a Mouse During OFT (A) Control, BoNT, PD, PD + BoNT. The Bar Graph Represents (B) the Number of Grids Crossed and (C) the Distance Travelled by the Experimental Mice. (*, **, *** Indicates P Value ≤0.05, ≤0.01 and ≤0.001 Respectively).
BoNT Improves Object Recognition Memory in MPTP-Induced Mouse Model of PD
To examine the object recognition-based memory functions, experimental mice were subjected to the NOR test. During the training phase, the mice were allowed to freely explore the objects kept in the arena. In the test phase, one of the familiar objects was replaced with a novel object. During the experiments, mice in BoNT showed an increased tendency to recognize the novel object more than the familiarized object compared to control, PD, and PD + BoNT groups. The BoNT-treated mice spent more time in the novel object zone compared to the control, PD, and PD + BoNT groups (Control = 5.6 ± 1, BoNT = 13.7 ± 3.5, PD = 1.8 ± 0.7, PD + BoNT = 5.5 ± 1.4). Eventually, the percentage of discrimination index was found to be significantly higher in the BoNT-treated group. The ability to recognize the novel object was reduced in mice in the PD group, with a lower discrimination index compared to the control, BoNT, and PD + BoNT groups. In contrast, PD + BoNT showed an improvement in recognizing the novel object when compared to the PD group (Control = 4.5 ± 1, BoNT = 10.8 ± 3.2, PD = 1.2 ± 0.9, PD + BoNT = 4.3 ± 1.3) (Figure 2). BoNT Enhances Object Recognition Memory in the MPTP-Induced Mouse Model of PD. Representative Tracking Images of the Mouse During NOR (A) Control, BoNT, PD, PD + BoNT. The Bar Graph Represents (B) Time Spent in the Novel Zone, and (C) the Discrimination Index of the Experimental Mice. (*, *** Indicates P Value ≤0.05 and ≤0.001 Respectively).
BoNT Enhances Spatial Memory in MPTP-Induced Mouse Model of PD
During the MWM learning task, mice from all groups gradually learned to locate the hidden platform, however with varying durations. The control group demonstrated a consistent improvement in learning to find the platform, whereas mice in the PD group took significantly longer time to locate it, compared to the control group. In contrast, BoNT-treated mice exhibited improved performance in identifying the hidden platform compared to all other groups. Furthermore, mice in the PD + BoNT group showed significantly better performance than those in the PD group. In the probe test, used as a measure of memory retention, BoNT-treated mice spent significantly more time in the platform zone compared to the control, PD, and PD + BoNT groups. Mice in the PD group spent the less time in the platform zone, whereas those in the PD + BoNT group exhibited an increased duration in the platform zone compared to the PD group (Control = 34.5 ± 2.9, BoNT = 41.4 ± 0.9, PD = 18 ± 2.8, PD + BoNT = 32.6 ± 2.8) (Figure 3), highlighting the memory-enhancing effects mediated by BoNT. BoNT Improves the Spatial Learning and Memory in the MPTP-Induced Mouse Model of PD. Representative Tracking Images of a Mouse From Each Group During MWM (A) Control, BoNT, PD, PD + BoNT. The Bar Graph Represents (B) Escape Latency to Find the Hidden Platform and (C) Time Spent in the Platform Zone During the Probe Trial by the Experimental Mice. (*, **, *** Indicates P Value ≤0.05, ≤0.01 and ≤0.001 Respectively).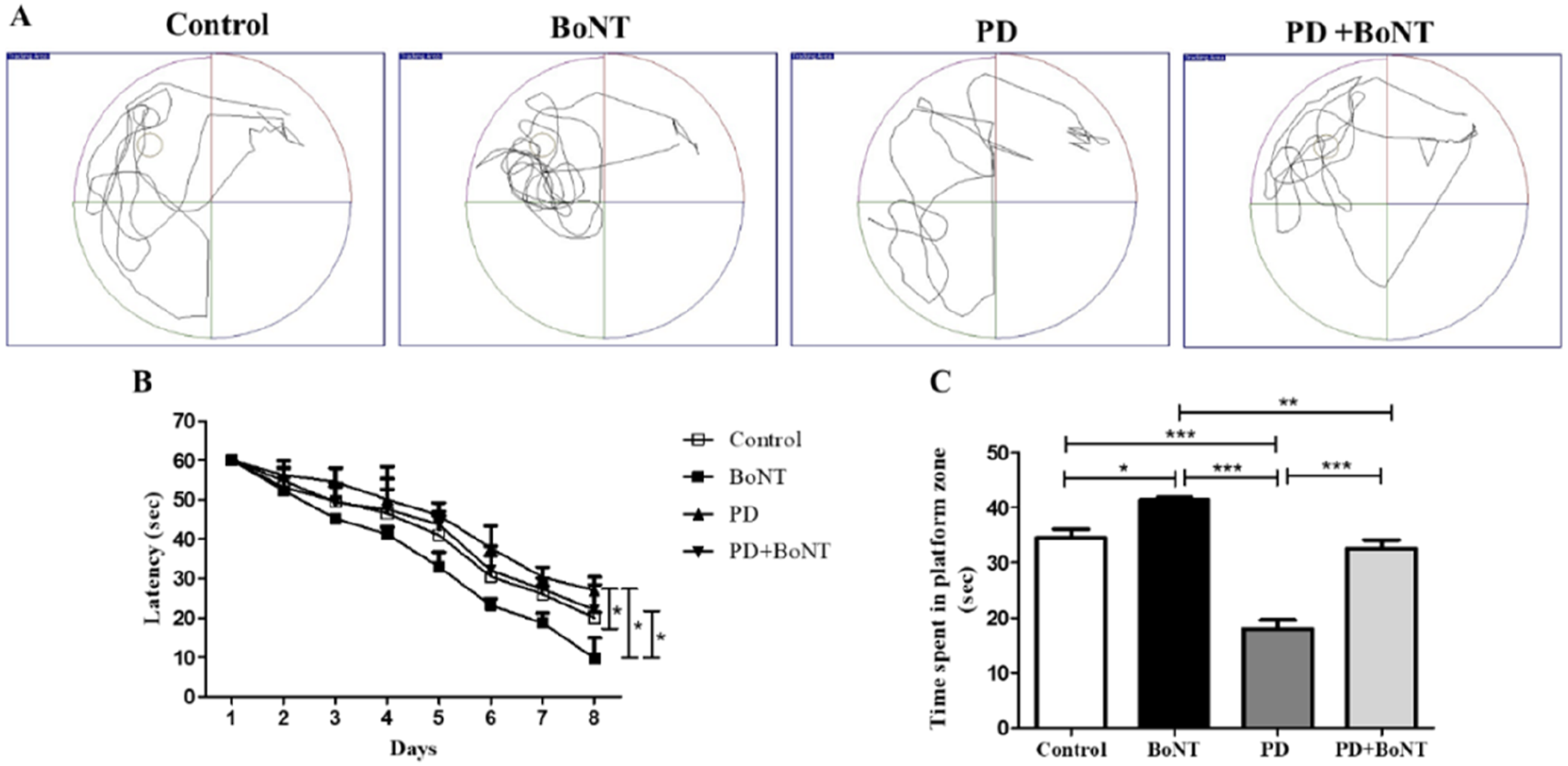
BoNT Attenuates the Impaired Hippocampal Neurogenesis in MPTP-Induced Mouse Model of PD
Hippocampal neurogenesis is a crucial component of neuroplasticity, playing a key role in learning and memory. While enhanced neurogenesis plays a pivotal role in memory encoding, PD has been characterized by altered hippocampal neurogenesis.
26
The rate of neurogenic process was assessed by quantifying the number of DCX-positive immature neurons in the hippocampus. PD mice exhibited a reduced number of DCX-positive cells in the DG of the hippocampus compared to the control, BoNT, and PD + BoNT groups. In contrast, the BoNT-treated group showed a significant increase in the number of DCX-positive cells. In the PD + BoNT group, a notable increase in DCX-positive cells was observed compared to the PD group (Control = 55.7 ± 15.4, BoNT = 62.7 ± 18.4, PD = 30 ± 3.6, PD + BoNT = 52.5 ± 13.6), thereby indicating a BoNT-mediated significant defense against MPTP-induced impairment of neurogenesis (Figure 4). BoNT Enhances the Number of Immature Neurons in the Hippocampus. The Images Represent the Immunohistochemical Assessment of DCX-Positive Cells in the DG of the Hippocampus in (A) Control, BoNT, PD, PD + BoNT. Scale bar = 25 μm. The Bar Graph Represents (B) Number of DCX-Positive Cells in the DG of the Hippocampus of Experimental Mice. (*, **Indicates P Value ≤0.05 and ≤0.01 Respectively).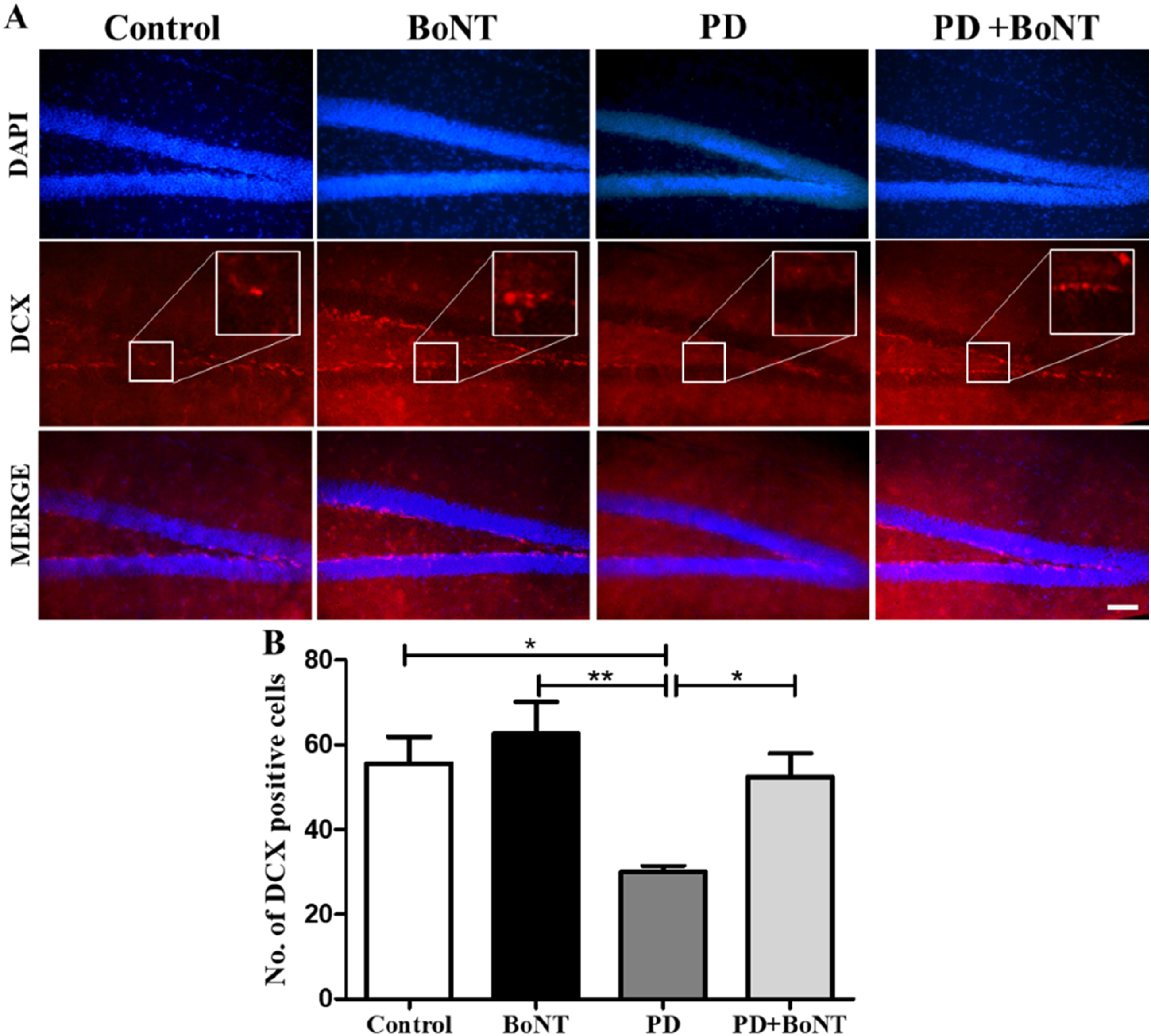
We further assessed neuronal differentiation in the hippocampus upon BoNT treatment using BrdU co-labeling with the mature neuronal marker NeuN. While the percentage of BrdU/NeuN double-positive cells was reduced in the PD group, it was significantly higher in the BoNT group compared to the control, PD, and PD + BoNT groups. Notably, the percentage of BrdU/NeuN double-positive cells was increased in the PD + BoNT group compared to the PD group (Control = 72 ± 3, BoNT = 84 ± 2, PD = 53 ± 6, PD + BoNT = 67 ± 4), indicating that BoNT facilitates neuronal differentiation of NSCs in PD (Figure 5). BoNT Promotes Neuronal Differentiation in the Hippocampus. The Image (A) Representative Image of BrdU/NeuN Positive Cells in the DG of Hippocampus Scale bar = 100 μm. The Bar Graph Represents (B) the Percentage of BrdU/NeuN Positive Cells in the DG of the Hippocampus of Experimental Mice. (*, **, *** Indicates P Value ≤0.05, ≤0.01 and ≤0.001 Respectively).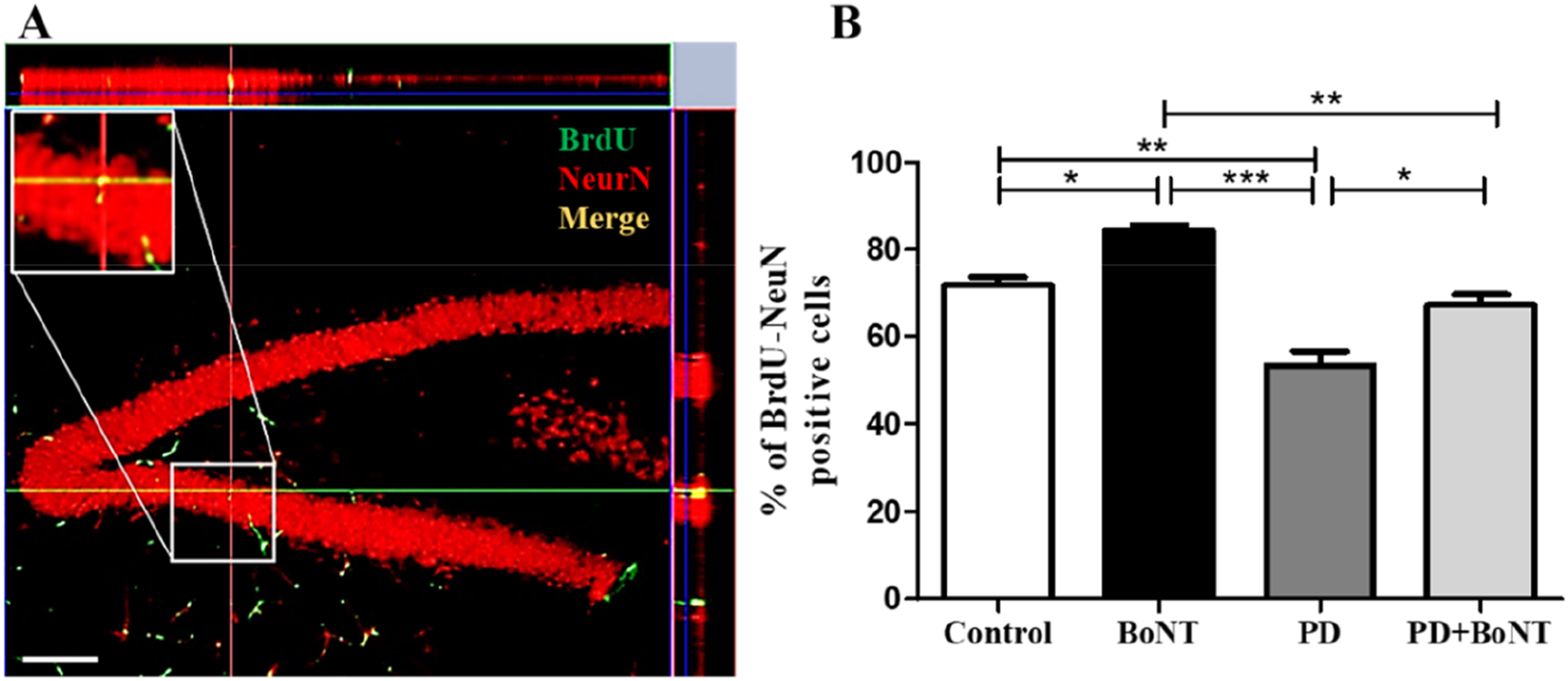
BoNT Regulates the Aberrant Microglial Count in MPTP-Induced Mouse Model of PD
Microglia are the most active immune cells in the brain, playing key roles in immunosurveillance and synaptic plasticity in the normal brain. Activated microglia mediate neuroinflammation, leading to the impairment in neurogenesis has been well documented in many neurodegenerative conditions including PD.
27
To determine whether BoNT treatment harnesses on MPTP-induced neuroinflammation at the level of microglia, the number of Iba-1-expressing cells in the brains of the experimental groups was compared. The results revealed that the number of Iba-1 positive cells in the hippocampal DG was significantly higher in the PD group compared to the control, BoNT, and PD + BoNT groups. In contrast, the number of Iba-1 positive cells in the hippocampus of the BoNT group was reduced compared to the PD group. The PD + BoNT-treated mice showed a decrease in Iba-1 positive cells in the DG of the hippocampus compared to the PD group (Control = 22 ± 4, BoNT = 18 ± 5, PD = 36 ± 10, PD + BoNT = 23 ± 4) (Figure 6). BoNT Reduces the Aberrant Neuroinflammation in the Hippocampus. The Images Represent the Immunohistochemical Assessment of Iba-1 Positive Cells in the DG of the Hippocampus in (A) Control, BoNT, PD, PD + BoNT. Scale bar = 25 μm. The Bar Graph Represents (B) Number of Iba-1 Positive Cells in the DG of the Hippocampus of Experimental Mice. (*, **, *** Indicates P Value ≤0.05, ≤0.01 and ≤0.001 Respectively)
BoNT Prevents the Deterioration of Pyramidal Neurons in the CA1 and CA3 Regions of the Hippocampus in MPTP Induced Mouse Model of PD
The hippocampus plays a crucial role in cognition and exhibits specialized neuroplasticity properties. Among the hippocampal subfields, the pyramidal neurons in the CA1 and CA3 regions are crucial for temporal, contextual, and spatial memory, while their disintegration and loss can lead to memory impairment.
28
To determine neuronal density, brain sections were subjected to cresyl violet staining. In the PD group, there was distinct cell loss in the CA1 and CA3 regions of the hippocampus compared to the control, BoNT, and PD + BoNT groups. In the BoNT-treated group, the number of pyramidal neurons in the CA1 and CA3 regions was significantly increased compared to the control, PD, and PD + BoNT groups. The PD + BoNT-treated group also exhibited a higher number of pyramidal neurons in the hippocampus than the PD group. These results indicate that BoNT treatment provides neuroprotection against MPTP-induced alterations in the CA1 (Control = 126 ± 7, BoNT = 145 ± 9, PD = 105 ± 9, PD + BoNT = 128 ± 8). and CA3 (Control = 124 ± 6, BoNT = 152 ± 22, PD = 94 ± 12, PD + BoNT = 125 ± 9) regions of the hippocampus (Figure 7). BoNT Protects Pyramidal Neurons in the CA1 and CA3 Regions of the Brain in MPTP Induced Mouse Model of PD. The Images Represent the Nissl-Stained CA-1 and CA-3 Region of the Hippocampus of (A) Control, BoNT, PD, PD + BoNT. The Bar Graph Represents (B) the Number of Pyramidal Neurons in the CA1 Region and (C) the Number of Pyramidal Neurons in the CA3 Region of the Hippocampus of the Experimental Mice. (**, *** Indicates P Value ≤0.05, ≤0.01 and ≤0.001 Respectively)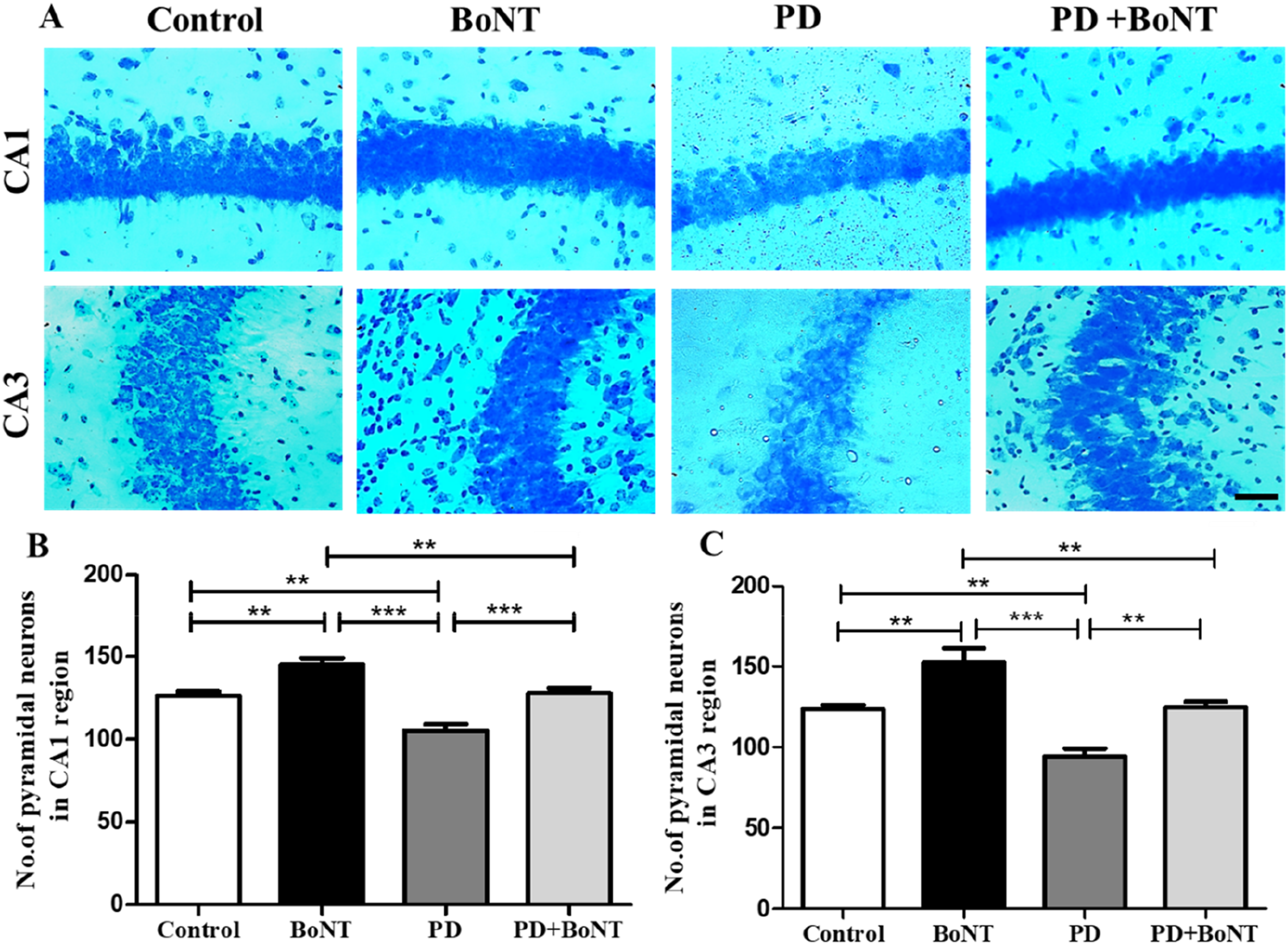
Discussion
PD is a devastating neurodegenerative disorder, increasingly characterized by chronic memory impairment in addition to motor deficits. 5 In the physiological state, hippocampal neurogenesis has been shown to facilitate memory, while its impairment during the neurological diseases is linked to the development of memory loss.9,29,30 Therefore, therapeutic strategies aimed at boosting hippocampal plasticity have become a leading focus in preclinical research to reverse the memory decline in aging and neurodegenerative disorders. Previously, Yesudhas et al 19 demonstrated that BoNT treatment improves hippocampal plasticity and memory in aging experimental animals. In this study, we extended these findings to provide evidence for the improvement of neurogenesis by BoNT as a potential mechanism for memory enhancement in the MPTP-induced mouse model of PD. The present study demonstrates that BoNT treatment enhances learning and memory, accompanied by improved hippocampal neurogenesis in the MPTP-induced experimental model of PD. This is evidenced by a better performance in behavioral tests, a significant increase in the number of DCX-positive immature neurons and enhanced frequency of neuronal differentiation as indicated by the increased percentage of BrdU/NeuN newborn neurons in the hippocampus of PD + BoNT treated mice compared to the PD group. Furthermore, an increased number of pyramidal neurons in the CA1 and CA3 regions of the hippocampus was observed in both BoNT- and PD + BoNT-treated mice, indicating BoNT-mediated neuroprotection. These findings suggest that the hippocampal plasticity enhancing the memory observed following BoNT treatment is likely attributable to its proneurogenic effects.
Depletion of dopamine in the forebrain has been a well-established pathogenic signature of PD, whereas BoNT has been shown to modulate the abnormal release of ACh.14,31,32 Although the most common treatment for PD is dopamine replacement therapy, which provides only symptomatic relief, few strategies have explored alternative pharmacological therapeutics such as enhancing neuroregeneration to compensate for the progressive degeneration of dopaminergic neurons. Despite its established ACh modulatory capacity, BoNT has been shown to exert pleiotropic effects in the brain.16,33 The mechanisms through which BoNT mediates these effects remain an area of active investigation. Notably, the increased discharge of neuroinflammatory molecules from activated microglia is known to induce neuronal loss and impair the neurogenic process in the hippocampus.34,35 Clinical and experimental data indicate that PD is closely linked to neuroinflammatory processes, including T-cells infiltration, microglial activation, and blood-brain barrier (BBB) disruption. 36 Several animal models of PD, such as those induced by MPTP, 6-hydroxydopamine, lipopolysaccharide (LPS), or rotenone, exhibit microglia-associated inflammation. 37 Strategies aimed at reducing neuroinflammation and promoting sustained neurogenesis could thus serve as promising therapeutic interventions in PD. BoNT has been shown to minimize neuroinflammatory markers and is well-established for its ability to attenuate pain and related symptoms. While cyclooxygenase (COX) is the primary inducer of pain and neuroinflammatory processes, its inhibition by BoNT has recently been noticed. 20 Notably, increased expression of COX in microglia has been reported to induce its activation, contributing to neuroinflammation. 38 In this study, BoNT-treated mice showed reduction in the number of Iba-1 positive microglial cells in the hippocampus which may partly indicate that BoNT mitigates neuroinflammation. This observation corroborates with a previous findings by Piotrowska et al, 39 which demonstrated reduced activation of cultured microglia upon treatment with BoNT, leading to a decreased release of proinflammatory molecules. Moreover, intratemporomandibular joint (TMJ) injection of BoNT-A has been shown to deactivate inflammatory pathways by reducing the P2X7 receptor/Cathepsin S (CatS)/Fractalkine (FKN) microglia-activation pathway in the trigeminal subnucleus caudalis in a rat model of arthritis. 40 Eventually, the effect of BoNT on microglial inactivation could also be related to its COX-reducing properties in the brain. 20 This reduction in the pathogenic signatures of microglia could effectively facilitate neuroprotection and create a more favourable environment for hippocampal neurogenesis. Recent reports showed the administration of BoNT aids in harnessing the microglial activation in many neurological and mood disorders. While depression has been known to induce memory impairment due to a reduction in neurogenesis, BoNT-A has been reported to reduce depressive-like symptoms in the reserpine-induced PD mouse model by decreasing hippocampal microglial engulfment and neuroinflammation. 41 Gene microarray analysis by Wang et al 42 reported upregulation of genes CHRM3, SPOCK1, and NELL1, responsible for neuronal development and survival in human SH-SY5Y neuroblastoma cells after treatment with BoNT for 4 and 48 h. Notably, BoNT treatment has been demonstrated to induce different neuritogenic activities in chicken nerve-muscle cocultures and cultured mouse motor neurons.43,44 This indicates that BoNT-mediated neural plasticity could originate at the neuronal differentiation of NSCs and survival with synaptogenic potentials. 45 BDNF, is a neurotrophic factor essential for synaptogenesis and the regulation of neurogenesis at the level of neuronal differentiation and survival of newborn neurons.45,46 Notably, reduced levels of BDNF have been known to be associated with the pathogenesis of PD. BoNT treatment has been shown to induce the expression of BDNF and provide nerve regeneration in an experimental rat model of sciatic nerve injury. 47 While decreased neurogenesis has been implicated in the onset and progression of depression, Li et al reported that the antidepressant-like effects of BoNT are mediated through enhanced serotonin signaling and modulation of the BDNF/ERK/CREB pathways.41,48 Our previous findings showed that BoNT alleviates anxiety-like behaviors, reduces neuroinflammation, and provides neuroprotection to hippocampal neurons in aging mice. 23 The current study demonstrates that BoNT also induces neurogenesis in the hippocampus thereby improving learning and memory in the preclinical model of PD. These findings, in conjunction with existing literature, further underscore the therapeutic potential of BoNT in promoting neuroregenerative plasticity during the pathogenesis of PD. Consequently, further research into the pro-neurogenic and pro-cognitive effects of BoNT is crucial to elucidate its underlying molecular mechanisms and refine its therapeutic applications. However, the use of BoNT has been associated with some adverse effects, and the long-term effects of BoNT remain unknown. Therefore, future studies delineating the long-term effects of BoNT on neurogenic capacity, as well as its pharmacological and toxicological impacts on the neurobiology of diseases, need to be considered.
Conclusion
The present study demonstrates that BoNT treatment ameliorated memory impairments in the MPTP-induced mouse model of PD. The BoNT-mediated enhancement of hippocampal neurogenesis and neuroprotection likely accounts for the observed improvements in memory performance. Additionally, the reduced microglial activation suggests that BoNT treatment creates a more favorable environment for effective neuronal differentiation and survival, mitigating the neuroinflammatory processes associated with the pathogenesis of PD. These findings highlight the potential dual mechanism of BoNT, involving both neuroprotection and the promotion of neuroplasticity, which are critical for addressing cognitive deficits in PD. While prior studies have demonstrated that BoNT can exhibit neurotrophic and long-term effects, future studies are required to investigate the sustained efficacy, potential delayed toxicity, and regulation of neuroplasticity associated with prolonged exposure of BoNT.
Ethical Statement
Ethical Approval
This work was reviewed and approved by the Institutional Animal Ethics Committee (IAEC), Bharathidasan University (Ref. No: BDU/IAEC/P26/2021) under the regulation of the Committee for the Purpose of Control and Supervision of Experiments on Animals (CPCSEA), India.
Footnotes
Acknowledgments
The authors also thank RUSA 2.0, Biological Sciences, Bharathidasan University, (TN RUSA: 311/RUSA (2.0)/2018 dt. 2 December 2020) and Anusandhan National Research Foundation (ANRF)/Science Engineering Research Board (SERB) (CRG/2023/005266), Department of Science and Technology (DST), Government of India for their financial support. The authors acknowledge University Science Instrumentation Centre (USIC)-BDU and DST-PURSE (Phases 1 and 2), BDU for the confocal analysis. The authors thank Dr. Muthu Kumar, USIC-BDU, for the excellent technical support with the confocal microscopy.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
The article contains all the data required to evaluate the findings.
